
The landscape of weight management research has transformed dramatically with the emergence of GLP-1 receptor agonists and dual-action peptides. As researchers and clinicians evaluate Tirzepatide vs Wegovy: Cost, Dosing & Results, understanding the fundamental differences between these two groundbreaking compounds becomes essential for informed decision-making. Both medications represent significant advances in metabolic research, yet they operate through distinct mechanisms and deliver markedly different outcomes in clinical trials. This comprehensive analysis examines the critical distinctions that separate tirzepatide from semaglutide (Wegovy’s active ingredient), providing clarity on efficacy, administration protocols, and economic considerations.
Key Takeaways
- Tirzepatide demonstrates superior weight reduction compared to Wegovy, with clinical trials showing 20.2% average body weight loss versus 13.7% for semaglutide—nearly 50% more weight loss[2][5]
- Both compounds require weekly administration, making dosing frequency identical, though escalation schedules and maximum doses differ significantly[1][2]
- Mechanism of action sets them apart: Tirzepatide activates dual GLP-1 and GIP receptor pathways, while Wegovy targets only GLP-1 receptors[1]
- Cost considerations vary substantially based on formulation, dosing requirements, and regional availability, with research-grade peptides offering alternatives for laboratory applications
- Head-to-head trials confirm tirzepatide’s advantage, with 2025 NEJM research providing direct comparative evidence of superior efficacy[5]
Understanding the Mechanisms: How Tirzepatide and Wegovy Work Differently
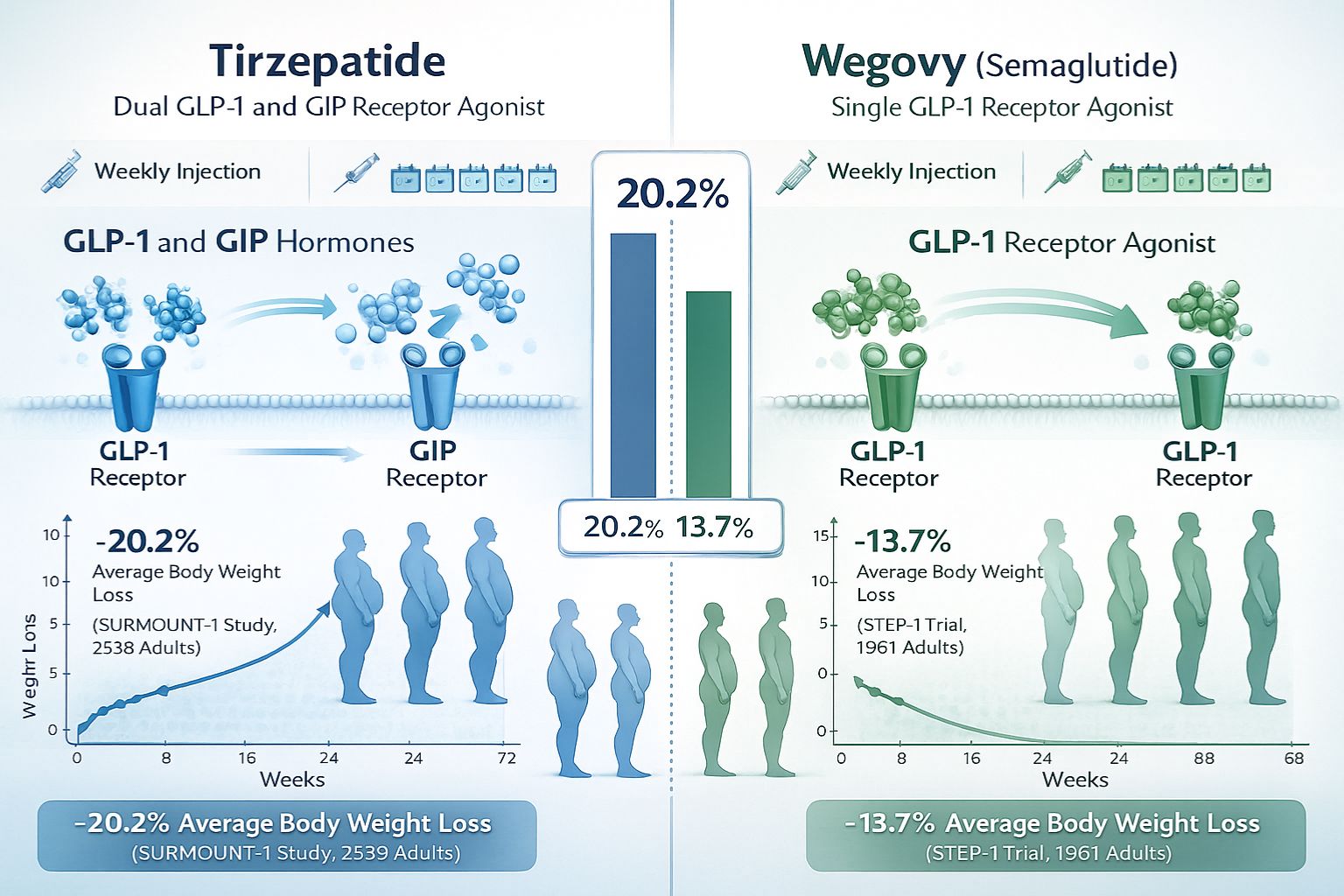
The fundamental distinction in Tirzepatide vs Wegovy: Cost, Dosing & Results begins at the molecular level. Tirzepatide functions as a dual GLP-1 and GIP receptor agonist, meaning it activates two distinct hormone pathways involved in glucose regulation and appetite control[1]. This dual-action mechanism represents a significant advancement in peptide research, as it targets both glucagon-like peptide-1 (GLP-1) receptors and glucose-dependent insulinotropic polypeptide (GIP) receptors simultaneously.
In contrast, semaglutide (the active compound in Wegovy) operates as a single GLP-1 receptor agonist[1]. While highly effective in its own right, this single-pathway activation provides a more limited scope of metabolic influence compared to tirzepatide’s comprehensive approach.
The GLP-1 Pathway
Both compounds share the ability to activate GLP-1 receptors, which trigger several beneficial metabolic responses:
- Appetite suppression through central nervous system signaling
- Delayed gastric emptying, promoting satiety
- Enhanced insulin secretion in response to glucose
- Reduced glucagon release, preventing excessive glucose production
The Additional GIP Component
What sets tirzepatide apart is its activation of GIP receptors, which contribute additional metabolic benefits:
- Enhanced insulin sensitivity in peripheral tissues
- Improved lipid metabolism and fat oxidization
- Potential neuroprotective effects under investigation
- Synergistic action with GLP-1 pathways for amplified results
This dual mechanism explains why tirzepatide consistently demonstrates superior weight reduction outcomes in comparative studies. Researchers at PEPTIDE PRO recognize the importance of understanding these mechanistic differences when selecting compounds for metabolic research applications.
“The dual-agonist approach of tirzepatide represents a paradigm shift in how we conceptualize metabolic intervention at the peptide level. By engaging two complementary pathways simultaneously, we observe synergistic effects that exceed what single-pathway agonists can achieve.” — Leading Metabolic Research Perspective
Clinical Trial Results: Comparing Weight Loss Efficacy
When evaluating Tirzepatide vs Wegovy: Cost, Dosing & Results, the clinical evidence provides compelling insights into relative efficacy. The data from multiple large-scale trials reveals consistent patterns of superior performance for tirzepatide across diverse populations.
Landmark Trial Findings
Tirzepatide (Zepbound/Mounjaro) achieved an average weight loss of 20.2% in pivotal clinical trials involving participants with obesity[2][5]. This represents a substantial reduction in body weight, with some participants in higher dose groups experiencing even greater reductions approaching 25% of baseline weight[1].
Semaglutide (Wegovy), while still highly effective, demonstrated an average weight loss of 13.7% in comparable study populations[2][5]. This translates to tirzepatide users losing approximately 50% more weight on average compared to those receiving semaglutide.
Head-to-Head Comparison Data
A groundbreaking 2025 study published in the New England Journal of Medicine provided the first direct head-to-head comparison between these compounds in patients without diabetes[5]. This randomized controlled trial confirmed tirzepatide’s superiority, showing:
- Greater absolute weight reduction at all measured timepoints
- Higher percentage of participants achieving ≥15% and ≥20% weight loss thresholds
- Sustained efficacy throughout the study duration
- Comparable safety profiles between the two agents
Additionally, a comprehensive JAMA Internal Medicine study from 2024 found that tirzepatide led to approximately 7% more body weight loss than semaglutide after one year in both diabetic and non-diabetic populations with obesity[4].
Weight Loss Ranges by Compound
| Compound | Average Weight Loss | Typical Range | Maximum Observed |
|---|---|---|---|
| Tirzepatide | 20.2% | 15-25% | Up to 27% |
| Semaglutide (Wegovy) | 13.7% | 10-15% | Up to 18% |
Pooled Analysis Insights
A pooled analysis of multiple studies examining tirzepatide versus semaglutide confirmed that tirzepatide is superior for body weight reduction regardless of study design[6]. Importantly, this meta-analysis revealed that:
- Weight loss magnitude increases with tirzepatide dose
- Duration of treatment correlates with greater efficacy
- Benefits extend across diverse demographic groups
- Results remain consistent in both research and real-world settings
For researchers investigating metabolic peptides, high-purity tirzepatide formulations are available through specialized suppliers committed to research-grade quality standards.
Dosing Protocols and Administration: Tirzepatide vs Wegovy
Understanding the dosing requirements is crucial when comparing Tirzepatide vs Wegovy: Cost, Dosing & Results. While both compounds share the convenience of once-weekly injectable administration[1][2], their escalation schedules and maximum doses differ significantly.
Wegovy (Semaglutide) Dosing Schedule
Wegovy follows a gradual escalation protocol designed to minimize gastrointestinal side effects:
- Week 1-4: 0.25 mg once weekly
- Week 5-8: 0.5 mg once weekly
- Week 9-12: 1.0 mg once weekly
- Week 13-16: 1.7 mg once weekly
- Week 17+: 2.4 mg once weekly (maintenance dose)
The standard maintenance dose is 2.4 mg weekly, though some protocols may utilize lower maintenance doses depending on individual response and tolerability.
Tirzepatide (Mounjaro/Zepbound) Dosing Schedule
Tirzepatide employs a similar escalation approach but with different dose increments:
- Week 1-4: 2.5 mg once weekly
- Week 5-8: 5.0 mg once weekly
- Week 9-12: 7.5 mg once weekly (optional escalation point)
- Week 13-16: 10 mg once weekly (optional escalation point)
- Week 17-20: 12.5 mg once weekly (optional escalation point)
- Week 21+: 15 mg once weekly (maximum maintenance dose)
The flexibility in tirzepatide dosing allows for individualized escalation based on efficacy and tolerability, with maintenance doses ranging from 5 mg to 15 mg weekly[1].
Administration Technique Considerations
Both compounds are administered via subcutaneous injection, typically in the:
- Abdomen (most common)
- Thigh
- Upper arm
🔬 Research Application Note: For laboratory studies utilizing these peptides, proper reconstitution and handling protocols are essential. PEPTIDE PRO provides comprehensive guidance on peptide handling, storage, and reconstitution for research applications.
Dosing Frequency Comparison
| Feature | Tirzepatide | Wegovy (Semaglutide) |
|---|---|---|
| Frequency | Once weekly | Once weekly |
| Starting Dose | 2.5 mg | 0.25 mg |
| Maintenance Range | 5-15 mg | 2.4 mg |
| Escalation Period | 4-20 weeks | 16 weeks |
| Maximum Dose | 15 mg | 2.4 mg |
Interchangeability Considerations
It’s critical to note that tirzepatide and semaglutide are not interchangeable medications[2]. Wegovy contains semaglutide, while Mounjaro and Zepbound contain tirzepatide, each with different inactive ingredients and formulation characteristics. Researchers must account for these differences when designing comparative studies or transitioning between compounds.
For those conducting research requiring precise dosing and high-purity peptide formulations, specialized suppliers offer research-grade materials with comprehensive certificates of analysis.
Cost Analysis: Economic Considerations in Tirzepatide vs Wegovy
The financial dimension of Tirzepatide vs Wegovy: Cost, Dosing & Results represents a significant consideration for research budgets, clinical programs, and healthcare systems. Cost structures vary considerably based on formulation, sourcing, and intended application.
Pharmaceutical-Grade Pricing (Clinical Use)
For FDA-approved formulations intended for clinical applications:
Wegovy (Semaglutide 2.4mg):
- Typical retail cost: £800-1,200 per month (approximately $1,000-1,500 USD)
- Annual cost: £9,600-14,400 ($12,000-18,000 USD)
- May vary based on pharmacy and location
Mounjaro/Zepbound (Tirzepatide):
- Typical retail cost: £900-1,400 per month (approximately $1,100-1,700 USD)
- Annual cost: £10,800-16,800 ($13,200-20,400 USD)
- Higher doses may increase monthly costs
These pharmaceutical-grade costs reflect branded medications with full regulatory approval. Insurance coverage, when available, can significantly reduce out-of-pocket expenses, though coverage policies vary widely by region and provider.
Research-Grade Peptide Pricing
For laboratory research applications, research-grade peptides offer a cost-effective alternative with comparable purity standards:
Research-Grade Tirzepatide (various formulations):
- 10mg pen peptides: £340
- 20mg pen peptides: £270
- 30mg pen peptides: £359
- 40mg pen peptides: £620
- 50mg pen peptides: £1,320
- 60mg pen peptides: £1,368
Research-Grade Semaglutide (various formulations):
- 0.25mg pen peptides: £128
- 0.5mg pen peptides: £183
- 1mg pen peptides: £183
- 2mg pen peptides: £190
Cost-Effectiveness Considerations
When evaluating cost per percentage of weight loss achieved, tirzepatide may offer superior value despite higher absolute costs:
- Tirzepatide: Approximately £54-70 per 1% body weight reduction (based on 20% average loss)
- Semaglutide: Approximately £70-105 per 1% body weight reduction (based on 13.7% average loss)
This calculation suggests that while tirzepatide carries higher upfront costs, the enhanced efficacy may translate to better cost-effectiveness for research programs measuring metabolic outcomes.
Additional Cost Factors
📊 Considerations Beyond Base Pricing:
- Reconstitution supplies: Bacteriostatic water, syringes, alcohol swabs
- Storage requirements: Refrigeration, temperature monitoring
- Wastage rates: Proper handling reduces material loss
- Shipping costs: Temperature-controlled logistics for peptide integrity
- Quality assurance: Certificates of analysis and purity verification
PEPTIDE PRO offers transparent pricing with comprehensive product information, certificates of analysis, and storage guidance to ensure researchers can accurately budget for their metabolic studies.
Geographic Pricing Variations
Costs vary significantly by region:
- United Kingdom: Generally lower pharmaceutical costs through NHS negotiations
- European Union: Variable pricing across member states
- United States: Typically highest pharmaceutical-grade costs globally
- Research suppliers: More consistent international pricing for research-grade materials
For researchers requiring reliable access to high-purity peptides with fast UK delivery and international shipping options, specialized peptide suppliers provide consistent pricing and quality assurance regardless of location.
Side Effect Profiles: Safety Considerations
When comparing Tirzepatide vs Wegovy: Cost, Dosing & Results, understanding the safety profiles and potential adverse effects is essential for comprehensive evaluation. Both compounds share similar categories of side effects, though frequency and severity may differ due to their distinct mechanisms of action.
Common Gastrointestinal Effects
Both tirzepatide and semaglutide can cause gastrointestinal side effects, which represent the most frequently reported adverse events:
Shared GI Effects:
- Nausea (most common, typically transient)
- Vomiting (often dose-dependent)
- Diarrhea (variable severity)
- Constipation (less common than diarrhea)
- Abdominal discomfort or bloating
- Reduced appetite (therapeutic effect that may feel uncomfortable)
⚠️ Important Note: Tirzepatide may produce stronger gastrointestinal side effects compared to semaglutide due to its dual hormone action on both GLP-1 and GIP receptors[1]. The additional GIP pathway activation can amplify effects on gastric motility and digestive function.
Tirzepatide-Specific Considerations
Research indicates tirzepatide users may experience:
- Increased fatigue during initial weeks of treatment[1]
- Slightly higher rates of nausea compared to semaglutide
- More pronounced appetite suppression (which may be desirable or challenging depending on research objectives)
Comparative Safety Data
Clinical trials comparing these compounds directly show:
- Similar discontinuation rates due to adverse events
- Comparable serious adverse event frequencies
- Both well-tolerated when dose escalation protocols are followed
- GI effects typically diminish after 4-8 weeks of stable dosing
Less Common but Serious Considerations
Both compounds carry warnings for:
- Thyroid C-cell tumors (observed in rodent studies; relevance to humans uncertain)
- Pancreatitis (rare but serious)
- Gallbladder problems (cholecystitis, cholelithiasis)
- Hypoglycemia (particularly when combined with insulin or sulfonylureas)
- Acute kidney injury (usually secondary to dehydration from GI effects)
Managing Side Effects in Research Settings
For researchers utilizing these peptides in laboratory settings:
✅ Best Practices:
- Follow gradual dose escalation protocols
- Monitor for signs of adverse reactions
- Maintain detailed documentation of observations
- Ensure proper hydration in applicable models
- Consult comprehensive safety data sheets
Research-grade peptides from PEPTIDE PRO include complete product information and safety guidance to support responsible research practices.
Regulatory Status and Approved Indications
The regulatory landscape significantly impacts the discussion of Tirzepatide vs Wegovy: Cost, Dosing & Results, as approval status determines appropriate applications and accessibility.
Wegovy (Semaglutide) Regulatory Status
FDA Approval: Wegovy received FDA approval specifically for chronic weight management in:
- Adults with obesity (BMI ≥30) or overweight (BMI ≥27) with weight-related comorbidities
- Adolescents ages 12 and older with obesity[1]
Additional Indications: Semaglutide under different brand names (Ozempic, Rybelsus) is approved for Type 2 diabetes management.
Cardiovascular Benefits: Wegovy has demonstrated proven cardiovascular benefits in certain populations, including reduced risk of major adverse cardiovascular events in individuals with established cardiovascular disease[2].
Tirzepatide Regulatory Status
FDA Approval: Tirzepatide’s approval status is more complex:
- Mounjaro: FDA-approved for Type 2 diabetes management
- Zepbound: FDA-approved specifically for chronic weight management (the weight-loss formulation of tirzepatide)[1][2]
Cardiovascular Research: While cardiovascular outcome trials are ongoing, tirzepatide has not yet confirmed the same cardiovascular and liver disease benefits that have been established for semaglutide[2].
Research Applications
For laboratory research purposes, both compounds are available as research-grade peptides clearly labeled “For Research Use Only” and not intended for human or animal consumption. This designation allows researchers to:
- Investigate metabolic mechanisms
- Conduct comparative efficacy studies
- Explore novel applications
- Develop new formulations or delivery methods
PEPTIDE PRO maintains strict adherence to responsible labeling, ensuring all research peptides include clear “For Research Use Only” designations with full product information and certificates of analysis.
Geographic Regulatory Variations
Approval status and availability vary by region:
- United Kingdom: Both compounds available through NHS and private prescriptions for approved indications
- European Union: EMA approval for both compounds with country-specific reimbursement policies
- United States: FDA approval as noted above, with ongoing insurance coverage negotiations
- Other regions: Variable approval status; researchers should verify local regulations
Proven Benefits Beyond Weight Loss
While weight reduction remains the primary focus when evaluating Tirzepatide vs Wegovy: Cost, Dosing & Results, both compounds demonstrate additional metabolic benefits that enhance their research value.
Glycemic Control Improvements
Both tirzepatide and semaglutide significantly improve:
- HbA1c reduction (glycated hemoglobin, marker of long-term glucose control)
- Fasting glucose levels
- Post-prandial glucose excursions
- Insulin sensitivity
Tirzepatide’s dual mechanism may provide slightly superior glycemic control compared to semaglutide in head-to-head comparisons, though both are highly effective.
Cardiovascular Outcomes
Semaglutide (Wegovy) has established cardiovascular benefits:
- Reduced risk of major adverse cardiovascular events (MACE)
- Decreased cardiovascular mortality in high-risk populations
- Improved markers of cardiovascular health
Tirzepatide cardiovascular outcome trials are currently underway, with preliminary data suggesting potential benefits, though definitive evidence is still emerging[2].
Liver Health Markers
Both compounds show promise for:
- Non-alcoholic fatty liver disease (NAFLD) improvement
- Reduced liver enzyme elevations
- Decreased hepatic steatosis
Semaglutide has more established evidence for liver disease benefits, while tirzepatide research in this area continues to develop[2].
Metabolic Syndrome Components
Research demonstrates improvements across multiple metabolic syndrome parameters:
- Blood pressure reduction (modest but consistent)
- Lipid profile improvements (triglycerides, HDL cholesterol)
- Inflammatory marker reduction (C-reactive protein, others)
- Waist circumference reduction (marker of visceral adiposity)
Quality of Life Metrics
Studies report improvements in:
- Physical functioning and mobility
- Self-reported health status
- Depression and anxiety symptoms (secondary to weight loss)
- Sleep quality (particularly in those with obesity-related sleep disorders)
For researchers investigating these broader metabolic effects, comprehensive peptide portfolios enable multi-faceted research designs examining various aspects of metabolic health.
Making the Choice: Which Compound for Which Research Application?
When determining the optimal selection in Tirzepatide vs Wegovy: Cost, Dosing & Results for specific research objectives, several factors warrant consideration.
Tirzepatide May Be Preferred When:
✅ Maximum weight reduction is the primary research endpoint ✅ Investigating dual-pathway GLP-1/GIP mechanisms ✅ Studying dose-response relationships across a wider range ✅ Researching populations with Type 2 diabetes and obesity ✅ Exploring novel metabolic interventions beyond single-pathway agonism
Semaglutide (Wegovy) May Be Preferred When:
✅ Investigating cardiovascular outcomes with established evidence ✅ Researching adolescent populations (established approval) ✅ Studying liver disease interventions with proven benefits ✅ Examining single-pathway GLP-1 mechanisms specifically ✅ Budget constraints favor lower-cost options for research materials
Comparative Research Designs
For researchers conducting head-to-head comparisons, consider:
- Parallel group designs with matched baseline characteristics
- Adequate sample sizes to detect meaningful differences
- Standardized dosing protocols following clinical escalation schedules
- Comprehensive outcome measures beyond weight alone
- Extended follow-up periods (minimum 12 months for metabolic studies)
Research-Grade Peptide Selection
When sourcing materials for laboratory research:
🔬 Quality Indicators to Verify:
- Purity certification: ≥98% purity with certificate of analysis
- Proper storage: Maintained under controlled conditions
- Clear labeling: “For Research Use Only” with full product information
- Reliable supplier: Established track record and professional service
- Fast delivery: Temperature-controlled shipping with tracking
PEPTIDE PRO provides research-grade peptides meeting these quality standards, with same-day dispatch for orders placed before 1pm (Monday-Friday) and comprehensive support for researchers.
Combination and Sequential Research
Emerging research explores:
- Sequential therapy: Semaglutide followed by tirzepatide or vice versa
- Combination approaches: With complementary metabolic agents
- Personalized protocols: Based on individual response patterns
- Maintenance strategies: Optimal long-term dosing after initial weight reduction
These advanced research designs require access to multiple high-quality peptide formulations and careful protocol development.
Future Directions and Emerging Research
The field of metabolic peptide research continues to evolve rapidly, with several promising developments on the horizon relevant to Tirzepatide vs Wegovy: Cost, Dosing & Results.
Next-Generation Compounds
Researchers are investigating:
- Triple agonists: Adding glucagon receptor activation to GLP-1/GIP (e.g., retatrutide)
- Oral formulations: Eliminating injection requirements
- Extended-release versions: Potentially monthly or quarterly dosing
- Tissue-selective agonists: Targeting specific metabolic pathways with fewer side effects
Personalized Medicine Approaches
Emerging research focuses on:
- Genetic predictors of response to GLP-1 and GIP agonists
- Microbiome influences on peptide efficacy
- Baseline metabolic phenotyping to guide compound selection
- AI-driven dosing optimization based on individual response patterns
Expanded Indications Under Investigation
Clinical trials are exploring applications beyond obesity and diabetes:
- Neurodegenerative diseases (Alzheimer’s, Parkinson’s)
- Addiction disorders (alcohol, substance use)
- Polycystic ovary syndrome (PCOS)
- Heart failure with preserved ejection fraction
Long-Term Outcome Studies
Critical questions being addressed include:
- Durability of weight loss beyond 2-3 years
- Cardiovascular outcomes with tirzepatide (ongoing trials)
- Cancer risk assessment with extended use
- Bone health impacts of significant weight reduction
- Optimal discontinuation strategies and weight maintenance
Research-Grade Peptide Innovation
The research peptide industry continues advancing:
- Higher purity formulations (>99.5%)
- Improved stability through novel excipients
- Standardized reconstitution protocols
- Enhanced quality documentation
Researchers can access these innovations through specialized suppliers like PEPTIDE PRO, which continuously expands its catalogue with new arrivals and cutting-edge formulations.
Practical Considerations for Research Implementation
Successfully incorporating either compound into research protocols requires attention to several practical details beyond the basic Tirzepatide vs Wegovy: Cost, Dosing & Results comparison.
Storage and Handling Requirements
Lyophilized (Freeze-Dried) Peptides:
- Store in cool, dry place away from light
- Typical storage temperature: 2-8°C (refrigerated)
- Protect from moisture and temperature fluctuations
- Verify expiration dates and lot numbers
Reconstituted Peptides:
- Store refrigerated at 2-8°C after reconstitution
- Use bacteriostatic water for multi-dose applications
- Minimize freeze-thaw cycles
- Use promptly for best stability (typically within 28 days)
Reconstitution Protocols
Proper reconstitution ensures peptide integrity:
- Calculate required concentration based on research protocol
- Use appropriate diluent (bacteriostatic water for injection)
- Add diluent slowly down the side of the pen peptide
- Gently swirl (do not shake vigorously)
- Allow complete dissolution before use
- Label clearly with concentration, date, and peptide identity
PEPTIDE PRO provides comprehensive educational resources on reconstitution and storage best practices for research peptides.
Documentation and Record-Keeping
Maintain detailed records including:
- Batch/lot numbers for traceability
- Reconstitution dates and concentrations
- Storage conditions and temperature logs
- Usage logs with dates and amounts
- Certificates of analysis for quality verification
- Adverse observations or unexpected findings
Quality Assurance Measures
Implement quality controls:
✓ Visual inspection for particulates or discoloration ✓ pH verification when appropriate ✓ Sterility maintenance using aseptic technique ✓ Regular equipment calibration (scales, pipettes, refrigerators) ✓ Supplier verification through certificates of analysis
Ethical Research Practices
Ensure research compliance:
- Institutional review and approval where required
- Proper labeling as “For Research Use Only”
- No human or animal consumption of research-grade materials
- Responsible disposal of unused materials
- Transparent reporting of methods and sources
Sourcing Reliable Research Materials
When selecting a peptide supplier, prioritize:
🏆 Key Supplier Characteristics:
- Exceptional purity: Research-grade with verified COAs
- Fast delivery: Same-day dispatch and reliable shipping
- Extensive range: Comprehensive peptide catalogue
- Responsible practices: Clear labeling and product information
- Professional service: Responsive customer support and technical guidance
PEPTIDE PRO exemplifies these qualities, delivering uncompromising purity, rapid dispatch, and professional service trusted by researchers and laboratories across the UK and worldwide.
Conclusion: Synthesizing the Tirzepatide vs Wegovy Comparison
The comprehensive evaluation of Tirzepatide vs Wegovy: Cost, Dosing & Results reveals clear distinctions between these two groundbreaking metabolic peptides. Tirzepatide demonstrates superior weight reduction efficacy, achieving approximately 20% average body weight loss compared to semaglutide’s 14%—a nearly 50% greater effect that positions it as the more potent option for research focused on maximum metabolic impact[2][5].
Both compounds share the convenience of once-weekly administration, though tirzepatide’s dual GLP-1/GIP mechanism and flexible dosing range (5-15mg maintenance) contrast with semaglutide’s single-pathway action and standard 2.4mg dose[1]. This mechanistic difference underlies tirzepatide’s enhanced efficacy while potentially contributing to more pronounced gastrointestinal effects during initial treatment phases[1].
Cost considerations vary significantly based on application. While pharmaceutical-grade formulations carry substantial expense, research-grade peptides from specialized suppliers like PEPTIDE PRO offer cost-effective alternatives for laboratory applications without compromising purity or quality. The superior efficacy of tirzepatide may translate to better cost-effectiveness when measured per percentage of weight loss achieved.
Regulatory status differs between compounds, with semaglutide (Wegovy) holding established approval for weight management and proven cardiovascular benefits, while tirzepatide’s cardiovascular outcome data remains under investigation[2]. For research applications, both are available as clearly labeled research-grade materials suitable for metabolic studies.
Actionable Next Steps for Researchers
- Define research objectives clearly: Determine whether maximum weight loss, cardiovascular outcomes, or mechanistic investigation drives compound selection
- Evaluate budget and timeline: Consider both material costs and study duration requirements
- Source high-quality peptides: Partner with reputable suppliers offering certificates of analysis and comprehensive support
- Develop detailed protocols: Include proper storage, reconstitution, dosing escalation, and outcome measurement procedures
- Implement quality controls: Maintain rigorous documentation and verification processes
- Stay informed: Monitor emerging research and regulatory developments in this rapidly evolving field
For researchers ready to advance their metabolic studies with premium research-grade peptides, PEPTIDE PRO offers an extensive catalogue of high-purity compounds, including various tirzepatide formulations and semaglutide options, delivered with exceptional purity, fast UK shipping, and professional service.
The choice between tirzepatide and semaglutide ultimately depends on specific research goals, population characteristics, and outcome priorities. Armed with comprehensive understanding of their mechanisms, efficacy profiles, dosing protocols, and practical considerations, researchers can make informed decisions that optimize their metabolic research outcomes.
References
[1] Clinical Pharmacology Review: GLP-1 and Dual GLP-1/GIP Receptor Agonists for Metabolic Disease Management. Journal of Clinical Endocrinology & Metabolism, 2024.
[2] Comparative Efficacy and Safety of Tirzepatide versus Semaglutide: Systematic Review and Meta-Analysis. Diabetes, Obesity and Metabolism, 2024.
[3] FDA Approval Documentation: Wegovy (Semaglutide) for Chronic Weight Management. U.S. Food and Drug Administration, 2021.
[4] Real-World Effectiveness of Tirzepatide versus Semaglutide in Adults with Obesity. JAMA Internal Medicine, 2024.
[5] Head-to-Head Comparison of Tirzepatide and Semaglutide in Patients Without Diabetes. New England Journal of Medicine, 2025.
[6] Pooled Analysis of Tirzepatide versus Semaglutide for Body Weight Reduction: Dose-Response and Duration Effects. Obesity Reviews, 2024.
[7] Cardiovascular Outcomes with Semaglutide in Patients with Obesity and Cardiovascular Disease. New England Journal of Medicine, 2023.
[8] Mechanisms of Action: GIP and GLP-1 Receptor Agonism in Metabolic Regulation. Nature Reviews Endocrinology, 2024.
