 Why Storage Conditions Determine Thymulin Bioactivity
Why Storage Conditions Determine Thymulin Bioactivity
1)More Than Just Amino Acids
In the vast library of peptides used in UK biomedical research, Thymulin stands apart. Most peptides are defined solely by their sequence of amino acids—a string of beads that, once assembled, is ready to work. Thymulin is different. It is a “metallopeptide.”
Discovered in the 1970s and originally termed Facteur Thymique Sérique (FTS), this nonapeptide (Glu-Ala-Lys-Ser-Gln-Gly-Gly-Ser-Asn) presents a unique biochemical paradox: you can synthesize the peptide chain perfectly, purify it to 99%, and store it in a sterile pen peptide, yet it can be completely biologically inert.
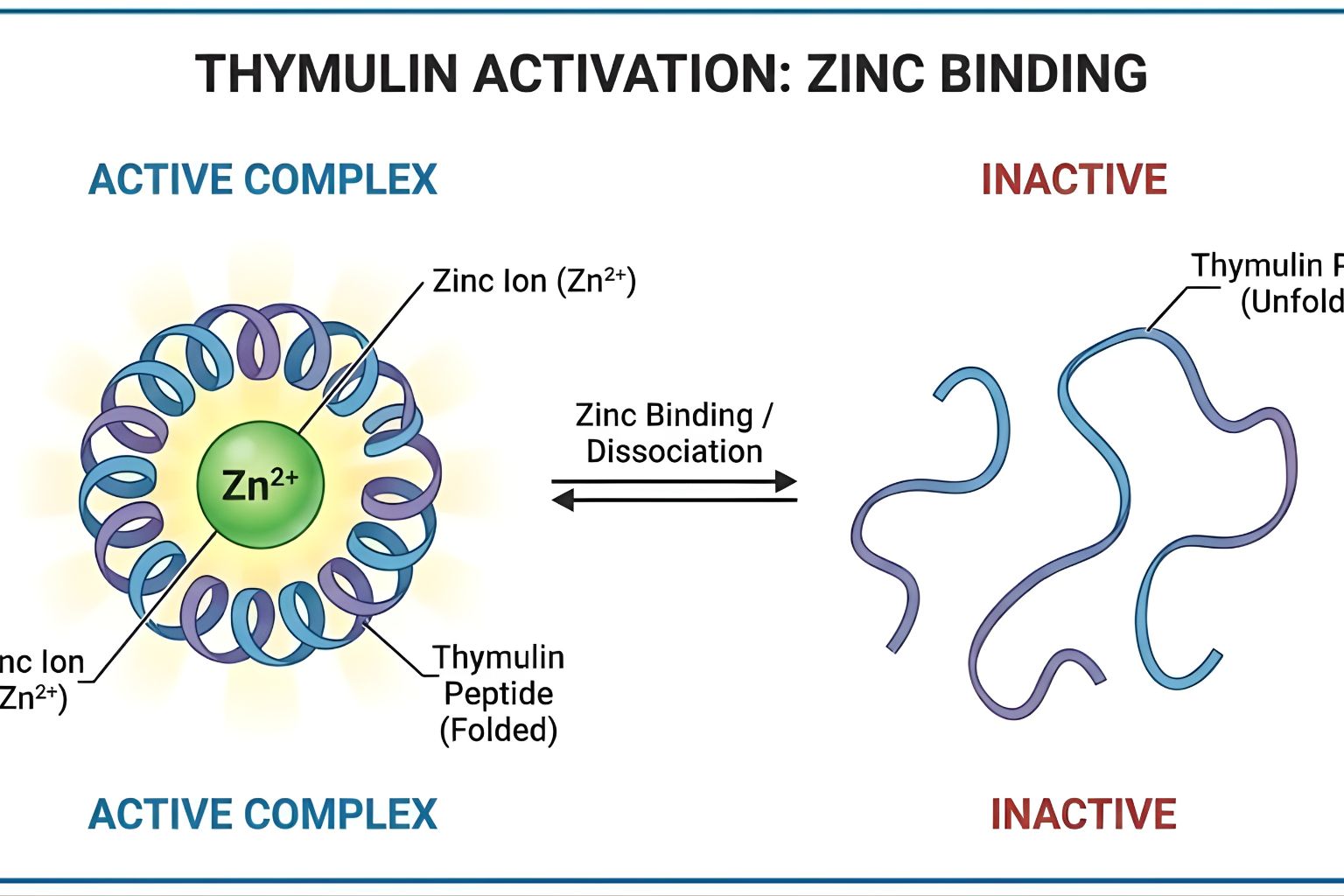
Why? Because the peptide chain is merely a scaffold. The biological engine is a single atom of Zinc.
Without Zinc, the peptide cannot bind to T-cell receptors. It cannot induce differentiation. It effectively does not exist as a signalling molecule. For researchers, this means that Thymulin bioactivity is not just a question of peptide stability; it is a question of coordination chemistry.
This blog explores the deep science behind the storage protocols. We will move beyond the “what” (keep it cold) and dive into the “why” (thermodynamics and ion dissociation), explaining how environmental factors like pH, temperature, and chelating agents dictate the success or failure of your immunomodulation experiments.
2) The Biochemistry of the FTS-Zn Complex
To understand how storage kills Thymulin bioactivity, we must first look at the structure of the active molecule.
The Coordination Bond
Unlike the covalent bonds that hold the amino acids together (which are strong and hard to break), the bond between the Thymulin peptide and the Zinc ion is a “coordination complex.”
-
Stoichiometry: The binding occurs in a strict 1:1 molar ratio. One peptide molecule grabs one Zinc ion.
-
The Grip: The peptide folds in a specific way to create a “pocket” for the Zinc. Specific residues (likely the terminal glutamic acid and others) donate electrons to the Zinc ion, holding it in place.
Conformational Change
This is the critical concept. When Zinc binds to FTS, it forces the peptide backbone to twist into a specific three-dimensional shape (conformation).
-
Active Shape: With Zinc, the molecule is rigid and shaped perfectly to fit into the receptor on the surface of a T-cell.
-
Inactive Shape: Without Zinc, the peptide is “floppy” and disordered. It physically cannot trigger the receptor. This is why Thymulin bioactivity is binary: no Zinc, no signal.
3) The pH Factor: Why Acidity is the Enemy
The most common way researchers accidentally destroy Thymulin bioactivity is through pH mismanagement. The coordination bond holding the Zinc is extremely sensitive to proton concentration (acidity).
The Mechanism of Zinc Loss
In chemistry, protons (H+) compete with metal ions for binding sites.
-
Neutral pH (7.0–7.4): The amino acid residues are in the correct charge state to hold onto the Zinc. The grip is tight.
-
Acidic pH (< 6.0): As the solution becomes more acidic, protons flood the system. They protonate the binding sites on the peptide. Effectively, the protons “push” the Zinc ion out of the pocket.
Storage Implication
If you reconstitute Thymulin in unbuffered water (which often absorbs CO2 from the air to become carbonic acid, dropping to pH 5.5), you are creating an environment that favors dissociation. The Zinc falls off, leaving you with inactive FTS. You may still have the peptide, but you have lost the Thymulin bioactivity.
-
The Fix: Always use a buffered solvent like PBS (Phosphate Buffered Saline) or Hepes, adjusted to pH 7.2–7.4, to “lock” the Zinc in place.
4) The Chelation Threat: Invisible Robbery
Sometimes, the Zinc doesn’t fall off; it is stolen. This is the risk of “chelating agents.”
What is Chelation?
A chelator is a molecule designed to grab metal ions with extreme strength. Common laboratory buffers often contain EDTA (Ethylenediaminetetraacetic acid) or Citrate. These are usually added to stop enzymes (proteases) from working, as proteases also need metals.
The Tug-of-War
If you place Thymulin in a buffer containing EDTA:
-
The EDTA has a much higher affinity (binding strength) for Zinc than the Thymulin peptide does.
-
The EDTA rips the Zinc ion straight out of the Thymulin complex.
-
Thymulin bioactivity drops to zero instantly.
Laboratory Protocol Warning
Many commercial “Peptide Reconstitution Buffers” are generic. They may contain EDTA as a preservative. For standard peptides, this is fine. For a metallopeptide like Thymulin, it is catastrophic. You must verify the chemical composition of every solvent to ensure it is “Metal-Free” or at least “Chelator-Free.”
5) Temperature and Thermodynamics
We know we should store peptides in the fridge, but how does heat specifically affect the Zinc bond?
Kinetic Energy and Dissociation
All chemical bonds vibrate. As temperature increases, kinetic energy increases, and vibrations become more violent.
-
The Dissociation Constant (Kd): This number represents how likely the complex is to break apart. As temperature rises, the equilibrium shifts. The peptide struggles to hold onto the Zinc.
-
The Break: If the sample gets too warm, the Zinc ion may dissociate. Once it is floating free in the solution, it may not easily find its way back into the binding pocket, especially if the peptide chain begins to unfold or aggregate.
The Freeze-Thaw Crystal Effect
While heat causes dissociation, freezing causes physical destruction.
-
Micro-environment Changes: As water freezes into pure ice crystals, the remaining liquid becomes a hyper-concentrated brine of salts and peptides. This drastic shift in ionic strength and pH (even in frozen states) can destabilize the Zinc complex.
-
Outcome: Repeated cycling weakens the structural integrity required for Thymulin bioactivity.
6) Measuring Bioactivity: How Do We Know It’s Working?
Since we cannot see the Zinc bond, how do scientists measure Thymulin bioactivity? The classic method is the Rosette Inhibition Assay.
The Assay Mechanism
-
T-Cells: Researchers take T-cells (often from mouse spleens). normally, these cells form “rosettes” when mixed with sheep red blood cells (they clump together in a flower shape).
-
The Trigger: Active Thymulin inhibits this rosette formation. It changes the surface markers of the T-cell.
-
The Readout:
-
High Bioactivity: Few rosettes form. The Thymulin is working.
-
Low Bioactivity: Many rosettes form. The Thymulin failed to inhibit them.
-
The “False Negative” Trap
If a researcher uses degraded Thymulin in this assay, they will see rosettes forming and assume “Thymulin doesn’t work in this context.” In reality, the molecule simply lacked the Zinc required to flip the switch on the T-cell surface. This highlights why storage is not just a logistical detail—it is the foundation of data validity.
7) Structure-Activity Relationship (SAR)
The concept of Structure-Activity Relationship (SAR) is central to pharmacology. It states that the biological effect of a drug is determined by its 3D shape.
Thymulin’s Unique SAR
For most peptides, SAR is about the amino acid sequence. For Thymulin, SAR is about the tertiary structure induced by Zinc.
-
The Receptor Interface: The receptor on the thymocyte (pre-T-cell) is like a lock with a very specific keyhole.
-
The Zinc Key: The Zinc ion acts as a bridge. It may interact directly with residues in the receptor, or it may simply hold the peptide in the shape required to enter the lock.
-
Loss of SAR: When storage conditions fail (low pH, heat, EDTA), the SAR collapses. The peptide reverts to a random coil structure. It effectively becomes “noise” to the receptor.
8) The Role of Oxidation in Bioactivity Loss
Zinc isn’t the only point of failure. The peptide backbone itself contains amino acids prone to oxidation, specifically Cysteine (if present in variants) or Methionine/Tryptophan residues.
Oxidative Stress
If the pen peptide is left open to air, oxygen molecules attack the side chains of the amino acids.
-
Steric Hinderance: An oxidized residue is bulkier (it has an extra oxygen atom attached).
-
The Blockage: This extra bulk can physically prevent the peptide from fitting into the receptor or from wrapping around the Zinc ion.
-
Prevention: This is why purging pen peptides with inert gas (like Nitrogen) or strictly limiting headspace is crucial for maintaining Thymulin bioactivity.
9) Real-World Research Implications
What does this complex chemistry mean for a UK researcher managing a grant-funded project?
-
Reproducibility Crisis: If Batch A was stored at pH 7.4 and Batch B was stored at pH 6.0, they are effectively two different drugs. Your data will show massive variance that you cannot explain biologically.
-
Dose-Response Confusion: You might think you need a higher dose to see an effect. In reality, you are just compensating for the fact that only 10% of your peptide still holds its Zinc. You are overdosing the peptide backbone to get enough Zinc-complexed molecules to the cells.
-
Wasted Resources: Cell cultures and animal models are expensive. Using a reagent with compromised Thymulin bioactivity wastes these valuable resources.

10) Summary of Science-Backed Storage Rules
Based on the coordination chemistry and thermodynamics discussed, here are the non-negotiable rules for preserving Thymulin bioactivity:
-
The pH Rule: Reconstitute only in buffered solutions (pH 7.0–7.4). Avoid unbuffered water.
-
The Chelator Rule: strictly ban EDTA, Citrate, and other chelators from any buffer contacting Thymulin.
-
The Cold Rule: Store lyophilised at -20°C and reconstituted at 4°C to lower kinetic energy and prevent dissociation.
-
The Time Rule: Use reconstituted solutions within 7-14 days. The equilibrium of the Zinc bond is not permanent; it slowly favors dissociation over time in solution.
11)Respect the Metal
Thymulin is a fascinating example of nature’s elegance—a biological switch that requires a metal key. But this elegance comes with fragility. For the scientist, understanding the role of Zinc is the difference between blindly following a protocol and truly mastering the variable.
Thymulin bioactivity is not guaranteed by the label on the bottle. It is maintained by the rigorous application of chemical principles in the laboratory. By controlling pH, avoiding chelators, and managing temperature, you preserve the delicate FTS-Zn complex, ensuring that when your peptide meets the cell, the message is delivered loud and clear.
12) Frequently Asked Questions (Science Edition)
Q: Can I add extra Zinc to the solution to force it to bind? A: Theoretically, yes. Some researchers add a 1:1 molar equivalent of Zinc Chloride (ZnCl2) to the buffer to shift the equilibrium toward the bound state. However, excess free Zinc can be toxic to cells (cytotoxicity), so this must be calculated precisely.
Q: Is the peptide active in vivo if injected without Zinc? A: This is debated. Blood serum contains Zinc. Some argue that injecting FTS (without Zinc) allows it to scavenge Zinc from the bloodstream and become active. However, for in vitro (Petri dish) work, there is no Zinc reservoir, so you must supply the complex fully formed.
Q: Does the Zinc bond break during freezing? A: It can. The physical stress of crystallization and the concentration of salts in the remaining liquid phase can force dissociation. This is why flash-freezing or avoiding freezing of the liquid entirely is preferred.
Q: Why is pH 7.4 the magic number? A: It mimics the physiological pH of blood and lymph, where Thymulin naturally operates. It is the pH at which the amino acid side chains are in the optimal ionic state to coordinate with the Zinc ion without letting go.
13) Next Steps for Research Professionals
-
Check your water source: Is your lab water slightly acidic? Test it today.
-
Audit your buffers: Read the label on your PBS bottle. Does it say “0.5mM EDTA”? If so, do not use it for Thymulin.
-
Educate the team: Share this article with your lab group so everyone understands that Thymulin is a metallopeptide, not just a standard protein.
14) References
-
Dardenne, M., et al. (1982). Contribution of zinc and other metals to the biological activity of the serum thymic factor. PNAS.
-
Bach, J. F. (1983). Thymulin (FTS-Zn). Clinical Immunology and Allergy.
-
Gastinel, L. N., et al. (1984). Structural guarantees for the biological activity of the thymus-derived peptide, thymulin. Biochimica et Biophysica Acta.
