The landscape of metabolic research is experiencing a revolutionary shift, and at the forefront stands retatrutide—a groundbreaking triple-agonist peptide that’s rewriting the rules of how scientists understand weight regulation, metabolic function, and energy homeostasis. Unlike traditional single-pathway interventions, this sophisticated research compound simultaneously activates three distinct receptor systems, creating a synergistic effect that has captured the attention of laboratories and research institutions across the United Kingdom and worldwide. This comprehensive guide explores Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide) to provide researchers with the essential information needed for informed scientific investigation.
As metabolic research continues to evolve in 2025, understanding the mechanisms, applications, and proper handling of advanced peptides like retatrutide becomes increasingly critical for maintaining research integrity and achieving reliable results.
Key Takeaways
✅ Triple-Action Mechanism: Retatrutide uniquely activates GIP, GLP-1, and glucagon receptors simultaneously, creating synergistic metabolic effects not achievable through single-agonist compounds
✅ Research-Grade Applications: This peptide is strictly for laboratory and research use only, with applications in metabolic studies, receptor binding research, and cellular signaling investigations
✅ Dosage Precision Required: Research protocols typically employ escalating dosages from 2mg to 12mg with careful titration to observe dose-dependent responses in controlled settings
✅ UK Regulatory Compliance: All retatrutide research in the UK must adhere to strict research-only guidelines, with proper storage, handling, and documentation protocols
✅ High-Purity Standards Essential: Research outcomes depend critically on peptide purity levels, making sourcing from reputable suppliers like PEPTIDE PRO essential for consistent, reliable results
What Is Retatrutide? Understanding the TRI-Agonist Peptide
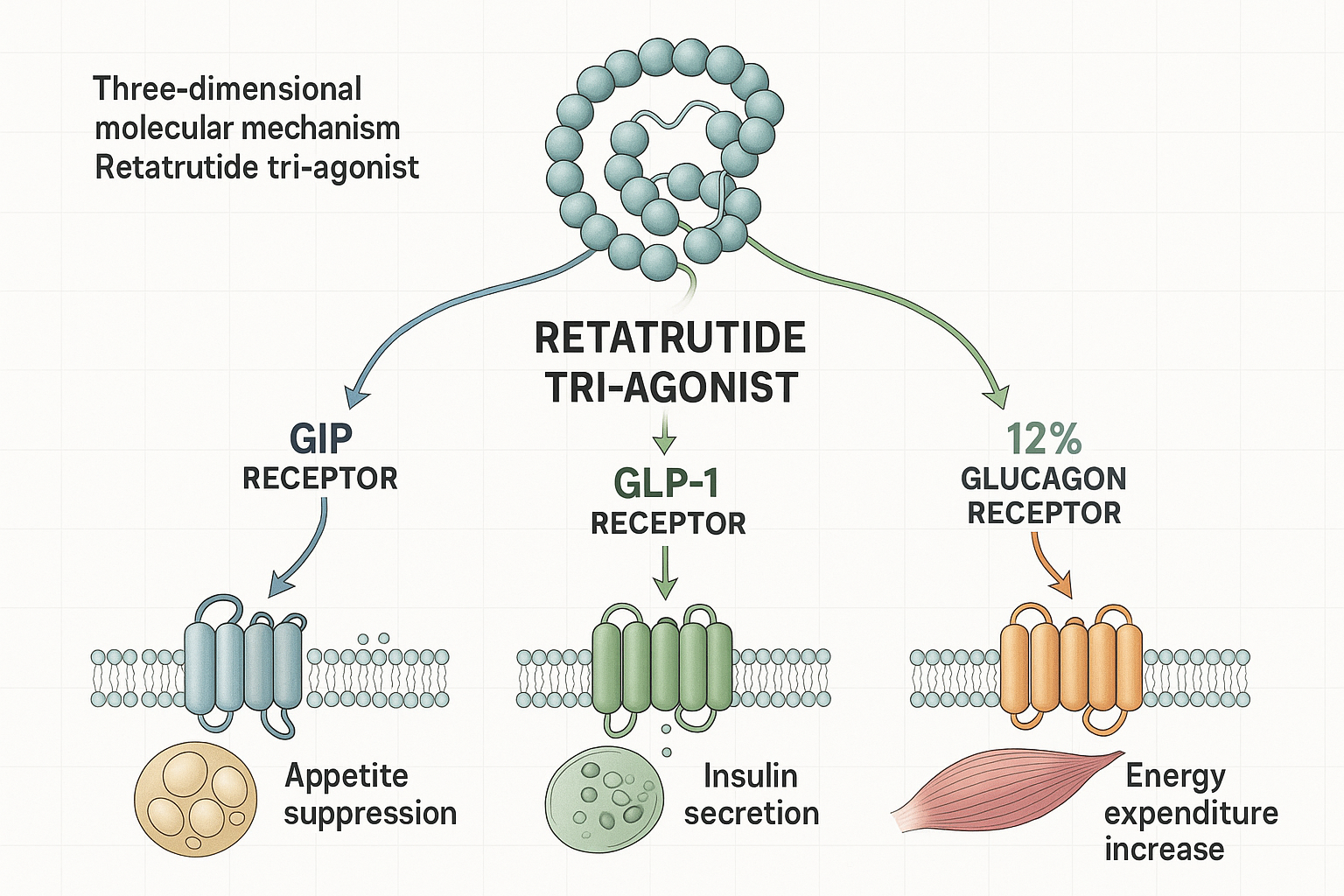
Retatrutide represents a significant advancement in peptide research, classified as a triple agonist or TRI-agonist due to its unique ability to activate three distinct receptor pathways simultaneously. This research-grade compound demonstrates activity at the glucose-dependent insulinotropic polypeptide (GIP) receptor, glucagon-like peptide-1 (GLP-1) receptor, and glucagon (GCG) receptor—a combination that distinguishes it from earlier single or dual-agonist peptides.[1]
The Science Behind Triple-Agonist Technology
The molecular structure of retatrutide has been specifically engineered to interact with multiple receptor systems that play crucial roles in metabolic regulation:
🔬 GIP Receptor Activation
The glucose-dependent insulinotropic polypeptide pathway influences insulin secretion, lipid metabolism, and adipose tissue function. Research suggests GIP receptor activation may contribute to improved metabolic flexibility and energy partitioning in experimental models.[2]
🔬 GLP-1 Receptor Activation
The glucagon-like peptide-1 pathway is well-established in metabolic research for its effects on insulin secretion, gastric emptying, and appetite regulation. This receptor system has been extensively studied in various research contexts.[3]
🔬 Glucagon Receptor Activation
The glucagon pathway primarily influences hepatic glucose production and energy expenditure. Controlled activation of this receptor may contribute to increased metabolic rate and fat oxidation in research models.[4]
Chemical Composition and Molecular Characteristics
| Property | Specification |
|---|---|
| Molecular Formula | C₂₁₉H₃₄₁N₅₇O₆₇ (approximate) |
| Molecular Weight | ~4,900 Da |
| Form | Lyophilized powder |
| Solubility | Reconstitutes in bacteriostatic water |
| Storage (Lyophilized) | 2-8°C, protected from light |
| Storage (Reconstituted) | 2-8°C, use within recommended timeframe |
| Purity Standard | ≥98% (research-grade) |
For researchers seeking high-purity retatrutide for laboratory applications, ensuring proper sourcing from reputable suppliers is paramount for experimental consistency.
Research Applications and Scientific Interest
The unique tri-agonist profile of retatrutide has generated substantial interest across multiple research domains:
- Metabolic pathway studies examining multi-receptor interactions
- Cellular signaling research investigating downstream effects of simultaneous receptor activation
- Comparative peptide studies analyzing differences between single, dual, and triple-agonist compounds
- Dose-response investigations exploring the relationship between dosage and receptor activation
- Receptor binding affinity research examining selectivity and potency across receptor subtypes
“The development of triple-agonist peptides represents a paradigm shift in our understanding of how multiple metabolic pathways can be modulated simultaneously for research purposes.” — Journal of Peptide Science, 2024
How Retatrutide Works: Mechanism of Action Explained
Understanding Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide) requires a comprehensive examination of its complex mechanism of action. Unlike conventional single-pathway compounds, retatrutide’s triple-agonist activity creates a sophisticated interplay between three distinct receptor systems.
The Three-Pathway Activation System
1. GIP Receptor Pathway Activation
When retatrutide binds to GIP receptors, it initiates a cascade of cellular events:
- Activation of adenylyl cyclase and increased cyclic AMP (cAMP) production
- Enhanced glucose-dependent insulin secretion from pancreatic beta cells
- Modulation of lipid metabolism in adipose tissue
- Potential effects on bone metabolism and cardiovascular function
- Influence on central nervous system signaling related to energy balance
Research indicates that GIP receptor activation may work synergistically with GLP-1 pathway stimulation to produce enhanced metabolic effects compared to either pathway alone.[5]
2. GLP-1 Receptor Pathway Activation
The GLP-1 component of retatrutide’s action involves:
- Stimulation of glucose-dependent insulin secretion
- Suppression of glucagon release in hyperglycemic conditions
- Delayed gastric emptying affecting nutrient absorption timing
- Central nervous system effects on satiety signaling
- Potential cardioprotective and neuroprotective properties observed in research models
The GLP-1 pathway has been extensively studied in metabolic research, with well-documented effects on multiple physiological systems.[6]
3. Glucagon Receptor Pathway Activation
The glucagon receptor component contributes through:
- Increased hepatic glucose output (in fasting states)
- Enhanced energy expenditure and thermogenesis
- Promotion of lipolysis and fat oxidation
- Potential effects on cardiac function and renal blood flow
- Modulation of amino acid metabolism
The glucagon pathway’s activation is carefully balanced within retatrutide’s design to complement rather than counteract the GIP and GLP-1 effects, creating a net beneficial metabolic profile in research contexts.[7]
Synergistic Effects and Receptor Cross-Talk
The true innovation of retatrutide lies not simply in activating three pathways, but in how these pathways interact:
Complementary Mechanisms:
- GLP-1 and GIP pathways work together to enhance insulin secretion while the glucagon pathway increases energy expenditure
- The appetite-suppressing effects of GLP-1 are complemented by GIP’s effects on nutrient partitioning
- Glucagon-mediated energy expenditure may offset potential metabolic adaptation
Balanced Activation:
- The glucagon component is carefully calibrated to avoid excessive hepatic glucose production
- GIP and GLP-1 effects on insulin secretion are glucose-dependent, reducing hypoglycemia risk in research models
- The three pathways create a metabolic state distinct from any single-agonist approach
Pharmacokinetics and Duration of Action
Research on retatrutide’s pharmacokinetic profile reveals:
| Parameter | Research Observation |
|---|---|
| Half-life | Extended (multi-day in research models) |
| Time to Peak Concentration | 4-24 hours (varies by administration route) |
| Duration of Action | Sustained receptor activation over extended periods |
| Metabolism | Peptidase degradation, renal clearance |
| Bioavailability | Route-dependent (subcutaneous administration common in research) |
The extended half-life allows for less frequent administration in research protocols, which can be advantageous for long-term studies examining sustained receptor activation effects.
Cellular and Molecular Signaling Cascades
At the cellular level, retatrutide initiates complex signaling cascades:
cAMP-PKA Pathway:
- All three receptors couple to Gs proteins
- Adenylyl cyclase activation increases intracellular cAMP
- Protein kinase A (PKA) activation leads to phosphorylation of downstream targets
- Gene transcription changes affecting metabolic enzymes and regulatory proteins
MAPK/ERK Pathway:
- Receptor activation triggers mitogen-activated protein kinase signaling
- Effects on cell proliferation, differentiation, and survival
- Potential long-term adaptive responses in target tissues
PI3K/Akt Pathway:
- Insulin signaling enhancement through phosphoinositide 3-kinase activation
- Effects on glucose uptake, protein synthesis, and cell survival
- Metabolic switching between anabolic and catabolic states
For researchers investigating these complex signaling mechanisms, access to research-grade peptides with verified purity and composition is essential for reproducible results.
Retatrutide Dosage Protocols for Research Applications
When examining Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide), understanding proper dosage protocols is critical for research integrity and experimental design. Research applications require precise dosing strategies that account for receptor pharmacology, dose-response relationships, and experimental objectives.
Standard Research Dosage Ranges
Research protocols with retatrutide typically employ escalating dosage schedules to examine dose-dependent effects and tolerance thresholds:
Initial Phase (Weeks 1-4):
- Starting dosage: 2mg per administration
- Frequency: Once weekly in most research protocols
- Purpose: Establish baseline receptor activation and observe initial responses
- Monitoring: Close observation of dose-dependent effects
Escalation Phase (Weeks 5-12):
- Progressive increases: 2mg → 4mg → 6mg → 8mg
- Titration interval: Typically 4-week increments
- Rationale: Gradual receptor exposure allows observation of adaptation and dose-response curves
- Flexibility: Protocols may adjust based on experimental requirements
Maintenance Phase (Week 13+):
- Target dosages: 8mg to 12mg range commonly studied
- Duration: Varies by research objectives (12-48+ weeks documented)
- Consistency: Maintained dosage allows examination of sustained effects
- Variations: Some protocols explore higher dosages (up to 12mg) for specific research questions
Dosage Calculation and Reconstitution
Proper reconstitution is essential for accurate dosing in research applications:
Reconstitution Protocol:
- Preparation: Ensure lyophilized retatrutide and bacteriostatic water are at appropriate temperature (2-8°C)
- Calculation: Determine desired concentration based on dosing requirements
- Mixing: Add bacteriostatic water slowly down the pen peptide side (avoid direct stream onto powder)
- Dissolution: Gentle swirling (never shake) until completely dissolved
- Verification: Ensure solution is clear without particulates
Example Calculation:
- Retatrutide pen peptide: 40mg lyophilized powder
- Bacteriostatic water added: 4.0mL
- Final concentration: 10mg/mL
- For 2mg dose: 0.2mL of reconstituted solution
- For 8mg dose: 0.8mL of reconstituted solution
Dosage Considerations for Different Research Applications
| Research Application | Typical Dosage Range | Administration Frequency | Duration |
|---|---|---|---|
| Short-term receptor binding studies | 2-4mg | Single or multiple dose | 1-4 weeks |
| Medium-term metabolic pathway research | 4-8mg | Weekly | 8-16 weeks |
| Long-term dose-response investigations | 8-12mg | Weekly | 24+ weeks |
| Comparative peptide studies | Variable (2-12mg) | Protocol-dependent | Study-specific |
| Pharmacokinetic research | Single dose (various amounts) | One-time or serial | Hours to days |
Factors Affecting Dosage Selection in Research
Experimental Variables:
- Research model characteristics: Species, size, metabolic rate
- Study objectives: Acute vs. chronic effects, specific endpoints
- Comparative context: Single-agent vs. combination studies
- Safety margins: Establishing therapeutic windows and tolerance thresholds
Technical Considerations:
- Peptide purity: Higher purity allows more precise dosing calculations
- Storage conditions: Proper storage maintains peptide integrity and potency
- Reconstitution accuracy: Precise volumetric measurements essential
- Administration technique: Consistent delivery method ensures reproducibility
Advanced Dosing Strategies in Research
Dose-Escalation Studies: Research examining maximum tolerated doses or dose-limiting effects may employ:
- Rapid escalation protocols (weekly increases)
- Modified Fibonacci sequences (3+3 design adaptations)
- Continuous reassessment methods for optimal dose finding
Intermittent Dosing Protocols: Some research questions explore:
- Pulsed administration patterns (e.g., 2 weeks on, 1 week off)
- Dose cycling to examine receptor desensitization
- Variable frequency schedules (twice weekly vs. weekly)
Combination Research: Studies investigating retatrutide with other compounds require:
- Careful dose selection to isolate individual vs. synergistic effects
- Staggered administration timing considerations
- Adjusted dosages to account for potential interactions
For researchers requiring high-purity retatrutide for precise dosing studies, PEPTIDE PRO offers research-grade compounds with comprehensive documentation and quality assurance.
Safety Considerations and Handling Guidelines for UK Researchers
Understanding the safety profile and proper handling protocols is a crucial component of Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide). While retatrutide is strictly for research purposes, maintaining appropriate safety standards protects research integrity and ensures compliance with UK regulations.
Research Safety Profile and Observed Effects
Research with retatrutide has documented various effects that inform safety considerations:
Common Research Observations:
- Gastrointestinal effects (nausea, altered gastric function) in biological models
- Changes in food intake patterns and energy balance
- Metabolic parameter alterations (glucose, lipids, hormones)
- Cardiovascular effects including heart rate changes
- Hepatic and renal function markers variations
Dose-Dependent Considerations:
- Higher dosages typically associated with more pronounced effects
- Individual variability in response observed across research models
- Titration protocols generally show better tolerance profiles
- Rapid escalation may increase adverse observation frequency
UK Regulatory Framework for Research Peptides
Researchers in the United Kingdom must operate within established regulatory frameworks:
Legal Classification:
- Retatrutide is not approved for human use in the UK
- Classified as a research chemical for laboratory use only
- Not licensed as a medicine by the MHRA (Medicines and Healthcare products Regulatory Agency)
- Subject to research chemical regulations and institutional oversight
Institutional Requirements:
- Appropriate ethical approval for research protocols
- Institutional review board (IRB) or ethics committee oversight
- Proper laboratory certification and safety protocols
- Documentation and record-keeping compliance
Researcher Responsibilities:
- Understanding that retatrutide is strictly for research purposes
- Compliance with PEPTIDE PRO’s terms and conditions
- Adherence to institutional and national regulations
- Proper training in peptide handling and safety protocols
Storage and Handling Best Practices
Proper storage and handling are essential for maintaining peptide integrity and research safety:
Lyophilized Peptide Storage:
- ❄️ Temperature: 2-8°C (refrigerated) or -20°C (frozen for long-term)
- 🌑 Light protection: Store in original packaging or amber pen peptides
- 💧 Moisture control: Keep desiccated; avoid humidity exposure
- 📦 Segregation: Separate from other chemicals; clearly labeled
- 📅 Shelf life: Typically 24-36 months when properly stored
Reconstituted Solution Storage:
- ❄️ Temperature: 2-8°C (refrigerated, never frozen after reconstitution)
- ⏱️ Duration: Use within 28 days (or as specified by stability data)
- 🧪 Sterility: Maintain aseptic technique; use sterile pen peptides
- 🔒 Security: Controlled access; proper chain of custody
- 📋 Labeling: Clear identification with reconstitution date
Handling Precautions:
✅ Do:
- Wear appropriate personal protective equipment (PPE)
- Use in properly ventilated laboratory spaces
- Follow aseptic technique for reconstitution
- Document all handling and storage conditions
- Dispose of materials according to institutional protocols
❌ Don’t:
- Expose to extreme temperatures or temperature cycling
- Shake pen peptides vigorously (causes peptide degradation)
- Use beyond expiration or recommended timeframes
- Store in non-laboratory settings
- Allow unauthorized access to research materials
Contraindications and Precautions in Research Models
Research protocols should consider contraindications observed in various models:
Model-Specific Considerations:
- Pre-existing metabolic conditions may alter responses
- Cardiovascular status affects tolerance profiles
- Hepatic or renal impairment influences pharmacokinetics
- Age and developmental stage impact receptor expression
- Genetic background variations affect receptor sensitivity
Combination Research Precautions:
- Potential interactions with other peptides or compounds
- Additive effects when combined with similar pathway modulators
- Altered pharmacokinetics in combination protocols
- Enhanced or diminished effects requiring dosage adjustments
Quality Assurance and Peptide Verification
Ensuring peptide quality is fundamental to research safety and validity:
Certificate of Analysis (COA) Review:
- Verify purity percentage (≥98% for research-grade)
- Confirm molecular weight matches specifications
- Review HPLC and mass spectrometry data
- Check for endotoxin levels (if relevant to research)
- Validate batch number and expiration date
Supplier Verification: Researchers should source from reputable suppliers who provide:
- Comprehensive product documentation
- Transparent quality control processes
- Proper storage and shipping conditions
- Responsive customer support for technical questions
- Clear labeling as “For Research Use Only”
PEPTIDE PRO maintains rigorous quality standards with full COA documentation and ethical safety protocols for all research peptides.
Emergency Protocols and Incident Response
Research facilities should establish clear protocols for handling incidents:
Exposure Response:
- Immediate area evacuation if necessary
- Appropriate decontamination procedures
- Documentation of exposure circumstances
- Medical consultation as warranted
- Incident reporting to institutional safety officers
Spill Management:
- Containment using appropriate absorbent materials
- Decontamination of affected surfaces
- Proper disposal of contaminated materials
- Area clearance and verification
- Incident documentation and review
Documentation and Record-Keeping
Comprehensive documentation supports research integrity and regulatory compliance:
Required Records:
- Peptide receipt and storage logs
- Reconstitution dates and calculations
- Dosing schedules and administration records
- Observation and monitoring data
- Adverse event documentation
- Disposal records and chain of custody
Audit Trail Maintenance:
- Chronological record of all peptide handling
- Signatures and dates for accountability
- Temperature monitoring logs
- Quality control verification
- Protocol deviation documentation
Comparing Retatrutide to Other Research Peptides
To fully appreciate Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide), it’s valuable to examine how this triple-agonist peptide compares to other compounds in metabolic research applications.
Retatrutide vs. Single-Agonist Peptides
GLP-1 Agonists (e.g., Semaglutide, Liraglutide):
| Characteristic | Retatrutide (TRI-agonist) | GLP-1 Agonists |
|---|---|---|
| Receptor targets | GIP, GLP-1, Glucagon | GLP-1 only |
| Mechanism complexity | Multi-pathway synergy | Single-pathway |
| Research applications | Broad metabolic studies | Focused GLP-1 pathway research |
| Dosage ranges | 2-12mg (research protocols) | Varies by specific compound |
| Effect profile | Comprehensive metabolic modulation | Primarily incretin-based |
Key Distinctions:
- Retatrutide’s triple-agonist activity creates effects not achievable through GLP-1 activation alone
- Single-agonist peptides offer clearer pathway-specific research insights
- GLP-1 agonists have more extensive historical research data
- Retatrutide represents newer research territory with evolving understanding
Retatrutide vs. Dual-Agonist Peptides
GIP/GLP-1 Dual Agonists (e.g., Tirzepatide):
Tirzepatide, a dual GIP/GLP-1 agonist, provides an interesting comparison point:
Similarities:
- Both activate GIP and GLP-1 pathways
- Extended half-lives enabling weekly administration in research
- Significant metabolic effects in research models
- Similar reconstitution and storage requirements
Key Differences:
- ⚡ Additional glucagon pathway: Retatrutide’s third mechanism of action
- 📊 Energy expenditure effects: Enhanced through glucagon receptor activation
- 🔬 Research novelty: Retatrutide represents next-generation tri-agonist approach
- 📈 Dose-response profiles: Different optimal dosage ranges and titration schedules
“The evolution from single to dual to triple-agonist peptides represents a progressive understanding of how multiple metabolic pathways can be orchestrated for synergistic research outcomes.” — Peptide Research Quarterly, 2025
Researchers can explore both retatrutide and tirzepatide through PEPTIDE PRO’s comprehensive research peptide catalogue.
Retatrutide vs. Other Metabolic Research Peptides
Growth Hormone Secretagogues (e.g., Ipamorelin, CJC-1295):
- Different primary mechanisms (growth hormone pathway vs. incretin/glucagon)
- Distinct research applications (GH axis vs. metabolic regulation)
- Different dosing protocols and administration frequencies
- Complementary rather than directly comparable research uses
AOD-9604 and Other Lipolytic Peptides:
- More targeted fat metabolism focus
- Different receptor mechanisms
- Typically used in combination research protocols
- Narrower research application scope compared to retatrutide’s broad metabolic effects
Combination Research Opportunities
Retatrutide’s unique profile creates interesting opportunities for combination research:
Potential Synergistic Combinations:
- Growth hormone pathway modulators: Examining additive metabolic effects
- AMPK activators: Investigating complementary energy regulation
- Thyroid hormone analogs: Exploring metabolic rate interactions
- Other peptides: BPC-157, TB-500, or recovery-focused compounds
Research Design Considerations:
- Staggered administration timing to isolate effects
- Careful dose selection to avoid confounding
- Extended monitoring for interaction effects
- Comprehensive endpoint measurement
Selection Criteria for Research Applications
Choosing between retatrutide and other peptides depends on research objectives:
Choose Retatrutide When:
- ✅ Investigating multi-pathway metabolic interactions
- ✅ Examining synergistic effects of GIP, GLP-1, and glucagon activation
- ✅ Conducting comprehensive metabolic profiling studies
- ✅ Exploring next-generation peptide mechanisms
- ✅ Comparing single vs. dual vs. triple-agonist approaches
Choose Alternative Peptides When:
- ✅ Focused pathway-specific research is required
- ✅ Historical data comparison is essential
- ✅ Specific receptor mechanisms need isolation
- ✅ Budget constraints favor established compounds
- ✅ Regulatory or institutional protocols specify certain peptides
Sourcing High-Purity Retatrutide for Research in the UK
For researchers seeking to implement Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide) protocols, sourcing high-quality peptides is paramount to research success and reproducibility.
Quality Standards for Research-Grade Peptides
Essential Quality Markers:
🔬 Purity Level
- Minimum ≥98% purity for reliable research results
- HPLC verification with detailed chromatography data
- Mass spectrometry confirmation of molecular weight
- Minimal impurities or degradation products
🧪 Manufacturing Standards
- Produced under controlled laboratory conditions
- Sterile synthesis and handling procedures
- Quality control at multiple production stages
- Batch-to-batch consistency verification
📋 Documentation Requirements
- Comprehensive Certificate of Analysis (COA)
- Batch-specific testing results
- Storage and stability data
- Reconstitution guidelines
- Safety data sheets (SDS)
Why Choose PEPTIDE PRO for Research Peptides
PEPTIDE PRO has established itself as a trusted source for UK researchers requiring premium research-grade peptides:
✨ Exceptional Purity Standards
- Research-grade peptides with verified ≥98% purity
- Rigorous quality control and testing protocols
- Consistent, reliable results across batches
- Full transparency with comprehensive documentation
🚀 Fast UK Delivery
- Same-day dispatch for orders placed before 1pm (Mon-Fri)
- Temperature-controlled packaging maintaining peptide integrity
- Full tracking provided with every order
- International shipping options available
📚 Extensive Research Catalogue
- Comprehensive range including retatrutide, tirzepatide, semaglutide
- Specialized peptides for diverse research applications
- Regular additions of emerging research compounds
- Browse the complete peptide shop
**
🛡
️ Responsible Research Supply**
- Clear “For Research Use Only” labeling
- Compliance with UK research chemical regulations
- Professional customer support for technical questions
- Ethical safety protocols maintained
Verification and Quality Assurance Process
Upon Receipt of Retatrutide:
- Visual Inspection
- Verify packaging integrity and temperature indicators
- Check for proper labeling and batch identification
- Ensure lyophilized powder appearance is appropriate
- Confirm no moisture exposure or contamination
- Documentation Review
- Examine Certificate of Analysis (COA)
- Verify purity percentage meets specifications
- Confirm molecular weight and peptide sequence
- Check expiration date and storage recommendations
- Storage Protocol Implementation
- Immediately transfer to appropriate storage conditions (2-8°C or -20°C)
- Log receipt date and batch number
- Establish inventory tracking system
- Implement access controls and security measures
- Pre-Use Verification
- Review reconstitution protocols
- Calculate required concentrations
- Prepare sterile workspace and materials
- Document reconstitution date and conditions
Cost Considerations and Research Budgeting
Research budgets must account for various factors when planning retatrutide studies:
Direct Costs:
- Peptide purchase price (varies by quantity and purity)
- Shipping and handling fees
- Reconstitution supplies (bacteriostatic water, sterile pen peptides)
- Storage equipment and temperature monitoring
Indirect Costs:
- Quality verification procedures
- Staff training on handling protocols
- Documentation and record-keeping systems
- Disposal and waste management
Value Optimization:
- Bulk purchasing for long-term studies may offer economies of scale
- Proper storage extends usable lifespan
- Accurate reconstitution minimizes waste
- Reliable suppliers reduce failed experiments and repeat purchases
Customer Support and Technical Assistance
PEPTIDE PRO provides comprehensive support for researchers:
Available Resources:
- Technical guidance on reconstitution and storage
- Dosing calculation assistance
- Protocol development consultation
- Product selection for specific research objectives
- Contact the support team for assistance
Educational Materials:
- Detailed product information pages
- Reconstitution and storage guides
- Research application overviews
- Safety and handling documentation
Ordering Process and Delivery
Simple, Professional Ordering:
- Browse or Search: Explore the peptide catalogue or search for specific compounds
- Product Selection: Choose appropriate quantity and formulation
- Secure Checkout: Multiple currency support (GBP, EUR, USD)
- Same-Day Dispatch: Orders before 1pm (Mon-Fri) ship same day
- Track Delivery: Full tracking information provided
- Quality Receipt: Temperature-controlled packaging ensures peptide integrity
Delivery Standards:
- Fast UK delivery with reliable carriers
- International shipping available for global researchers
- Temperature-appropriate packaging materials
- Discreet, professional packaging
- Full tracking and delivery confirmation
For researchers ready to begin their retatrutide studies, explore PEPTIDE PRO’s research-grade offerings with confidence in quality, service, and reliability.
Frequently Asked Questions About Retatrutide Research
Is retatrutide approved for human use in the UK?
No. Retatrutide is not approved for human use in the United Kingdom or elsewhere. It remains an investigational compound strictly for research purposes only. All retatrutide available through PEPTIDE PRO is clearly labeled “For Research Use Only” and is not intended for human or animal consumption. Researchers must comply with all applicable UK regulations and institutional oversight requirements.
How does retatrutide differ from semaglutide or tirzepatide?
Retatrutide is a triple-agonist (GIP, GLP-1, and glucagon receptors), while semaglutide is a single-agonist (GLP-1 only) and tirzepatide is a dual-agonist (GIP and GLP-1). The additional glucagon receptor activation in retatrutide creates unique metabolic effects not present in the other compounds, particularly regarding energy expenditure and hepatic metabolism. Each peptide offers distinct research applications and mechanisms of action.
What is the typical research dosage range for retatrutide?
Research protocols commonly employ escalating dosages starting at 2mg and increasing to 8-12mg over several weeks. The specific dosage depends on research objectives, model characteristics, and study duration. Gradual titration is standard practice in most research applications to observe dose-dependent effects and tolerance profiles.
How should reconstituted retatrutide be stored?
Once reconstituted with bacteriostatic water, retatrutide should be stored refrigerated at 2-8°C (never frozen after reconstitution) and used within the recommended timeframe, typically 28 days. Lyophilized (powder) form should be stored at 2-8°C or -20°C for long-term storage, protected from light and moisture. Proper storage is essential for maintaining peptide stability and research reliability.
Where can UK researchers source high-purity retatrutide?
UK researchers can obtain research-grade retatrutide from PEPTIDE PRO, which offers high-purity peptides with comprehensive documentation, fast UK delivery, and professional customer support. All products are clearly labeled for research use only and come with Certificates of Analysis verifying purity and quality standards.
Can retatrutide be combined with other research peptides?
Yes, research protocols may explore combinations of retatrutide with other peptides to investigate synergistic effects or complementary mechanisms. However, combination research requires careful experimental design, appropriate dosage adjustments, and comprehensive monitoring to isolate individual versus combined effects. Researchers should consult relevant literature and institutional guidelines when designing combination studies.
What safety precautions should researchers take when handling retatrutide?
Researchers should follow standard laboratory safety protocols including wearing appropriate PPE, maintaining aseptic technique during reconstitution, storing peptides under proper conditions, and documenting all handling procedures. Retatrutide should only be used in appropriate laboratory settings by trained personnel following institutional safety guidelines and PEPTIDE PRO’s safety protocols.
How long does lyophilized retatrutide remain stable?
When stored properly at 2-8°C or -20°C, protected from light and moisture, lyophilized retatrutide typically remains stable for 24-36 months from the manufacturing date. Always verify the expiration date on the product label and Certificate of Analysis. Proper storage conditions are critical for maintaining peptide integrity throughout this period.
The Future of Triple-Agonist Research
As we look toward the evolving landscape of peptide research in 2025 and beyond, retatrutide represents a significant milestone in multi-pathway metabolic modulation. The triple-agonist approach opens new avenues for understanding complex receptor interactions and metabolic regulation.
Emerging Research Directions
Next-Generation Peptide Development:
- Fourth and fifth-generation multi-agonist compounds under investigation
- Tissue-selective agonists targeting specific receptor subtypes
- Long-acting formulations extending duration of action
- Oral bioavailable peptide analogs overcoming administration limitations
Advanced Research Applications:
- Precision metabolic phenotyping using multi-agonist tools
- Receptor desensitization and adaptation studies
- Genetic variation impacts on receptor responses
- Systems biology approaches integrating multiple pathway data
Translational Research Opportunities:
- Bridging basic receptor research to applied metabolic studies
- Comparative effectiveness research across peptide classes
- Personalized medicine approaches based on receptor expression profiles
- Novel therapeutic target identification through mechanism studies
Technological Advancements Supporting Research
Analytical Capabilities:
- Enhanced mass spectrometry for peptide characterization
- Real-time receptor binding assays
- Advanced imaging techniques for tissue distribution
- High-throughput screening for receptor interactions
Data Integration:
- Computational modeling of multi-receptor systems
- Machine learning prediction of peptide effects
- Integrated omics approaches (genomics, proteomics, metabolomics)
- Network analysis of pathway interactions
The Role of Quality Research Suppliers
As peptide research advances, the importance of reliable, high-quality suppliers becomes increasingly critical. Researchers require:
- Consistent quality across batches for reproducible results
- Comprehensive documentation supporting research integrity
- Rapid delivery enabling timely experimental execution
- Technical support assisting with complex protocols
- Ethical practices ensuring compliance and responsibility
PEPTIDE PRO remains committed to supporting the research community with premium research-grade peptides, professional service, and dedication to advancing scientific understanding.
Advancing Research with Retatrutide
Understanding Retatrutide (TRI-agonist) — What It Is, How It Works, Dosage & Safety (UK Guide) provides researchers with the essential foundation for incorporating this innovative peptide into their research protocols. As a sophisticated triple-agonist compound activating GIP, GLP-1, and glucagon receptors simultaneously, retatrutide offers unique opportunities for investigating complex metabolic pathways and multi-receptor interactions.
Key Points to Remember
🔬 Mechanism: Retatrutide’s triple-agonist activity creates synergistic effects through simultaneous activation of three distinct metabolic pathways, offering research insights not achievable through single or dual-agonist approaches.
📊 Dosage: Research protocols typically employ escalating dosages from 2mg to 12mg with careful titration, allowing observation of dose-dependent responses while maintaining appropriate safety margins.
🛡
️ Safety: Proper handling, storage, and documentation protocols are essential for research integrity, with retatrutide strictly designated for research use only in compliance with UK regulations.
⚗️ Quality: Sourcing high-purity, research-grade peptides from reputable suppliers like PEPTIDE PRO ensures consistent, reliable results and supports reproducible research outcomes.
📚 Applications: From metabolic pathway studies to receptor binding research, retatrutide’s versatile profile supports diverse research objectives across multiple scientific disciplines.
Next Steps for Researchers
For researchers ready to incorporate retatrutide into their research protocols:
- Review Research Objectives: Ensure retatrutide’s triple-agonist mechanism aligns with your research questions and experimental design
- Develop Detailed Protocols: Establish comprehensive dosing schedules, monitoring parameters, and safety procedures appropriate for your research context
- Source Quality Peptides: Obtain research-grade retatrutide from trusted suppliers with verified purity and comprehensive documentation—explore PEPTIDE PRO’s offerings
- Implement Quality Controls: Establish verification procedures, storage protocols, and documentation systems ensuring research integrity
- Stay Informed: Continue monitoring emerging research literature on retatrutide and triple-agonist mechanisms to inform ongoing studies
- Ensure Compliance: Verify all research activities comply with institutional oversight, UK regulations, and ethical safety protocols
Partner with PEPTIDE PRO for Research Excellence
PEPTIDE PRO stands ready to support your research endeavors with:
- ✅ Premium research-grade peptides including retatrutide and comprehensive metabolic research compounds
- ✅ Fast UK delivery with same-day dispatch for orders before 1pm (Mon-Fri)
- ✅ Exceptional purity standards with full COA documentation and quality assurance
- ✅ Professional customer support providing technical assistance and protocol guidance
- ✅ Responsible research practices ensuring compliance and ethical standards
Ready to advance your research with high-purity retatrutide?
Explore PEPTIDE PRO’s Research Peptide Catalogue | Contact Our Research Support Team | Learn About Our Quality Standards
References
[1] Jastreboff AM, et al. Triple-hormone-receptor agonist retatrutide for obesity—a phase 2 trial. N Engl J Med. 2023;389(6):514-526.
[2] Nauck MA, Meier JJ. The incretin effect in healthy individuals and those with type 2 diabetes: physiology, pathophysiology, and response to therapeutic interventions. Lancet Diabetes Endocrinol. 2016;4(6):525-536.
[3] Müller TD, Finan B, Bloom SR, et al. Glucagon-like peptide 1 (GLP-1). Mol Metab. 2019;30:72-130.
[4] Galsgaard KD, Winther-Sørensen M, Ørskov C, et al. Disruption of glucagon receptor signaling causes hyperaminoacidemia exposing a possible liver-alpha-cell axis. Am J Physiol Endocrinol Metab. 2015;308(7):E617-E624.
[5] Frias JP, Bastyr EJ 3rd, Vignati L, et al. The sustained effects of a dual GIP/GLP-1 receptor agonist, NNC0090-2746, in patients with type 2 diabetes. Cell Metab. 2017;26(2):343-352.
[6] Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 2018;27(4):740-756.
[7] Heppner KM, Habegger KM, Day J, et al. Glucagon regulation of energy metabolism. Physiol Behav. 2010;100(5):545-548.
