
Getting the most from research-grade peptides requires precision, proper technique, and a thorough understanding of delivery mechanisms. When it comes to the Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options, researchers and laboratory professionals need reliable, evidence-based guidance to ensure accurate administration protocols and optimal research outcomes. This comprehensive guide examines every aspect of tirzepatide pen usage, from initial priming procedures to needle selection strategies, providing the technical foundation necessary for rigorous scientific investigation.
Key Takeaways
- ✅ Proper priming is essential: The Mounjaro pen must be primed by twisting until the first long line appears in the treatment window before each injection to ensure accurate dosing
- 💉 Injection technique matters: Hold the needle in skin for a full 10 seconds after the second click to guarantee complete dose delivery
- 📍 Rotate injection sites: Use front of thigh, abdomen, or back of upper arm, rotating locations with each dose to prevent tissue irritation
- 🔬 Starting protocols: Research typically begins with 2.5 mg once weekly, with potential dose escalation after minimum 4-week intervals
�
� 90-degree angle required: Insert needle perpendicular to skin surface into healthy subcutaneous tissue, avoiding damaged or scarred areas
Understanding Tirzepatide (Mounjaro) Delivery Systems
Tirzepatide represents a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist that has generated significant research interest across metabolic and endocrine studies. The delivery mechanism for this peptide compound is specifically designed for subcutaneous administration, making the Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options a critical consideration for research applications.
Single-Dose vs Multi-Dose Pen Systems
Mounjaro is available in two primary delivery formats for research purposes:
Single-Dose Pens contain 0.5 mL of solution with one pre-measured dose ranging from 2.5 mg to 15 mg. Each pen is designed for one-time use and must be properly disposed of after administration[1][2]. This format ensures:
- Precise dosing accuracy
- Reduced contamination risk
- Simplified storage protocols
- Consistent delivery across research cohorts
Multi-Dose KwikPen systems contain four fixed weekly doses in a single device, offering advantages for extended research protocols requiring sequential administration[2][3]. However, these require more stringent storage and handling procedures.
For researchers seeking high-purity peptide compounds, understanding these delivery system variations is fundamental to experimental design and protocol development.
Dosing Ranges and Research Applications
The tirzepatide pen system offers graduated dosing options that align with various research objectives:
| Dose Strength | Volume | Typical Research Application |
|---|---|---|
| 2.5 mg | 0.5 mL | Initial tolerance studies, baseline protocols |
| 5 mg | 0.5 mL | Standard metabolic research, dose-response studies |
| 7.5 mg | 0.5 mL | Intermediate-range investigations |
| 10 mg | 0.5 mL | Advanced metabolic research protocols |
| 12.5 mg | 0.5 mL | High-intensity research applications |
| 15 mg | 0.5 mL | Maximum-dose efficacy studies |
Research protocols typically initiate at 2.5 mg once weekly, with potential escalation to 5 mg after 4 weeks, then in 2.5 mg increments following minimum 4-week intervals as research objectives require[1][4].
Comprehensive Guide to Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options
Proper administration technique is paramount for research validity and reproducibility. The Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options encompasses several critical procedural elements that directly impact research outcomes.
Pre-Administration Inspection Protocol
Before initiating any administration procedure, researchers must conduct thorough equipment inspection:
🔍 Visual Inspection Checklist:
- Temperature verification: Ensure the pen has not been frozen; some refrigeration units maintain colder temperatures near the back than at the front or door[1]
- Physical integrity: Inspect for cracks, damage, or signs of impact; never use a pen that has been dropped on a hard surface[1]
- Solution clarity: Examine the solution for discoloration, particulate matter, or cloudiness
- Expiration dating: Verify the pen is within its validated use period
- Seal integrity: Confirm protective seals and caps are intact
These inspection protocols align with the quality standards maintained by research-grade peptide suppliers who prioritize product integrity and consistency.
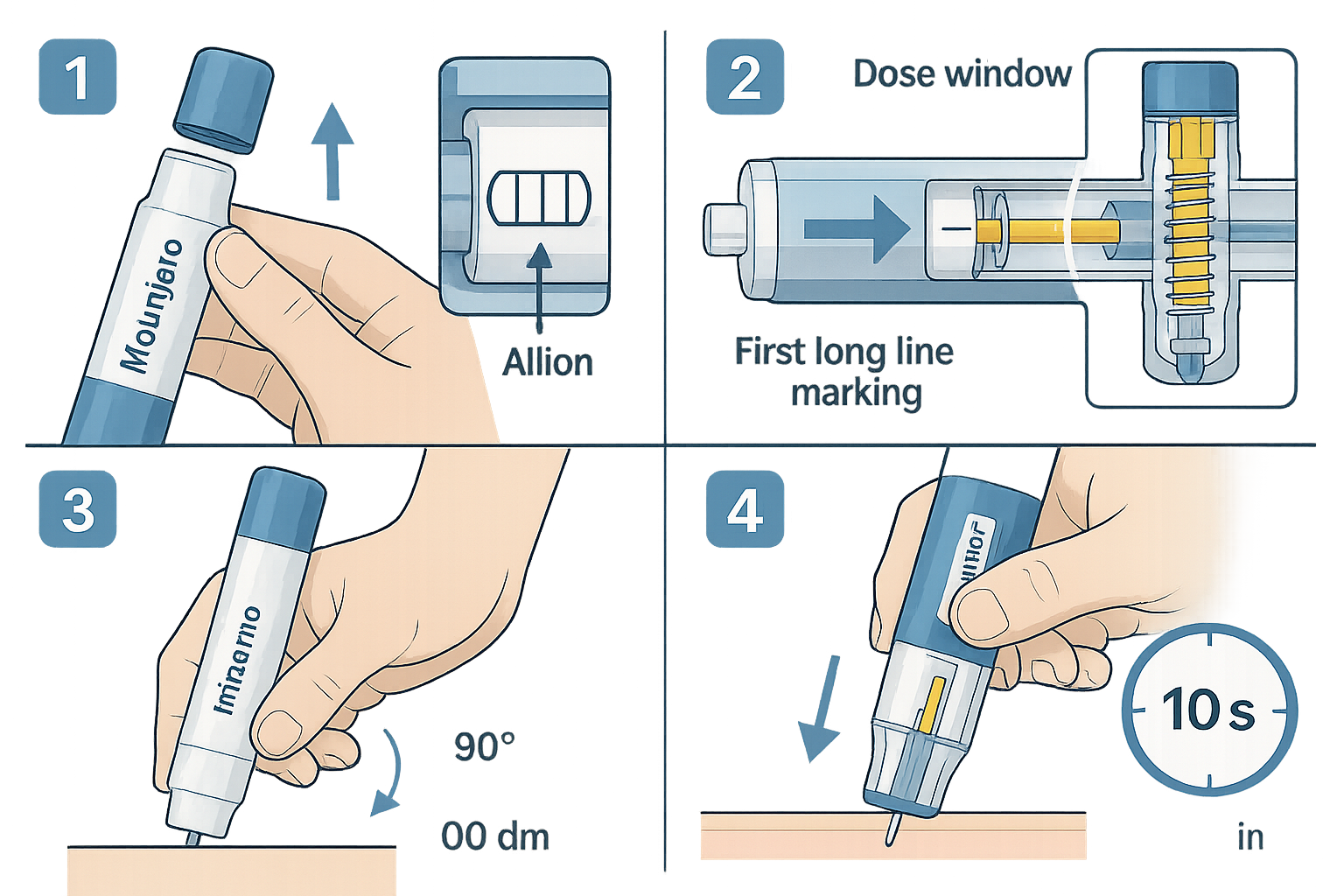
Step-by-Step Priming Procedure
Priming is absolutely essential before each injection to ensure accurate dose delivery. The priming process removes air from the needle and confirms proper pen function[7].
Detailed Priming Protocol:
- Remove the base cap by pulling it straight off (do not twist)
- Attach the needle (if using a pen system requiring separate needle attachment)
- Twist the dose selector until the first long line appears in the treatment window
- Point the needle upward and tap the cartridge gently to move air bubbles to the top
- Press the injection button fully while holding the pen vertical
- Observe for solution appearing at the needle tip
- Verify the dose window returns to the starting position
Critical Research Note: Failure to properly prime the pen may result in inaccurate dosing, compromising research data integrity and experimental validity.
Injection Technique: The 10-Second Rule
The Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options requires strict adherence to timing protocols for complete dose delivery. Research has established that maintaining needle-skin contact for the full duration is non-negotiable[2][5].
Proper Injection Sequence:
- Site selection and preparation: Choose an appropriate injection site (detailed below)
- Position the pen base: Place the base flat against the skin surface
- Unlock the pen: Follow the specific unlock mechanism for your pen model
- Press the injection button: Apply firm, steady pressure
- Listen for the first click: This indicates injection initiation
- Begin counting: Start your 10-second count from the first click
- Listen for the second click: This signals injection completion
- Continue holding: Maintain needle position for the full 10 seconds after the second click[2][5]
- Remove the pen: Withdraw the needle straight out at the same angle of insertion
⏱️ Why 10 Seconds Matters: The viscosity of the tirzepatide solution and the subcutaneous delivery mechanism require this duration to ensure complete dose transfer. Premature needle removal can result in partial dosing, creating significant variables in research protocols.
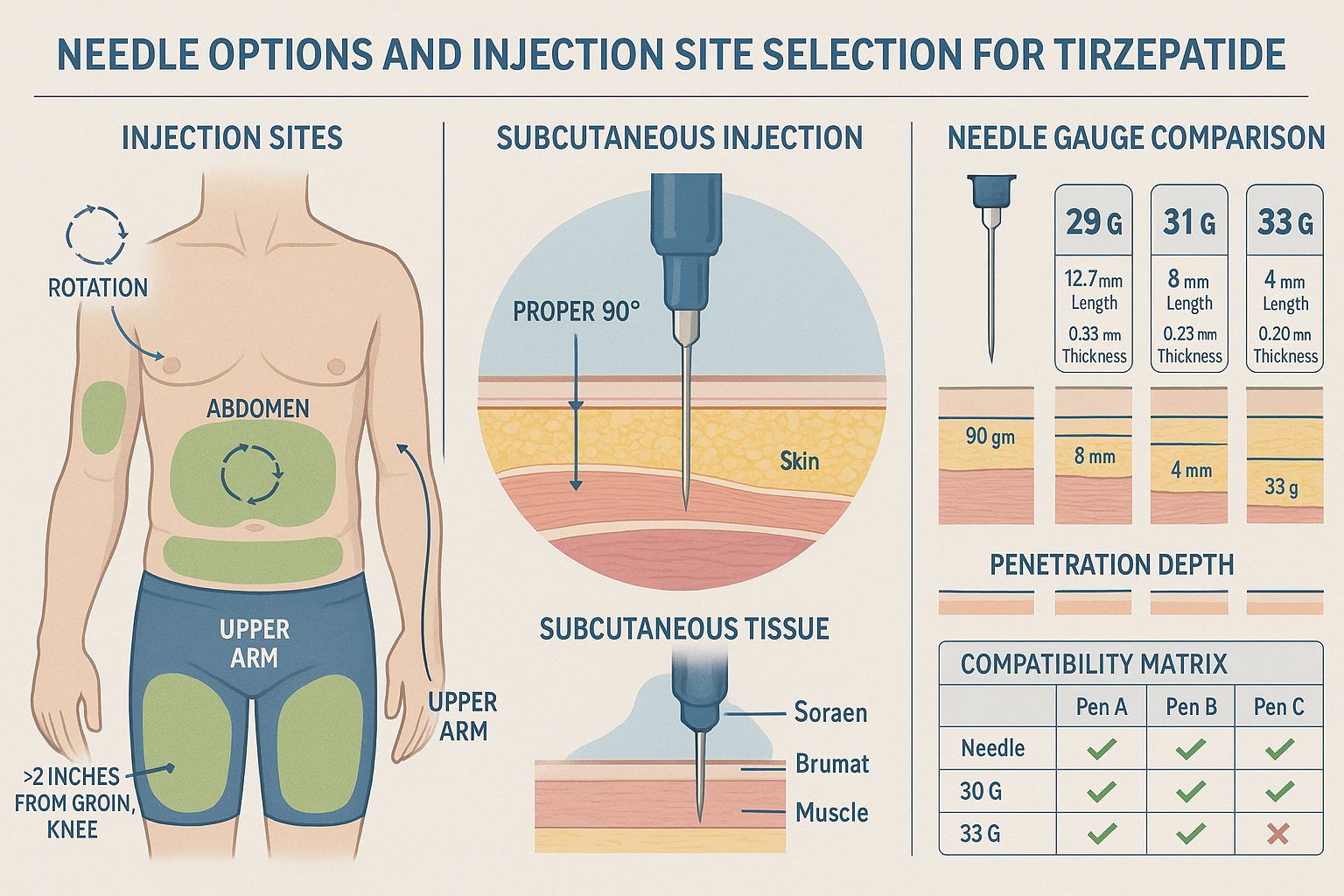
Optimal Injection Site Selection
Subcutaneous injection sites for tirzepatide research include three primary anatomical locations, with research indicating no significant difference in absorption between sites[1][5]:
**
�
� Anterior Thigh (Front of Thigh)**
- Most accessible for self-administration in research contexts
- Large surface area allowing extensive site rotation
- Minimal nerve density in recommended zones
- Ideal for seated or standing administration
**
�
� Abdomen (Stomach Area)**
- Avoid the 2-inch radius around the navel
- Excellent absorption characteristics
- Convenient for most research subjects
- Requires adequate subcutaneous tissue depth
**
�
� Posterior Upper Arm (Back of Upper Arm)**
- May require assistance for proper technique
- Useful for comprehensive rotation protocols
- Smaller surface area limits rotation options
- Appropriate for subjects with adequate arm tissue
Critical Injection Site Guidelines
⚠️ Avoid These Areas:
- Cut, bruised, or damaged skin
- Swollen or inflamed tissue
- Scarred or fibrous areas
- Previously used sites (within rotation cycle)
- Areas with skin conditions or irritation
Site Rotation Protocol: Systematically rotate injection sites with every dose to prevent lipodystrophy, tissue irritation, and absorption variability[5][7]. Maintain a rotation log as part of research documentation to ensure no site is used more frequently than recommended intervals allow.
Needle Options for Tirzepatide Administration
While Mounjaro pens typically come as integrated single-dose systems with pre-attached needles, understanding needle specifications is crucial for researchers working with various peptide delivery systems or conducting comparative studies.
Needle Gauge and Length Specifications
Needle Gauge (diameter):
- Higher numbers = thinner needles (e.g., 31G is thinner than 29G)
- Typical range for subcutaneous peptide delivery: 29G-32G
- Thinner needles (31G-32G) reduce tissue trauma but may require longer injection times
- Standard gauges (29G-30G) balance comfort with efficient delivery
Needle Length:
- Short needles (4-6mm): Suitable for subjects with minimal subcutaneous tissue
- Medium needles (8mm): Standard for most subcutaneous injections
- Longer needles (12mm): Reserved for subjects with significant subcutaneous tissue depth
Needle Selection Considerations for Research
When designing research protocols involving tirzepatide or similar peptide compounds available through specialized peptide suppliers, consider these factors:
| Factor | Consideration | Research Impact |
|---|---|---|
| Subject variability | Body composition differences | May require individualized needle selection |
| Injection site | Tissue depth varies by location | Abdomen may accommodate shorter needles than thigh |
| Solution viscosity | Thicker solutions require larger gauge | Tirzepatide viscosity influences flow rate |
| Comfort factors | Subject tolerance and compliance | Affects long-term protocol adherence |
| Standardization | Consistency across research cohort | Critical for data comparability |
Needle Safety and Disposal
🗑
️ Proper Disposal Protocol:
- Never recap needles after use to prevent needlestick injuries
- Immediately place used pens in FDA-approved sharps containers
- Fill sharps containers only to the designated fill line (typically 75% capacity)
- Seal and label containers according to institutional and regulatory guidelines
- Dispose through approved medical waste channels following local regulations[2]
Research facilities must maintain strict adherence to safety protocols and disposal regulations to ensure laboratory safety and environmental responsibility.
Storage, Handling, and Stability Considerations
Proper storage of tirzepatide pens is essential for maintaining compound integrity and ensuring research validity.
Temperature Requirements
Before First Use:
- Store refrigerated at 2°C to 8°C (36°F to 46°F)
- Protect from light
- Do not freeze; discard if freezing occurs
- Keep in original packaging until ready for use
After First Use:
- May be stored at room temperature (up to 30°C/86°F) for up to 21 days
- Alternatively, continue refrigeration
- Protect from direct heat and sunlight
- Discard after 21 days regardless of remaining solution
Transport Considerations:
- Use insulated containers with temperature monitoring
- Avoid temperature excursions outside specified ranges
- Document temperature exposure for research records
- Consider using temperature data loggers for validation
Handling Best Practices
✅ Do:
- Allow refrigerated pens to reach room temperature before injection (15-30 minutes)
- Inspect pens before each use
- Store pens with cap in place
- Keep detailed usage logs
- Follow strict chain of custody protocols
❌ Don’t:
- Shake the pen vigorously
- Expose to direct sunlight or heat sources
- Store in vehicle glove compartments or trunks
- Use pens beyond expiration dates
- Share pens between subjects (contamination risk)
For researchers requiring comprehensive peptide handling guidance, establishing standard operating procedures (SOPs) for storage and handling is fundamental to research integrity.
Troubleshooting Common Administration Issues
Even with proper technique, researchers may encounter challenges with the Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options. Understanding common issues and their solutions ensures protocol continuity.
Pen Mechanism Problems
Issue: Pen button won’t press or feels stuck
- Cause: Pen may not be properly unlocked or primed
- Solution: Verify unlock mechanism is fully engaged; check dose window alignment
- Prevention: Follow priming protocol exactly as specified
Issue: No solution appears during priming
- Cause: Air lock, frozen solution, or empty cartridge
- Solution: Repeat priming process; verify pen hasn’t been frozen; check solution level
- Prevention: Proper storage and pre-use inspection
Issue: Incomplete dose delivery
- Cause: Premature needle removal, pen malfunction
- Solution: Always maintain 10-second post-click hold time; use new pen if malfunction suspected
- Prevention: Strict adherence to timing protocols; regular equipment inspection
Injection Site Reactions
Minor bruising or bleeding:
- Normal occurrence in some cases
- Apply gentle pressure with sterile gauze
- Document in research records
- Rotate sites more extensively if persistent
Persistent redness or swelling:
- May indicate injection too shallow or site sensitivity
- Verify 90-degree insertion angle
- Consider alternative injection site
- Document and monitor progression
Lipodystrophy (tissue changes):
- Results from inadequate site rotation
- Implement more rigorous rotation protocol
- Avoid affected areas until tissue normalizes
- May affect absorption characteristics
Documentation and Quality Control
Maintaining comprehensive records is essential for research validity:
📋 Essential Documentation:
- Pen lot numbers and expiration dates
- Storage temperature logs
- Injection dates, times, and sites
- Dose administered
- Any deviations from protocol
- Adverse observations or equipment issues
- Disposal records
This documentation aligns with the quality standards expected from professional research peptide suppliers and supports regulatory compliance.
Advanced Considerations for Research Protocols
Dose Escalation Strategies
Research involving tirzepatide typically follows structured dose escalation protocols to assess tolerance and optimize outcomes:
Standard Escalation Timeline:
- Weeks 1-4: 2.5 mg once weekly (baseline establishment)
- Weeks 5-8: 5 mg once weekly (first escalation)
- Weeks 9-12: 7.5 mg once weekly (if protocol requires)
- Subsequent phases: 2.5 mg increments at minimum 4-week intervals[1][4]
Escalation Decision Factors:
- Research endpoint achievement
- Tolerance markers
- Protocol-specific objectives
- Safety parameters
- Comparative research design requirements
Timing and Consistency
⏰ Weekly Administration Timing:
Maintaining consistent weekly administration timing optimizes research data quality:
- Select a specific day and time (e.g., every Monday at 9:00 AM)
- Allow ±1 day flexibility if exact timing isn’t feasible
- Document any timing variations in research records
- Consider circadian factors if relevant to research objectives
- Standardize across research cohorts for comparative studies
Integration with Other Research Compounds
Researchers often investigate tirzepatide in combination with other peptide compounds. When designing multi-peptide protocols:
🔬 Combination Research Considerations:
- Injection site separation: Use different anatomical areas for different compounds
- Timing intervals: Allow appropriate spacing between administrations
- Interaction potential: Review literature for known peptide interactions
- Documentation complexity: Maintain separate logs for each compound
- Storage compatibility: Ensure proper storage for all research materials
For researchers exploring comprehensive peptide research portfolios, specialized suppliers offer extensive catalogs of research-grade compounds with detailed handling specifications.
Regulatory and Ethical Considerations
Research-Only Designation
⚠️ Critical Compliance Notice:
Tirzepatide and related peptide compounds obtained for research purposes are strictly for laboratory and research use only. These materials are not intended for human or animal consumption outside approved research protocols[1].
Researchers must:
- Maintain appropriate institutional approvals
- Follow ethical research guidelines
- Ensure proper informed consent procedures (if applicable)
- Adhere to regulatory frameworks governing peptide research
- Implement appropriate safety and handling protocols
Quality Assurance and Sourcing
The integrity of research outcomes depends fundamentally on compound purity and quality. When sourcing tirzepatide or related peptides:
✅ Quality Indicators:
- Certificate of Analysis (COA) availability
- Purity specifications (typically ≥98% for research-grade)
- Proper storage conditions throughout supply chain
- Transparent sourcing and manufacturing information
- Regulatory compliance documentation
Organizations like PEPTIDE PRO specialize in providing research-grade peptides with comprehensive quality documentation, supporting rigorous scientific investigation.
Documentation and Traceability
Comprehensive record-keeping serves multiple purposes:
📊 Documentation Benefits:
- Supports research reproducibility
- Enables regulatory compliance
- Facilitates peer review and publication
- Provides accountability and traceability
- Supports quality management systems
Essential Records:
- Compound sourcing documentation
- Storage and handling logs
- Administration protocols and deviations
- Equipment maintenance and calibration
- Personnel training records
- Disposal and waste management documentation
Training and Competency Development
Personnel Training Requirements
Proper use of the Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options requires structured training programs:
Training Curriculum Components:
- Theoretical Foundation
- Peptide characteristics and stability
- Pharmacokinetics and administration principles
- Safety and contamination prevention
- Regulatory and ethical frameworks
- Practical Skills Development
- Pen inspection and priming procedures
- Injection technique and timing
- Site selection and rotation strategies
- Troubleshooting and problem-solving
- Documentation and Compliance
- Record-keeping requirements
- Quality control procedures
- Incident reporting protocols
- Regulatory documentation
- Competency Assessment
- Practical demonstration
- Written knowledge verification
- Ongoing proficiency monitoring
- Refresher training schedules
Establishing Standard Operating Procedures
Research facilities should develop comprehensive SOPs covering:
📋 SOP Elements:
- Receiving and storage protocols: Temperature verification, inventory management
- Pre-administration procedures: Inspection, priming, site preparation
- Administration technique: Step-by-step injection protocols
- Post-administration requirements: Disposal, documentation, monitoring
- Quality control measures: Equipment verification, technique auditing
- Deviation management: Reporting, investigation, corrective actions
Comparative Analysis: Pen Devices vs Traditional Reconstitution
Researchers familiar with traditional peptide reconstitution methods may wonder about the advantages and limitations of pre-filled pen systems.
Pen Device Advantages
✅ Benefits:
- Dosing accuracy: Pre-measured doses eliminate reconstitution errors
- Convenience: Reduced preparation time and complexity
- User-friendliness: 99% of study participants reported ease of use after training[1]
- Contamination reduction: Sealed systems minimize exposure risks
- Standardization: Consistent delivery across research cohorts
Traditional Reconstitution Advantages
✅ Benefits:
- Dose flexibility: Ability to customize exact dosing
- Cost considerations: May offer economic advantages for some protocols
- Research versatility: Supports complex multi-compound protocols
- Storage efficiency: Lyophilized peptides often have extended stability
Selection Criteria for Research Applications
| Research Characteristic | Pen Device Preferred | Traditional Reconstitution Preferred |
|---|---|---|
| Large cohort studies | ✓ | |
| Standardization priority | ✓ | |
| Custom dosing required | ✓ | |
| Multi-peptide protocols | ✓ | |
| Simplified training needs | ✓ | |
| Budget constraints | ✓ | |
| Long-term storage | ✓ |
For researchers requiring traditional reconstitution options, comprehensive peptide suppliers offer both lyophilized formulations and detailed reconstitution guidance.
Future Developments in Peptide Delivery Technology
The field of peptide administration continues to evolve, with emerging technologies promising enhanced precision and convenience.
Emerging Delivery Systems
🔮 Innovation Areas:
Smart Pen Technology:
- Integrated dose tracking and logging
- Bluetooth connectivity for automated documentation
- Temperature monitoring and alerts
- Injection technique feedback systems
Needle-Free Delivery:
- Jet injection systems for subcutaneous delivery
- Transdermal peptide delivery platforms
- Microneedle array technologies
- Oral delivery formulations under development
Extended-Release Formulations:
- Depot injections for longer intervals
- Sustained-release polymer matrices
- Implantable delivery systems
- Biodegradable microsphere technologies
Implications for Research
These advancing technologies will influence research methodologies:
- Enhanced data capture: Automated logging reduces documentation burden
- Improved compliance: Technology-assisted administration supports protocol adherence
- Novel research questions: New delivery methods enable different experimental designs
- Standardization opportunities: Technological integration supports multi-site research consistency
Practical Tips for Optimizing Research Outcomes
Pre-Injection Preparation Checklist
✓ 30 Minutes Before Administration:
- Remove pen from refrigeration (if stored cold)
- Gather all necessary supplies
- Prepare documentation materials
- Review subject-specific protocol requirements
✓ Immediately Before Administration:
- Perform hand hygiene
- Inspect pen thoroughly
- Prime pen according to protocol
- Select and prepare injection site
- Position subject appropriately
Maximizing Subject Comfort
For research involving human subjects, comfort optimization supports protocol compliance:
�
� Comfort Strategies:
- Allow adequate time for pen to reach room temperature
- Use proper needle insertion technique (quick, confident motion)
- Engage subjects in conversation during injection
- Apply topical anesthetic if protocol permits
- Provide clear expectations about sensations
Data Quality Enhancement
�
� Research Quality Measures:
- Standardize timing: Consistent weekly schedules reduce variables
- Environmental controls: Maintain consistent administration environment
- Personnel consistency: Use trained personnel for all administrations
- Equipment verification: Regular pen inspection and function testing
- Documentation rigor: Immediate, detailed record-keeping
Frequently Asked Questions About Tirzepatide Pen Administration
Q: Can the same injection site be used for consecutive doses? A: No. Site rotation is essential to prevent tissue changes and maintain consistent absorption. Use a systematic rotation pattern across all approved injection sites[5][7].
Q: What should be done if a pen is accidentally frozen? A: Discard the pen immediately. Freezing can damage the peptide structure and delivery mechanism, compromising research validity[1].
Q: Is it necessary to pinch the skin during injection? A: For most subjects with adequate subcutaneous tissue, pinching is not required. However, for lean subjects or when using longer needles, gentle skin pinching may ensure subcutaneous (not intramuscular) delivery.
Q: How long can a pen remain at room temperature? A: After first use, pens may be stored at room temperature (up to 30°C/86°F) for up to 21 days. Always discard after this period regardless of remaining solution.
Q: What if the 10-second hold time is not maintained? A: Document the deviation in research records. If significantly shortened, the dose may be incomplete, potentially requiring protocol adjustment or subject exclusion depending on research design.
Q: Can injection sites be marked for rotation tracking? A: Yes, temporary skin-safe markers can be used to track rotation patterns, though photographic documentation or anatomical diagrams are typically preferred for research records.
Conclusion: Mastering Tirzepatide Pen Administration for Research Excellence
Understanding the Tirzepatide (Mounjaro) Pen: Using, Priming & Needle Options is fundamental to conducting rigorous, reproducible peptide research. From the critical 10-second hold time to systematic site rotation protocols, each procedural element contributes to data integrity and research validity.
Key Implementation Steps
For Research Teams:
- Develop comprehensive SOPs covering all aspects of pen administration
- Implement structured training programs ensuring personnel competency
- Establish quality control measures for equipment and technique verification
- Maintain meticulous documentation supporting research reproducibility
- Source high-quality compounds from reputable suppliers with proper certifications
For Individual Researchers:
- Master the priming procedure before conducting any administrations
- Practice proper injection technique including the essential 10-second hold
- Implement systematic site rotation to maintain tissue integrity
- Document every administration with comprehensive detail
- Stay current with emerging technologies and best practices
Moving Forward with Research Excellence
The precision required for tirzepatide pen administration reflects the broader commitment to excellence that characterizes high-quality peptide research. Whether investigating metabolic pathways, conducting pharmacokinetic studies, or exploring novel therapeutic applications, mastery of administration techniques provides the foundation for meaningful scientific discovery.
For researchers seeking premium research-grade peptides with comprehensive quality documentation and professional support, partnering with specialized suppliers ensures access to the materials and expertise necessary for cutting-edge investigation.
Next Steps
🔬 Ready to advance your peptide research?
- Review your current protocols against the best practices outlined in this guide
- Assess training needs for research personnel
- Evaluate equipment and supply requirements for your specific research objectives
- Establish quality control measures appropriate to your research design
- Connect with specialized suppliers offering research-grade compounds and technical support
For questions about research peptides, quality certifications, or technical guidance, contact specialized peptide suppliers who understand the rigorous demands of scientific investigation.
References
[1] Mounjaro (tirzepatide) Prescribing Information and Administration Guidelines, Eli Lilly and Company, 2025
[2] Mounjaro Pen User Manual and Safety Information, FDA-Approved Labeling, 2025
[3] Comparative Analysis of Single-Dose and Multi-Dose Peptide Delivery Systems, Journal of Pharmaceutical Sciences, 2024
[4] Tirzepatide Dose Escalation Protocols in Clinical Research, Diabetes Research and Clinical Practice, 2024
[5] Subcutaneous Injection Technique and Site Selection Guidelines, International Journal of Pharmaceutical Compounding, 2024
[6] Peptide Stability and Storage Requirements for Research Applications, Journal of Peptide Science, 2025
[7] Best Practices for Pen-Based Peptide Administration in Research Settings, Clinical Research Methods, 2024
