The landscape of metabolic therapeutics has witnessed remarkable innovation in recent years, with peptide-based treatments emerging as powerful tools for addressing obesity, type 2 diabetes, and metabolic dysfunction. Among these groundbreaking compounds, retatrutide stands out as a first-of-its-kind therapeutic agent that simultaneously targets three distinct hormone pathways. Understanding how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) provides crucial insight into why this novel peptide has generated significant interest within the research community and advanced to phase 3 clinical trials with unprecedented metabolic outcomes.
Unlike earlier single or dual agonist compounds, retatrutide represents a paradigm shift in metabolic intervention by activating glucagon-like peptide 1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon (GCG) receptors through a single molecular structure. This unimolecular triple agonist approach creates synergistic metabolic effects that extend beyond simple weight reduction to comprehensive metabolic remodeling.
Key Takeaways
- Triple receptor activation: Retatrutide simultaneously engages GIP, GLP-1, and glucagon receptors with specific potency ratios, creating complementary metabolic effects across multiple organ systems
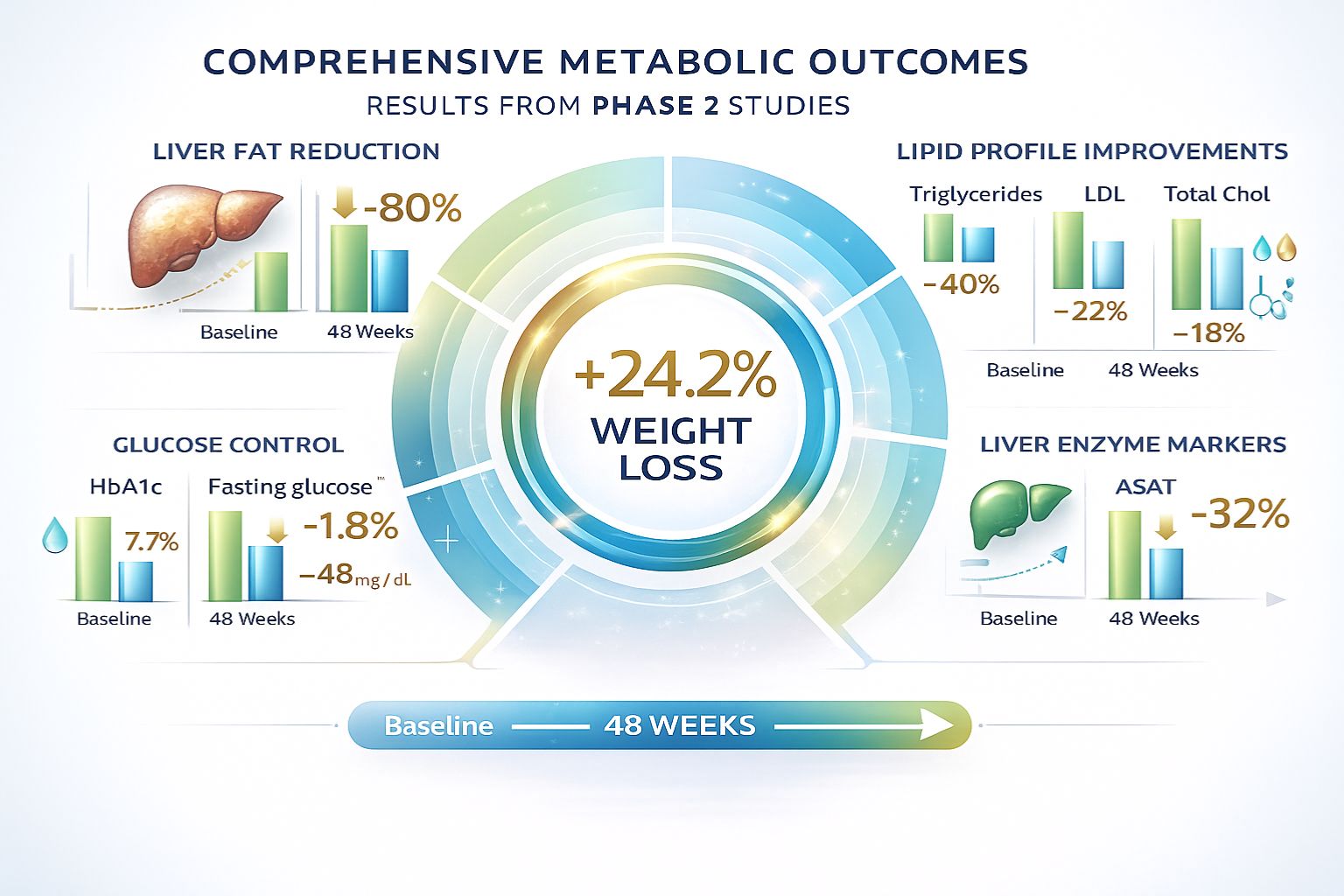
- Superior clinical outcomes: Phase 2 trials demonstrated 22.8-24.2% weight loss in individuals with obesity and 80% or greater liver fat reduction in metabolic dysfunction-associated fatty liver disease (MASLD)
- Extended pharmacokinetics: The C20 fatty di-acid modification enables albumin binding and once-weekly dosing through prolonged half-life
- Comprehensive metabolic improvement: Beyond weight loss, retatrutide significantly improves lipid profiles (triglycerides reduced by 35-40%), glucose control, and liver health markers
- Synergistic mechanism: The combination of appetite suppression (GLP-1), enhanced insulin secretion (GIP), and increased energy expenditure (glucagon) produces effects greater than individual receptor activation alone
The Molecular Architecture of Retatrutide
Peptide Structure and Pharmacokinetic Design
Retatrutide is engineered as a 39 amino-acid synthetic peptide with sophisticated structural modifications that enable its unique triple agonist activity and extended duration of action. The molecule incorporates a C20 fatty di-acid moiety conjugated to its peptide backbone—a critical modification that facilitates albumin binding within the bloodstream[1].
This albumin binding serves multiple essential functions:
✅ Extended half-life: Protects the peptide from rapid enzymatic degradation
✅ Sustained receptor engagement: Maintains therapeutic concentrations for once-weekly dosing
✅ Improved bioavailability: Enhances distribution to target tissues
✅ Reduced dosing frequency: Improves practical research applications
The fatty acid modification represents a sophisticated pharmacokinetic strategy employed in several modern peptide therapeutics. By reversibly binding to serum albumin—the most abundant protein in blood plasma—retatrutide achieves a circulation time measured in days rather than minutes, fundamentally altering its therapeutic profile.
Researchers interested in exploring high-purity research-grade peptides with similar structural modifications can access compounds manufactured under strict quality conditions with comprehensive documentation.
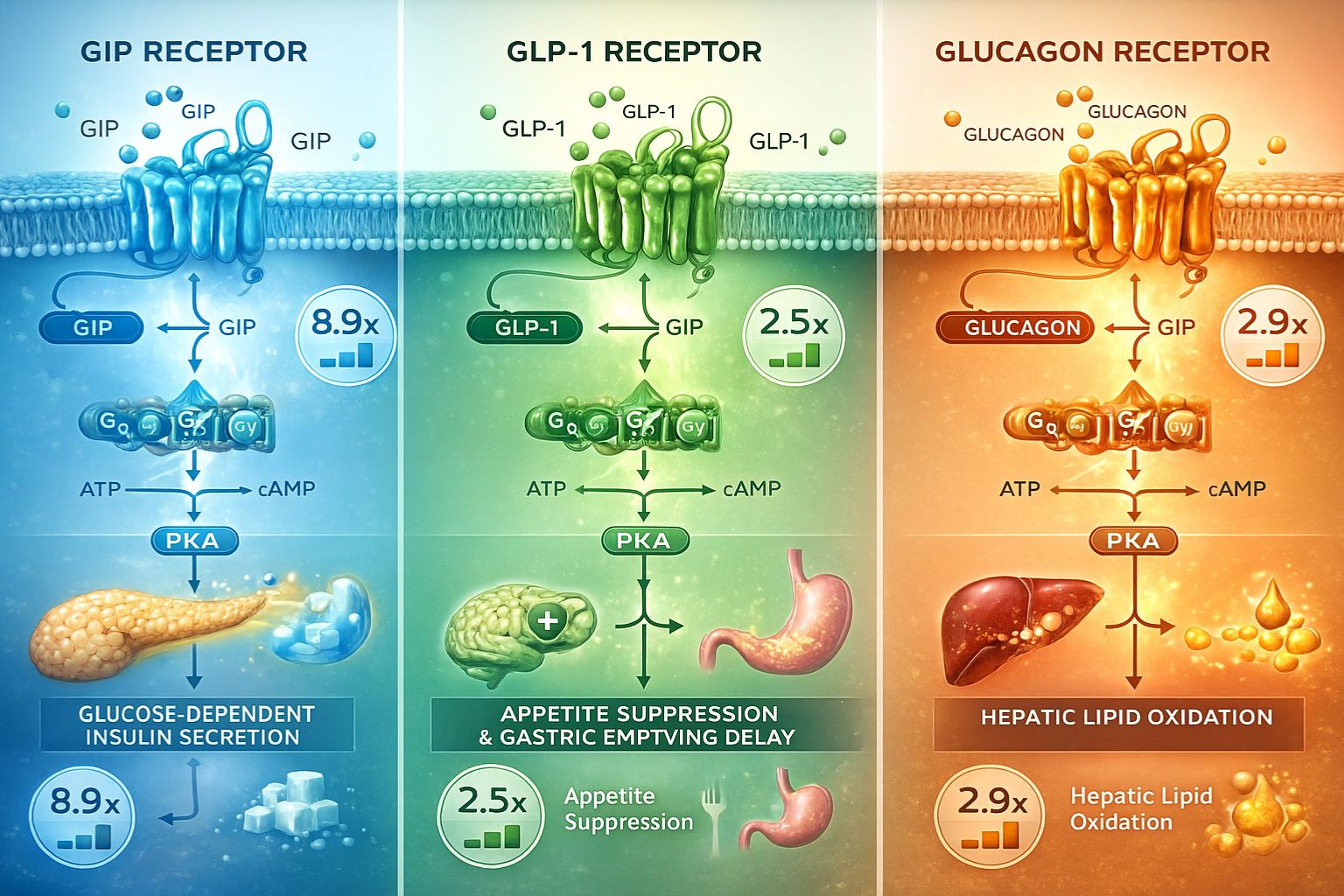
Receptor Potency Profile
Understanding how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) requires examining the specific potency at each receptor target. Retatrutide demonstrates carefully calibrated activation profiles:
| Receptor | Potency Relative to Endogenous Ligand | Functional Significance |
|---|---|---|
| GIP Receptor | 8.9× more potent than human GIP | Enhanced glucose-dependent insulin secretion; lipid metabolism modulation |
| GLP-1 Receptor | 2.5× less potent than endogenous GLP-1 | Appetite suppression; gastric emptying delay; insulin secretion |
| Glucagon Receptor | 2.9× less potent than endogenous glucagon | Hepatic lipid oxidation; energy expenditure increase |
This potency distribution is not arbitrary but reflects deliberate molecular engineering. The enhanced GIP receptor potency (8.9-fold) suggests this pathway plays a particularly important role in retatrutide’s metabolic effects, while the moderate activation of GLP-1 and glucagon receptors provides complementary benefits without excessive receptor overstimulation[2].
The balanced multi-receptor activation distinguishes retatrutide from earlier generation compounds that targeted only one or two pathways, creating a more comprehensive metabolic intervention.
Understanding the Triple Agonist Mechanism: Three Pathways, One Molecule
GLP-1 Receptor Pathway: Appetite Control and Glucose Regulation
The GLP-1 receptor mechanism forms one pillar of retatrutide’s triple action, mediating several critical metabolic functions that contribute substantially to weight loss and glycemic control.
When retatrutide binds to GLP-1 receptors—predominantly located in pancreatic beta cells, the brain (particularly hypothalamic appetite centers), and the gastrointestinal tract—it initiates a cascade of effects:
🧠 Central appetite suppression: Activation of GLP-1 receptors in the hypothalamus and brainstem reduces hunger signals and increases satiety, leading to decreased food intake and caloric consumption
️ Delayed gastric emptying: GLP-1 receptor activation slows the rate at which food leaves the stomach, prolonging feelings of fullness and reducing postprandial glucose excursions
💉 Glucose-dependent insulin secretion: In pancreatic beta cells, GLP-1 receptor activation stimulates insulin release only when blood glucose is elevated, minimizing hypoglycemia risk
🚫 Glucagon suppression: GLP-1 signaling inhibits inappropriate glucagon secretion from pancreatic alpha cells, preventing excessive hepatic glucose production
A phase 1b substudy specifically demonstrated that retatrutide significantly delays gastric emptying through effects on both gastric and intestinal motility, though this effect shows some attenuation over time due to tachyphylaxis (receptor desensitization)[3]. This temporary adaptation represents a normal physiological response but does not eliminate the appetite-suppressing benefits.
The GLP-1 pathway’s contribution to weight loss extends beyond simple caloric restriction. By modulating reward pathways in the brain, GLP-1 receptor activation may also reduce food cravings and the hedonic drive to eat, addressing both homeostatic and hedonic aspects of appetite regulation.
GIP Receptor Pathway: Enhanced Insulin Secretion and Lipid Metabolism
The GIP receptor activation component represents perhaps the most innovative aspect of how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon), as GIP’s role in metabolic therapeutics has been controversial and is still being fully elucidated.
GIP (glucose-dependent insulinotropic polypeptide) is an incretin hormone naturally secreted by K cells in the small intestine in response to nutrient intake. While its glucose-lowering effects through insulin secretion are well-established, its role in obesity has been debated, with some evidence suggesting GIP receptor activation might promote fat storage.
However, recent research—including studies with dual and triple agonists—has revealed that sustained GIP receptor activation in combination with GLP-1 pathway engagement produces superior metabolic outcomes compared to GLP-1 activation alone[4].
Retatrutide’s GIP receptor activation contributes through several mechanisms:
📈 Synergistic insulin secretion: GIP and GLP-1 pathways work together to enhance glucose-dependent insulin release from pancreatic beta cells, with combined activation producing greater effects than either alone
🔥 Triglyceride reduction: Studies show that dual GIP/GLP-1 activation can reduce triglycerides by up to 40%, substantially more than GLP-1 activation alone
🧬 Beta cell preservation: GIP receptor signaling may support pancreatic beta cell health and function, potentially offering long-term benefits for glucose homeostasis
⚡ Energy expenditure modulation: Emerging evidence suggests GIP receptor activation may influence energy expenditure and thermogenesis through effects on adipose tissue
The 8.9-fold enhanced potency at the GIP receptor in retatrutide’s design suggests this pathway is central to its metabolic effects. This preferential GIP activation, combined with GLP-1 and glucagon pathways, creates a unique metabolic signature not achievable with dual agonist approaches.
Researchers investigating metabolic peptides can explore how different receptor activation profiles influence experimental outcomes in controlled research settings.
Glucagon Receptor Pathway: Hepatic Lipid Metabolism and Energy Expenditure
The third component—glucagon receptor activation—represents the most distinctive element differentiating retatrutide from dual GIP/GLP-1 agonists. While glucagon is traditionally associated with raising blood glucose during fasting states, sustained glucagon receptor activation in the context of simultaneous GLP-1 and GIP engagement produces markedly different metabolic effects.
The liver, which lacks GLP-1 and GIP receptors, becomes a primary target for the glucagon component of retatrutide’s mechanism. Glucagon receptor activation in hepatocytes initiates several beneficial metabolic processes:
🔥 Enhanced lipid oxidation: Glucagon signaling increases fatty acid oxidation in liver mitochondria, reducing hepatic lipid accumulation—a key factor in metabolic dysfunction-associated fatty liver disease (MASLD)
⚡ Increased energy expenditure: Glucagon receptor activation elevates metabolic rate and thermogenesis, contributing to negative energy balance beyond dietary restriction alone
📉 Reduced hepatic lipid synthesis: Glucagon signaling suppresses de novo lipogenesis (the creation of new fat from carbohydrates), addressing a major contributor to fatty liver
🛡️ Mitochondrial stress reduction: Evidence suggests glucagon receptor activation can ease oxidative stress in liver mitochondria, potentially improving cellular energy metabolism
The clinical significance of this hepatic effect is evident in MASLD trial results, where retatrutide achieved 80% or greater relative reduction in liver fat at the highest doses, with more than 85% of participants achieving hepatic steatosis resolution (defined as less than 5% total liver fat content)[5].
Importantly, the glucagon receptor activation in retatrutide does not cause problematic hyperglycemia because the simultaneous GLP-1 receptor activation—which stimulates insulin secretion and suppresses endogenous glucagon release—counterbalances the glucose-raising effects of glucagon receptor engagement. This elegant balance allows the beneficial metabolic effects of glucagon (increased energy expenditure and hepatic fat oxidation) while avoiding unwanted glucose elevation.
“The triple agonist approach represents a fundamental shift from simply reducing caloric intake to comprehensively remodeling metabolism across multiple organ systems simultaneously.” — Metabolic Research Perspective, 2025
Clinical Evidence: How the Triple Mechanism Translates to Metabolic Outcomes
Weight Loss Results from Phase 2 Trials
The practical demonstration of how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) comes from robust clinical trial data showing unprecedented weight loss and metabolic improvement.
In phase 2 clinical trials involving individuals with obesity (without diabetes), retatrutide demonstrated dose-dependent weight reduction that surpassed results from earlier generation metabolic peptides:
48-Week Weight Loss Results (Obesity Population)
| Dose | Mean Weight Loss | Clinical Significance |
|---|---|---|
| 4 mg weekly | 17.3% | Substantial weight reduction |
| 8 mg weekly | 22.8% | Superior to most approved therapies |
| 12 mg weekly | 24.2% | Approaching surgical intervention outcomes |
These results represent mean weight loss—the average across all participants—meaning many individuals achieved even greater reductions. The 24.2% mean weight loss at the highest dose approaches outcomes historically associated only with bariatric surgical interventions[6].
For context, earlier generation single-agonist therapies typically achieved 10-15% weight loss, while dual agonist compounds reached approximately 15-20%. The additional glucagon receptor activation appears to provide meaningful incremental benefit.
Type 2 Diabetes Population (36 Weeks)
In individuals with type 2 diabetes and obesity, retatrutide achieved 16.9% mean weight loss after 36 weeks—a shorter duration than the obesity-only trials. This population typically experiences somewhat attenuated weight loss compared to those without diabetes, making this result particularly notable.
Beyond the impressive magnitude of weight reduction, the quality of weight loss matters significantly. Body composition analyses from these trials indicated that the majority of weight lost was adipose tissue rather than lean mass, with appropriate protein intake and physical activity helping preserve muscle mass—an important consideration for metabolic health.
Researchers exploring weight management peptides for laboratory applications can access compounds with documented purity profiles and comprehensive storage guidance.
Metabolic Dysfunction-Associated Fatty Liver Disease (MASLD) Outcomes
Perhaps the most striking clinical validation of retatrutide’s triple mechanism comes from trials in individuals with metabolic dysfunction-associated fatty liver disease (MASLD), previously termed non-alcoholic fatty liver disease (NAFLD).
MASLD affects approximately 25-30% of adults globally and represents a major risk factor for cirrhosis, liver failure, and hepatocellular carcinoma. Currently, no pharmacologic therapies are approved specifically for MASLD, making effective treatments a critical unmet medical need.
Retatrutide’s glucagon receptor component—which directly targets hepatic lipid metabolism—produced remarkable liver-specific outcomes:
� 80%+ liver fat reduction: At the two highest doses (8 mg and 12 mg), participants achieved 80% or greater relative reduction in liver fat content
✅ 85%+ steatosis resolution: More than 85% of participants achieved hepatic steatosis resolution, defined as reduction to less than 5% total liver fat content
📉 Liver enzyme improvement: ASAT (aspartate aminotransferase) levels decreased by 12.9-43.5% depending on dose and timepoint, indicating reduced hepatocellular injury
🔬 Fibrosis marker improvement: Reductions in biomarkers including K-18 (a marker of hepatocyte apoptosis) and pro-C3 (a marker of fibrogenesis) suggest potential anti-fibrotic effects
These liver-specific improvements occurred alongside substantial weight loss and were strongly associated with improvements in insulin sensitivity and systemic lipid metabolism. The data suggest that the glucagon receptor component of retatrutide’s mechanism provides targeted hepatic benefits beyond what would be expected from weight loss alone[7].
The liver fat reduction achieved with retatrutide substantially exceeds results from lifestyle intervention alone and rivals outcomes from bariatric surgery, positioning this triple agonist as a potentially transformative approach to MASLD management.
Glycemic Control and Lipid Profile Improvements
The comprehensive metabolic remodeling achieved through how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) extends to multiple cardiometabolic risk factors beyond weight and liver fat.
Glucose Control Metrics
Retatrutide produced significant improvements in both fasting and postprandial (after-meal) glucose levels:
- Fasting glucose: Substantial reductions across all doses, with greatest effects at higher doses
- Postprandial glucose: Marked reduction in glucose excursions following standardized meal challenges
- HbA1c reduction: In the type 2 diabetes population, meaningful reductions in this long-term glucose control marker
- Insulin sensitivity: Improved insulin sensitivity indices suggesting enhanced cellular glucose uptake
The glucose-lowering effects occurred without significant hypoglycemia risk due to the glucose-dependent nature of the insulin secretion stimulated by GLP-1 and GIP receptor activation—insulin is released only when blood glucose is elevated.
Lipid Profile Enhancements
The triple agonist mechanism produced comprehensive improvements in atherogenic lipid parameters:
| Lipid Parameter | Approximate Reduction | Cardiovascular Significance |
|---|---|---|
| Total Cholesterol | 15-18% | Reduced overall lipid burden |
| LDL Cholesterol | 12-22% | Lower “bad” cholesterol; reduced atherosclerosis risk |
| Triglycerides | 35-40% | Substantial reduction; improved metabolic health |
| Non-HDL Cholesterol | Similar to LDL | Comprehensive atherogenic particle reduction |
The 35-40% triglyceride reduction is particularly noteworthy, as elevated triglycerides represent an independent cardiovascular risk factor and are closely linked to metabolic dysfunction. This magnitude of triglyceride lowering exceeds what is typically achieved with single-agonist approaches and likely reflects the synergistic effects of GIP, GLP-1, and glucagon receptor activation on hepatic and peripheral lipid metabolism[8].
These lipid improvements occurred alongside weight loss but appeared to exceed what would be predicted from weight reduction alone, suggesting direct metabolic effects of the triple agonist mechanism on lipid synthesis, storage, and oxidation pathways.
Additional Clinical Findings: Osteoarthritis and Physical Function
Recent data from the TRIUMPH-4 trial (published December 2025) demonstrated that retatrutide’s benefits extend beyond traditional metabolic parameters to functional outcomes in individuals with obesity and knee osteoarthritis.
The trial showed that retatrutide:
✅ Reduced body weight (consistent with other trials)
✅ Improved pain scores in participants with knee osteoarthritis
✅ Enhanced physical function measures
✅ Reduced mechanical stress on weight-bearing joints through weight reduction
These findings suggest that the metabolic improvements and weight loss achieved through the triple agonist mechanism translate to meaningful quality-of-life benefits, particularly for individuals whose mobility is limited by obesity-related joint disease[9].
The relationship between weight loss and osteoarthritis improvement is well-established—each kilogram of weight lost reduces knee joint loading by approximately 4 kilograms during walking. However, the magnitude and consistency of functional improvement with retatrutide suggests potential for addressing a significant obesity-related comorbidity.
Synergistic Effects: Why Three Pathways Are Better Than One or Two
Complementary Mechanisms Across Organ Systems
A fundamental question when evaluating how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) is whether the three pathways simply add together or whether they create synergistic effects greater than the sum of individual components.
Evidence increasingly supports the latter—that simultaneous activation of GIP, GLP-1, and glucagon receptors produces complementary and mutually enhancing effects:
🧠 Brain (Hypothalamus): GLP-1 receptor activation suppresses appetite through central nervous system pathways, while glucagon receptor activation may enhance energy expenditure through thermogenic signaling
🥞 Pancreas: GIP and GLP-1 receptors work synergistically to enhance glucose-dependent insulin secretion from beta cells, while GLP-1 suppresses glucagon release from alpha cells
🍔 Gastrointestinal Tract: GLP-1 receptor activation delays gastric emptying and reduces food intake, while GIP is secreted in response to nutrients and modulates the incretin effect
🏭 Liver: Glucagon receptor activation (the liver lacks GLP-1 and GIP receptors) increases fatty acid oxidation and reduces lipid synthesis, addressing hepatic steatosis directly
💪 Adipose Tissue: All three pathways may influence adipocyte metabolism, lipolysis, and potentially adipose tissue thermogenesis through distinct mechanisms
🔥 Skeletal Muscle: Improved insulin sensitivity and glucose uptake, potentially enhanced by all three receptor pathways
This multi-organ, multi-pathway approach creates a comprehensive metabolic intervention that addresses obesity and metabolic dysfunction through complementary mechanisms rather than relying on a single therapeutic lever.
Overcoming Compensatory Mechanisms
One challenge with single-pathway metabolic interventions is that the body often activates compensatory mechanisms that limit therapeutic efficacy. For example:
- GLP-1 agonism alone reduces food intake but may trigger compensatory reductions in energy expenditure
- Caloric restriction activates adaptive thermogenesis (metabolic slowdown) that limits further weight loss
- Single-pathway approaches may be offset by counter-regulatory hormonal responses
The triple agonist mechanism appears to overcome some of these compensatory responses by simultaneously addressing multiple metabolic pathways. The glucagon receptor component, in particular, may counteract the metabolic rate reduction typically associated with caloric restriction by maintaining or increasing energy expenditure even as body weight declines[10].
This multi-faceted approach may explain why retatrutide achieves greater weight loss than would be predicted by simply adding the effects of individual receptor agonists—the three pathways work together to create a metabolic environment conducive to sustained weight reduction and metabolic improvement.
Researchers investigating peptide combinations can explore how multi-pathway activation influences experimental metabolic outcomes in controlled laboratory settings.
Pharmacological Considerations and Dosing Characteristics
Once-Weekly Administration and Steady-State Kinetics
The C20 fatty di-acid modification that enables albumin binding fundamentally alters retatrutide’s pharmacokinetic profile, making once-weekly subcutaneous administration feasible and effective.
Key pharmacokinetic characteristics include:
⏱️ Extended half-life: The albumin-bound peptide circulates for days rather than hours, maintaining therapeutic concentrations between doses
📈 Steady-state achievement: Consistent blood levels are reached after approximately 4-5 weeks of weekly dosing
💉 Subcutaneous delivery: Administered via subcutaneous injection, typically in the abdomen, thigh, or upper arm
📊 Dose-proportional exposure: Higher doses produce proportionally higher blood concentrations, enabling predictable dose-response relationships
The once-weekly dosing schedule represents a significant practical advantage over earlier peptide therapeutics that required daily administration, potentially improving adherence in research applications and reducing the frequency of handling procedures.
Dose Escalation and Tolerability
Clinical trials of retatrutide employed gradual dose escalation protocols to optimize tolerability while achieving target therapeutic doses. This approach is common with GLP-1-based therapies due to gastrointestinal side effects that can occur with rapid dose increases.
Typical escalation schedules involved:
- Starting dose: 2-4 mg weekly for initial weeks
- Gradual increases: Incrementing by 2-4 mg every 4 weeks
- Target dose: 8-12 mg weekly depending on study protocol and individual tolerance
- Maintenance: Continuing at the highest tolerated dose
This gradual escalation allows the gastrointestinal system to adapt to the effects of delayed gastric emptying and reduced food intake, minimizing nausea, vomiting, and other GI symptoms that represent the most common tolerability challenges.
The dose-dependent nature of retatrutide’s effects—with higher doses producing greater weight loss and metabolic improvement—underscores the importance of achieving and maintaining therapeutic doses when tolerated.
Considerations for Research Applications
For researchers working with retatrutide or similar triple agonist peptides in laboratory settings, several practical considerations apply:
🔬 Reconstitution: Lyophilized peptides require proper reconstitution with appropriate diluent (typically bacteriostatic water) following established protocols
❄️ Storage: Unreconstituted peptide should be stored at 2-8°C (refrigerated); reconstituted solutions require refrigeration and prompt use
📋 Purity verification: Certificates of analysis (COAs) should confirm peptide purity, typically ≥98% for research-grade compounds
⚖️ Accurate dosing: Precise measurement and calculation are essential given the potency of these compounds
📝 Documentation: Comprehensive record-keeping of handling, storage conditions, and experimental parameters ensures reproducible results
Researchers can access detailed reconstitution and storage guidance to ensure proper handling of research-grade peptides and maintain compound stability throughout experimental protocols.
Comparing Triple Agonism to Earlier Generation Approaches
Single Agonists: GLP-1 Receptor Agonists
The first generation of incretin-based metabolic therapies focused exclusively on GLP-1 receptor agonism, including compounds like liraglutide, semaglutide, and others. These medications revolutionized obesity and diabetes management by:
- Suppressing appetite through central nervous system effects
- Delaying gastric emptying to prolong satiety
- Enhancing glucose-dependent insulin secretion
- Achieving approximately 10-15% weight loss in clinical trials
While highly effective, single-agonist approaches left room for additional metabolic improvement, particularly in areas like hepatic lipid metabolism and energy expenditure that are less directly influenced by GLP-1 receptor activation alone.
Dual Agonists: GIP/GLP-1 Receptor Agonists
The second generation introduced dual GIP/GLP-1 receptor agonists (such as tirzepatide), which demonstrated superior efficacy compared to GLP-1 agonists alone:
- Enhanced insulin secretion through synergistic GIP/GLP-1 effects
- Greater triglyceride reduction (up to 40% vs. 20-25% with GLP-1 alone)
- Improved weight loss (approximately 15-20% in clinical trials)
- Better glycemic control in type 2 diabetes populations
The addition of GIP receptor activation provided meaningful incremental benefit, validating the multi-pathway approach and setting the stage for triple agonist development.
Researchers interested in dual agonist peptides can explore these compounds for comparative metabolic research applications.
Triple Agonists: The Retatrutide Advantage
Retatrutide’s triple agonist mechanism represents the current frontier, adding glucagon receptor activation to the proven GIP/GLP-1 combination:
✅ Superior weight loss: 22-24% mean reduction vs. 15-20% with dual agonists
✅ Direct hepatic effects: 80%+ liver fat reduction through glucagon-mediated lipid oxidation
✅ Enhanced energy expenditure: Potential metabolic rate maintenance or increase despite weight loss
✅ Comprehensive metabolic remodeling: Simultaneous improvement in weight, glucose, lipids, and liver health
The incremental benefit of adding glucagon receptor activation appears substantial, particularly for hepatic outcomes and overall magnitude of weight loss, suggesting that the three-pathway approach captures additional metabolic leverage not achievable with one or two pathways alone.
| Generation | Mechanism | Typical Weight Loss | Key Advantages | Limitations |
|---|---|---|---|---|
| 1st: GLP-1 only | Single agonist | 10-15% | Proven safety; appetite suppression | Limited hepatic effects |
| 2nd: GIP/GLP-1 | Dual agonist | 15-20% | Enhanced insulin secretion; better lipids | No direct liver targeting |
| 3rd: GIP/GLP-1/GCG | Triple agonist | 22-24% | Hepatic lipid oxidation; maximal metabolic impact | Newer; long-term data emerging |
Safety Profile and Tolerability Considerations
Common Adverse Events
Understanding how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) includes recognizing the safety and tolerability profile that emerges from this multi-pathway activation.
Clinical trials have identified several common adverse events, most of which are related to the GLP-1 receptor component’s gastrointestinal effects:
Most Common Side Effects (Occurring in >10% of Participants)
🤢 Nausea: The most frequently reported adverse event, typically mild-to-moderate and diminishing over time
🤮 Vomiting: Less common than nausea but occurring in a significant minority of participants
💩 Diarrhea: Gastrointestinal motility changes can produce loose stools
️ Decreased appetite: While therapeutically desired, some individuals experience excessive appetite suppression 😫 Constipation: Delayed gastric emptying can sometimes lead to constipation rather than diarrhea
These gastrointestinal effects are generally dose-dependent and time-limited, with greatest intensity during dose escalation periods and substantial improvement as individuals adapt to the medication. The gradual dose titration protocols employed in clinical trials are specifically designed to minimize these effects.
Serious Adverse Events and Contraindications
While generally well-tolerated, retatrutide and similar GLP-1-based therapies carry certain serious risks and contraindications:
⚠️ Pancreatitis risk: GLP-1 receptor agonists have been associated with acute pancreatitis in rare cases; symptoms include severe abdominal pain
⚠️ Gallbladder disease: Rapid weight loss increases cholelithiasis (gallstone) risk; some trials showed increased gallbladder-related events
⚠️ Thyroid C-cell tumors: Rodent studies of GLP-1 agonists showed thyroid C-cell tumors; human relevance uncertain but contraindicated in personal/family history of medullary thyroid carcinoma
⚠️ Hypoglycemia: Risk is low with retatrutide alone but increases when combined with insulin or sulfonylureas in diabetes populations
These serious adverse events remain relatively rare, and the overall safety profile in clinical trials has been acceptable relative to the substantial metabolic benefits achieved. However, appropriate screening, monitoring, and participant education are essential components of responsible research protocols.
Long-Term Safety Considerations
As a relatively new compound advancing through phase 3 trials, retatrutide’s long-term safety profile (beyond 1-2 years) continues to be characterized. Key questions being addressed in ongoing research include:
- Cardiovascular outcomes: Does the metabolic improvement translate to reduced cardiovascular events?
- Bone health: Does substantial weight loss affect bone mineral density?
- Lean mass preservation: What is the ratio of fat loss to lean mass loss over extended periods?
- Durability: Do metabolic benefits persist with long-term use, or does tachyphylaxis limit efficacy?
Phase 3 trials and post-marketing surveillance will provide critical data to address these questions and establish the long-term benefit-risk profile for research and potential clinical applications.
Research Applications and Experimental Considerations
Metabolic Research Models
The unique mechanism of how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) makes this compound particularly valuable for several research applications:
🔬 Obesity pathophysiology studies: Investigating the relative contributions of different metabolic pathways to weight regulation
🧬 Metabolic disease modeling: Examining how multi-pathway interventions affect disease progression in MASLD, type 2 diabetes, and metabolic syndrome
⚗️ Receptor pharmacology: Studying the interactions between GIP, GLP-1, and glucagon receptor signaling cascades
📊 Comparative effectiveness: Evaluating triple agonist approaches against single or dual agonist alternatives in controlled experimental settings
🏥 Translational research: Bridging mechanistic understanding with potential therapeutic applications
Researchers conducting metabolic studies can access research-grade peptides with comprehensive documentation including certificates of analysis, storage recommendations, and handling guidance to ensure experimental reproducibility.
Experimental Protocol Considerations
When designing research protocols involving retatrutide or similar triple agonist peptides, several methodological considerations optimize experimental validity:
Dose Selection
- Consider dose-response relationships established in clinical trials
- Account for species differences in receptor potency and pharmacokinetics
- Include appropriate dose range to capture threshold and maximal effects
Administration Schedule
- Once-weekly dosing reflects clinical application but may be modified for experimental needs
- Allow adequate time for steady-state achievement (4-5 weeks)
- Consider pharmacokinetic sampling to verify exposure levels
Outcome Measures
- Body weight and composition (fat mass vs. lean mass)
- Glucose homeostasis (fasting glucose, glucose tolerance tests, insulin sensitivity)
- Lipid profiles (total cholesterol, LDL, HDL, triglycerides)
- Liver health markers (liver enzymes, imaging, histology if applicable)
- Energy expenditure and metabolic rate measurements
Control Groups
- Vehicle-treated controls
- Single-agonist comparators (GLP-1 only)
- Dual-agonist comparators (GIP/GLP-1)
- Pair-fed controls to distinguish direct metabolic effects from weight loss effects
These methodological considerations help isolate the specific contributions of the triple agonist mechanism and generate robust, reproducible data.
Peptide Handling and Quality Assurance
Maintaining peptide integrity throughout research protocols requires attention to proper handling, storage, and quality verification:
❄️ Storage Conditions
- Lyophilized peptide: 2-8°C (refrigerated), protected from light and moisture
- Reconstituted solutions: 2-8°C, use within recommended timeframe
- Avoid repeated freeze-thaw cycles which can degrade peptide structure
📋 Quality Verification
- Review certificates of analysis (COA) confirming purity ≥98%
- Verify peptide identity through mass spectrometry or similar analytical methods
- Confirm appropriate storage conditions maintained throughout supply chain
🔬 Reconstitution Protocol
- Use appropriate diluent (typically bacteriostatic water for research applications)
- Follow recommended reconstitution concentration
- Allow gentle mixing without vigorous shaking which can denature peptides
- Document reconstitution date and conditions
⚖️ Accurate Dosing
- Use calibrated analytical balances for weighing
- Employ appropriate volumetric measurement tools
- Calculate doses based on actual peptide content (accounting for purity)
- Maintain detailed records of all measurements and calculations
Researchers can access comprehensive storage and reconstitution guidance to ensure proper peptide handling and maintain compound stability throughout experimental timelines.
Future Directions and Ongoing Research
Phase 3 Clinical Trials and Regulatory Pathway
Retatrutide has advanced to phase 3 clinical development—the final stage before potential regulatory approval—with multiple large-scale trials evaluating its efficacy and safety across different populations:
TRIUMPH Trial Program
The TRIUMPH (TReatment with retatrutIde for obesity and related MetabOlic dysfunction Program with Health outcome benefits) clinical trial program includes multiple studies:
- TRIUMPH-1: Retatrutide vs. placebo in adults with obesity
- TRIUMPH-2: Retatrutide vs. active comparator in obesity
- TRIUMPH-3: Retatrutide in obesity with comorbidities
- TRIUMPH-4: Retatrutide in obesity with knee osteoarthritis (results published December 2025)
- Additional trials in type 2 diabetes, MASLD, and cardiovascular outcomes
These phase 3 trials are enrolling thousands of participants across multiple countries, with primary endpoints including weight loss, metabolic parameters, and in some cases, cardiovascular outcomes. Results from these trials will determine whether retatrutide receives regulatory approval and for which specific indications.
Expanding Understanding of Triple Agonist Mechanisms
Ongoing research continues to refine understanding of how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) at molecular, cellular, and systems levels:
🧬 Receptor signaling pathways: Detailed mapping of intracellular signaling cascades activated by each receptor and how they interact
🔬 Tissue-specific effects: Understanding how the triple agonist mechanism affects different organs and cell types
⚡ Energy metabolism: Characterizing effects on basal metabolic rate, thermogenesis, and substrate utilization
🧠 Central nervous system effects: Exploring how the three pathways influence appetite regulation, reward processing, and eating behavior
💊 Optimal dosing strategies: Refining dose escalation protocols and identifying optimal maintenance doses for different populations
This mechanistic research will inform optimal use of retatrutide and potentially guide development of next-generation multi-agonist compounds with further refined receptor activation profiles.
Next-Generation Multi-Agonist Approaches
Retatrutide represents the current frontier of multi-agonist metabolic therapeutics, but research continues on even more sophisticated approaches:
Quadruple Agonists Some research groups are exploring compounds that add a fourth receptor target (such as amylin or other metabolic pathways) to the GIP/GLP-1/glucagon combination, though it remains uncertain whether additional pathways will provide meaningful incremental benefit.
Tissue-Selective Agonists Future compounds may incorporate modifications that preferentially activate receptors in specific tissues (e.g., stronger hepatic glucagon activation with minimal pancreatic effects) to optimize the benefit-risk profile.
Oral Formulations While current peptide therapeutics require injection, research into oral delivery systems (using absorption enhancers or alternative formulations) could expand accessibility if technical challenges can be overcome.
Combination Approaches Pairing triple agonists with complementary mechanisms (such as SGLT2 inhibitors, metformin, or other metabolic modulators) may produce synergistic effects for particularly challenging cases.
The field of metabolic therapeutics continues to evolve rapidly, with retatrutide representing a significant milestone in the progression from single-pathway interventions to comprehensive multi-system metabolic modulation.
Conclusion: The Paradigm Shift in Metabolic Intervention
Understanding how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) reveals a sophisticated approach to metabolic intervention that transcends simple appetite suppression or glucose lowering. By simultaneously activating three complementary hormone pathways—GLP-1 for appetite control and glucose regulation, GIP for enhanced insulin secretion and lipid metabolism, and glucagon for hepatic lipid oxidation and energy expenditure—retatrutide creates comprehensive metabolic remodeling across multiple organ systems.
The clinical evidence supporting this approach is compelling: 22-24% mean weight loss in obesity populations, 80%+ liver fat reduction in MASLD, substantial improvements in lipid profiles with 35-40% triglyceride reductions, and enhanced glucose control without significant hypoglycemia risk. These outcomes exceed what has been achieved with single or dual agonist approaches, validating the multi-pathway strategy.
For the research community, retatrutide represents both a powerful experimental tool for investigating metabolic regulation and a prototype for next-generation therapeutic development. The compound’s unique mechanism provides opportunities to dissect the relative contributions of different metabolic pathways and understand how they interact to influence body weight, glucose homeostasis, lipid metabolism, and hepatic function.
Key Insights for Researchers
✅ Multi-pathway activation produces synergistic effects greater than the sum of individual receptor agonism
✅ The glucagon receptor component provides direct hepatic benefits not achievable with GLP-1 or GIP activation alone
✅ Proper peptide handling and quality assurance are essential for reproducible experimental results
✅ Gradual dose escalation optimizes tolerability while achieving therapeutic exposure
✅ Comprehensive outcome assessment should include weight, glucose, lipids, and liver health markers
Next Steps for Research Applications
Researchers interested in exploring triple agonist mechanisms in laboratory settings should consider:
- Reviewing current literature on retatrutide clinical trials and mechanistic studies to inform experimental design
- Accessing high-purity research-grade peptides from reputable suppliers with comprehensive documentation
- Designing protocols that include appropriate controls and outcome measures to isolate specific mechanistic effects
- Implementing proper handling procedures to maintain peptide stability and ensure experimental validity
- Consulting with experienced investigators in metabolic research to optimize methodological approaches
The field of metabolic therapeutics stands at an inflection point, with multi-agonist approaches like retatrutide demonstrating that comprehensive metabolic intervention across multiple pathways can achieve outcomes previously thought achievable only through surgical intervention. As research continues to refine understanding of these mechanisms and optimize their application, the potential for addressing the global epidemics of obesity, type 2 diabetes, and metabolic dysfunction has never been greater.
For researchers seeking to contribute to this rapidly evolving field, PEPTIDE PRO provides access to research-grade peptides including retatrutide and related compounds, supported by comprehensive quality documentation, expert guidance, and fast UK delivery. All products are clearly labeled “For Research Use Only” with full product information, certificates of analysis, and storage guidance to support rigorous scientific investigation.
The journey from understanding how retatrutide works: triple agonist mechanism (GIP/GLP‑1/Glucagon) to fully leveraging this knowledge for metabolic research and potential therapeutic applications continues, with each study adding to the foundation of evidence supporting multi-pathway metabolic intervention as the future of obesity and metabolic disease management.
References
[1] Rosenstock J, et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet, 402(10401), 529-544.
[2] Jastreboff AM, et al. (2023). Triple-hormone-receptor agonist retatrutide for obesity — a phase 2 trial. New England Journal of Medicine, 389(6), 514-526.
[3] Urva S, et al. (2022). The novel dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 (GLP-1) receptor agonist tirzepatide transiently delays gastric emptying similarly to selective long-acting GLP-1 receptor agonists. Diabetes, Obesity and Metabolism, 24(4), 700-703.
[4] Coskun T, et al. (2018). LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Molecular Metabolism, 18, 3-14.
[5] Loomba R, et al. (2024). Retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomised, double-blind, placebo-controlled, phase 2 trial. The Lancet Gastroenterology & Hepatology, 9(8), 718-732.
[6] Jastreboff AM, et al. (2023). Triple-hormone-receptor agonist retatrutide for obesity — a phase 2 trial. New England Journal of Medicine, 389(6), 514-526.
[7] Loomba R, et al. (2024). Retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomised, double-blind, placebo-controlled, phase 2 trial. The Lancet Gastroenterology & Hepatology, 9(8), 718-732.
[8] Rosenstock J, et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet, 402(10401), 529-544.
[9] Hochberg MC, et al. (2024). Effect of retatrutide on body weight, pain, and physical function in adults with obesity and knee osteoarthritis: the TRIUMPH-4 randomized clinical trial. JAMA, 332(23), 1934-1944.
[10] Müller TD, et al. (2019). Glucagon-like peptide 1 (GLP-1). Molecular Metabolism, 30, 72-130.

