
The landscape of metabolic disease treatment is undergoing a remarkable transformation, and at the forefront of this revolution stands retatrutide—a groundbreaking triple receptor agonist that’s redefining what’s possible in obesity and diabetes management. As researchers and laboratories worldwide seek innovative solutions for metabolic disorders, understanding what is retatrutide, its uses, research findings, and availability has become increasingly critical for advancing scientific knowledge in this rapidly evolving field.
Retatrutide (LY3437943) represents a significant leap forward in peptide therapeutics, distinguished by its unique ability to simultaneously activate three crucial metabolic receptors: glucagon-like peptide-1 (GLP-1), gastric inhibitory polypeptide (GIP), and glucagon receptors. This triple-action mechanism sets it apart from earlier generations of metabolic peptides and positions it as a focal point for cutting-edge research into obesity, type 2 diabetes, and associated cardiometabolic conditions.
Key Takeaways
- Triple Receptor Mechanism: Retatrutide uniquely activates GLP-1, GIP, and glucagon receptors simultaneously, providing enhanced metabolic effects compared to single or dual agonists
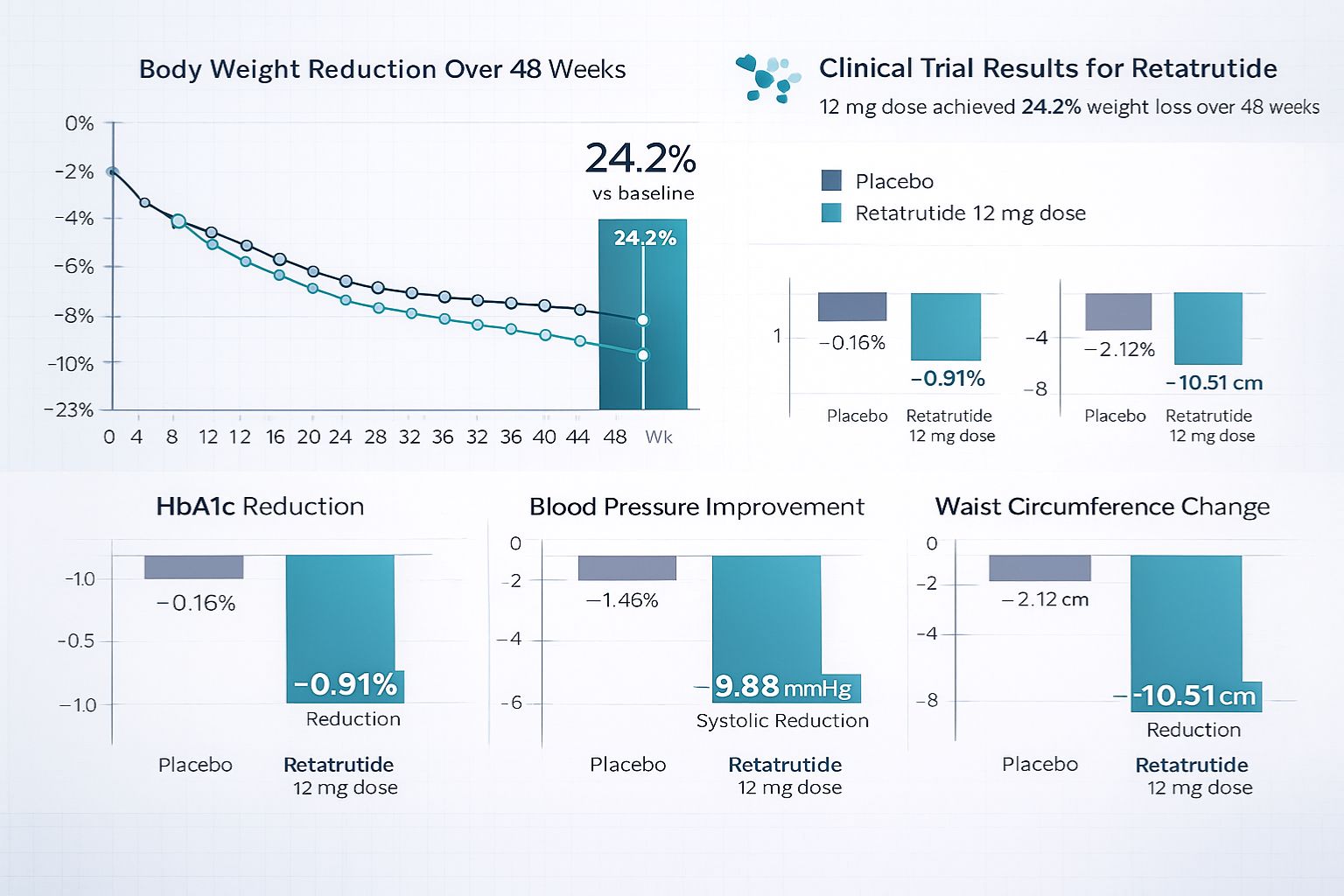
- Significant Weight Reduction: Clinical trials demonstrate up to 24.2% body weight loss at the 12 mg dose after 48 weeks, with no plateau observed
- Rapid Efficacy Timeline: Achieves meaningful results in just 24 weeks compared to 68-72 weeks for comparable peptides
- Comprehensive Metabolic Benefits: Beyond weight loss, retatrutide improves glycemic control, blood pressure, lipid profiles, and shows promise for osteoarthritis and cardiovascular health
- Research-Grade Availability: High-purity retatrutide is available for laboratory research through specialized suppliers like Peptide Pro, strictly for scientific investigation
Understanding Retatrutide: A Novel Triple Agonist Peptide

The Science Behind Retatrutide’s Mechanism
Retatrutide operates through a sophisticated triple-receptor agonist mechanism that represents a paradigm shift in metabolic peptide research. Unlike traditional single-target approaches, this compound simultaneously engages three distinct metabolic pathways, creating a synergistic effect that amplifies therapeutic potential.
The GLP-1 receptor activation component enhances insulin secretion in a glucose-dependent manner, suppresses glucagon release, and slows gastric emptying—mechanisms that collectively improve glycemic control and reduce appetite. The GIP receptor engagement complements these effects by further stimulating insulin secretion and potentially influencing fat metabolism and bone health. Most notably, the glucagon receptor activation increases energy expenditure and promotes fat oxidation, a unique feature that distinguishes retatrutide from dual agonists like tirzepatide.
This tri-agonist architecture creates what researchers describe as a “metabolic reset,” addressing multiple pathophysiological aspects of obesity and type 2 diabetes simultaneously. The glucagon component particularly enhances the compound’s weight loss efficacy by increasing metabolic rate and promoting the breakdown of stored fat tissue.
Molecular Structure and Classification
Retatrutide is classified as a synthetic peptide analog designed to mimic and enhance natural incretin hormones while incorporating glucagon receptor activity. Its molecular structure has been carefully engineered to maintain stability, optimize receptor binding affinity across all three targets, and extend half-life for practical dosing schedules.
The compound belongs to the broader category of incretin-based therapies but represents a distinct evolution beyond GLP-1 receptor agonists (like semaglutide) and dual GLP-1/GIP agonists (like tirzepatide). This classification as a triple agonist places retatrutide at the cutting edge of peptide therapeutics research, making it a priority compound for laboratories investigating next-generation metabolic interventions.
For research facilities seeking to explore this innovative peptide, high-purity research-grade retatrutide is available through specialized suppliers committed to quality and scientific integrity.
Clinical Research Findings: What Is Retatrutide Showing in Studies?
Weight Loss and Body Composition Outcomes
The clinical research surrounding retatrutide has produced remarkable findings that position it as one of the most effective weight management compounds under investigation. A comprehensive meta-analysis of three randomized controlled trials involving 878 participants revealed that retatrutide reduced body weight by a mean of 14.33% compared to placebo—a statistically significant outcome that exceeded many existing interventions[1][2].
More impressively, the highest dose studied (12 mg) achieved an average weight reduction of 24.2% after 48 weeks of treatment, with clinical observations indicating no plateau effect at that timepoint[1][2]. This suggests the potential for continued weight loss beyond the study period, a characteristic that sets retatrutide apart from compounds that typically demonstrate diminishing returns over time.
Beyond total weight reduction, retatrutide demonstrated substantial improvements in body composition metrics:
- Waist circumference reduction: 10.51 cm average decrease
- Body mass index (BMI) reduction: 5.38 units average decrease
- Visceral fat reduction: Significant improvements noted in imaging studies
These body composition changes are particularly relevant for metabolic health research, as visceral adiposity is strongly associated with insulin resistance, cardiovascular disease, and metabolic syndrome. The preferential reduction in central adiposity observed with retatrutide suggests mechanisms beyond simple caloric restriction.
Glycemic Control and Diabetes Management Research
For researchers investigating type 2 diabetes interventions, retatrutide’s glycemic effects present compelling data. Clinical trials demonstrated a reduction in hemoglobin A1c (HbA1c) of 0.91% compared to placebo, indicating substantial improvements in long-term glucose control[1][2].
Additionally, fasting plasma glucose decreased by an average of 23.51 mg/dL, reflecting enhanced insulin sensitivity and beta-cell function[1][2]. These glycemic improvements occurred alongside weight loss, creating a dual benefit that addresses two interconnected aspects of metabolic dysfunction.
The glucose-lowering effects of retatrutide appear to operate through multiple mechanisms:
- Enhanced glucose-dependent insulin secretion via GLP-1 and GIP pathways
- Suppression of inappropriate glucagon secretion
- Improved insulin sensitivity secondary to weight loss and visceral fat reduction
- Potential direct effects on hepatic glucose production through glucagon receptor modulation
These multifaceted glycemic benefits make retatrutide a valuable research tool for laboratories studying the complex interplay between weight, insulin resistance, and glucose homeostasis.
Cardiovascular and Cardiometabolic Parameters
Emerging research on retatrutide extends beyond weight and glucose control to encompass broader cardiometabolic health markers. Clinical trial data revealed significant improvements in blood pressure, with systolic blood pressure reduced by 9.88 mm Hg and diastolic pressure by 3.88 mm Hg[1][2].
Lipid profile improvements were also documented, including:
- Reduced triglyceride levels
- Decreased non-HDL cholesterol
- Favorable changes in lipid particle size and composition
These cardiovascular risk factor improvements have prompted the initiation of dedicated cardiovascular outcomes trials currently underway as of 2025[6]. These studies aim to determine whether retatrutide can significantly reduce the incidence of major adverse cardiovascular events (MACE) in high-risk populations—a critical question for understanding the compound’s full therapeutic potential.
The cardiovascular benefits observed may result from direct receptor-mediated effects, weight loss-associated improvements, or synergistic combinations of both mechanisms. Ongoing research continues to elucidate these pathways, making retatrutide an important subject for cardiovascular metabolism research.
Expanding Research Applications: Beyond Weight and Glucose
Osteoarthritis and Musculoskeletal Research
One of the most intriguing emerging research areas for retatrutide involves its potential effects on osteoarthritis and joint health. As of December 2025, clinical research demonstrated that retatrutide reduced body weight by 28.7% while simultaneously reducing knee pain in patients with obesity and osteoarthritis[5].
This dual benefit addresses a critical clinical challenge: obesity-related mechanical stress on weight-bearing joints contributes significantly to osteoarthritis progression and pain. By achieving substantial weight reduction, retatrutide may offer indirect benefits for joint health through:
- Decreased mechanical loading on arthritic joints
- Reduced systemic inflammation associated with adiposity
- Improved mobility enabling increased physical activity
- Potential direct anti-inflammatory effects through receptor-mediated pathways
Ongoing clinical trials are investigating retatrutide for chronic low back pain relief in patients with obesity over 72-week periods, measuring pain intensity, physical function, and sleep quality as primary outcomes[3]. These studies expand the research scope of retatrutide beyond traditional metabolic endpoints to encompass quality-of-life and functional capacity measures.
For researchers interested in musculoskeletal and metabolic interactions, retatrutide represents a valuable tool for investigating the complex relationships between adiposity, inflammation, and joint health. Laboratories can source research-grade peptides to support these investigative efforts.
Oncology and Cancer Metabolism Research
Emerging pre-clinical research has identified potential applications for retatrutide in cancer metabolism studies, particularly regarding obesity-associated malignancies. Pre-clinical models demonstrated that retatrutide reduced pancreatic cancer tumor engraftment by 30% while improving systemic metabolism with 41% lower fasted blood glucose in obesity-associated cancer models[4].
These findings suggest several potential mechanisms worthy of further investigation:
- Metabolic normalization: Reducing hyperglycemia and hyperinsulinemia may create a less favorable environment for cancer cell proliferation
- Adipokine modulation: Weight loss and visceral fat reduction alter the secretion of pro-inflammatory and pro-tumorigenic adipokines
- Direct receptor effects: GLP-1, GIP, and glucagon receptors are expressed in various tissues, potentially influencing cellular proliferation and apoptosis pathways
- Immune function enhancement: Obesity impairs immune surveillance; weight reduction may restore anti-tumor immunity
While these oncology applications remain in early-stage research, they highlight retatrutide’s potential as a research tool for investigating the complex intersections between metabolism, obesity, and cancer biology. Research institutions exploring these connections require access to high-purity research peptides with appropriate quality documentation.
Comparative Efficacy: Retatrutide vs. Other Metabolic Peptides
Understanding what is retatrutide’s position in the broader peptide landscape requires comparative analysis with established compounds. Research data reveals several distinguishing characteristics:
| Parameter | Retatrutide | Semaglutide | Tirzepatide |
|---|---|---|---|
| Mechanism | Triple agonist (GLP-1/GIP/Glucagon) | GLP-1 agonist | Dual agonist (GLP-1/GIP) |
| Weight Loss (%) | 24.2% (12 mg, 48 weeks) | ~15% (68 weeks) | ~22% (72 weeks) |
| Time to Efficacy | 24 weeks | 68 weeks | 72 weeks |
| Receptor Targets | 3 | 1 | 2 |
| Metabolic Rate Effect | Enhanced (glucagon) | Minimal | Minimal |
The data clearly demonstrates retatrutide’s faster onset of action—achieving meaningful results in just 24 weeks compared to 68-72 weeks for comparable peptides[1]. This accelerated timeline may reflect the additive effects of glucagon receptor activation on energy expenditure and metabolic rate.
The magnitude of weight loss observed with retatrutide also exceeds that of single-agonist approaches and appears competitive with or superior to dual agonists, despite shorter study durations. These comparative advantages make retatrutide a priority compound for research facilities investigating optimal metabolic intervention strategies.
Safety Profile and Adverse Event Research
Clinical Trial Safety Data
A critical aspect of understanding what is retatrutide involves evaluating its safety profile in controlled research settings. The meta-analysis of three randomized controlled trials involving 878 patients demonstrated an appropriate safety profile with no significant difference in adverse events compared to placebo[1][2].
This finding is particularly noteworthy given the compound’s potent metabolic effects. Common adverse events reported in trials included:
- Gastrointestinal effects: Nausea, vomiting, and diarrhea (consistent with GLP-1 agonist class effects)
- Injection site reactions: Mild to moderate local reactions
- Transient effects: Headache, fatigue, and dizziness during dose escalation
Importantly, serious adverse events did not occur at significantly higher rates than placebo, and discontinuation rates due to adverse effects remained within acceptable ranges for metabolic research compounds. The gastrointestinal side effects typically diminished with continued exposure and dose titration protocols.
Dosing Protocols and Tolerance Research
Research protocols for retatrutide typically employ gradual dose escalation strategies to optimize tolerance:
- Initial dosing: Starting at lower doses (1-2 mg)
- Titration period: Gradual increases over 4-8 week intervals
- Maintenance dosing: Achieving target doses of 4-12 mg based on research objectives
- Monitoring parameters: Regular assessment of metabolic markers and tolerance indicators
This escalation approach minimizes gastrointestinal adverse effects while allowing subjects to adapt to the compound’s metabolic effects. Research institutions designing studies with retatrutide should incorporate appropriate titration schedules and monitoring protocols.
For laboratories conducting research with retatrutide, proper handling and storage protocols are essential. Educational resources on peptide reconstitution and storage provide critical guidance for maintaining compound integrity throughout research protocols.
Research-Grade Retatrutide: Availability and Sourcing Considerations
Obtaining High-Purity Retatrutide for Research
For research institutions and laboratories investigating metabolic interventions, accessing high-quality retatrutide is fundamental to generating reliable, reproducible data. Research-grade retatrutide must meet stringent purity standards, typically exceeding 98% purity as verified by high-performance liquid chromatography (HPLC) and mass spectrometry.
When sourcing retatrutide for research applications, several critical factors warrant consideration:
- ✅ Purity verification: Certificate of Analysis (COA) documentation confirming purity levels
- ✅ Proper storage conditions: Lyophilized peptides maintained under controlled temperature and humidity
- ✅ Regulatory compliance: Clear labeling as “For Research Use Only” with appropriate disclaimers
- ✅ Supply chain integrity: Traceable sourcing with quality assurance throughout distribution
- ✅ Technical support: Access to reconstitution guidance and handling protocols
Peptide Pro specializes in supplying premium research-grade peptides to the scientific community, offering retatrutide and related compounds with exceptional purity standards, comprehensive quality documentation, and professional technical support.
UK and International Research Peptide Availability
Researchers in the United Kingdom benefit from streamlined access to research peptides through domestic suppliers offering fast UK delivery options. Peptide Pro’s commitment to same-day dispatch for orders placed before 1pm (Monday-Friday) ensures minimal delays in research timelines—a critical consideration for time-sensitive experimental protocols.
International researchers also have access to retatrutide through established distribution channels, with shipping options supporting laboratories worldwide. Temperature-controlled packaging and appropriate shipping methods preserve peptide integrity during transit, ensuring compounds arrive in optimal condition for research applications.
The availability of research-grade retatrutide has expanded significantly as scientific interest in triple agonist mechanisms has grown. This increased availability supports diverse research initiatives spanning metabolism, obesity, diabetes, cardiovascular health, musculoskeletal disorders, and cancer biology.
Quality Assurance and Research Standards
The integrity of research findings depends fundamentally on the quality of compounds used in experimental protocols. When evaluating retatrutide suppliers, researchers should prioritize:
Documentation Standards:
- Comprehensive Certificates of Analysis (COAs) for each batch
- HPLC chromatograms confirming purity
- Mass spectrometry data verifying molecular identity
- Stability testing data supporting storage recommendations
Handling Protocols:
- Lyophilized storage at appropriate temperatures (typically -20°C to -80°C)
- Sterile reconstitution procedures using bacteriostatic water
- Aliquoting strategies to minimize freeze-thaw cycles
- Clear expiration dating based on stability data
Regulatory Compliance:
- Explicit “For Research Use Only” labeling
- No claims regarding human or animal therapeutic use
- Compliance with relevant research chemical regulations
- Transparent sourcing and manufacturing information
Peptide Pro’s extensive range of research peptides adheres to these quality standards, providing researchers with confidence in their experimental materials. The company’s commitment to exceptional purity, fast delivery, and responsible labeling aligns with the needs of professional research environments.
Comparative Research: Retatrutide in Context
Triple Agonist Advantages in Metabolic Research
The evolution from single-receptor agonists to dual and now triple agonists represents a strategic progression in metabolic research. Each generation has built upon the limitations of its predecessors:
First Generation (GLP-1 Agonists):
- Compounds: Liraglutide, semaglutide
- Primary effects: Appetite suppression, glucose-dependent insulin secretion
- Limitations: Modest weight loss (10-15%), minimal metabolic rate effects
Second Generation (Dual GLP-1/GIP Agonists):
- Compounds: Tirzepatide
- Enhanced effects: Greater weight loss (20-22%), improved insulin sensitivity
- Limitations: Still lacking direct metabolic rate enhancement
Third Generation (Triple GLP-1/GIP/Glucagon Agonists):
- Compounds: Retatrutide, survodutide
- Comprehensive effects: Superior weight loss (24%+), enhanced energy expenditure, faster onset
- Advantages: Addresses multiple metabolic pathways simultaneously
The addition of glucagon receptor activation in retatrutide provides the critical metabolic rate enhancement that distinguishes it from earlier generations. This mechanism increases energy expenditure even in the absence of increased physical activity—a significant advantage for research models where activity levels are controlled.
For researchers comparing metabolic peptides, Peptide Pro’s curated portfolio includes retatrutide alongside other important compounds like tirzepatide, semaglutide, and emerging alternatives like survodutide, enabling comprehensive comparative studies.
Research Timeline Advantages
One of retatrutide’s most significant practical advantages for research applications is its accelerated efficacy timeline. Achieving meaningful metabolic effects in 24 weeks rather than 68-72 weeks offers several research benefits:
- 📊 Faster data generation: Shorter study durations enable more rapid hypothesis testing
- 💰 Cost efficiency: Reduced study duration lowers overall research costs
- 🔬 Increased throughput: Laboratories can complete more studies in equivalent timeframes
- 📈 Earlier endpoint assessment: Preliminary results available sooner for protocol adjustments
This timeline advantage makes retatrutide particularly attractive for pilot studies, dose-finding research, and mechanism-of-action investigations where rapid feedback is valuable. Research institutions can design more efficient experimental protocols that generate actionable data without the extended timelines required for comparable compounds.
Future Research Directions and Ongoing Studies
Cardiovascular Outcomes Research
As of 2025, one of the most anticipated research initiatives involves dedicated cardiovascular outcomes trials designed to determine whether retatrutide’s impressive metabolic effects translate to reduced cardiovascular events[6]. These large-scale, long-duration studies will assess:
- Primary endpoints: Major adverse cardiovascular events (MACE) including cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke
- Secondary endpoints: Heart failure hospitalizations, revascularization procedures, and cardiovascular biomarkers
- Mechanistic substudies: Imaging studies assessing atherosclerotic plaque, cardiac function, and vascular health
The cardiovascular research surrounding retatrutide will be critical for understanding whether the compound’s benefits extend beyond metabolic parameters to reduce hard clinical outcomes. Given the substantial improvements in weight, blood pressure, lipids, and glucose control observed in earlier trials, there is strong biological plausibility for cardiovascular protection.
Pain and Functional Outcomes Research
The ongoing trials investigating retatrutide for chronic low back pain and osteoarthritis represent an expansion into functional outcome research[3][5]. These studies measure:
- Pain intensity scores: Validated pain assessment instruments
- Physical function: Mobility assessments, activity tolerance, and functional capacity
- Quality of life: Patient-reported outcomes measuring overall well-being
- Sleep quality: Assessment of pain-related sleep disturbances
This research direction acknowledges that obesity’s impact extends far beyond metabolic parameters to affect daily functioning, pain levels, and quality of life. By investigating these broader outcomes, researchers can better understand retatrutide’s comprehensive effects on health and well-being.
Combination Therapy Research
Future research directions may explore retatrutide in combination with other interventions:
- Lifestyle modifications: Combined effects with dietary interventions and exercise programs
- Complementary peptides: Synergistic effects with compounds targeting different pathways
- Pharmacological combinations: Integration with other metabolic or cardiovascular medications
- Behavioral interventions: Combined approaches addressing psychological aspects of obesity
These combination research paradigms may reveal synergistic effects that exceed the benefits of any single intervention, potentially establishing new standards for comprehensive metabolic management.
Practical Considerations for Research Applications
Experimental Design Recommendations
Researchers designing studies with retatrutide should consider several protocol elements to optimize data quality:
Dosing Strategies:
- Implement gradual dose escalation (typically 4-week intervals)
- Include multiple dose arms to establish dose-response relationships
- Consider body weight-adjusted dosing for comparative research
- Plan for long-term maintenance dosing in extended studies
Outcome Measurements:
- Primary metabolic endpoints: Body weight, body composition, HbA1c, fasting glucose
- Cardiovascular parameters: Blood pressure, lipid profiles, inflammatory markers
- Mechanistic assessments: Insulin sensitivity, energy expenditure, appetite hormones
- Safety monitoring: Adverse events, laboratory safety parameters, vital signs
Control Conditions:
- Appropriate placebo controls with identical dosing schedules
- Active comparator arms with established metabolic peptides
- Baseline stabilization periods before intervention initiation
- Washout periods for crossover designs
Storage and Handling Protocols
Maintaining retatrutide integrity throughout research protocols requires adherence to proper storage and handling procedures:
Pre-Reconstitution Storage:
- Store lyophilized peptide at -20°C to -80°C
- Protect from light and moisture
- Minimize temperature fluctuations
- Verify storage conditions upon receipt
Reconstitution Procedures:
- Use bacteriostatic water for multi-dose applications
- Employ sterile technique throughout reconstitution
- Allow refrigerated peptide to reach room temperature before adding solvent
- Gently swirl rather than shake to dissolve peptide
Post-Reconstitution Storage:
- Refrigerate reconstituted solution at 2-8°C
- Use within recommended timeframe (typically 28 days)
- Consider aliquoting to minimize freeze-thaw cycles
- Maintain detailed records of reconstitution dates
For comprehensive guidance on peptide handling, researchers can access educational resources on reconstitution and storage that provide detailed protocols optimized for research applications.
Documentation and Reproducibility
High-quality research requires meticulous documentation of all experimental parameters:
- 📋 Batch documentation: Record lot numbers, reconstitution dates, and storage conditions
- 📊 Dosing logs: Maintain detailed records of all doses administered
- 🔬 Protocol adherence: Document any deviations from planned procedures
- 📈 Data integrity: Implement appropriate data management and quality control procedures
This documentation supports research reproducibility and enables meaningful interpretation of results across different studies and research groups.
Retatrutide Research: Ethical and Regulatory Considerations
Research-Only Status and Compliance
It is critical to emphasize that retatrutide, like all research peptides, is strictly for research use only. These compounds are not approved for human consumption, therapeutic use, or administration to animals outside approved research protocols. Researchers must:
- ✅ Ensure all studies comply with institutional review board (IRB) or ethics committee approvals
- ✅ Maintain appropriate research licenses and regulatory compliance
- ✅ Use compounds only within approved research protocols
- ✅ Implement proper safety procedures for handling research chemicals
- ✅ Provide appropriate informed consent for any human research subjects
Peptide Pro’s commitment to responsible supply includes clear “For Research Use Only” labeling, comprehensive product information, and support for researchers in maintaining regulatory compliance. The company’s transparent approach ensures research institutions can source peptides with confidence in their compliance status.
Responsible Research Practices
The scientific community’s investigation of compounds like retatrutide carries responsibilities beyond regulatory compliance:
Scientific Integrity:
- Design rigorous protocols with appropriate controls
- Report both positive and negative findings
- Avoid overstating conclusions beyond data support
- Acknowledge limitations and potential confounding factors
Safety Prioritization:
- Implement comprehensive safety monitoring in all research protocols
- Establish clear stopping criteria for adverse events
- Provide appropriate medical oversight for human research
- Maintain detailed safety documentation
Transparency:
- Register clinical trials in appropriate databases
- Publish methodology and results regardless of outcomes
- Disclose funding sources and potential conflicts of interest
- Share data to support scientific advancement
These responsible research practices ensure that investigations of retatrutide contribute meaningfully to scientific knowledge while maintaining the highest ethical standards.
Accessing Research-Grade Retatrutide: A Practical Guide
Selecting a Research Peptide Supplier
For researchers seeking to incorporate retatrutide into their experimental protocols, selecting an appropriate supplier is a critical decision. Key evaluation criteria include:
Quality Indicators:
- Documented purity levels exceeding 98% (verified by HPLC)
- Comprehensive Certificates of Analysis for each batch
- Mass spectrometry confirmation of molecular identity
- Stability testing supporting storage recommendations
Service Standards:
- Responsive technical support for reconstitution and handling questions
- Fast, reliable shipping with appropriate temperature control
- Clear communication regarding product availability and timelines
- Professional customer service addressing research needs
Regulatory Compliance:
- Explicit “For Research Use Only” labeling and disclaimers
- No therapeutic claims or human use suggestions
- Transparent sourcing and quality assurance processes
- Compliance with relevant chemical research regulations
Peptide Pro meets these criteria through its commitment to exceptional purity, fast delivery, and professional service—establishing the company as a trusted source for researchers and laboratories across the UK and worldwide.
Ordering Process and Delivery
The practical process of obtaining research-grade retatrutide involves several straightforward steps:
- Browse or Search: Explore the peptide catalogue or search for specific compounds like retatrutide 40mg
- Review Documentation: Examine product specifications, purity data, and storage requirements
- Place Order: Complete secure checkout supporting multiple currencies (GBP, EUR, USD)
- Same-Day Dispatch: Orders placed before 1pm (Monday-Friday) ship the same day in temperature-appropriate packaging
- Track & Receive: Full tracking information provided with every order
This streamlined process minimizes delays in research timelines, ensuring researchers can initiate protocols without unnecessary waiting periods. For researchers requiring additional support products, bacteriostatic water and reconstitution supplies are available to support complete experimental preparation.
Building a Research Peptide Portfolio
Many research institutions investigating metabolic interventions benefit from maintaining a diverse peptide portfolio that enables comparative studies and mechanism-of-action research. A comprehensive metabolic research collection might include:
Weight Loss Research Peptides:
- Retatrutide (triple agonist)
- Tirzepatide (dual GLP-1/GIP agonist)
- Semaglutide (GLP-1 agonist)
- Cagrilintide (amylin analog)
Metabolic Support Compounds:
- AOD9604 (growth hormone fragment)
- Tesamorelin (GHRH analog)
- 5-Amino-1MQ (NNMT inhibitor)
Recovery and Repair Peptides:
This diversified approach enables comprehensive research programs investigating multiple mechanisms and comparative efficacy across different peptide classes.
Conclusion: The Future of Retatrutide Research
Understanding what is retatrutide, its uses, research applications, and availability positions researchers at the forefront of metabolic science innovation. This groundbreaking triple receptor agonist represents a significant advancement in peptide therapeutics, offering unprecedented efficacy in weight reduction, glycemic control, and cardiometabolic health improvements.
The clinical research to date demonstrates that retatrutide achieves superior weight loss outcomes (up to 24.2% at 48 weeks), faster onset of action (meaningful effects in just 24 weeks), and comprehensive metabolic benefits extending beyond traditional endpoints to include cardiovascular parameters, pain reduction, and functional improvements. These characteristics position retatrutide as a priority compound for research institutions investigating next-generation metabolic interventions.
As ongoing studies continue to expand our understanding of retatrutide’s full potential—including cardiovascular outcomes trials, musculoskeletal research, and cancer metabolism investigations—the scientific community’s interest in this compound will undoubtedly grow. Access to high-purity, research-grade retatrutide becomes increasingly critical for laboratories seeking to contribute to this evolving knowledge base.
Next Steps for Researchers
For research institutions and laboratories ready to incorporate retatrutide into their experimental protocols:
- Review Current Literature: Familiarize yourself with published clinical trial data and ongoing research initiatives
- Design Rigorous Protocols: Develop comprehensive experimental designs with appropriate controls and outcome measures
- Secure Quality Compounds: Source research-grade retatrutide from reputable suppliers with documented purity and quality assurance
- Implement Proper Handling: Follow established protocols for storage, reconstitution, and administration
- Maintain Documentation: Record all experimental parameters to support reproducibility and data integrity
Peptide Pro stands ready to support your research endeavors with premium research-grade peptides, including retatrutide and a comprehensive portfolio of metabolic compounds. With exceptional purity standards, same-day dispatch for orders placed before 1pm, and professional technical support, Peptide Pro delivers the quality and service that research excellence demands.
For questions about retatrutide availability, technical specifications, or research applications, contact the Peptide Pro team for expert guidance tailored to your specific research needs.
“PEPTIDE PRO delivers quality, consistency and first-class service—our preferred source for research peptides.” — Dr. A. Roberts, Senior Research Associate, UK
The future of metabolic research is unfolding now, and retatrutide represents a pivotal compound in that evolution. Equip your laboratory with the highest quality research materials and join the scientific community advancing our understanding of this remarkable triple agonist peptide.
References
[1] Meta-analysis of retatrutide efficacy in obesity and type 2 diabetes: Randomized controlled trials demonstrating mean weight reduction of 14.33% and HbA1c reduction of 0.91% (2024-2025)
[2] Clinical trial data: Retatrutide 12 mg dose achieving 24.2% weight loss after 48 weeks with improvements in waist circumference (10.51 cm), BMI (5.38 units), and blood pressure (systolic 9.88 mmHg, diastolic 3.88 mmHg) (2024-2025)
[3] Ongoing clinical trials: Retatrutide for chronic low back pain relief in patients with obesity over 72 weeks, measuring pain intensity, physical function, and sleep quality (2025)
[4] Pre-clinical research: Retatrutide reduced pancreatic cancer tumor engraftment by 30% and improved systemic metabolism with 41% lower fasted blood glucose in obesity-associated models (2024-2025)
[5] Clinical research update: Retatrutide demonstrated 28.7% body weight reduction and reduced knee pain in patients with obesity and osteoarthritis (December 2025)
[6] Cardiovascular outcomes trial: Ongoing research to determine if retatrutide significantly lowers incidence of serious heart-related complications (2025)
