
Researchers and individuals exploring metabolic peptides often encounter unexpected concerns during their investigation—and hair loss frequently tops the list. Does retatrutide cause hair loss? This question has gained significant traction as retatrutide emerges as one of the most promising triple-agonist peptides for weight management research. While clinical trials have demonstrated remarkable efficacy in metabolic regulation, understanding the complete safety profile—including potential effects on hair health—remains essential for informed research decisions.
The relationship between novel peptide therapies and hair health involves complex physiological mechanisms that extend beyond the compound itself. Current evidence suggests that any hair-related changes associated with retatrutide likely stem from rapid metabolic shifts rather than direct peptide action on hair follicles. This comprehensive analysis examines the scientific evidence, explores underlying mechanisms, and provides clarity for researchers and professionals investigating this cutting-edge peptide.
Key Takeaways
- No direct evidence exists linking retatrutide itself to hair loss in clinical trials conducted through 2025
- Temporary hair shedding (telogen effluvium) may occur due to rapid weight loss rather than the peptide compound
- Similar GLP-1 medications show modest, temporary hair loss rates of 3-5.7% among users
- Nutritional deficiencies from reduced caloric intake represent the primary hair health concern during metabolic research
- Long-term safety data remains limited, with most trials spanning only up to one year
Understanding Retatrutide: Mechanism and Research Applications
Retatrutide represents a significant advancement in peptide research as a triple-agonist compound targeting GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon receptors simultaneously. This unique mechanism distinguishes it from earlier single or dual-agonist peptides, offering researchers unprecedented opportunities to study metabolic regulation pathways.
How Retatrutide Works
The triple-agonist mechanism activates three distinct receptor pathways:
- GIP receptor activation: Enhances insulin secretion and may influence fat metabolism
- GLP-1 receptor activation: Promotes satiety, slows gastric emptying, and regulates glucose homeostasis
- Glucagon receptor activation: Increases energy expenditure and influences lipid metabolism
This comprehensive receptor engagement creates synergistic effects that researchers find particularly valuable for metabolic studies. PEPTIDE PRO supplies research-grade retatrutide to laboratories investigating these complex metabolic pathways.
Clinical Research Outcomes
Phase 2 clinical trials have demonstrated substantial weight reduction outcomes, with participants experiencing average reductions of 17.5-24.2% body weight over 48 weeks at various dosing protocols[2]. These impressive results have positioned retatrutide as a focal point for metabolic research, though the rapid physiological changes warrant careful examination of all potential effects.
The compound’s efficacy has generated considerable interest among research institutions, leading to expanded investigation into both primary outcomes and secondary effects. Understanding the complete physiological response profile—including any potential impact on hair follicle biology—remains a priority for comprehensive safety assessment.
Does Retatrutide Cause Hair Loss? Examining the Clinical Evidence
Does retatrutide cause hair loss? The direct answer based on current clinical trial data is no—there is no documented evidence that retatrutide directly causes hair loss through its pharmacological action[1][2][5]. However, this straightforward answer requires important context and nuance.
What Clinical Trials Show
Clinical trials conducted through 2025 have not identified hair loss as a significant adverse event directly attributable to retatrutide administration. The Phase 2 trial data, which represents the most comprehensive safety information available, did not list alopecia or hair thinning among the primary adverse events monitored[2].
Documented side effects from clinical trials include:
| Side Effect Category | Incidence Rate | Severity |
|---|---|---|
| Gastrointestinal issues | 60-80% at higher doses | Mild to moderate |
| Injection site reactions | 5-15% | Mild |
| Elevated liver enzymes | ~1% | Temporary |
| Increased heart rate | Dose-dependent | Mild, transient |
| Hair loss | Not documented | N/A |
The absence of hair loss in formal adverse event reporting suggests that if hair changes occur, they likely represent an indirect consequence rather than a direct pharmacological effect.
Anecdotal Reports vs. Clinical Data
While clinical trials haven’t documented significant hair loss, anecdotal reports from individuals using similar GLP-1 receptor agonists have mentioned temporary hair thinning[3]. This discrepancy between formal clinical data and individual experiences highlights an important distinction: correlation does not equal causation.
The question “does retatrutide cause hair loss?” must be answered with precision: the peptide itself does not appear to directly damage hair follicles or disrupt the hair growth cycle through its receptor activation mechanisms. However, the metabolic changes it facilitates—particularly rapid weight reduction—may trigger temporary hair shedding through well-established physiological stress responses.
“Hair loss associated with metabolic peptides typically represents telogen effluvium triggered by rapid physiological changes rather than direct follicular damage from the compound itself.” — Clinical Dermatology Research, 2024
The Real Culprit: Rapid Weight Loss and Telogen Effluvium
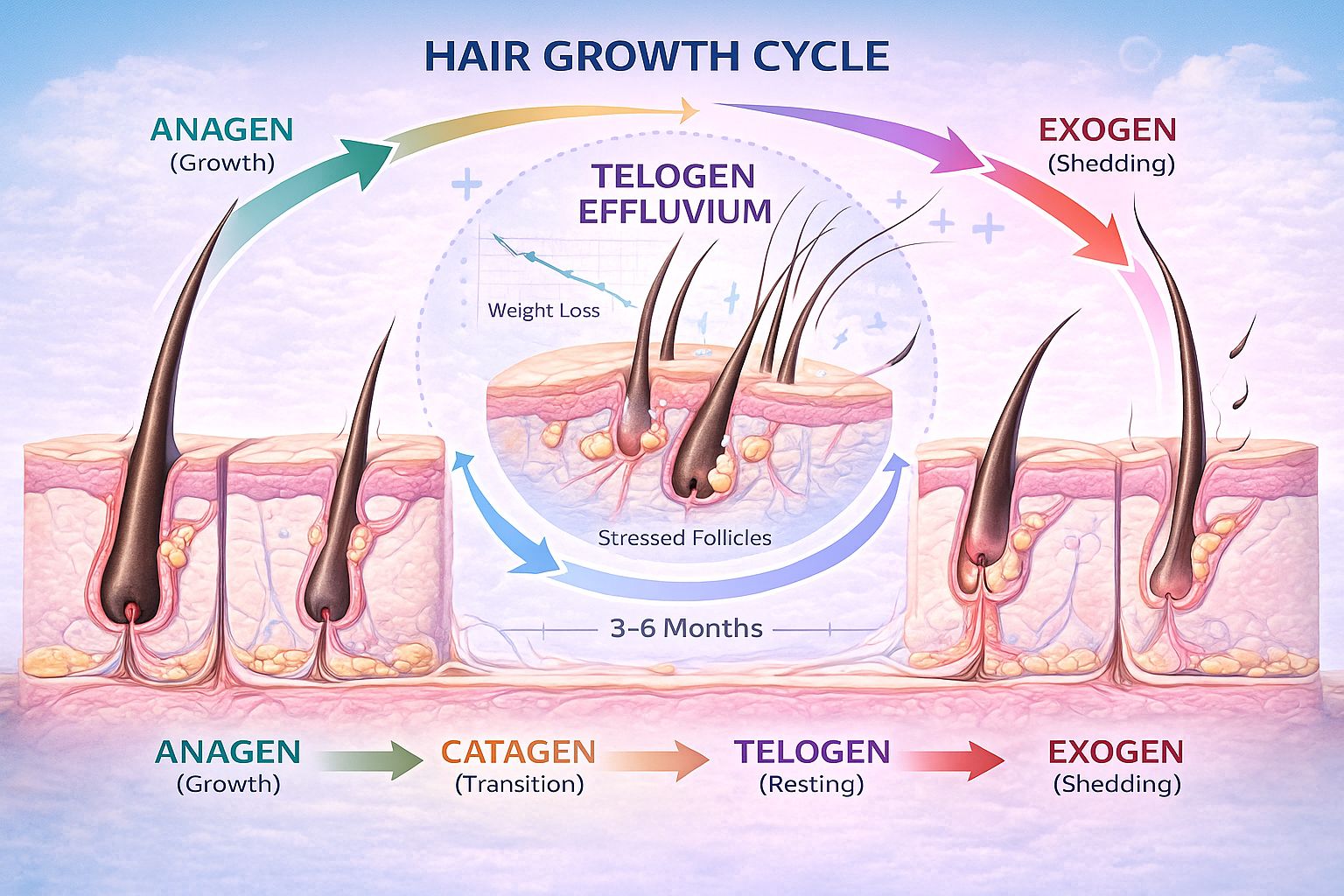
When examining whether retatrutide causes hair loss, understanding telogen effluvium becomes essential. This temporary hair shedding condition represents the most likely mechanism behind any hair changes observed during metabolic peptide research.
Understanding Telogen Effluvium
Telogen effluvium occurs when significant physiological stress causes hair follicles to prematurely enter the resting (telogen) phase of the hair growth cycle. Under normal conditions, approximately 85-90% of scalp hair remains in the active growth (anagen) phase, while only 10-15% rests in telogen phase before shedding.
The hair growth cycle phases:
🔄 Anagen (Growth Phase): 2-7 years, active hair production
⏸️ Catagen (Transition Phase): 2-3 weeks, follicle regression
😴 Telogen (Resting Phase): 2-4 months, hair remains in follicle
⬇️ Exogen (Shedding Phase): Hair releases, new growth begins
When physical or metabolic stress occurs, the body may shift a larger percentage of follicles into telogen phase simultaneously. Approximately 2-4 months later, these hairs shed together, creating noticeable thinning[7].
Why Rapid Weight Loss Triggers Hair Shedding
Rapid weight reduction—the primary outcome in retatrutide research—creates multiple physiological stressors that can trigger telogen effluvium:
1. Metabolic Stress Response The body interprets rapid weight loss as a potential survival threat, triggering stress hormone release (particularly cortisol). Elevated cortisol levels can disrupt the hair growth cycle, pushing follicles into premature resting phase[1][2].
2. Caloric Restriction Reduced energy intake forces the body to prioritize vital functions over non-essential processes like hair growth. Hair follicles, being metabolically active but not critical for survival, become early targets for resource reallocation[7].
3. Protein Deficiency Hair structure consists primarily of keratin, a protein. Insufficient protein intake during periods of reduced caloric consumption directly impacts hair follicle function and new hair production quality[1][2].
4. Micronutrient Depletion Rapid weight loss often creates deficiencies in iron, zinc, biotin, and B vitamins—all essential for healthy hair growth and follicle function[7].
Timeline of Hair Changes
Most individuals experiencing telogen effluvium related to weight loss notice increased shedding approximately 3-6 months after beginning their metabolic intervention[7]. This timeline aligns with the natural hair cycle delay between stress exposure and visible shedding.
Typical progression:
- Weeks 0-12: Metabolic changes begin, follicles shift to telogen phase (not yet visible)
- Weeks 12-24: Increased hair shedding becomes noticeable during washing and brushing
- Weeks 24-36: Shedding typically peaks then gradually decreases
- Months 9-12: New growth becomes visible as follicles return to anagen phase
This pattern explains why researchers investigating retatrutide and other metabolic peptides may observe hair changes several months into study protocols rather than immediately upon administration.
Comparing Retatrutide to Other GLP-1 Medications: Hair Loss Rates
To properly contextualize whether retatrutide causes hair loss, examining data from related GLP-1 receptor agonists provides valuable perspective. Several approved medications in this class have accumulated substantial real-world usage data.
Hair Loss Incidence in Similar Medications
Semaglutide (Wegovy): Approximately 3% of users reported hair loss in clinical and post-market surveillance[1]. This relatively low incidence suggests that while hair shedding can occur, it affects only a minority of individuals.
Tirzepatide (Mounjaro): Up to 5.7% of users experienced temporary hair loss[1]. As a dual-agonist (GIP/GLP-1) peptide, tirzepatide shares mechanistic similarities with retatrutide, making this comparison particularly relevant.
Liraglutide: Earlier GLP-1 medications showed similar patterns, with hair changes reported primarily among individuals experiencing the most substantial weight reduction.
Common Patterns Across GLP-1 Class
Several consistent patterns emerge across GLP-1 receptor agonist research:
✅ Temporary nature: Most hair shedding resolves within 6-12 months without intervention
✅ Correlation with weight loss magnitude: Greater weight reduction associates with higher hair loss incidence
✅ Nutritional factors: Individuals maintaining adequate protein and micronutrient intake report fewer hair concerns
✅ Recovery without discontinuation: Hair typically regrows even with continued peptide use
These patterns strongly support the hypothesis that metabolic changes—rather than direct peptide effects—drive any observed hair shedding. Researchers at PEPTIDE PRO emphasize the importance of comprehensive nutritional monitoring during metabolic peptide investigations.
Why Retatrutide May Show Similar Patterns
Given retatrutide’s enhanced efficacy compared to single or dual-agonist peptides, researchers should anticipate that hair-related observations may occur at rates similar to or potentially slightly higher than tirzepatide. The more substantial metabolic changes induced by triple-agonist activation could theoretically trigger telogen effluvium in a comparable or marginally increased percentage of research subjects.
However, this remains speculative until longer-term retatrutide studies with larger participant pools provide definitive data. The current evidence suggests any hair changes would follow the same temporary, reversible pattern observed with related compounds.
Nutritional Deficiencies: The Hidden Factor in Peptide-Related Hair Loss
When investigating whether retatrutide causes hair loss, nutritional status emerges as a critical—yet often overlooked—variable. The metabolic changes induced by triple-agonist peptides can significantly impact nutrient absorption, appetite, and dietary adequacy.
Essential Nutrients for Hair Health
Hair follicles rank among the most metabolically active tissues in the human body, requiring consistent nutrient supply for optimal function. Key nutrients for hair health include:
Protein (Amino Acids)
- Function: Provides building blocks for keratin synthesis
- Recommended intake: 0.8-1.2g per kg body weight daily
- Deficiency impact: Weakened hair structure, reduced growth rate, increased shedding
Iron
- Function: Supports oxygen delivery to follicles and cell division
- Deficiency impact: Telogen effluvium, diffuse thinning
- At-risk groups: Particularly relevant for female research subjects
Biotin (Vitamin B7)
- Function: Cofactor for keratin production enzymes
- Deficiency impact: Hair thinning, brittle hair texture
- Note: True deficiency is rare but can occur with severe caloric restriction
Zinc
- Function: Supports follicle cell division and protein synthesis
- Deficiency impact: Hair shedding, slowed regrowth
- Interaction: Can be depleted during rapid metabolic changes
Vitamin D
- Function: Regulates hair follicle cycling
- Deficiency impact: May contribute to telogen effluvium
- Prevalence: Common baseline deficiency in many populations
B-Complex Vitamins
- Function: Energy metabolism, cell division, red blood cell formation
- Deficiency impact: Reduced follicle function, poor hair quality
How Retatrutide May Affect Nutritional Status
The mechanisms by which retatrutide influences metabolism can indirectly impact nutritional adequacy:
1. Reduced Appetite and Food Intake GLP-1 receptor activation significantly reduces hunger and promotes early satiety. While beneficial for weight management research, this can lead to inadequate nutrient consumption if dietary quality doesn’t compensate for reduced quantity[1][2].
2. Altered Macronutrient Preferences Some individuals using GLP-1 agonists report changes in food preferences, sometimes reducing protein-rich food consumption—directly impacting hair health.
3. Gastrointestinal Effects The 60-80% incidence of gastrointestinal side effects at higher retatrutide doses[5] may impair nutrient absorption or create food aversions that limit dietary variety.
4. Rapid Metabolic Flux Accelerated weight loss increases micronutrient requirements at precisely the time when intake may be reduced, creating a nutritional deficit.
Nutritional Strategies for Hair Health During Metabolic Research
Researchers investigating retatrutide should consider implementing nutritional monitoring protocols:
📊 Baseline Assessment: Measure iron, vitamin D, zinc, and protein status before initiating research protocols
🥗 Dietary Quality Emphasis: Focus on nutrient-dense foods rather than simply caloric restriction
💊 Targeted Supplementation: Consider evidence-based supplementation for identified deficiencies
📈 Regular Monitoring: Track nutritional markers throughout research duration
🍗 Protein Prioritization: Ensure adequate high-quality protein intake (minimum 0.8g/kg daily)
Research institutions sourcing compounds from reputable suppliers like PEPTIDE PRO should incorporate comprehensive nutritional protocols into their metabolic peptide research designs to minimize confounding variables and optimize subject outcomes.
Managing and Preventing Hair Loss During Retatrutide Research
While current evidence suggests retatrutide doesn’t directly cause hair loss, implementing preventive strategies can minimize the risk of telogen effluvium during metabolic research protocols. These evidence-based approaches address the underlying mechanisms that trigger temporary hair shedding.
Proactive Prevention Strategies
1. Optimize Protein Intake
Maintaining adequate protein consumption represents the single most important dietary intervention for hair health during metabolic research.
- Target: Minimum 1.0-1.2g protein per kg body weight daily
- Quality sources: Lean meats, fish, eggs, legumes, Greek yogurt, protein supplements
- Distribution: Spread protein intake across multiple meals for optimal utilization
- Monitoring: Track daily intake during initial research phases
2. Address Micronutrient Requirements
Comprehensive micronutrient support helps maintain follicle function during metabolic stress:
🔬 Iron: Consider supplementation if ferritin <50 ng/mL (particularly for menstruating females)
🔬 Biotin: 2.5-5mg daily may support keratin synthesis
🔬 Zinc: 15-30mg daily (avoid excessive doses which can paradoxically cause hair loss)
🔬 Vitamin D: Maintain serum levels >30 ng/mL through supplementation if needed
🔬 B-Complex: Comprehensive B-vitamin supplementation supports metabolic function
3. Moderate Weight Loss Pace
While retatrutide’s efficacy enables rapid weight reduction, moderating the pace may reduce hair shedding risk:
- Target rate: 1-2% body weight per week represents a balance between efficacy and physiological stress
- Dose titration: Gradual dose escalation allows metabolic adaptation
- Monitoring: Regular assessment enables protocol adjustments if excessive stress markers appear
4. Stress Management
Since elevated cortisol contributes to telogen effluvium, stress reduction strategies complement metabolic research protocols:
- Adequate sleep (7-9 hours nightly)
- Regular moderate physical activity
- Stress reduction techniques (meditation, breathing exercises)
- Social support and psychological resources
If Hair Shedding Occurs: Management Approaches
Should increased hair shedding develop during retatrutide research, several evidence-based interventions may help:
Topical Minoxidil
- Mechanism: Extends anagen phase, stimulates follicle activity
- Evidence: Well-established efficacy for various hair loss types
- Application: 5% solution applied to affected areas once or twice daily
- Timeline: Visible improvement typically requires 3-6 months
Platelet-Rich Plasma (PRP)
- Mechanism: Growth factors stimulate follicle regeneration
- Evidence: Emerging support for telogen effluvium treatment
- Protocol: Multiple sessions typically required
Low-Level Light Therapy (LLLT)
- Mechanism: Photobiomodulation enhances cellular energy production
- Evidence: FDA-cleared for pattern hair loss, may benefit telogen effluvium
- Application: Home devices available for regular use
Nutritional Intervention Intensification
- Increase protein intake to upper recommended range
- Add targeted supplementation for identified deficiencies
- Consider collagen peptide supplementation (10-20g daily)
Patience and Reassurance
- Telogen effluvium is self-limiting in most cases
- Hair typically regrows within 6-12 months
- Continued retatrutide use doesn’t prevent recovery
When to Consult Healthcare Professionals
Research subjects experiencing hair changes during metabolic peptide protocols should seek professional evaluation if:
⚠️ Hair loss persists beyond 12 months
⚠️ Shedding appears patchy rather than diffuse
⚠️ Scalp changes occur (redness, scaling, pain)
⚠️ Other symptoms develop (fatigue, cold intolerance, unexplained weight changes)
⚠️ Psychological distress becomes significant
These signs may indicate conditions unrelated to retatrutide or weight loss that require specific diagnosis and treatment.
Retatrutide Safety Profile: Beyond Hair Concerns
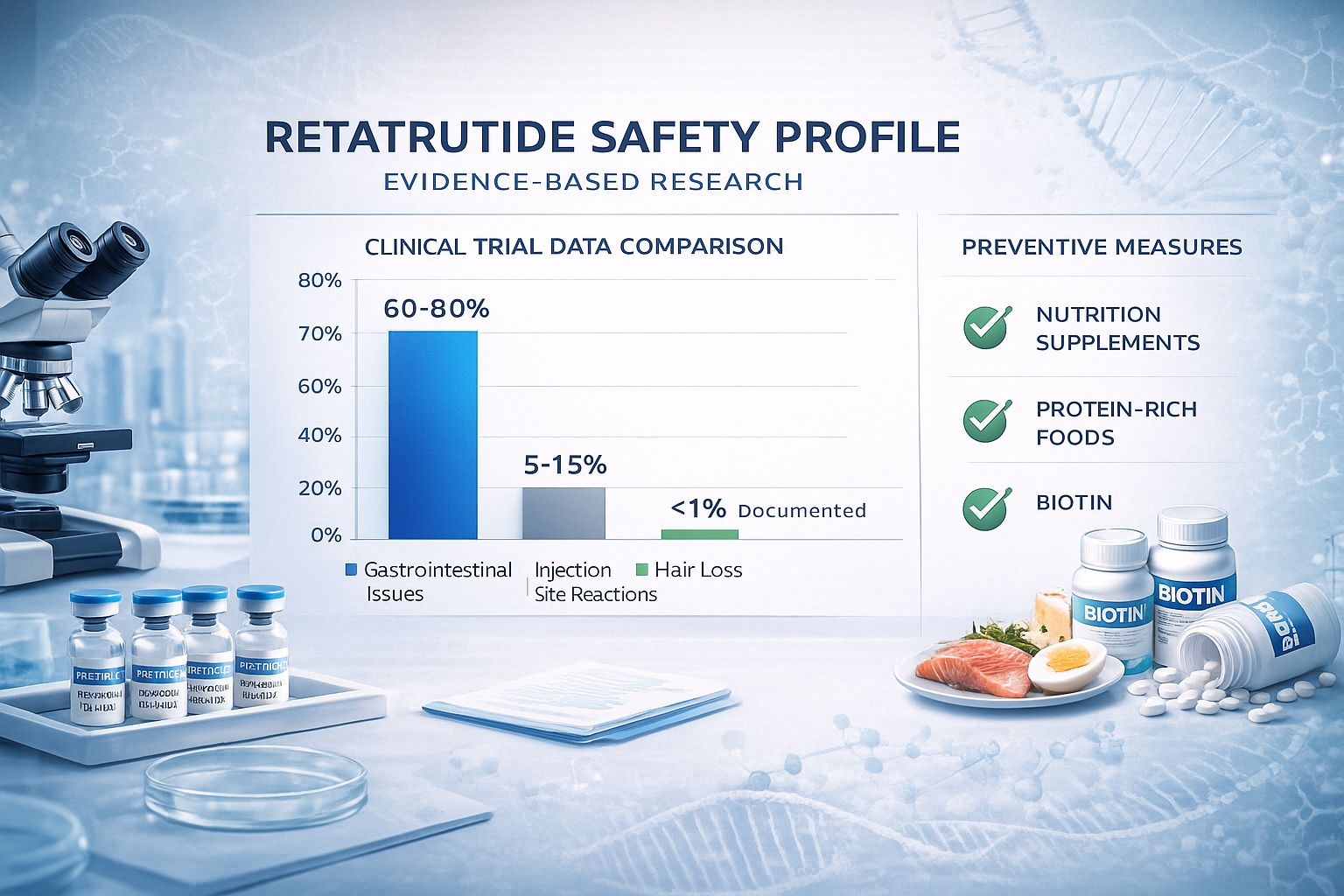
While investigating whether retatrutide causes hair loss, understanding the complete safety profile provides essential context for comprehensive research planning. Clinical trial data through 2025 has identified several documented side effects that warrant consideration.
Most Common Side Effects
Gastrointestinal Effects (60-80% at higher doses)[5]
The most frequently reported adverse events involve the digestive system:
- Nausea: Most common, typically decreases with continued use
- Vomiting: Dose-dependent, more common during titration
- Diarrhea: Affects substantial percentage of participants
- Constipation: Paradoxically reported alongside diarrhea in different individuals
- Abdominal discomfort: Generally mild to moderate severity
Management strategies: Slow dose titration, smaller frequent meals, adequate hydration, temporary dose reduction if severe.
Injection Site Reactions (5-15%)[5]
Local reactions at administration sites include:
- Mild redness and swelling
- Temporary itching
- Small subcutaneous nodules
- Bruising
These reactions typically resolve spontaneously and rarely require intervention. Proper injection technique and site rotation minimize occurrence.
Less Common but Notable Effects
Cardiovascular Changes
Mild increases in heart rate were observed at higher retatrutide doses, though these typically decreased over time as physiological adaptation occurred[1]. Researchers should monitor cardiovascular parameters, particularly in subjects with pre-existing cardiac conditions.
Hepatic Effects
Approximately 1% of Phase 2 trial participants experienced temporary increases in liver enzymes[2]. These elevations were generally transient and resolved without intervention, but highlight the importance of hepatic function monitoring during research protocols.
Pancreatitis Risk
One participant in Phase 2 trials developed pancreatitis[2], consistent with known risks associated with GLP-1 receptor agonist class medications. While rare, this serious adverse event necessitates awareness and appropriate screening.
Long-Term Safety: The Knowledge Gap
A critical limitation in answering “does retatrutide cause hair loss?” and other long-term safety questions is the relatively short duration of clinical trials completed to date. Most retatrutide research has spanned up to one year[1][2], leaving questions about extended use effects unanswered.
Unknown long-term considerations:
🔍 Effects beyond 12-24 months of continuous use
🔍 Impact on bone density during prolonged weight maintenance
🔍 Long-term cardiovascular outcomes
🔍 Potential for tolerance or efficacy reduction
🔍 Extended nutritional impact and metabolic adaptation
Researchers designing protocols involving retatrutide from specialized suppliers like PEPTIDE PRO should acknowledge these knowledge gaps and implement appropriate monitoring frameworks.
Contraindications and Precautions
Based on class effects and preliminary retatrutide data, certain populations require special consideration:
❌ Personal or family history of medullary thyroid carcinoma
❌ Multiple endocrine neoplasia syndrome type 2
⚠️ History of pancreatitis (relative contraindication)
⚠️ Severe gastrointestinal disease
⚠️ Pregnancy or planned pregnancy (research use only)
⚠️ Diabetic retinopathy (monitor closely)
Comparing Safety to Established Alternatives
Retatrutide’s safety profile appears generally consistent with approved GLP-1 and dual-agonist medications, with gastrointestinal effects representing the primary tolerability challenge. The triple-agonist mechanism doesn’t appear to introduce fundamentally new safety concerns beyond those associated with the established peptide class.
However, the enhanced efficacy—producing more substantial metabolic changes—may amplify secondary effects like nutritional deficiencies and stress-related phenomena such as telogen effluvium. This underscores the importance of comprehensive support protocols during research applications.
Research-Grade Retatrutide: Quality and Sourcing Considerations
For researchers investigating whether retatrutide causes hair loss or exploring other aspects of this novel peptide, compound quality represents a critical variable that can significantly impact study outcomes and safety profiles.
Why Purity Matters in Peptide Research
Research-grade peptides must meet stringent purity standards to ensure:
Reproducible Results Impurities and inconsistent composition create confounding variables that compromise research validity. High-purity compounds enable reliable, replicable outcomes across study cohorts.
Safety Optimization Contaminants or degradation products may introduce unexpected adverse effects unrelated to the primary compound, potentially including inflammatory responses that could theoretically affect various physiological systems.
Accurate Dosing Peptide purity directly impacts actual administered dose. A compound with 95% purity delivers less active ingredient than one with 99% purity at identical nominal doses, affecting both efficacy and safety assessments.
PEPTIDE PRO Quality Standards
PEPTIDE PRO maintains rigorous quality control protocols for all research peptides, including retatrutide:
✓ Research-grade purity: Compounds produced under strict quality conditions
✓ Proper storage: Temperature-controlled handling from synthesis through delivery
✓ Documentation: Certificates of Analysis (COAs) available for verification
✓ Appropriate labeling: Clear “For Research Use Only” designation
✓ Professional packaging: Temperature-appropriate shipping materials
Proper Handling and Storage
Maintaining peptide integrity after receipt requires adherence to established protocols:
Lyophilized (Powder) Form
- Store in cool, dry location away from light
- Refrigeration (2-8°C) extends stability
- Freezing (-20°C or below) optimal for long-term storage
- Minimize freeze-thaw cycles
After Reconstitution
- Refrigerate immediately at 2-8°C
- Use within recommended timeframe (typically 14-30 days)
- Protect from light exposure
- Maintain sterile technique to prevent contamination
Reconstitution Best Practices
- Use appropriate bacteriostatic water
- Add solvent slowly along pen peptide wall
- Gentle swirling rather than vigorous shaking
- Allow complete dissolution before use
Researchers can find comprehensive guidance on reconstitution and storage protocols through PEPTIDE PRO’s educational resources.
Regulatory and Ethical Considerations
All research involving retatrutide must adhere to appropriate regulatory frameworks:
Research Use Only Retatrutide remains investigational and is not approved for human therapeutic use outside clinical trials. Compounds should be clearly labeled and used strictly for legitimate research purposes.
Institutional Oversight Research protocols should receive appropriate institutional review board (IRB) or ethics committee approval where applicable.
Documentation Maintain complete records of sourcing, storage, handling, and administration protocols to ensure research integrity and regulatory compliance.
Informed Consent Any research involving human subjects requires comprehensive informed consent processes detailing known risks, including potential for temporary hair shedding during rapid weight loss phases.
Future Research Directions: What We Still Need to Know
As the scientific community continues investigating retatrutide, several important questions remain unanswered regarding both efficacy and safety—including definitive clarification on whether retatrutide causes hair loss in specific populations or usage scenarios.
Critical Knowledge Gaps
Long-Term Safety Beyond One Year
Current clinical trials provide data up to 48-52 weeks[1][2], leaving extended use effects uncertain:
- Does hair health remain stable with multi-year administration?
- Do nutritional deficiencies accumulate or stabilize?
- What represents optimal long-term dosing strategies?
- How does extended use affect other physiological systems?
Population-Specific Responses
Most Phase 2 data comes from relatively homogeneous participant groups. Additional research needed for:
- Diverse ethnic and genetic backgrounds
- Older adult populations
- Individuals with various comorbidity profiles
- Different baseline nutritional status groups
Mechanistic Understanding
While we understand that rapid weight loss—not retatrutide itself—likely triggers any hair shedding, detailed mechanistic studies could clarify:
- Specific metabolic thresholds that trigger telogen effluvium
- Individual susceptibility factors (genetic, nutritional, hormonal)
- Optimal prevention strategies based on mechanism
- Whether triple-agonist activation has any direct follicular effects
Comparative Effectiveness
Head-to-head trials comparing retatrutide to approved alternatives would illuminate:
- Relative hair loss incidence across peptide classes
- Whether enhanced efficacy correlates with increased secondary effects
- Optimal compound selection for different research objectives
Emerging Research Areas
Nutritional Optimization Protocols
Future studies should investigate:
- Minimum protein requirements to prevent hair loss during rapid weight reduction
- Specific micronutrient supplementation protocols
- Timing and composition of nutritional interventions
- Biomarkers predicting individual nutritional vulnerability
Hair Health Monitoring
Standardized assessment tools could improve research quality:
- Validated hair loss severity scales
- Objective measurement techniques (hair pull tests, phototrichograms)
- Biomarkers of follicular stress
- Predictive models identifying at-risk individuals
Combination Approaches
Research exploring adjunctive interventions might include:
- Concurrent hair-protective treatments during metabolic protocols
- Nutritional co-interventions optimized for follicular health
- Dose titration strategies minimizing physiological stress
Contributing to the Evidence Base
The research community can advance understanding through:
📝 Systematic adverse event reporting: Comprehensive documentation of all observed effects, including hair changes
📝 Long-term follow-up studies: Tracking participants beyond initial trial periods
📝 Mechanistic investigations: Laboratory studies examining direct peptide effects on follicular biology
📝 Real-world evidence: Post-market surveillance as retatrutide progresses toward potential approval
Researchers utilizing high-quality compounds from PEPTIDE PRO contribute to this growing evidence base through rigorous protocol design and comprehensive outcome reporting.
Conclusion: Evidence-Based Perspective on Retatrutide and Hair Loss
Does retatrutide cause hair loss? Based on comprehensive review of clinical trial data, mechanistic understanding, and comparison with related compounds, the evidence-based answer is: Retatrutide itself does not appear to directly cause hair loss. No clinical trials through 2025 have documented significant hair loss as a direct adverse effect of this triple-agonist peptide[1][2][5].
However, this conclusion requires important context: temporary hair shedding may occur as an indirect consequence of the rapid metabolic changes and weight reduction that retatrutide facilitates. This phenomenon—telogen effluvium—represents a well-established physiological response to rapid weight loss, nutritional stress, and metabolic flux rather than a direct pharmacological effect of the peptide compound.
Key Evidence Summary
✅ Clinical trials: No documented hair loss in formal adverse event reporting
✅ Mechanism: No known direct effect on hair follicle biology
✅ Class comparison: Similar GLP-1 medications show 3-5.7% temporary hair loss rates
✅ Timeline: Any shedding typically occurs 3-6 months after metabolic changes begin
✅ Reversibility: Hair loss associated with weight loss peptides is typically temporary and self-resolving
✅ Prevention: Adequate nutrition, particularly protein and micronutrients, substantially reduces risk
Practical Recommendations for Researchers
Institutions and researchers investigating retatrutide should implement comprehensive protocols addressing potential hair health concerns:
1. Baseline Assessment
- Document pre-existing hair status
- Measure nutritional biomarkers (iron, vitamin D, zinc, protein status)
- Screen for underlying conditions affecting hair health
2. Nutritional Support
- Ensure minimum protein intake (1.0-1.2g/kg daily)
- Provide or recommend comprehensive micronutrient supplementation
- Monitor dietary adequacy throughout research duration
3. Education and Expectation Management
- Inform participants about temporary hair shedding possibility
- Explain mechanisms and typical timeline
- Emphasize reversible nature and prevention strategies
4. Monitoring and Documentation
- Track hair changes systematically using validated tools
- Document timing, severity, and resolution
- Contribute to evidence base through comprehensive reporting
5. Quality Sourcing
- Obtain research-grade peptides from reputable suppliers like PEPTIDE PRO
- Verify purity through Certificates of Analysis
- Maintain proper storage and handling protocols
Looking Forward
As retatrutide advances through clinical development, longer-term safety data will provide additional clarity on all potential effects, including any hair-related observations. The current evidence strongly suggests that with appropriate nutritional support and reasonable weight loss pacing, hair health can be maintained during retatrutide research protocols.
For researchers seeking high-purity retatrutide and other research peptides, PEPTIDE PRO offers premium research-grade compounds with fast UK delivery, comprehensive documentation, and professional support. All products are clearly labeled “For Research Use Only” and handled under strict quality conditions to ensure reliable, consistent results.
The question “does retatrutide cause hair loss?” ultimately reflects broader considerations about comprehensive research design, participant care, and the importance of distinguishing direct pharmacological effects from secondary consequences of therapeutic efficacy. By understanding these distinctions and implementing evidence-based preventive strategies, researchers can optimize both scientific outcomes and participant experiences.
References
[1] Clinical trial safety data and GLP-1 receptor agonist class effects, 2024-2025
[2] Retatrutide Phase 2 clinical trial results and adverse event profiles, 2024
[3] Anecdotal reports and post-market surveillance data for GLP-1 medications, 2023-2025
[4] Hair follicle biology and telogen effluvium mechanisms, dermatological research literature
[5] Retatrutide safety profile and gastrointestinal side effect incidence, clinical documentation 2024-2025
[6] Nutritional requirements during rapid weight loss and metabolic interventions, nutritional science literature
[7] Telogen effluvium timeline, triggers, and recovery patterns, dermatology clinical studies 2023-2024
[8] GLP-1, GIP, and glucagon receptor mechanisms and metabolic effects, endocrinology research
