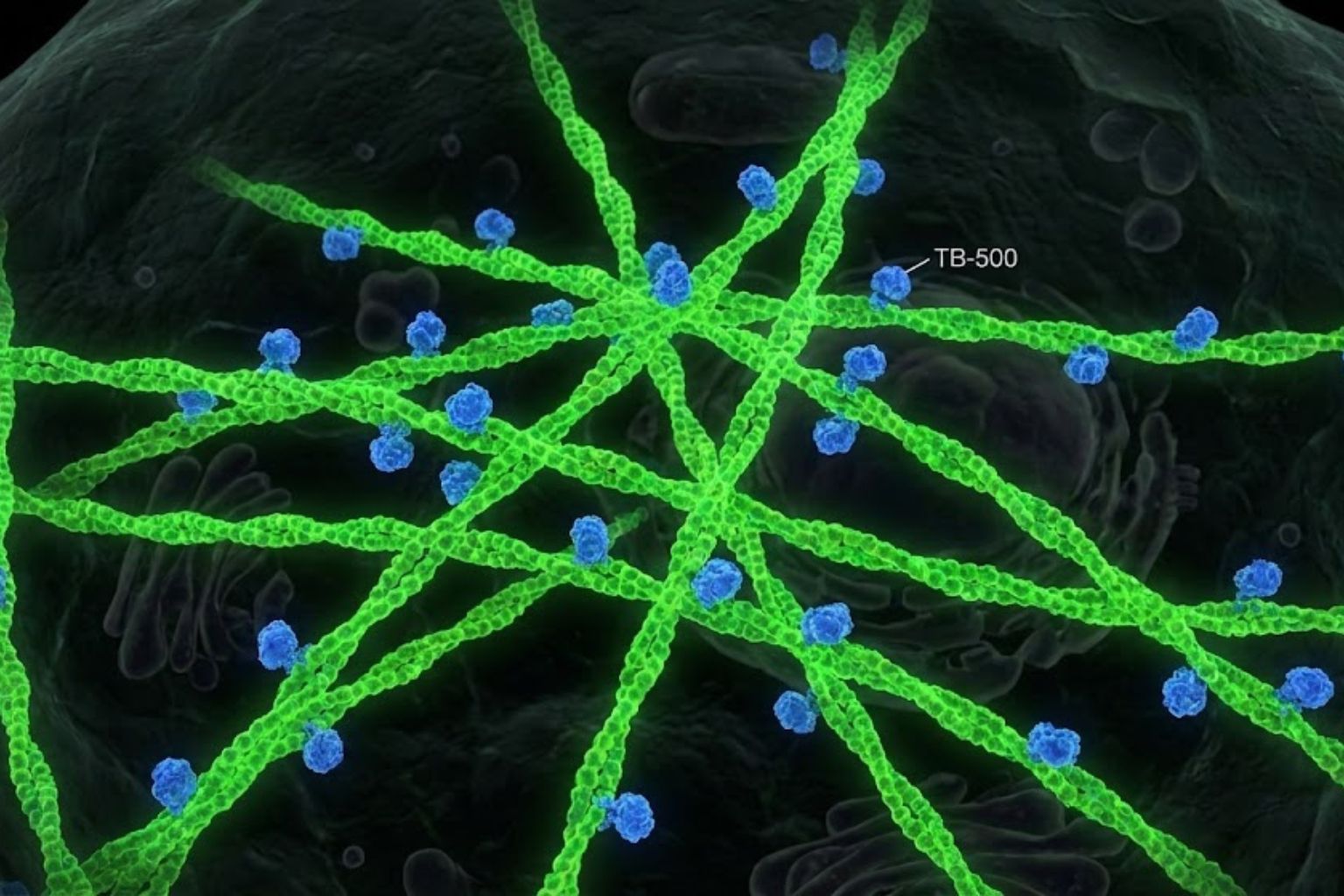
Unlocking the Cellular Mechanics of TB-500 (Thymosin Beta-4)
In the rapidly evolving landscape of regenerative medicine, few peptides have garnered as much interest as Thymosin Beta-4, and its synthetic derivative, TB-500. However, to truly understand the potential of this peptide in a research setting, one must look beyond the macroscopic results—the healed wounds or the recovered muscle tissue—and peer into the microscopic world of cellular drivers.
The TB-500 mechanism is not merely a hormonal signal; it is a fascinating study of cytoskeletal dynamics. Unlike growth factors that bind to surface receptors to trigger a downstream signaling cascade (like flipping a switch), TB-500 functions largely by entering the cell and modulating the very building blocks of the cell’s physical structure: actin.
This article provides a deep dive into the biophysics of this interaction, exploring how a simple chain of amino acids can orchestrate complex physiological processes ranging from cell migration to the formation of new blood vessels.
1. The Foundation: The Role of G-Actin Sequestration
To understand TB-500, one must first understand actin. Actin is the most abundant protein in eukaryotic cells, forming the cytoskeleton—the scaffolding that gives a cell its shape, allows it to divide, and enables it to move. Actin exists in two states:
-
G-Actin (Globular Actin): The free, monomeric form floating in the cytoplasm.
-
F-Actin (Filamentous Actin): The polymerised form, arranged in long chains or meshworks that create the cell’s skeleton.
The Polymerisation Paradox
For a cell to move or change shape, it must rapidly disassemble its F-actin structure and reassemble it in a new direction. This requires a ready supply of G-actin. However, G-actin has a natural tendency to polymerise spontaneously into F-actin if left unchecked. If all G-actin turned into F-actin, the cell would become rigid and static, unable to respond to injury.
The TB-500 “Buffer” Mechanism
This is where the TB-500 mechanism becomes critical. TB-500 acts as an actin-sequestering molecule. It binds to G-actin monomers in a 1:1 ratio. By wrapping itself around the actin monomer, it acts as a molecular “lock,” preventing the actin from adding itself to a growing filament prematurely.
This creates a “buffer pool” of high-concentration G-actin stored within the cell. Under normal conditions, TB-500 keeps this pool dormant. However, when a cell receives a signal to migrate or repair—such as during the acute phase of wound healing—TB-500 releases this sequestered actin.
Because the cell now has a massive, instantly available supply of raw materials (G-actin), it can engage in explosive polymerisation. This allows for rapid cytoskeletal remodeling, enabling the cell to extend protrusions and crawl toward a wound site at speeds that would be impossible if it had to synthesize new actin from scratch. In essence, TB-500 transforms the cell from a static structure into a rapid-response unit.
2. Cell Migration: The Engine of Tissue Repair
The primary physiological outcome of actin sequestration is enhanced cell migration. This is the cornerstone of tissue repair. If cells cannot move, wounds cannot close.
The Mechanics of “Crawling”
Cell migration is a cyclical process powered by the cytoskeleton. To move, a cell must:
-
Protrude: Extend the leading edge of the cell (creating structures called lamellipodia and filopodia).
-
Adhere: Grip the extracellular matrix at the front.
-
Contract: Pull the cell body forward.
-
Retract: Release the grip at the rear.
Step 1, protrusion, is driven entirely by the rapid polymerisation of actin filaments pushing against the cell membrane. Research indicates that the TB-500 mechanism is pivotal here. By delivering G-actin to the leading edge of the cell, TB-500 fuels the “treadmilling” of actin filaments, where new monomers are added to the front while old ones fall off the back.
Clinical Implications: Corneal and Dermal Healing
Studies on corneal healing have provided some of the most visible evidence of this mechanism. The cornea requires perfect transparency, which means scarring must be minimized. TB-500 has been shown to accelerate the migration of keratinocytes (skin cells) and epithelial cells to bridge the gap in damaged tissue. By speeding up the closure of the wound, the window of opportunity for bacterial infection is reduced, and the likelihood of aberrant scarring is diminished.
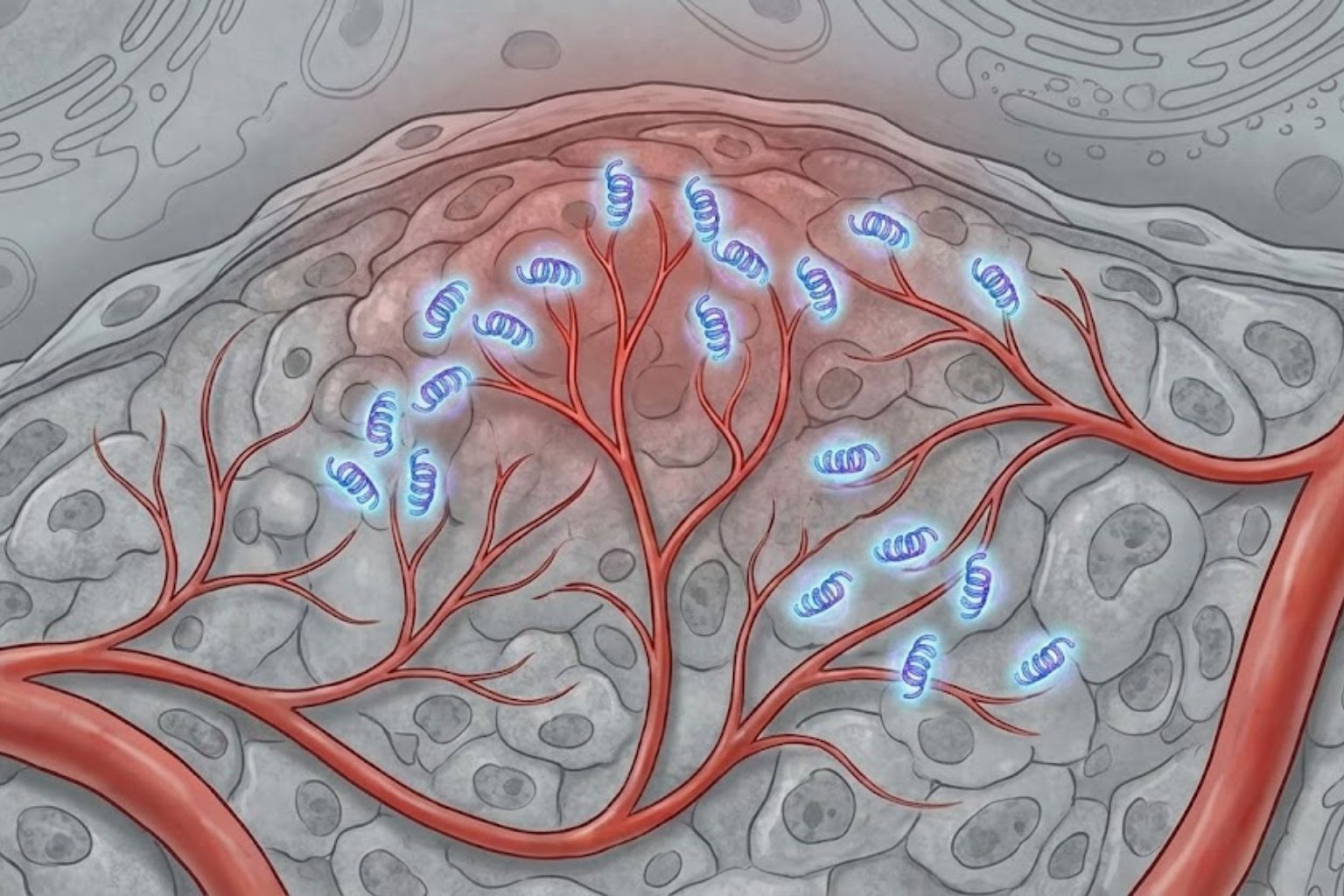
3. Angiogenesis: Building the Supply Lines
A tissue cannot repair itself if it is starved of oxygen and nutrients. Therefore, the repair process is inextricably linked to angiogenesis—the physiological process through which new blood vessels form from pre-existing vessels.
The Endothelial Driver
Angiogenesis relies on the behavior of endothelial cells, which line the interior of blood vessels. To form a new capillary, endothelial cells must break out of their existing vessel wall, migrate into the damaged tissue, and proliferate to form a new tube.
Research indicates that TB-500 is a potent pro-angiogenic factor. It operates on two levels:
-
Direct Stimulation: Through the actin-binding mechanism described above, it physically enables endothelial cells to migrate faster and farther into the hypoxic (oxygen-starved) tissue.
-
Signaling Pathways: TB-500 has been observed to upregulate the expression of Vascular Endothelial Growth Factor (VEGF), the master regulator of blood vessel growth.
Cardiac Tissue and Ischemia
This mechanism is of particular interest in cardiac research. Following a myocardial infarction (heart attack), heart muscle dies due to a lack of blood flow. The “scar” that forms is non-conductive and non-contractile, leading to heart failure. Studies suggest that TB-500 can induce angiogenesis in the border zone of the infarct, preserving heart tissue that would otherwise die and potentially allowing for functional recovery rather than just scar formation.
4. The “Short Segment” Hypothesis: Why LKKTET Matters
TB-500 is not the full Thymosin Beta-4 protein; it is a synthetic fraction. Full-length Thymosin Beta-4 consists of 43 amino acids. TB-500 typically replicates the N-terminal sequence, specifically amino acids 17 through 23. This sequence is LKKTET (Leucine-Lysine-Lysine-Threonine-Glutamic Acid-Threonine).
The Active Site
Why use a fragment instead of the whole protein? Research suggests that the LKKTET sequence is the specific active site responsible for the actin-binding properties. This is the “business end” of the molecule.
-
Targeted Action: By isolating this sequence, researchers can utilize the specific actin-modulating effects without necessarily triggering other, less understood pathways associated with the C-terminus of the full protein.
-
Bioavailability: Smaller peptides generally have better tissue penetration. In a research setting, the lower molecular weight of TB-500 allows it to navigate the extracellular matrix more easily to reach target cells.
-
Stereochemistry: The LKKTET sequence fits into the cleft of the G-actin monomer like a key in a lock. This precise steric fit is what allows the peptide to inhibit salt-induced polymerisation, maintaining the liquid pool of actin.
This “Short Segment” hypothesis validates the idea that one does not need the entire protein to replicate its most vital reparative functions.
5. Anti-Inflammatory Pathways: The Silent Healer
While cytoskeletal mechanics are the “hardware” of repair, inflammation is the “software.” Chronic inflammation acts as a roadblock to healing. If the immune system remains in an attack phase (clearing debris and fighting pathogens), the regenerative phase (rebuilding tissue) cannot begin.
Modulating Cytokines
Beyond structure, the TB-500 mechanism influences the chemical environment of the wound. It has been observed to downregulate pro-inflammatory cytokines, specifically TNF-alpha (Tumor Necrosis Factor) and IL-1 beta.
By lowering the volume of these inflammatory signals, TB-500 helps transition the wound environment from a hostile, inflammatory state to a permissive, remodeling state. This is crucial for preventing fibrosis.
The Anti-Fibrotic Effect
Fibrosis (scarring) is essentially a chaotic over-reaction to injury. It involves the excessive deposition of collagen by fibroblasts. TB-500 appears to modulate fibroblast behavior. By ensuring that cells migrate in an organized fashion and by reducing the inflammatory signaling that drives panic-level collagen dumping, TB-500 promotes the alignment of collagen fibers. This results in tissue that more closely resembles the original, uninjured tissue, rather than a thick, non-functional scar.
6. Differentiation vs. Recruitment: The Homing Beacon
A common misconception in regenerative medicine is that peptides like TB-500 turn stem cells into new tissue (differentiation). The reality is more nuanced and perhaps more clever.
TB-500 acts less like a factory manager and more like a recruiter. An important distinction in the TB-500 mechanism is that it promotes the survival and migration of existing cells rather than forcing the differentiation of stem cells directly.
The “Signal Flare”
When tissue is injured, stem cells and progenitor cells (like Epicardial Progenitor Cells in the heart) act as a reserve force. TB-500 acts as a chemoattractant—a chemical homing beacon. It creates a concentration gradient that guides these stem cells from their niches (like the bone marrow or vessel walls) to the site of injury.
Once the stem cells arrive, TB-500 promotes their survival in the harsh, low-oxygen environment of the wound (an effect known as anti-apoptosis). By keeping these progenitor cells alive and moving them to the right location, TB-500 allows the body’s natural signaling pathways to take over and direct their differentiation into the specific tissue types needed (muscle, skin, or blood vessels).
The TB-500 mechanism represents a sophisticated interplay of cytoskeletal management and cellular signaling. It is not a “magic bullet” that simply grows tissue; rather, it is a master regulator of cellular logistics.
-
It manages inventory: By sequestering G-actin, it ensures cells have the raw materials for movement.
-
It facilitates transport: By driving angiogenesis, it ensures the repair site has blood flow.
-
It manages the workforce: By recruiting stem cells and reducing inflammation, it creates an environment where repair can actually occur.
For researchers, understanding this actin-binding capability is key. It moves the conversation from “does it work?” to “how can we leverage this specific mechanism?” Whether investigating cardiac recovery, soft tissue injury, or corneal repair, the biophysics of TB-500 offer a compelling roadmap for the future of regenerative science.
