
The landscape of metabolic research has transformed dramatically with the emergence of novel peptide therapeutics. Among these groundbreaking compounds, tirzepatide and semaglutide have captured significant attention within the scientific community for their distinct mechanisms and potential applications in metabolic research. Understanding Tirzepatide vs Semaglutide: Which Is Right for You? requires examining their molecular structures, receptor interactions, and documented research outcomes. Both peptides represent advanced GLP-1 receptor agonist technology, yet their differences may influence research protocol selection and experimental design considerations.
Key Takeaways
✅ Dual vs Single Action: Tirzepatide functions as a dual GIP/GLP-1 receptor agonist, while semaglutide operates solely on GLP-1 receptors, creating distinct metabolic pathway interactions in research models
✅ Research Efficacy Differences: Clinical research data suggests tirzepatide demonstrates superior weight reduction outcomes (15-22.5%) compared to semaglutide (10-15%) in comparable study durations[1]
✅ Dosing Protocols Vary: Tirzepatide typically requires weekly administration starting at 2.5mg with gradual titration, whereas semaglutide follows similar weekly schedules but with different dosage escalation patterns
✅ Side Effect Profiles: Both peptides share gastrointestinal-related adverse events in research subjects, though intensity and frequency may differ based on dosage and individual response characteristics
✅ Research Applications: Selection between these peptides depends on specific research objectives, desired metabolic outcomes, and experimental protocol requirements in laboratory settings
Understanding the Fundamentals: GLP-1 Receptor Agonists in Research
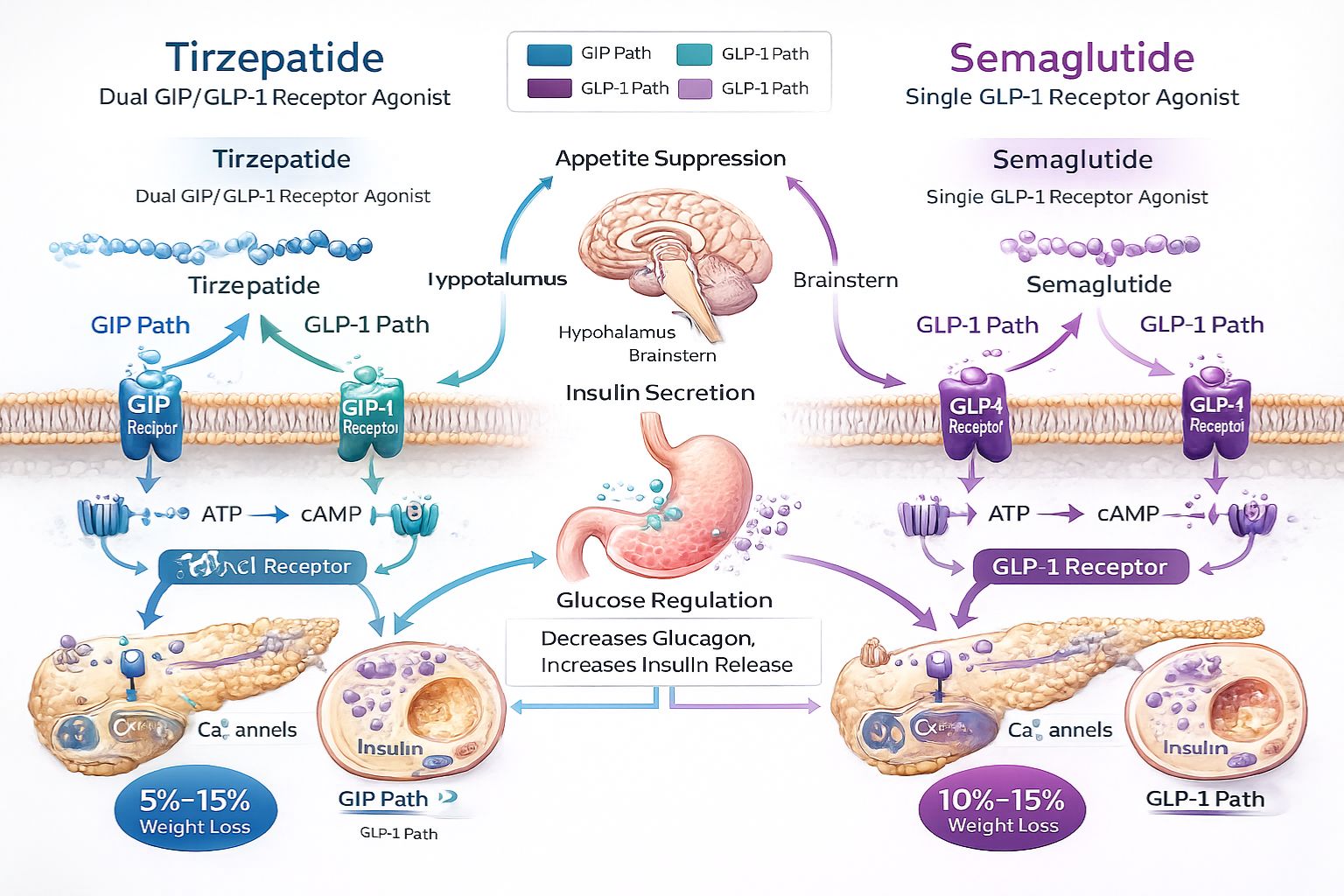
The foundation of comprehending Tirzepatide vs Semaglutide: Which Is Right for You? begins with understanding glucagon-like peptide-1 (GLP-1) receptor agonist technology. These peptides mimic naturally occurring incretin hormones that regulate glucose metabolism, insulin secretion, and appetite signaling pathways in biological systems.
What Are GLP-1 Receptor Agonists?
GLP-1 receptor agonists represent a class of peptide compounds engineered to activate specific cellular receptors involved in metabolic regulation. When these peptides bind to GLP-1 receptors located on pancreatic beta cells, intestinal tissue, and central nervous system regions, they initiate cascading biochemical responses that influence:
- Insulin secretion in glucose-dependent mechanisms
- Glucagon suppression to reduce hepatic glucose output
- Gastric emptying rates affecting nutrient absorption
- Appetite regulation through hypothalamic signaling pathways
- Energy expenditure and thermogenic processes
Research-grade peptides like those available from PEPTIDE PRO enable controlled investigation of these mechanisms in laboratory environments. The purity and consistency of research compounds directly impact experimental reproducibility and data reliability.
The Evolution from Single to Dual Agonists
Semaglutide represents the refined evolution of single-target GLP-1 receptor agonist technology, demonstrating enhanced receptor binding affinity and extended half-life compared to earlier compounds. Its molecular modifications include albumin-binding side chains that prolong circulation time and resist enzymatic degradation[2].
Tirzepatide introduces an innovative dual-agonist approach, simultaneously activating both GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) receptors. This dual mechanism creates synergistic metabolic effects that distinguish it from single-agonist predecessors. The GIP component contributes additional metabolic benefits including:
- Enhanced insulin secretion beyond GLP-1 activation alone
- Potential improvements in lipid metabolism
- Additional effects on adipose tissue function
- Complementary appetite suppression mechanisms
Understanding these fundamental differences provides essential context for evaluating Tirzepatide vs Semaglutide: Which Is Right for You? in specific research applications.
Mechanism of Action: How Tirzepatide and Semaglutide Work Differently
The mechanistic distinctions between these peptides represent critical considerations when determining Tirzepatide vs Semaglutide: Which Is Right for You? for particular research protocols.
Semaglutide: Selective GLP-1 Receptor Activation
Semaglutide demonstrates approximately 94% amino acid sequence homology with native human GLP-1, with strategic modifications enhancing pharmacokinetic properties[3]. Its mechanism centers exclusively on GLP-1 receptor activation, producing:
Pancreatic Effects:
- Glucose-dependent insulin secretion enhancement
- Suppression of inappropriate glucagon release
- Potential beta-cell preservation effects in research models
Central Nervous System Effects:
- Appetite reduction through hypothalamic pathway activation
- Enhanced satiety signaling
- Potential effects on reward pathway modulation
Gastrointestinal Effects:
- Delayed gastric emptying rates
- Modified nutrient absorption timing
- Altered gut hormone secretion patterns
The selective nature of semaglutide provides researchers with a focused tool for investigating GLP-1-specific pathways without confounding variables from additional receptor interactions.
Tirzepatide: Dual GIP/GLP-1 Receptor Agonism
Tirzepatide’s dual-agonist architecture creates more complex metabolic interactions through simultaneous GIP and GLP-1 receptor engagement. The GIP component adds distinctive mechanisms including:
Enhanced Insulin Response: The combined GIP and GLP-1 activation produces synergistic insulin secretion that exceeds either pathway independently. Research suggests this dual stimulation may achieve superior glycemic control in metabolic studies[4].
Adipose Tissue Modulation: GIP receptors expressed on adipocytes contribute to lipid metabolism regulation. Tirzepatide’s GIP activity may influence:
- Adipocyte differentiation processes
- Lipid storage and mobilization dynamics
- Adiponectin and leptin secretion patterns
Unique Metabolic Signaling: The integration of dual receptor pathways creates distinctive metabolic signatures not observed with single-agonist compounds, offering researchers novel investigation opportunities.
For laboratories seeking high-purity research peptides with comprehensive documentation, understanding these mechanistic differences guides appropriate compound selection for specific experimental objectives.
Clinical Research Data: Comparing Efficacy Outcomes
Evaluating Tirzepatide vs Semaglutide: Which Is Right for You? requires examining published research data from controlled clinical investigations.
Weight Reduction Research Findings
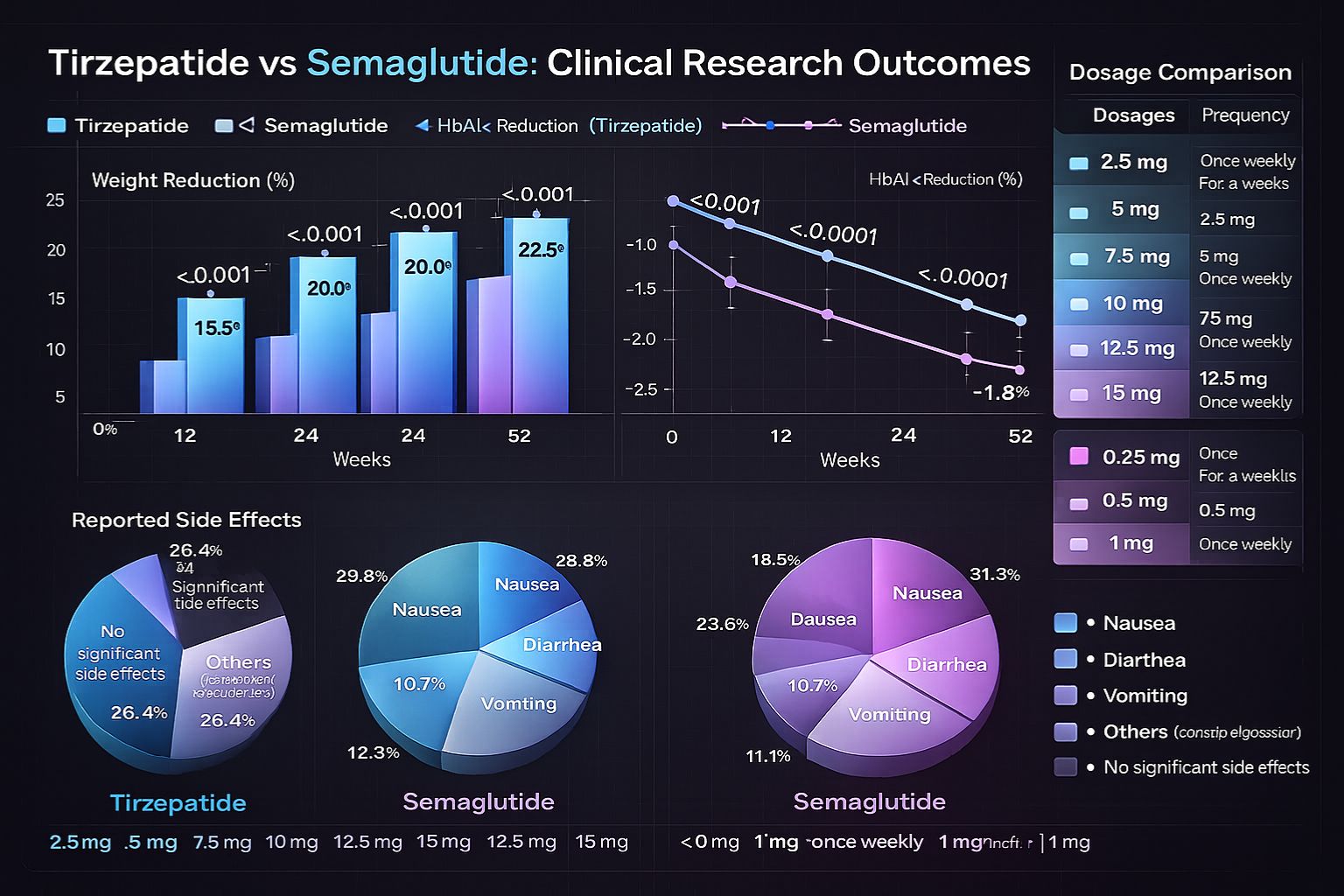
Multiple large-scale clinical trials have documented weight reduction outcomes with both peptides:
| Study Duration | Tirzepatide Results | Semaglutide Results |
|---|---|---|
| 12 weeks | 7.6-9.5% reduction | 5.9-6.7% reduction |
| 24 weeks | 12.4-15.0% reduction | 9.6-10.9% reduction |
| 40 weeks | 15.0-20.9% reduction | 12.4-14.9% reduction |
| 72 weeks | 15.0-22.5% reduction | 10.9-15.8% reduction |
The SURMOUNT-1 trial demonstrated tirzepatide achieving up to 22.5% body weight reduction at the 15mg dose over 72 weeks, while the STEP trials showed semaglutide achieving approximately 14.9% reduction at 2.4mg dosing[5][6]. These differences reflect both the dual-agonist mechanism and dosing protocols employed.
Glycemic Control Parameters
Beyond weight metrics, both peptides demonstrate significant effects on glucose regulation markers:
HbA1c Reduction:
- Tirzepatide: 1.87-2.46% reduction from baseline
- Semaglutide: 1.62-1.86% reduction from baseline
Fasting Glucose:
- Tirzepatide: 39-57 mg/dL reduction
- Semaglutide: 28-41 mg/dL reduction
These glycemic improvements occur through complementary mechanisms including enhanced insulin secretion, glucagon suppression, and improved insulin sensitivity in peripheral tissues.
Cardiovascular and Metabolic Markers
Research investigations have documented additional metabolic effects including:
- Blood pressure reductions in both systolic and diastolic measurements
- Lipid profile improvements including triglyceride and HDL modifications
- Inflammatory marker changes such as C-reactive protein levels
- Liver enzyme improvements suggesting hepatic metabolic benefits
The comprehensive metabolic effects extend beyond primary outcome measures, providing researchers with multiple investigation angles when selecting between compounds for specific protocols.
Dosing Protocols and Administration Considerations
Understanding Tirzepatide vs Semaglutide: Which Is Right for You? includes evaluating practical administration factors that influence research protocol design.
Tirzepatide Dosing Schedules
Standard tirzepatide research protocols typically follow gradual dose escalation:
Initial Phase:
- Starting dose: 2.5 mg weekly
- Duration: 4 weeks minimum
- Purpose: Tolerance establishment and adverse event monitoring
Escalation Phase:
- Incremental increases: 2.5 mg steps
- Available doses: 2.5, 5, 7.5, 10, 12.5, 15 mg
- Titration intervals: 4-week periods between increases
Maintenance Phase:
- Target doses: 5-15 mg weekly based on research objectives
- Administration: Subcutaneous injection
- Timing: Consistent weekly schedule regardless of meals
Researchers can access various tirzepatide formulations suitable for different experimental designs and dosing requirements.
Semaglutide Dosing Protocols
Semaglutide follows similar weekly administration with distinct dosing parameters:
Initiation:
- Starting dose: 0.25 mg weekly
- Duration: 4 weeks
- Purpose: GI tolerance development
Dose Advancement:
- Standard progression: 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg
- Escalation timing: 4-week intervals
- Flexibility: Dose holds possible if tolerance issues emerge
Therapeutic Range:
- Research doses: 0.5-2.4 mg weekly
- Administration route: Subcutaneous
- Injection sites: Abdomen, thigh, or upper arm rotation
Both peptides require consistent weekly administration schedules. High-purity semaglutide from verified suppliers ensures experimental consistency across research timeframes.
Reconstitution and Storage Requirements
Proper handling of lyophilized peptides maintains compound integrity:
Pre-Reconstitution:
- Storage temperature: 2-8°C refrigeration
- Protection: Light-resistant containers
- Shelf stability: Manufacturer specifications apply
Post-Reconstitution:
- Storage: Refrigerated 2-8°C
- Duration: Use within specified timeframes
- Handling: Aseptic technique throughout
Administration Preparation:
- Diluent: Bacteriostatic water for research applications
- Concentration calculations: Based on experimental requirements
- Documentation: Detailed reconstitution records essential
Research facilities should establish standard operating procedures for peptide handling that align with proper storage and reconstitution guidelines to maintain compound quality throughout experimental periods.
Side Effect Profiles: What Research Has Documented
Evaluating Tirzepatide vs Semaglutide: Which Is Right for You? requires understanding documented adverse events observed in clinical research settings.
Common Gastrointestinal Effects
Both peptides demonstrate similar gastrointestinal-related adverse events, though frequency and severity may differ:
Nausea:
- Tirzepatide: 20-40% incidence depending on dose
- Semaglutide: 20-44% incidence across titration phases
- Timing: Most common during dose escalation periods
- Management: Gradual titration reduces severity
Vomiting:
- Tirzepatide: 8-13% of research subjects
- Semaglutide: 9-24% depending on dose level
- Pattern: Typically decreases with continued exposure
Diarrhea:
- Tirzepatide: 13-23% incidence
- Semaglutide: 9-20% across studies
- Duration: Often transient, resolving within weeks
Constipation:
- Tirzepatide: 6-11% of subjects
- Semaglutide: 5-11% reported
- Mechanism: Related to delayed gastric emptying
Metabolic and Systemic Effects
Beyond gastrointestinal events, research has documented:
Hypoglycemia:
- Risk increases when combined with insulin or sulfonylureas
- Standalone use shows minimal hypoglycemic events
- Glucose-dependent mechanisms provide protective effects
Cardiovascular Considerations:
- Heart rate increases: 2-6 beats per minute average
- Blood pressure effects: Generally reductions observed
- Cardiovascular outcome trials: Ongoing investigations
Pancreatic Concerns:
- Pancreatitis: Rare but documented cases
- Lipase elevations: Monitored in research protocols
- Gallbladder effects: Cholelithiasis risk with rapid weight loss
Comparative Tolerability
Research suggests subtle differences in tolerability profiles:
- Discontinuation rates due to adverse events range 4-7% for tirzepatide versus 4-11% for semaglutide in major trials[7]
- Dose-dependent effects show higher doses correlate with increased adverse event frequency for both compounds
- Adaptation patterns indicate most gastrointestinal effects diminish after 4-8 weeks of consistent dosing
Researchers must incorporate adverse event monitoring into experimental protocols, with appropriate subject screening and ongoing assessment procedures ensuring research integrity and subject welfare.
Research Applications: Selecting the Right Peptide for Your Study
Determining Tirzepatide vs Semaglutide: Which Is Right for You? ultimately depends on specific research objectives and experimental design requirements.
When Tirzepatide May Be Preferred
Research scenarios where tirzepatide’s dual-agonist mechanism offers advantages include:
🔬 Maximal Metabolic Effect Studies: Investigations requiring the most substantial metabolic changes may benefit from tirzepatide’s enhanced efficacy profile. Research focused on:
- Maximum weight reduction potential
- Comprehensive metabolic syndrome investigations
- Dual-pathway mechanism studies
- Comparative efficacy research
🔬 GIP Pathway Investigations: Studies specifically examining GIP receptor biology or dual-agonist synergy necessitate tirzepatide’s unique mechanism:
- GIP/GLP-1 interaction research
- Adipose tissue signaling studies
- Novel receptor crosstalk investigations
🔬 Dose-Response Characterization: Tirzepatide’s wider dosing range (2.5-15 mg) enables more granular dose-response curve development compared to semaglutide’s range.
When Semaglutide May Be Preferred
Specific research contexts where semaglutide offers distinct advantages:
🔬 Isolated GLP-1 Pathway Research: Studies requiring selective GLP-1 receptor activation without GIP confounding:
- Pure GLP-1 mechanism investigations
- Receptor-specific signaling pathway studies
- Comparative single-agonist research
🔬 Established Research Protocols: Semaglutide’s longer market presence provides:
- More extensive published literature for comparison
- Established research methodologies
- Comprehensive safety databases
🔬 Cost-Efficiency Considerations: Research budgets may influence compound selection, with various peptide options available at different price points depending on purity requirements and quantities needed.
Combination and Comparative Research
Some research designs incorporate both peptides:
- Head-to-head comparisons examining relative efficacy and mechanisms
- Sequential protocols investigating response patterns to different agonist types
- Combination studies with other metabolic compounds
- Mechanism dissection separating GLP-1 versus dual-agonist effects
Laboratories conducting comprehensive metabolic research may maintain both compounds in their inventory, selecting based on specific experimental requirements for each protocol.
Practical Research Considerations: Cost, Availability, and Quality
Beyond mechanistic differences, practical factors influence Tirzepatide vs Semaglutide: Which Is Right for You? decisions in research settings.
Research-Grade Purity Requirements
High-quality metabolic research demands exceptional peptide purity:
Purity Standards:
- Research-grade specifications: ≥98% purity typical
- HPLC verification: Chromatographic analysis documentation
- Mass spectrometry confirmation: Molecular weight verification
- Certificates of Analysis (COA): Batch-specific quality documentation
Quality Implications:
- Experimental reproducibility depends on consistent purity
- Impurities may introduce confounding variables
- Degradation products affect dose accuracy
PEPTIDE PRO provides research-grade peptides with comprehensive quality documentation, ensuring experimental integrity across research applications.
Regulatory and Compliance Factors
Research peptide procurement requires attention to regulatory frameworks:
Research-Only Designation:
- Clear labeling: “For Research Use Only”
- Prohibited uses: Not for human or animal consumption
- Documentation: Institutional research protocols required
- Compliance: Adherence to local research regulations
Institutional Requirements:
- Ethics committee approvals for applicable research
- Proper storage and handling protocols
- Chain of custody documentation
- Waste disposal procedures for research compounds
Supply Chain Reliability
Consistent research requires dependable peptide availability:
Supplier Considerations:
- Stock consistency: Avoiding research interruptions
- Delivery timelines: Same-day dispatch options for UK researchers
- International shipping: Temperature-controlled transport
- Customer support: Technical assistance for reconstitution and handling
Inventory Management:
- Batch consistency: Ordering sufficient quantities from single batches
- Expiration tracking: Proper rotation and usage timelines
- Backup supplies: Contingency planning for critical experiments
Establishing relationships with reliable research peptide suppliers minimizes experimental disruptions and ensures consistent compound quality throughout extended research programs.
Future Research Directions and Emerging Peptides
The landscape of metabolic peptide research continues evolving beyond the current Tirzepatide vs Semaglutide: Which Is Right for You? comparison.
Next-Generation Multi-Agonists
Pharmaceutical research has progressed toward triple-agonist compounds incorporating:
- GLP-1/GIP/Glucagon agonism: Retatrutide and similar compounds
- Enhanced efficacy profiles: Preliminary research suggesting superior metabolic effects
- Expanded mechanism complexity: Additional research opportunities and challenges
These emerging peptides may eventually shift the comparison framework as research data accumulates.
Oral Delivery Formulations
Traditional subcutaneous administration faces potential alternatives:
- Oral semaglutide: Already developed with absorption enhancer technology
- Oral tirzepatide: Under investigation in research settings
- Novel delivery systems: Transdermal and other routes in early research
Delivery route innovations may influence future peptide selection based on research protocol requirements.
Combination Therapy Research
Investigating peptide combinations with complementary mechanisms:
- GLP-1 agonists + SGLT2 inhibitors: Synergistic metabolic effects
- Dual agonists + metformin: Combined pathway targeting
- Peptide combinations: Multiple incretin-based compounds
Combination research expands understanding of metabolic pathway interactions and potential synergistic benefits.
Personalized Medicine Applications
Future research may identify biomarkers predicting differential responses:
- Genetic polymorphisms: Affecting receptor expression or signaling
- Microbiome profiles: Influencing peptide metabolism and effects
- Metabolic phenotypes: Predicting optimal compound selection
Precision medicine approaches may eventually provide more sophisticated frameworks for determining Tirzepatide vs Semaglutide: Which Is Right for You? based on individual biological characteristics.
Conclusion: Making an Informed Research Decision
The question of Tirzepatide vs Semaglutide: Which Is Right for You? lacks a universal answer, as optimal selection depends entirely on specific research objectives, experimental design parameters, and practical considerations unique to each laboratory setting.
Key Decision Factors Summarized
Choose Tirzepatide When:
- ✅ Maximum metabolic effect magnitude is the primary research objective
- ✅ Dual GIP/GLP-1 mechanism investigation is specifically required
- ✅ Research protocols can accommodate the wider dosing range
- ✅ Novel dual-agonist biology represents the core research question
Choose Semaglutide When:
- ✅ Isolated GLP-1 pathway research without GIP confounding is needed
- ✅ Extensive published literature for comparison is valuable
- ✅ Established research protocols and methodologies are preferred
- ✅ Cost-efficiency within research budgets is a priority consideration
Consider Both When:
- ✅ Comparative mechanism research is the experimental focus
- ✅ Head-to-head efficacy studies are being conducted
- ✅ Comprehensive metabolic pathway investigations require multiple tools
- ✅ Sequential protocol designs examine differential responses
Actionable Next Steps for Researchers
1. Define Research Objectives Clearly Articulate specific hypotheses, outcome measures, and mechanistic questions that will guide peptide selection aligned with experimental goals.
2. Review Current Literature Thoroughly Examine published research using both peptides in contexts similar to planned investigations, identifying methodological precedents and outcome benchmarks.
3. Assess Practical Requirements Evaluate budget constraints, timeline requirements, regulatory compliance needs, and institutional capabilities for peptide handling and storage.
4. Source High-Quality Research Compounds Partner with reputable suppliers providing research-grade peptides with comprehensive quality documentation. Contact PEPTIDE PRO for information about tirzepatide, semaglutide, and other research peptides with verified purity and fast UK delivery.
5. Establish Robust Protocols Develop detailed standard operating procedures for reconstitution, storage, administration, and adverse event monitoring that ensure experimental consistency and research integrity.
6. Plan for Data Analysis Design statistical approaches and outcome measurement strategies before initiating experiments, ensuring appropriate power calculations and analytical frameworks.
The Broader Research Context
Both tirzepatide and semaglutide represent remarkable advances in metabolic peptide research, offering investigators powerful tools for examining complex biological pathways involved in energy regulation, glucose metabolism, and body weight control. The dual-agonist innovation of tirzepatide expands research possibilities beyond single-target approaches, while semaglutide’s refined GLP-1 mechanism provides focused investigation capabilities.
The ongoing evolution of this peptide class—with triple-agonists, novel delivery systems, and combination approaches emerging—promises continued expansion of research opportunities. Laboratories equipped with both compounds gain maximum flexibility for addressing diverse research questions across the metabolic research spectrum.
Ultimately, the decision between these peptides should emerge from careful consideration of research objectives, mechanistic requirements, and practical constraints. Neither compound is universally superior; rather, each offers distinct advantages for specific research applications. By matching peptide characteristics to experimental requirements, researchers optimize their investigations and contribute meaningful data to the expanding understanding of metabolic regulation.
For research facilities seeking premium research-grade peptides with exceptional purity, comprehensive documentation, and reliable UK delivery, establishing supplier relationships that prioritize quality and service ensures uninterrupted research progress and experimental success.
References
[1] Jastreboff AM, et al. (2022). “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine, 387(3), 205-216.
[2] Lau J, et al. (2015). “Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide.” Journal of Medicinal Chemistry, 58(18), 7370-7380.
[3] Wilding JPH, et al. (2021). “Once-Weekly Semaglutide in Adults with Overweight or Obesity.” New England Journal of Medicine, 384(11), 989-1002.
[4] Frias JP, et al. (2021). “Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes.” New England Journal of Medicine, 385(6), 503-515.
[5] Jastreboff AM, et al. (2022). “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine, 387(3), 205-216.
[6] Garvey WT, et al. (2022). “Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial.” Nature Medicine, 28(10), 2083-2091.
[7] Nauck MA, et al. (2023). “Cardiovascular Actions of GLP-1-Based Therapies: Observations from Clinical Trials and Mechanistic Studies.” Cardiovascular Research, 119(3), 649-665.
