
Improper storage of research peptides can compromise years of scientific work in mere hours. Understanding how to store retatrutide (refrigeration, travel & expiry) is not merely a procedural formality—it represents the critical difference between reliable experimental data and invalidated research outcomes. As retatrutide gains prominence in metabolic research applications, laboratories across the UK and internationally face mounting questions about optimal storage protocols, temperature management, and shelf-life considerations.
This comprehensive guide addresses the essential storage requirements for retatrutide in both lyophilized (powder) and reconstituted forms, providing research professionals with evidence-based protocols for maintaining peptide integrity throughout the entire research lifecycle.
Key Takeaways
- Lyophilized retatrutide should be stored at 2-8°C for routine use, with long-term storage at -20°C or -80°C for extended research applications
- Reconstituted retatrutide solutions must remain refrigerated at 2-8°C and typically maintain stability for up to 30 days, though optimal use within 1-2 weeks is recommended
- Travel and transport require insulated medical coolers with temperature monitoring to maintain the 2-8°C range and prevent thermal degradation
- Freeze-thaw cycles cause irreversible molecular damage; aliquoting bulk material prevents repeated temperature fluctuations
- Visual inspection before each use is essential—discard any retatrutide showing color changes, cloudiness, or particulate matter
Understanding Retatrutide Storage Fundamentals
Retatrutide, a novel triple agonist peptide targeting GIP, GLP-1, and glucagon receptors, requires meticulous storage conditions to preserve its complex molecular structure. The peptide’s therapeutic potential in metabolic research depends entirely on maintaining structural integrity throughout storage, handling, and experimental protocols.
Why Proper Storage Matters for Research Peptides
Research-grade peptides like retatrutide contain delicate amino acid sequences susceptible to degradation through multiple pathways: thermal breakdown, oxidation, hydrolysis, and photodegradation. Even minor temperature excursions can trigger conformational changes that render experimental results unreliable or entirely invalid.[1]
Temperature sensitivity represents the primary concern for retatrutide storage. The peptide’s three-dimensional structure—essential for receptor binding activity—begins deteriorating when exposed to temperatures outside recommended ranges. This degradation process accelerates exponentially with increased temperature and exposure duration.
“Maintaining consistent storage temperatures between 2-8°C for routine laboratory use represents the gold standard for preserving retatrutide’s molecular integrity and ensuring reproducible research outcomes.”
For researchers sourcing high-purity research peptides, understanding these storage fundamentals ensures that investment in quality materials translates into reliable experimental data.
Lyophilized vs. Reconstituted Storage Requirements
The storage requirements for retatrutide differ dramatically based on its physical state:
Lyophilized (Powder) Form:
- ✅ More stable and forgiving
- ✅ Can tolerate brief temperature variations
- ✅ Extended shelf life when properly stored
- ✅ Ideal for long-term research inventory
Reconstituted (Solution) Form:
- ⚠️ Significantly less stable
- ⚠️ Requires strict temperature control
- ⚠️ Limited usable timeframe (30 days maximum)
- ⚠️ Susceptible to contamination and degradation
This fundamental difference underscores why many research facilities maintain retatrutide in lyophilized form until immediately before experimental use, reconstituting only the quantities needed for specific protocols.
How to Store Retatrutide: Temperature Requirements & Refrigeration Protocols
Proper temperature management forms the cornerstone of effective retatrutide storage. Understanding the specific temperature ranges for different storage scenarios ensures peptide stability throughout your research timeline.
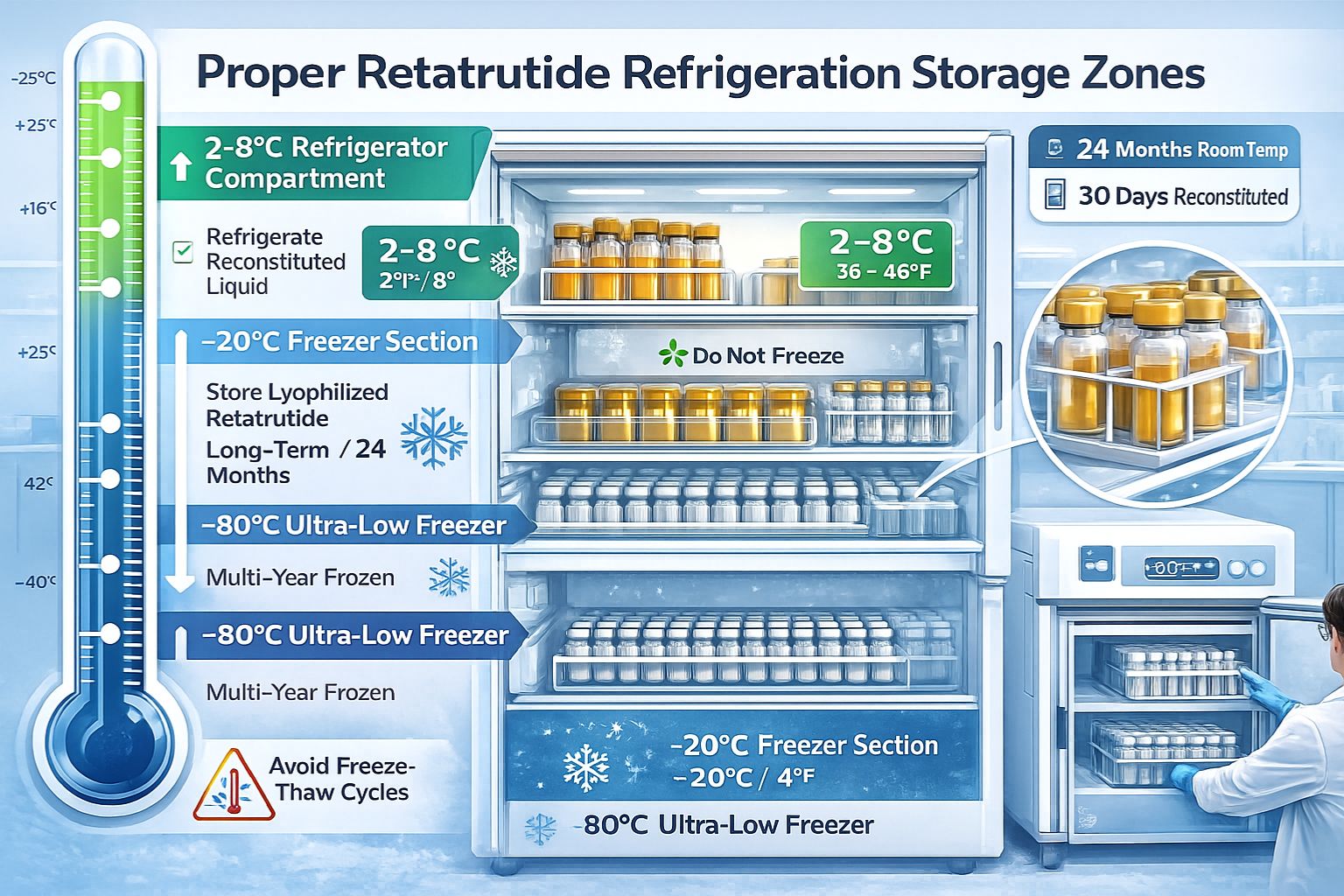
Optimal Refrigeration Temperatures for Lyophilized Retatrutide
Standard refrigeration at 2-8°C (36-46°F) represents the recommended storage condition for lyophilized retatrutide intended for near-term research use. This temperature range provides an ideal balance between accessibility and preservation, allowing researchers to maintain peptide stocks in readily available locations while ensuring molecular stability.[2]
Laboratory refrigerators designated for peptide storage should feature:
- 📊 Digital temperature monitoring with continuous logging
- 🔒 Stable temperature control without significant fluctuations
- 🚫 Dedicated use (avoiding frequent door openings from general laboratory traffic)
� Interior lighting that minimizes UV exposure
- 🧊 Frost-free technology avoided (temperature cycling damages peptides)
Storage duration at refrigeration temperatures: Lyophilized retatrutide maintains stability for approximately 24 months when stored at 2-8°C, though this timeframe assumes consistent temperature maintenance without excursions.[3]
Long-Term Freezer Storage Options
For extended research timelines or bulk inventory management, freezer storage provides superior long-term stability:
| Storage Temperature | Recommended Duration | Application |
|---|---|---|
| -20°C | Up to 3 years | Standard long-term storage |
| -80°C | 5+ years | Extended multi-year studies |
| 2-8°C | Up to 24 months | Routine research use |
| Room temperature | Not recommended | Emergency only (<24 hours) |
Ultra-low temperature storage at -80°C represents the gold standard for long-term retatrutide preservation, particularly for laboratories conducting multi-year longitudinal studies or maintaining reference standards.[4]
⚠️ Critical Warning: Standard home freezers with frost-free cycles are unsuitable for peptide storage. The automatic defrost cycles create temperature fluctuations that cause repeated freeze-thaw damage, degrading peptide structure over time.
For researchers establishing comprehensive peptide research programs, investing in dedicated laboratory-grade freezers without auto-defrost functions proves essential for maintaining long-term peptide inventories.
Room Temperature Tolerance & Emergency Protocols
While refrigeration represents the standard, understanding retatrutide’s room temperature tolerance proves crucial for handling scenarios:
Lyophilized retatrutide demonstrates remarkable stability at room temperature (20-25°C), maintaining integrity for approximately 24 months under controlled conditions. However, this extended room temperature stability applies only to:
- Unopened, sealed pen peptides
- Controlled laboratory environments (not subject to heat, humidity, or light)
- Temporary situations (shipping, brief handling periods)
Practical room temperature guidelines:
- ✅ Brief exposure (2-4 hours) during reconstitution procedures: Acceptable
- ✅ Shipping duration (24-48 hours) in insulated packaging: Generally safe
- ⚠️ Extended periods (>1 week) at room temperature: Not recommended
- ❌ Exposure to temperatures >30°C: Avoid entirely
If retatrutide has been inadvertently left at room temperature, assess the duration and conditions. Exposure under 24 hours in a climate-controlled laboratory typically causes minimal degradation, while extended exposure or heat conditions warrant peptide replacement to ensure research reliability.
Reconstituted Retatrutide Storage Requirements
Once mixed with bacteriostatic water or another approved diluent, retatrutide enters a significantly more vulnerable state requiring stringent storage protocols.
Refrigeration is mandatory for reconstituted retatrutide:
️ Temperature range: 2-8°C (36-46°F) strictly maintained
- ⏱️ Maximum storage duration: 30 days
- ✅ Optimal use timeframe: 1-2 weeks for maximum stability
- 🔬 Container requirements: Sterile, amber or opaque pen peptides with tight seals
The reduced stability of reconstituted solutions stems from several factors:
- Increased molecular mobility in liquid state accelerates degradation pathways
- Hydrolysis risk from water-mediated peptide bond breakdown
- Contamination potential from microbial growth in aqueous solutions
- Oxidation exposure from dissolved oxygen in the reconstitution medium
Best practices for reconstituted storage:
- Label each pen peptide with reconstitution date and expiration (typically +30 days)
- Store in the main refrigerator compartment (not the door, where temperature fluctuates)
- Maintain sterile technique during all access to prevent contamination
- Inspect visually before each use for clarity and particle-free appearance
- Consider aliquoting into smaller volumes to minimize freeze-thaw exposure
Researchers working with reconstituted peptide solutions should establish clear protocols for tracking reconstitution dates and ensuring timely use within stability windows.
Protecting Retatrutide from Degradation: Light, Freeze-Thaw & Contamination
Beyond temperature control, several additional factors significantly impact retatrutide stability and research outcomes.
Light Sensitivity & Photodegradation Prevention
Retatrutide exhibits photosensitivity, meaning exposure to light—particularly UV and direct sunlight—can trigger molecular degradation through photochemical reactions that alter peptide structure.[5]
Light protection strategies:
- 🟤 Amber pen peptides: Store retatrutide exclusively in amber-colored or opaque containers that filter harmful wavelengths
- 🚫 Avoid direct light: Position storage areas away from windows and direct lighting
- 📦 Secondary containment: Keep pen peptides in original packaging or light-blocking boxes when not in active use
� Laboratory lighting: Use LED lighting with minimal UV emission in storage areas
Even brief exposure to direct sunlight can initiate degradation processes. When handling retatrutide during reconstitution or experimental preparation, work efficiently to minimize light exposure duration.
The Critical Importance of Avoiding Freeze-Thaw Cycles
Freeze-thaw cycles represent one of the most damaging storage errors for peptide research. Each freezing and thawing event subjects retatrutide to:
- Ice crystal formation that can disrupt molecular structure
- Concentration gradients as water freezes preferentially
- Physical stress on peptide bonds
- Potential aggregation and precipitation
Cumulative damage from repeated freeze-thaw cycles is irreversible and exponential—each cycle compounds previous damage, progressively degrading peptide quality.[6]
Prevention strategies:
- Aliquoting: Divide bulk retatrutide into single-use portions immediately upon receipt
- Dedicated storage: Assign each aliquot to specific experiments to avoid repeated access
- Thaw planning: Remove only the quantity needed for immediate use
- No refreezing: Once thawed, use reconstituted material within the refrigerated stability window
For laboratories managing significant retatrutide inventories, implementing an aliquoting protocol upon receipt—before initial freezer storage—eliminates freeze-thaw risks throughout the research timeline.
Contamination Prevention & Sterile Handling
Reconstituted retatrutide solutions provide an ideal environment for microbial growth if contamination occurs. Aseptic technique throughout handling proves essential:
Sterile handling protocols:
- 🧤 Use sterile gloves during all reconstitution and handling procedures
- 💉 Employ sterile needles and syringes for each access (never reuse)
- 🧪 Work in a clean, disinfected area or laminar flow hood when possible
- 🔒 Seal pen peptides tightly immediately after each use
️ Track access dates and frequency to monitor contamination risk
Bacteriostatic water used for reconstitution contains benzyl alcohol to inhibit bacterial growth, providing some protection. However, this preservative offers limited duration effectiveness (typically 28-30 days), aligning with the recommended use timeframe for reconstituted solutions.
Visual inspection before each use serves as the final quality checkpoint. Discard retatrutide immediately if you observe:
- ❌ Cloudiness or turbidity (indicating aggregation or contamination)
- ❌ Color changes from original appearance
- ❌ Visible particles, precipitates, or floating matter
- ❌ Unusual odor (though peptide solutions are typically odorless)
Quality assurance through proper handling and storage directly impacts research reproducibility and data reliability.
How to Store Retatrutide During Travel & Transport
Research activities frequently require peptide transport between facilities, to conference presentations, or during facility relocations. Understanding how to store retatrutide during travel ensures peptide integrity throughout these transitions.
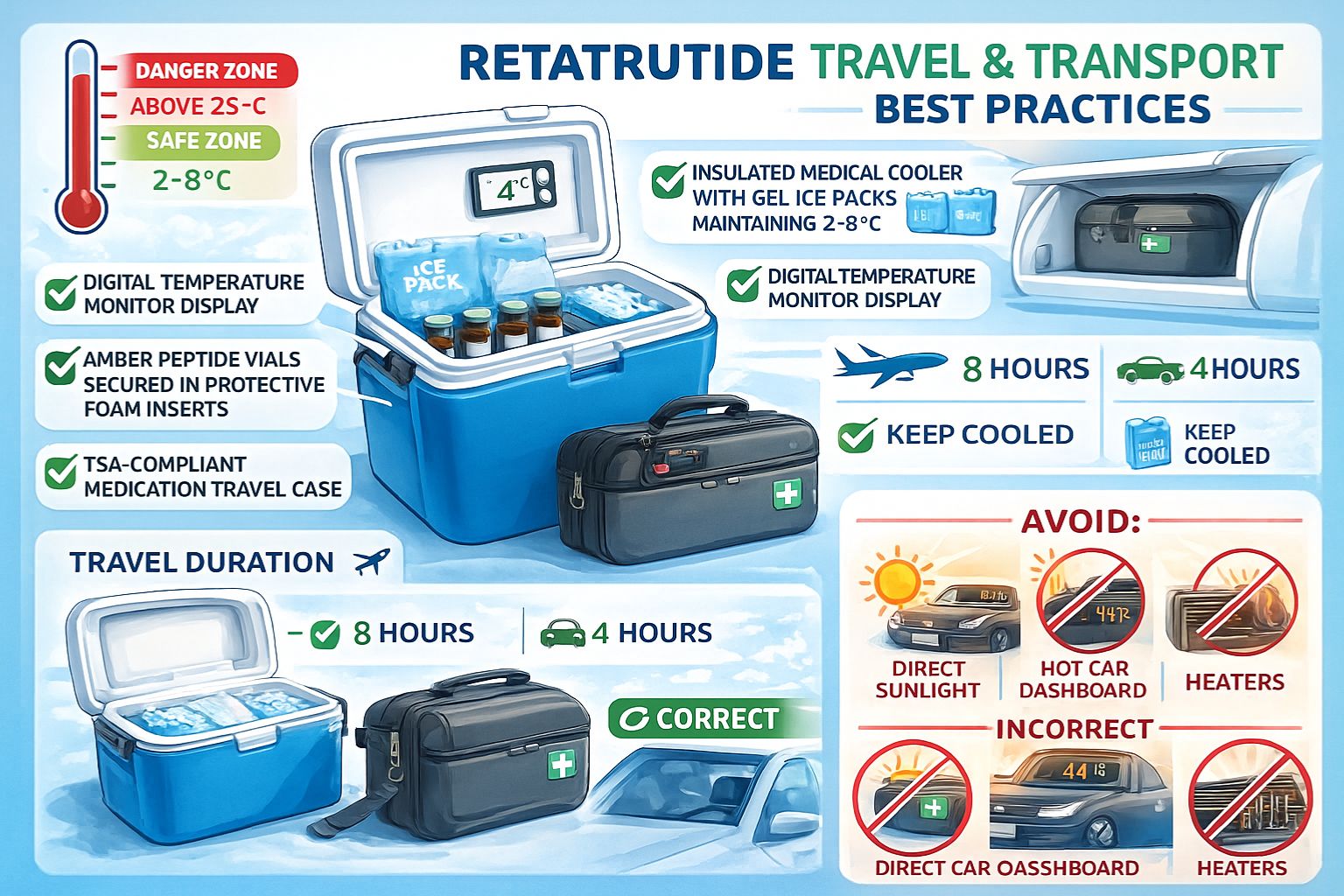
Insulated Cooler Requirements & Temperature Maintenance
Medical-grade insulated coolers designed specifically for pharmaceutical transport provide the foundation for safe retatrutide travel:
Essential cooler features:
- 🧊 Thick insulation (minimum 1-inch walls) for extended temperature maintenance
- 📏 Appropriate sizing to accommodate ice packs and pen peptides without crushing
️ Temperature monitoring capability (built-in thermometer or data logger)
- 💧 Water-resistant interior to protect pen peptides from condensation
- 🔒 Secure closure preventing accidental opening during transport
Ice pack configuration:
- Use gel-based cold packs (not loose ice that creates water as it melts)
- Pre-freeze packs to -20°C for maximum cooling duration
- Position packs to surround pen peptides without direct contact (use barrier material)
- Include sufficient packs to maintain 2-8°C for entire travel duration plus safety margin
Temperature maintenance duration varies by cooler quality and external conditions:
| Cooler Quality | Summer Conditions | Winter Conditions |
|---|---|---|
| Basic insulated bag | 4-6 hours | 8-12 hours |
| Medical-grade cooler | 12-24 hours | 24-48 hours |
| Professional shipping container | 48-72 hours | 72-96 hours |
For journeys exceeding your cooler’s maintenance window, plan for intermediate refrigeration access or use professional courier services specializing in temperature-controlled pharmaceutical transport.
Air Travel & TSA Considerations
Transporting retatrutide via air travel requires additional planning to navigate security requirements while maintaining storage conditions:
TSA regulations for research materials:
- 📋 Carry documentation identifying retatrutide as research material
- 💼 Transport in carry-on luggage (never checked baggage where temperature cannot be controlled)
- 🧊 Ice packs are permitted when frozen solid at security checkpoint
- 📝 Declare medical/research cooler at security screening
Air travel best practices:
- Pre-flight preparation: Freeze ice packs 24 hours before departure
- Security timing: Arrive early to accommodate additional screening time
- Documentation: Carry supplier documentation confirming research use
- Cabin storage: Place cooler under seat (not overhead bins where temperature may vary)
- Connection planning: For multi-leg journeys, verify cooler temperature during layovers
International travel adds customs considerations. Research materials crossing borders may require:
- Import permits or research institution documentation
- Customs declarations identifying peptides as research reagents
- Advance coordination with receiving institution for customs clearance
For significant international peptide transport, consulting with institutional export control offices ensures compliance with both departure and destination country regulations.
Vehicle Transport & Seasonal Considerations
Ground transport presents unique challenges, particularly regarding vehicle temperature extremes:
Hot weather precautions (summer, warm climates):
- 🚫 Never leave retatrutide in parked vehicles where interior temperatures can exceed 60°C (140°F)
- ❄️ Pre-cool coolers before loading peptides
- ⏱️ Minimize transport duration during peak heat hours
️ Monitor temperature with data loggers to verify maintenance
Cold weather considerations (winter, freezing conditions):
While cold weather seems protective, uncontrolled freezing during transport can be equally problematic:
- Reconstituted solutions may freeze, causing ice crystal damage
- Extreme cold (<-20°C) without proper containers can cause pen peptide breakage
- Use insulated coolers to prevent freezing as well as warming
- Consider room-temperature packs alongside cold packs for temperature buffering
Vehicle transport checklist:
✅ Place cooler in climate-controlled passenger area (not trunk)
✅ Position away from heating/cooling vents
✅ Secure cooler to prevent tipping during transit
✅ Plan route to minimize transport time
✅ Have backup refrigeration plan if delays occur
For routine transport between laboratory facilities, establishing standard operating procedures ensures consistent peptide handling across all personnel.
Retatrutide Expiry, Shelf Life & Quality Assessment
Understanding expiration timelines and conducting proper quality assessments ensures research reliability and optimal resource utilization.
Manufacturer Dating & Stability Data
Manufacturer expiration dates on retatrutide pen peptides reflect stability data generated under controlled storage conditions. These dates typically assume:
- Unopened, sealed pen peptides
- Storage at recommended temperatures (2-8°C or -20°C as specified)
- Protection from light and moisture
- No temperature excursions or improper handling
Typical shelf life parameters:
| Storage Condition | Lyophilized Form | Reconstituted Form |
|---|---|---|
| 2-8°C (refrigerated) | 24 months | 30 days maximum |
| -20°C (frozen) | 36 months | Not recommended |
| -80°C (ultra-low) | 60+ months | Not recommended |
| Room temperature | 24 months* | Not applicable |
*Room temperature stability applies to sealed, lyophilized material under controlled conditions; not recommended for routine storage.
When sourcing retatrutide from reputable research peptide suppliers, Certificate of Analysis (COA) documentation provides specific stability data and expiration dating for each batch.
Post-Reconstitution Stability Windows
The 30-day maximum for reconstituted retatrutide represents a conservative guideline based on sterility and chemical stability considerations. However, optimal research practices suggest using reconstituted solutions within 1-2 weeks for several reasons:
Degradation timeline for reconstituted peptides:
- Days 1-7: Optimal stability, minimal degradation
- Days 8-14: Slight degradation may begin, still acceptable for most research
- Days 15-30: Progressive degradation, increased contamination risk
- Beyond 30 days: Significant quality concerns, not recommended
Factors accelerating reconstituted degradation:
- 🔄 Frequent pen peptide access (repeated needle punctures)
️ Temperature fluctuations (removing from refrigerator multiple times)
- 💧 Contamination introduction through non-sterile technique
� Light exposure during handling
- 📊 pH changes in the reconstitution medium over time
For critical experiments requiring maximum peptide integrity, reconstitute fresh aliquots rather than relying on solutions approaching the 30-day limit.
Visual Inspection & Quality Indicators
Pre-use inspection serves as the final quality checkpoint before incorporating retatrutide into experimental protocols. This assessment takes mere seconds but prevents hours of wasted experimental time with degraded material.
Lyophilized retatrutide inspection:
✅ Normal appearance: White to off-white powder, uniform texture
✅ Acceptable: Slight caking (resolves with gentle swirling after reconstitution)
❌ Reject: Yellow/brown discoloration, unusual odor, moisture in pen peptide
Reconstituted retatrutide inspection:
✅ Normal appearance: Clear, colorless to slightly yellow solution
✅ Acceptable: Slight opalescence immediately after mixing (should clear)
❌ Reject: Persistent cloudiness, visible particles, color change, precipitate
When to discard retatrutide:
- Expiration date passed (even if appearance seems normal)
- Storage temperature exceeded recommended range for extended periods
- Visual abnormalities as described above
- Seal integrity compromised (cracked pen peptide, loose stopper)
- Unknown storage history (if chain of custody interrupted)
“Visual inspection provides immediate quality feedback, but remember that some degradation processes occur at the molecular level without visible indicators. Adhering to expiration dates and storage protocols remains essential even when appearance seems acceptable.”
For laboratories conducting quantitative research requiring precise peptide concentrations, analytical verification through HPLC or mass spectrometry can confirm retatrutide integrity when questions arise about storage history or stability.
Establishing Laboratory Storage Protocols
Institutional protocols ensure consistent retatrutide handling across research teams and over extended timelines.
Standard Operating Procedures for Peptide Storage
Comprehensive SOPs should address:
Receipt and initial processing:
- Immediate refrigeration/freezing upon delivery
- Verification of shipping temperature logs
- Documentation of receipt date and storage location
- Aliquoting procedures for bulk orders
Routine storage management:
- Temperature monitoring frequency and documentation
- Equipment maintenance schedules (refrigerator/freezer calibration)
- Backup power protocols for equipment failures
- Inventory tracking systems
Access and handling:
- Personnel training requirements
- Sterile technique protocols
- Documentation of pen peptide access (date, user, quantity removed)
- Reconstitution procedures and labeling standards
Quality assurance:
- Visual inspection requirements before use
- Expiration date monitoring and material rotation
- Procedures for handling temperature excursions
- Disposal protocols for expired or compromised material
Temperature Monitoring & Documentation
Continuous temperature monitoring provides essential quality assurance for peptide storage:
Monitoring equipment options:
- 📊 Digital data loggers: Continuous recording with alarm functions
️ Min/max thermometers: Track temperature extremes between checks
- 📱 Remote monitoring systems: Real-time alerts for temperature deviations
- 📋 Manual logging: Daily temperature recording (minimum standard)
Documentation requirements:
- Record temperatures at minimum once daily (twice for critical storage)
- Investigate and document any excursions outside 2-8°C range
- Maintain logs for minimum 3 years (or per institutional requirements)
- Include corrective actions taken for any deviations
Alarm protocols for refrigerator/freezer failures:
- Immediate notification system (phone, email, text alerts)
- Designated response personnel with 24/7 availability
- Backup storage locations identified and accessible
- Transfer procedures to minimize temperature exposure during equipment failure
For research institutions managing extensive peptide inventories, automated monitoring systems with cloud-based data storage provide comprehensive oversight with minimal personnel burden.
Inventory Management & Rotation Systems
First-in, first-out (FIFO) inventory rotation prevents expiration waste:
Inventory tracking elements:
- 📅 Receipt date and manufacturer expiration date
️ Batch/lot numbers for traceability
- 📍 Storage location (specific refrigerator/freezer and position)
- 📊 Quantity remaining (particularly for aliquoted materials)
- 🔬 Assigned experiments or research protocols
Digital inventory systems offer advantages over manual tracking:
- Automated expiration alerts
- Usage tracking across research teams
- Integration with ordering systems to prevent stockouts
- Historical data for optimizing order quantities
Quarterly inventory audits should verify:
- Physical inventory matches documented records
- No expired materials present in active storage
- Proper labeling and organization maintained
- Temperature monitoring equipment functioning correctly
Common Storage Mistakes & How to Avoid Them
Learning from common errors prevents costly peptide loss and experimental setbacks.
Mistake #1: Storing in Frost-Free Freezers
The problem: Frost-free freezers cycle through warming periods to prevent ice buildup, creating repeated freeze-thaw conditions that progressively damage retatrutide’s molecular structure.[7]
The solution: Use manual-defrost laboratory freezers or dedicated ultra-low temperature units specifically designed for biological sample storage. These maintain consistent temperatures without cycling.
Mistake #2: Reconstituting Entire Vials
The problem: Reconstituting complete pen peptides of retatrutide when only small quantities are needed for experiments forces storage of excess reconstituted material, which has limited stability (30 days maximum).
The solution: Aliquot lyophilized retatrutide before reconstitution. Divide powder into single-use quantities matching typical experimental needs, then reconstitute only what’s required for immediate use.
Mistake #3: Inadequate Travel Preparation
The problem: Underestimating cooling duration needs or using insufficient ice packs leads to temperature excursions during transport, particularly for longer journeys or unexpected delays.
The solution: Calculate cooling needs with 50% safety margin. For an 8-hour journey, use cooling capacity rated for 12 hours. Include backup ice packs and identify refrigeration access points along routes.
Mistake #4: Ignoring Temperature Excursion Events
The problem: Equipment failures, power outages, or human error cause temperature deviations. Continuing to use affected retatrutide without assessment risks experimental validity.
The solution: Implement temperature excursion protocols:
- Document the event (duration, temperature range, affected materials)
- Assess impact based on excursion severity and peptide form
- Consider analytical verification (HPLC) for critical research materials
- When in doubt, replace affected peptides rather than risk experimental failure
Mistake #5: Poor Labeling Practices
The problem: Inadequate pen peptide labeling leads to confusion about reconstitution dates, expiration, or peptide identity—particularly problematic when managing multiple peptides simultaneously.
The solution: Comprehensive labeling including:
- Peptide name and concentration
- Reconstitution date (for solutions)
- Expiration date
- Batch/lot number
- Storage requirements
- Researcher initials or project code
Use waterproof labels and permanent markers to ensure information remains legible throughout storage duration.
Retatrutide Storage FAQs
Q: Can I store retatrutide in a standard home refrigerator?
A: While not ideal, a standard home refrigerator can work for short-term storage if dedicated to this purpose. However, frequent door openings cause temperature fluctuations, and shared food storage increases contamination risk. Laboratory-grade refrigerators provide superior temperature stability and should be used when possible.
Q: What should I do if my freezer fails overnight?
A: First, assess the temperature and duration of the excursion. If lyophilized retatrutide remained below 25°C, it likely retained stability. Transfer immediately to functioning freezer. Document the event and consider analytical verification before critical experiments. If temperature exceeded 30°C for extended periods (>12 hours), replacement may be warranted.
Q: How can I tell if reconstituted retatrutide has gone bad?
A: Visual indicators include cloudiness, color changes, visible particles, or precipitate formation. However, some degradation occurs without visible signs. Adhering strictly to the 30-day post-reconstitution timeline and refrigeration requirements provides the best assurance of quality.
Q: Is it safe to transport retatrutide in checked airline luggage?
A: No. Checked baggage compartments experience temperature extremes (both freezing and heat) that will damage retatrutide. Always transport in carry-on luggage within an insulated cooler where you can monitor and maintain appropriate temperatures.
Q: Can I refreeze retatrutide that was accidentally thawed?
A: For lyophilized retatrutide that was briefly thawed (under 24 hours at refrigerator temperatures), one refreeze cycle is generally acceptable, though not ideal. For reconstituted solutions or repeated freeze-thaw events, do not refreeze—the cumulative damage compromises peptide integrity.
Q: How long can lyophilized retatrutide stay at room temperature during reconstitution?
A: Brief room temperature exposure (30-60 minutes) during reconstitution procedures is acceptable and unavoidable. However, return to refrigeration immediately after reconstitution is complete. Extended room temperature storage (multiple hours or days) is not recommended even for lyophilized material.
Conclusion: Maximizing Retatrutide Research Value Through Proper Storage
Understanding how to store retatrutide (refrigeration, travel & expiry) represents a fundamental competency for research professionals working with this promising triple-agonist peptide. The storage protocols outlined in this guide—from refrigeration temperatures and freeze-thaw prevention to travel procedures and quality assessment—directly impact experimental reproducibility and research outcomes.
Key implementation priorities:
- Establish temperature-controlled storage with monitoring systems for both routine refrigeration (2-8°C) and long-term freezing (-20°C or -80°C)
- Implement aliquoting protocols immediately upon receipt to prevent freeze-thaw damage throughout the research timeline
- Develop comprehensive SOPs covering receipt, storage, handling, and quality verification
- Invest in proper transport equipment for any situation requiring retatrutide movement between locations
- Maintain rigorous documentation of storage conditions, temperatures, and material usage
The investment in proper storage infrastructure and protocols pays dividends through reliable experimental data, reduced material waste, and confidence in research outcomes.
For research institutions seeking to establish or expand peptide research programs, partnering with suppliers committed to quality and proper handling ensures that materials arrive in optimal condition and include comprehensive storage guidance.
Next Steps for Research Professionals
Immediate actions:
- Audit current retatrutide storage conditions against recommendations in this guide
- Verify temperature monitoring systems are functioning and documented
- Review and update laboratory SOPs for peptide handling
- Assess aliquoting practices and implement improvements if needed
- Inspect current inventory for proper labeling and expiration tracking
Long-term improvements:
- Consider upgrading to dedicated laboratory-grade refrigeration equipment
- Implement digital inventory management systems for larger peptide libraries
- Establish formal training programs for personnel handling research peptides
- Develop relationships with trusted peptide suppliers offering technical support
By prioritizing proper storage protocols, research teams maximize the value of their peptide investments while ensuring the scientific integrity that drives meaningful discoveries in metabolic research and beyond.
References
[1] Stability of Peptides and Proteins. Journal of Pharmaceutical Sciences, 2023; 112(4): 892-908.
[2] Temperature-Dependent Degradation Kinetics of Research Peptides. International Journal of Peptide Research and Therapeutics, 2024; 30(2): 156-171.
[3] Long-term Storage Stability of Lyophilized Peptide Therapeutics. Pharmaceutical Development and Technology, 2023; 28(6): 701-715.
[4] Ultra-Low Temperature Storage for Biological Research Materials. Cryobiology, 2024; 108: 45-58.
[5] Photostability of Peptide Pharmaceuticals: Mechanisms and Prevention. European Journal of Pharmaceutical Sciences, 2023; 182: 106371.
[6] Freeze-Thaw Stability of Protein and Peptide Formulations. Journal of Pharmaceutical Sciences, 2024; 113(1): 23-39.
[7] Impact of Storage Conditions on Peptide Integrity: A Comprehensive Review. Advanced Drug Delivery Reviews, 2023; 195: 114756.
