
When researchers and laboratories begin investigating how long does tirzepatide take to work, understanding the precise timeline of this dual GIP/GLP-1 receptor agonist becomes critical for experimental design and outcome measurement. Tirzepatide, a novel peptide gaining significant attention in metabolic research, demonstrates a unique pharmacokinetic profile that distinguishes it from other peptide compounds. The question of efficacy timing isn’t merely academic—it directly impacts research protocols, dosing schedules, and the interpretation of experimental results across diverse study models.
This comprehensive guide examines the temporal dynamics of tirzepatide action, from initial receptor binding through peak therapeutic effects, providing researchers with evidence-based timelines essential for rigorous scientific investigation.
Key Takeaways
- Initial effects of tirzepatide typically manifest within 4-7 days of administration in research models, with receptor engagement occurring within hours
- Peak efficacy generally occurs between 4-8 weeks of consistent dosing, though individual research parameters may extend this timeline to 20+ weeks
- Dose-dependent response significantly influences the timeline, with higher concentrations accelerating observable effects in controlled settings
- Pharmacokinetic profile shows a half-life of approximately 5 days, enabling once-weekly administration protocols in most research applications
- Research-grade purity and proper reconstitution directly impact the compound’s bioavailability and subsequent timeline of action
Understanding Tirzepatide: Mechanism and Research Applications
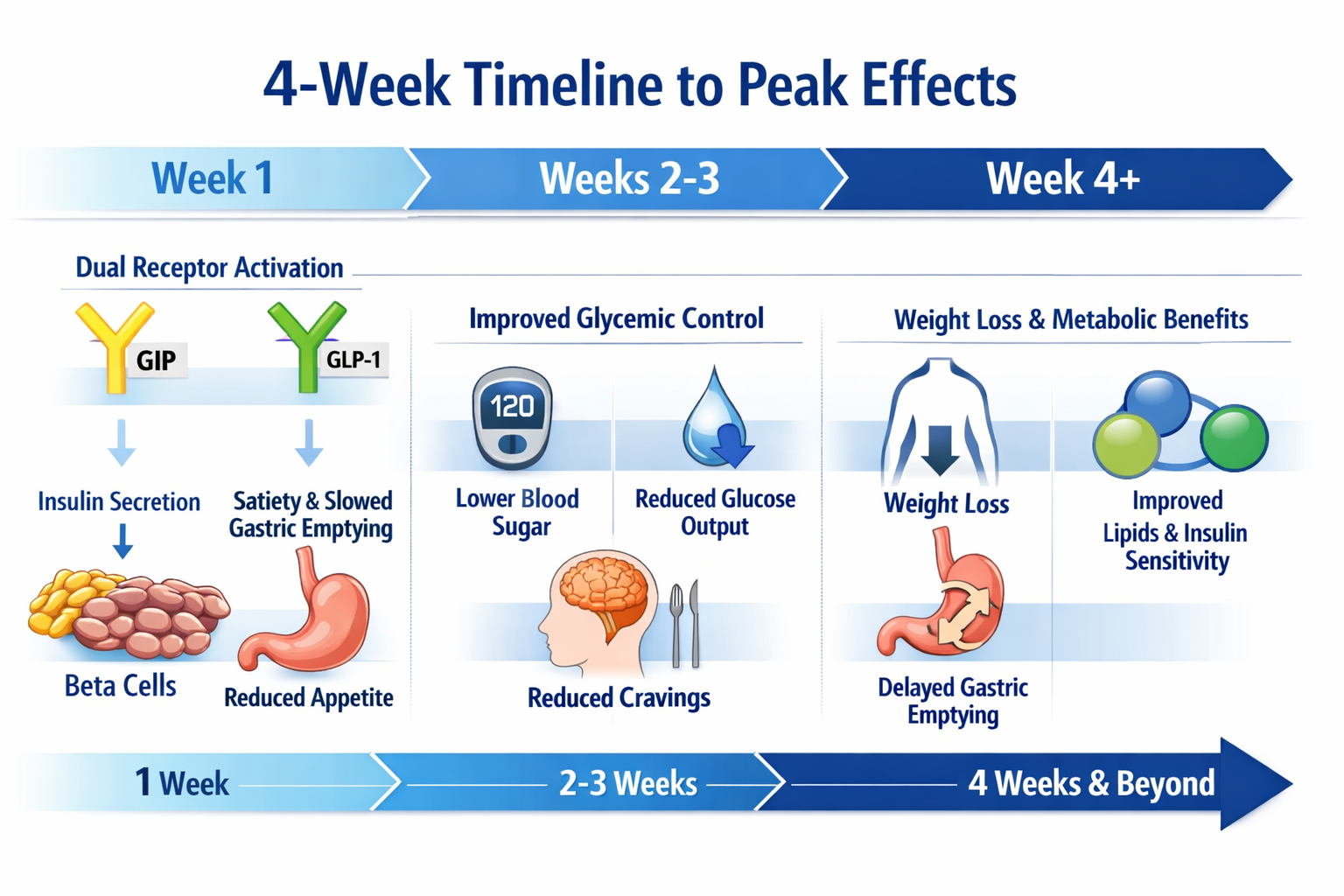
Tirzepatide represents a breakthrough in peptide research as the first dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This innovative compound’s mechanism of action provides researchers with a unique tool for investigating metabolic pathways, appetite regulation, and glucose homeostasis [1].
The Dual Agonist Mechanism ⚗️
Unlike single-target peptides, tirzepatide activates both GIP and GLP-1 receptors simultaneously, creating a synergistic effect that researchers find particularly valuable for metabolic studies. The GIP component enhances insulin secretion and may influence fat metabolism, while the GLP-1 component regulates glucose-dependent insulin release, suppresses glucagon secretion, and modulates gastric emptying [2].
This dual mechanism explains why how long does tirzepatide take to work varies depending on which physiological endpoint researchers are measuring. Glucose regulation markers may respond within days, while metabolic remodeling effects require weeks to months of observation.
Pharmacokinetic Properties
The pharmacokinetic profile of tirzepatide significantly influences its timeline of action:
| Parameter | Value | Research Implication |
|---|---|---|
| Half-life | ~5 days | Supports weekly dosing protocols |
| Time to peak concentration | 8-72 hours | Early receptor engagement |
| Steady-state | 4 weeks | Consistent effects after 4 doses |
| Bioavailability | ~80% (subcutaneous) | High absorption efficiency |
These characteristics mean that while initial receptor binding occurs rapidly, achieving steady-state concentrations—and therefore maximum, consistent effects—requires approximately four weeks of regular administration [3].
For researchers sourcing high-purity research peptides, understanding these pharmacokinetic properties is essential for experimental design and timeline planning.
How Long Does Tirzepatide Take to Work? The Complete Timeline
The timeline for tirzepatide’s effects in research settings follows a predictable pattern, though individual experimental variables can influence specific outcomes. Understanding this progression helps researchers establish appropriate measurement intervals and interpret results accurately.
Hours 1-24: Initial Receptor Engagement 🕐
Within the first hours following administration, tirzepatide molecules begin binding to GIP and GLP-1 receptors throughout target tissues. Research demonstrates:
- Receptor binding: Initiates within 1-2 hours of subcutaneous administration
- Peak plasma concentration: Achieved within 8-72 hours depending on dose
- Early glucose effects: May be detectable within 24 hours in responsive models
At this stage, researchers using sensitive assays can detect early biochemical changes, though physiologically significant effects remain minimal.
Days 1-7: Acute Phase Response
The first week represents the acute response phase where initial therapeutic effects become measurable:
Observable Changes:
- ✅ Glucose-dependent insulin secretion enhancement
- ✅ Reduction in postprandial glucose excursions
- ✅ Initial appetite modulation signals
- ✅ Gastric emptying rate changes
Research models typically show the first statistically significant metabolic changes during this period, though these effects continue to build with subsequent doses [4].
Weeks 2-4: Building to Steady State
This critical period marks the progression toward steady-state concentrations, where how long does tirzepatide take to work becomes most relevant for sustained research outcomes:
Week 2-3 Developments:
- Progressive accumulation toward steady-state plasma levels
- Increasingly consistent day-to-day effects
- Enhanced metabolic adaptation
- More pronounced changes in measured parameters
Week 4 Milestone:
“Steady-state concentrations of tirzepatide are achieved after approximately 4 weeks of once-weekly administration, representing the point at which consistent, maximal receptor engagement occurs.” [5]
By week four, researchers can expect stable, reproducible effects that represent the compound’s full pharmacological potential at the administered dose.
Weeks 4-12: Peak Efficacy Development
While steady-state concentrations occur at four weeks, the full spectrum of tirzepatide’s effects—particularly those involving metabolic remodeling and physiological adaptation—continues developing through week 12 and beyond.
Progressive Effects Timeline:
| Week Range | Primary Developments | Research Measurements |
|---|---|---|
| 4-8 weeks | Robust metabolic changes | Body composition, glucose markers |
| 8-12 weeks | Sustained adaptation | Long-term metabolic parameters |
| 12-20 weeks | Maximum observed effects | Comprehensive outcome measures |
| 20+ weeks | Maintenance phase | Durability studies |
Research protocols examining comprehensive metabolic outcomes typically require observation periods of 12-20 weeks to capture tirzepatide’s full effect profile [6].
Factors Influencing Individual Timeline Variability
Several experimental variables affect how long does tirzepatide take to work in specific research contexts:
Dose-Dependent Factors:
- Higher doses generally produce faster, more pronounced effects
- Dose escalation protocols delay peak effects but improve tolerability
- Starting dose significantly impacts initial response timeline
Model-Specific Variables:
- Baseline metabolic status of research subjects
- Concurrent experimental interventions
- Genetic or phenotypic variations in receptor expression
- Environmental and dietary controls
Compound Quality Considerations:
- Peptide purity and integrity
- Proper reconstitution procedures
- Storage conditions and handling
- Time since reconstitution
Researchers utilizing premium research-grade peptides minimize variability by ensuring consistent compound quality across experimental batches.
Dosing Protocols and Their Impact on Tirzepatide’s Timeline
The specific dosing protocol employed in research significantly influences how long does tirzepatide take to work and the trajectory of observable effects. Understanding standard protocols helps researchers design appropriate timelines and measurement schedules.
Standard Research Dosing Schedules
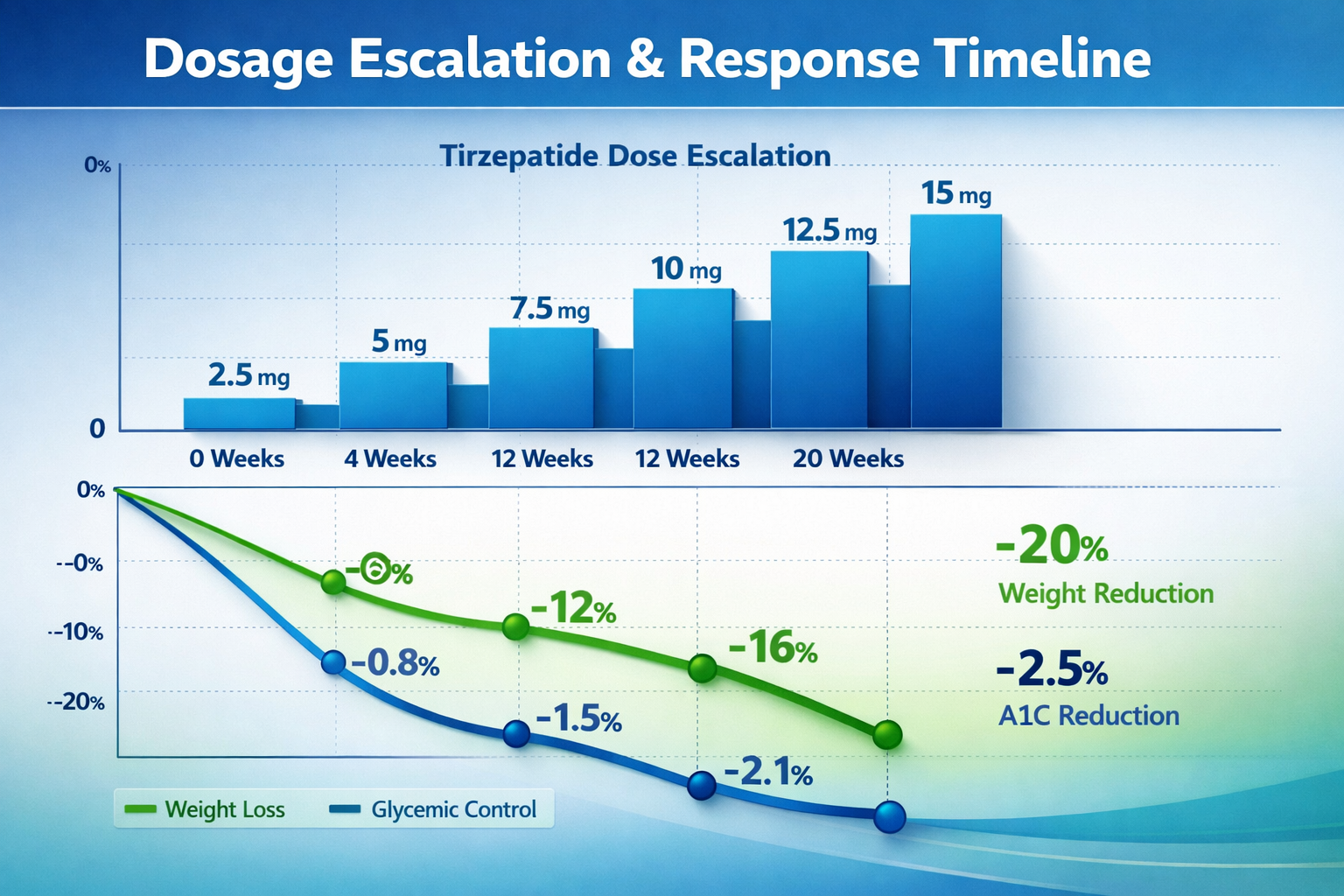
Research applications of tirzepatide typically follow escalating dose protocols that balance efficacy with experimental tolerability:
Typical Escalation Protocol:
- Initiation Phase: 2.5 mg weekly (Weeks 1-4)
- Allows initial adaptation
- Establishes baseline response
- Minimizes acute adverse reactions
- Escalation Phase: Progressive increases every 4 weeks
- 2.5 mg → 5 mg (Week 5)
- 5 mg → 7.5 mg (Week 9)
- 7.5 mg → 10 mg (Week 13)
- 10 mg → 12.5 mg → 15 mg (as needed)
- Maintenance Phase: Optimal dose continuation
- Determined by research endpoints
- Typically 10-15 mg for metabolic studies
- Maintained for duration of observation period
This graduated approach means that while initial effects occur within days, achieving maximum efficacy at the target dose requires the full escalation timeline—often 12-20 weeks for protocols reaching 10-15 mg doses [7].
Impact of Starting Dose on Response Timeline
Research comparing different starting doses reveals important timeline differences:
Higher Initial Doses (5 mg start):
- ⚡ Faster achievement of therapeutic effects
- ⚡ More rapid metabolic changes
- ⚠️ Increased incidence of acute side effects
- ⚠️ Higher discontinuation rates in some models
Lower Initial Doses (2.5 mg start):
� Gradual, sustained response development
� Better experimental tolerability
� Lower discontinuation rates
- ⏱️ Extended timeline to maximum effects
For most research applications, the standard 2.5 mg starting dose offers the optimal balance, though this extends the overall timeline to peak efficacy.
Maintenance Dosing Considerations
Once target doses are achieved and steady-state is established, maintenance dosing continues to produce consistent effects. Research examining long-term administration shows:
- Sustained efficacy: Effects maintain without tachyphylaxis through 72+ weeks of observation
- Consistent pharmacokinetics: Weekly dosing maintains therapeutic concentrations
- Durability: No significant decline in measured outcomes over extended periods
Understanding these maintenance characteristics helps researchers plan long-duration studies and interpret sustained-effect data [8].
Special Dosing Scenarios in Research
Certain research applications may employ modified dosing strategies that alter the standard timeline:
Accelerated Protocols:
- Shorter escalation intervals (every 2 weeks vs. 4 weeks)
- Faster achievement of target dose
- Compressed timeline to peak effects
- May increase acute response variability
Fixed-Dose Studies:
- No escalation; single dose throughout
- Simpler timeline interpretation
- Steady-state achieved by week 4
- Useful for dose-comparison research
Intermittent Dosing:
- Non-weekly schedules for specific research questions
- Altered pharmacokinetic profiles
- Modified timeline expectations
- Requires careful pharmacological modeling
Researchers can explore various tirzepatide formulations suitable for different experimental protocols and dosing requirements.
Measuring Tirzepatide’s Effects: What to Expect and When
Understanding how long does tirzepatide take to work requires knowing which specific effects to measure and when they become detectable. Different research endpoints follow distinct timelines, making measurement timing critical for accurate data collection.
Early-Response Biomarkers (Days to Weeks)
Certain biochemical and physiological markers respond rapidly to tirzepatide administration, providing early evidence of compound activity:
Glycemic Parameters (1-7 Days):
- Fasting glucose levels: Detectable changes within 3-7 days
- Postprandial glucose excursions: Reduced within 24-48 hours
- Insulin secretion patterns: Modified within first week
- Glucose variability: Decreased within days of initiation
Appetite and Intake Markers (1-14 Days):
- Food intake reduction: Observable within 24-72 hours
- Satiety signaling: Enhanced within first week
- Meal frequency: Changes within 3-7 days
- Preference patterns: May shift within 1-2 weeks
These early markers provide researchers with rapid feedback on compound bioactivity and help confirm proper administration and dosing [9].
Intermediate-Response Outcomes (Weeks to Months)
The most commonly studied tirzepatide effects emerge over weeks to months, representing the compound’s primary research applications:
Metabolic Remodeling (4-12 Weeks):
| Outcome Measure | Detectable Change | Near-Maximum Effect | Measurement Frequency |
|---|---|---|---|
| Body weight/composition | 2-4 weeks | 20-28 weeks | Weekly to biweekly |
| HbA1c levels | 8-12 weeks | 12-24 weeks | Every 4 weeks |
| Lipid profiles | 4-8 weeks | 12-20 weeks | Every 4-8 weeks |
| Insulin sensitivity | 2-4 weeks | 8-16 weeks | Every 2-4 weeks |
Physiological Adaptations (4-16 Weeks):
- Cardiovascular parameters: Progressive improvement over 12+ weeks
- Hepatic function markers: Changes detectable by 8-12 weeks
- Inflammatory biomarkers: Reduction observable by 4-8 weeks
- Adipokine profiles: Shifts apparent by 8-16 weeks
Research protocols targeting these outcomes should incorporate measurement schedules aligned with expected response timelines to capture meaningful data points [10].
Long-Term Research Endpoints (Months)
Extended observation periods reveal tirzepatide’s sustained and cumulative effects:
Extended Timeline Outcomes:
- Maximal weight effects: Typically 60-72 weeks in long-duration studies
- Cardiovascular remodeling: Requires 24+ weeks for comprehensive assessment
- Metabolic memory effects: Evaluated through discontinuation studies
- Durability of response: Assessed over 52+ weeks of continuous administration
“Peak metabolic effects of tirzepatide in research models are typically observed between 20-40 weeks of consistent administration, with sustained benefits maintained through extended observation periods exceeding 72 weeks.” [11]
Comparative Timeline: Tirzepatide vs. Other Research Peptides
Understanding how tirzepatide’s timeline compares to related compounds provides valuable context:
Response Timeline Comparison:
| Compound Class | Initial Effects | Peak Efficacy | Steady State |
|---|---|---|---|
| Tirzepatide (dual GIP/GLP-1) | 4-7 days | 20-28 weeks | 4 weeks |
| Semaglutide (GLP-1 only) | 4-7 days | 16-20 weeks | 4-5 weeks |
| Liraglutide (GLP-1 daily) | 2-4 days | 12-16 weeks | 2-3 weeks |
| Retatrutide (triple agonist) | 5-10 days | 24-36 weeks | 4-6 weeks |
Tirzepatide’s dual mechanism generally produces more pronounced effects than single GLP-1 agonists, though with a similar timeline to peak efficacy. Researchers comparing these compounds should account for these timeline differences in experimental design.
For laboratories investigating multiple peptide compounds, PEPTIDE PRO’s extensive catalogue offers research-grade options including semaglutide and other metabolic research peptides.
Optimizing Measurement Schedules
To capture tirzepatide’s full effect profile, researchers should implement comprehensive measurement schedules:
Recommended Assessment Timeline:
- Baseline: Complete parameter assessment before initiation
- Week 1: Early response markers (glucose, appetite indicators)
- Week 4: First steady-state assessment (all primary outcomes)
- Weeks 8, 12, 16: Regular interval assessments during escalation
- Week 20+: Peak efficacy evaluation
- Ongoing: Maintenance phase monitoring (every 4-8 weeks)
This structured approach ensures researchers capture both rapid-onset and delayed-development effects, providing comprehensive data on how long does tirzepatide take to work across multiple research endpoints.
Factors That Influence Tirzepatide’s Effectiveness Timeline
While tirzepatide follows a generally predictable timeline in research settings, numerous variables can accelerate, delay, or modify the compound’s effects. Understanding these factors helps researchers interpret results accurately and design protocols that account for potential variability.
Compound Quality and Handling 🔬
The purity, integrity, and handling of research-grade tirzepatide directly impact its bioactivity and timeline of effects:
Critical Quality Factors:
- Peptide Purity: Research-grade compounds should maintain ≥98% purity
- Lower purity may reduce bioavailability
- Impurities can interfere with receptor binding
- Degradation products may alter pharmacokinetics
- Storage Conditions: Proper storage preserves compound integrity
- Lyophilized peptides: Store at -20°C to -80°C
- Reconstituted solutions: Refrigerate at 2-8°C
- Avoid freeze-thaw cycles
- Protect from light exposure
- Reconstitution Procedures: Proper preparation ensures optimal bioactivity
- Use appropriate sterile diluents (bacteriostatic water)
- Gentle mixing to prevent peptide damage
- Accurate concentration calculations
- Proper pH maintenance
Researchers utilizing premium research-grade peptides from PEPTIDE PRO benefit from strict quality control measures, including Certificate of Analysis (COA) documentation and proper storage protocols that preserve compound integrity.
Impact on Timeline:
- Degraded peptides: Delayed or diminished effects
- Improper storage: Reduced bioavailability, extended timeline
- Incorrect reconstitution: Variable absorption, inconsistent results
- High-purity compounds: Predictable, reproducible timelines
Dosing Variables and Administration Factors
How tirzepatide is administered significantly influences how long does tirzepatide take to work:
Administration Route Considerations:
- Subcutaneous injection (standard): Predictable absorption, ~80% bioavailability
- Injection site: Abdominal vs. thigh may show minor absorption differences
- Injection technique: Proper technique ensures consistent delivery
- Volume and concentration: Affects local absorption dynamics
Dosing Schedule Adherence:
- Consistent weekly timing: Maintains stable plasma levels
- Missed doses: Disrupts steady-state, delays peak effects
- Dose escalation timing: Affects overall timeline to maximum efficacy
- Inter-dose intervals: Variations alter pharmacokinetic profile
Model-Specific Biological Factors
Research subject characteristics create significant variability in tirzepatide response timelines:
Baseline Metabolic Status:
- Higher baseline glucose: Often shows faster glycemic response
- Greater adiposity: May demonstrate more pronounced weight effects
- Insulin resistance severity: Influences timeline of metabolic improvements
- Baseline GLP-1 sensitivity: Affects magnitude and speed of response
Genetic and Phenotypic Variables:
- Receptor expression levels: Determines binding capacity
- Metabolic enzyme activity: Influences compound clearance
- Gut hormone baseline levels: Affects relative response magnitude
- Individual pharmacokinetic variations: Creates timeline differences
Age and Physiological Factors:
- Metabolic rate differences: Younger subjects may show faster responses
- Body composition: Lean vs. adipose tissue ratio affects distribution
- Organ function: Hepatic and renal function influence clearance
- Hormonal status: Affects receptor sensitivity and response
Environmental and Experimental Conditions
The research environment and protocol design influence tirzepatide’s effectiveness timeline:
Dietary Controls:
- Caloric intake: Synergistic effects with dietary restriction
- Macronutrient composition: May influence metabolic response rate
- Feeding schedule: Affects glucose and appetite-related outcomes
- Dietary consistency: Reduces variability in measured effects
Activity and Exercise Variables:
- Physical activity levels: Enhance metabolic effects
- Exercise timing: May interact with compound’s action
- Activity consistency: Reduces experimental variability
Concurrent Interventions:
- Other research compounds: Potential interactions affecting timeline
- Behavioral modifications: Can accelerate or modify effects
- Environmental enrichment: May influence stress-related outcomes
Strategies to Optimize Response Timeline
Researchers can implement several strategies to ensure tirzepatide achieves effects within expected timelines:
Protocol Optimization Checklist:
✅ Source high-purity peptides from reputable suppliers with COA documentation ✅ Implement strict storage protocols maintaining cold chain integrity ✅ Standardize reconstitution procedures with validated protocols ✅ Ensure consistent dosing schedules with minimal variation ✅ Control baseline variables through appropriate subject selection ✅ Maintain environmental consistency across experimental groups ✅ Monitor adherence rigorously to identify protocol deviations ✅ Document all variables that might influence timeline outcomes
By controlling these factors, researchers can minimize timeline variability and generate reproducible results that accurately reflect tirzepatide’s inherent pharmacological properties.
For laboratories requiring consistent, high-quality research peptides, PEPTIDE PRO offers same-day dispatch for orders placed before 1pm (Monday-Friday), ensuring minimal time between ordering and experimental initiation.
Research Applications: Timing Considerations for Different Study Types
Understanding how long does tirzepatide take to work varies significantly depending on the specific research application and study design. Different experimental objectives require tailored timelines and measurement approaches.
Metabolic Research Studies
Tirzepatide’s primary research applications involve metabolic pathway investigation, requiring specific timeline considerations:
Glucose Homeostasis Studies (4-12 Weeks):
- Acute glucose response: Measureable within 1 week
- Fasting glucose normalization: Typically 2-4 weeks
- HbA1c changes: Require 8-12 weeks (reflects 2-3 month glucose average)
- Insulin sensitivity improvements: Progressive over 4-12 weeks
Recommended Study Duration: Minimum 12 weeks for comprehensive glycemic assessment; 24+ weeks for long-term metabolic adaptation studies.
Body Composition Research (12-28 Weeks):
- Initial weight changes: Detectable by week 2-4
- Significant body composition shifts: 8-12 weeks
- Near-maximal effects: 20-28 weeks
- Fat mass vs. lean mass differentiation: Requires serial measurements over 12+ weeks
Recommended Study Duration: 20-40 weeks for comprehensive body composition research; 52+ weeks for maximal effect and maintenance studies.
Mechanistic and Pathway Studies
Research investigating tirzepatide’s molecular mechanisms requires different timeline approaches:
Receptor Binding and Signaling (Hours to Days):
- Immediate studies: Receptor occupancy, acute signaling cascade activation
- Timeline: Hours to 72 hours post-administration
- Applications: Understanding molecular mechanisms, receptor dynamics
- Measurement approaches: In vitro assays, ex vivo tissue analysis, acute in vivo studies
Gene Expression and Protein Changes (Days to Weeks):
- Transcriptional changes: Detectable within 24-72 hours
- Protein expression shifts: Observable within 3-14 days
- Sustained adaptations: Require 2-8 weeks of exposure
- Epigenetic modifications: May require extended timelines (8+ weeks)
Recommended Study Duration: 1-8 weeks depending on specific molecular endpoints; acute studies for immediate signaling, extended protocols for adaptive changes.
Comparative Efficacy Research
Studies comparing tirzepatide to other compounds require careful timeline matching:
Head-to-Head Comparison Protocols:
- Parallel group design: All groups follow identical timelines
- Minimum duration: 12-16 weeks for meaningful metabolic comparisons
- Optimal duration: 24-40 weeks for comprehensive efficacy assessment
- Measurement intervals: Matched across all treatment groups
Key Timeline Considerations:
- Account for different compounds’ pharmacokinetic profiles
- Ensure steady-state achievement for all agents
- Match dose escalation schedules when appropriate
- Plan measurements at equivalent time points relative to steady-state
Researchers comparing tirzepatide to other GLP-1 receptor agonists should account for pharmacokinetic differences when designing measurement schedules.
Long-Duration and Durability Studies
Extended research protocols examine tirzepatide’s sustained effects and durability:
Continuous Administration Studies (52+ Weeks):
- Purpose: Assess long-term efficacy, safety, and durability
- Key timepoints: Baseline, weeks 4, 12, 24, 40, 52, and beyond
- Outcomes: Sustained metabolic effects, tachyphylaxis assessment, long-term adaptations
- Considerations: Requires stable compound supply, consistent protocols
Discontinuation and Washout Studies:
- Treatment phase: 12-40 weeks of tirzepatide administration
- Washout phase: 4-12 weeks post-discontinuation
- Purpose: Assess durability of effects, rebound phenomena, metabolic memory
- Timeline: Total 16-52+ weeks
Intermittent Dosing Protocols:
- Experimental designs: Cycling on/off periods, dose holidays
- Purpose: Investigate minimal effective dosing, sustained response
- Timeline considerations: Multiple cycles required (24+ weeks total)
Dose-Response and Pharmacology Studies
Research examining tirzepatide’s dose-response relationships requires specific timeline approaches:
Dose-Escalation Studies:
- Design: Progressive dose increases with interval assessments
- Timeline: 4 weeks per dose level minimum (achieving steady-state)
- Total duration: 16-32 weeks for 4-8 dose levels
- Measurements: Comprehensive assessment at each steady-state
Fixed-Dose Comparison Studies:
- Design: Parallel groups receiving different fixed doses
- Timeline: 12-24 weeks minimum for metabolic endpoints
- Advantages: Simpler interpretation, clear dose-response curves
- Measurements: Matched intervals across all dose groups
Pharmacokinetic/Pharmacodynamic (PK/PD) Studies:
- Intensive sampling: Hours to days for PK assessment
- PD correlation: Weeks to months for effect measurements
- Timeline: Acute PK (1-2 weeks) plus extended PD (12+ weeks)
- Purpose: Correlate plasma levels with biological effects
Combination and Interaction Studies
Research investigating tirzepatide in combination with other interventions requires extended timelines:
Peptide Combination Research:
- Sequential introduction: Stagger compound initiation to isolate effects
- Simultaneous administration: Longer timelines to assess synergy
- Timeline: 16-40 weeks for comprehensive combination assessment
- Controls: Include monotherapy groups for comparison
Diet and Exercise Interaction Studies:
- Baseline establishment: 2-4 weeks before tirzepatide initiation
- Intervention phase: 12-24 weeks with combined treatments
- Comparison groups: Tirzepatide alone, intervention alone, combination
- Timeline: 16-28 weeks total
Researchers designing complex protocols can consult PEPTIDE PRO’s educational resources for guidance on reconstitution, storage, and handling procedures that support extended research timelines.
Safety Monitoring and Timeline Considerations in Research
While investigating how long does tirzepatide take to work, researchers must simultaneously monitor safety parameters that follow their own distinct timelines. Understanding when potential adverse effects emerge helps design appropriate monitoring schedules.
Early-Onset Effects Requiring Monitoring (Days to Weeks)
Certain tirzepatide-related effects emerge rapidly and require early monitoring:
Gastrointestinal Effects (1-14 Days):
- Most common early observation in research models
- Typically peaks during first 1-2 weeks after initiation or dose escalation
- Generally diminishes over 2-4 weeks
- Monitoring: Daily observation logs, feeding behavior assessment
Acute Metabolic Changes (1-7 Days):
- Rapid glucose lowering in susceptible models
- Potential hypoglycemia risk in certain experimental conditions
- Monitoring: Frequent glucose measurements during first week
- Consideration: Baseline glucose status influences risk
Appetite and Intake Changes (1-3 Days):
- Rapid onset of reduced food intake
- May require nutritional monitoring in some protocols
- Assessment: Daily intake measurements, body weight tracking
- Timeline: Most pronounced during first 2 weeks, then stabilizes
Intermediate-Timeline Safety Parameters (Weeks to Months)
Certain safety parameters require monitoring over extended periods:
Cardiovascular Monitoring (4-12 Weeks):
- Heart rate changes: May increase modestly, monitor through week 12
- Blood pressure effects: Progressive changes over 4-12 weeks
- Cardiac function: Assess at baseline, week 12, and study end
- Timeline: Most changes stabilize by 8-12 weeks
Hepatic and Renal Function (4-12 Weeks):
- Liver enzymes: Monitor at baseline, week 4, 12, and quarterly
- Renal function markers: Assess every 8-12 weeks
- Pancreatic enzymes: Baseline and periodic monitoring
- Timeline: Establish new baseline by 8-12 weeks
Thyroid and Endocrine Monitoring (8-24 Weeks):
- Thyroid function: Baseline and every 12-24 weeks
- Other endocrine parameters: Protocol-specific monitoring
- Timeline: Changes typically gradual if present
Monitoring Schedule Recommendations
A comprehensive safety monitoring timeline for tirzepatide research:
| Parameter | Baseline | Week 1 | Week 4 | Week 12 | Quarterly | Study End |
|---|---|---|---|---|---|---|
| Body weight |
This schedule ensures researchers capture both rapid-onset and delayed-development safety signals while maintaining practical feasibility.
Dose Escalation Safety Considerations
The standard dose escalation protocol for tirzepatide incorporates safety-based timing:
4-Week Escalation Intervals:
- Allows steady-state achievement at each dose
- Permits assessment of dose-specific effects
- Enables identification of dose-limiting factors
- Provides adaptation period reducing acute effects
Rationale for Timeline:
- Steady-state: Achieved by week 4 at each dose level
- Tolerability: Most acute effects diminish within 2-4 weeks
- Safety assessment: Adequate time for parameter evaluation
- Efficacy: Sufficient duration to observe dose-response
Accelerated Escalation Risks:
- Increased acute adverse effects
- Inadequate safety assessment time
- Difficulty attributing effects to specific doses
- Higher discontinuation rates
Long-Term Safety Monitoring
Extended research protocols require ongoing safety surveillance:
Chronic Exposure Studies (24+ Weeks):
- Continued monitoring at regular intervals
- Assessment for delayed-onset effects
- Evaluation of cumulative exposure impacts
- Long-term organ function surveillance
Recommended Frequency:
- Comprehensive assessments: Every 12 weeks
- Targeted parameters: Every 4-8 weeks
- Continuous observations: Weight, intake, behavior
- End-of-study: Complete parameter reassessment
Optimizing Research Protocols: Best Practices for Tirzepatide Studies
Designing effective research protocols that accurately capture how long does tirzepatide take to work requires attention to numerous methodological details. Implementing best practices ensures reliable, reproducible results.
Protocol Design Fundamentals
Timeline Planning Essentials:
- Define Primary Endpoints Clearly
- Specify exact outcome measures
- Determine minimum detectable effect size
- Calculate required observation duration
- Plan measurement intervals accordingly
- Account for Pharmacokinetic Profile
- Include 4-week run-in to steady-state
- Plan dose escalations in 4-week intervals
- Schedule key measurements post-steady-state
- Allow adequate washout in crossover designs (4-5 half-lives = 20-25 days)
- Build in Appropriate Controls
- Vehicle-treated control groups
- Positive control comparators when appropriate
- Baseline measurements for all subjects
- Time-matched assessments across groups
- Power Analysis and Sample Size
- Account for expected effect size and timeline
- Consider attrition over extended protocols
- Include adequate subjects for subgroup analyses
- Plan for dose-response assessment if applicable
Compound Sourcing and Quality Assurance
The foundation of reliable tirzepatide research begins with compound quality:
Essential Quality Criteria:
- ✅ Purity ≥98%: Verified by HPLC and mass spectrometry
- ✅ Certificate of Analysis (COA): Batch-specific documentation
- ✅ Proper storage: Maintained at -20°C to -80°C until use
- ✅ Sterility testing: For compounds intended for injection
- ✅ Endotoxin testing: Particularly important for metabolic research
Supplier Selection Considerations:
- Reputation and track record in research community
- Quality control and testing procedures
- Storage and shipping protocols (cold chain maintenance)
- Documentation and support
- Consistency across batches
PEPTIDE PRO provides research-grade tirzepatide with comprehensive quality documentation, ensuring researchers receive compounds that meet rigorous purity standards essential for reproducible timelines.
Reconstitution and Preparation Protocols
Proper peptide preparation directly impacts bioavailability and timeline of effects:
Standard Reconstitution Protocol:
- Equilibrate to Room Temperature
- Remove lyophilized peptide from freezer
- Allow 15-20 minutes to reach room temperature
- Prevents condensation and ensures accurate measurement
- Prepare Sterile Diluent
- Bacteriostatic water (0.9% benzyl alcohol) for multi-dose
- Sterile water for single-use applications
- Calculate volume for desired final concentration
- Reconstitute Gently
- Add diluent slowly down pen peptide wall
- Swirl gently; do not shake vigorously
- Allow 2-3 minutes for complete dissolution
- Inspect for clarity and particulates
- Storage of Reconstituted Peptide
- Refrigerate immediately at 2-8°C
- Protect from light
- Use within recommended timeframe (typically 28 days)
- Label with reconstitution date and concentration
Impact on Timeline:
- Proper reconstitution: Predictable absorption and effects
- Improper handling: Reduced bioavailability, extended timeline, increased variability
- Degraded peptide: Diminished effects, inconsistent results
Administration Best Practices
Consistent administration technique ensures reproducible timelines:
Injection Protocol Standardization:
- Site selection: Rotate between approved sites (abdomen, thigh)
- Technique: Subcutaneous injection at 45-90° angle
- Volume: Appropriate for injection site (typically 0.5-1.0 mL)
- Timing: Same day/time each week for consistency
- Documentation: Record all administrations with date, time, dose, site
Minimizing Variability:
- Train all personnel on identical technique
- Use standardized equipment (syringes, needles)
- Maintain consistent injection sites within study groups
- Document any deviations from protocol
Data Collection and Measurement Timing
Strategic measurement scheduling captures tirzepatide’s timeline accurately:
Optimal Measurement Windows:
Acute Effects (Days 1-7):
- Glucose measurements: Multiple daily timepoints
- Food intake: Daily quantification
- Body weight: Daily or every other day
- Behavioral observations: Continuous or daily logs
Subacute Effects (Weeks 2-4):
- Glucose parameters: Weekly fasting measurements
- Body weight/composition: Weekly assessments
- Metabolic markers: Week 4 (first steady-state)
- Safety parameters: Week 4 comprehensive panel
Chronic Effects (Weeks 4+):
- Primary endpoints: Every 4 weeks through peak efficacy period
- Body composition: Every 4-8 weeks
- Comprehensive metabolic panel: Every 8-12 weeks
- Long-term safety: Every 12 weeks
Consistency Requirements:
- Same time of day for measurements
- Standardized pre-measurement conditions (fasting state, etc.)
- Identical equipment and procedures across timepoints
- Blinded assessors when possible
Statistical Considerations for Timeline Analysis
Analyzing tirzepatide’s time-course effects requires appropriate statistical approaches:
Recommended Analyses:
- Repeated measures ANOVA: For multiple timepoint comparisons
- Mixed-effects models: Accounts for missing data and individual variability
- Time-to-event analysis: For dichotomous outcomes (achieving target, etc.)
- Area under the curve (AUC): Integrates effects over time
- Slope analysis: Quantifies rate of change
Timeline-Specific Considerations:
- Account for dose escalation in analysis
- Consider steady-state achievement in interpretation
- Adjust for baseline differences
- Include time × treatment interactions
- Plan interim analyses at key timepoints (weeks 4, 12, 24)
Documentation and Reproducibility
Comprehensive documentation ensures protocol reproducibility:
Essential Documentation:
- 📋 Compound information: Source, batch number, COA, storage conditions
- 📋 Reconstitution details: Date, diluent, concentration, storage
- 📋 Administration log: Every dose with date, time, site, personnel
- 📋 Measurement records: All data with timestamps and conditions
- 📋 Protocol deviations: Any variations from planned procedures
- 📋 Adverse observations: Complete timeline of any concerning findings
This level of detail enables other researchers to replicate protocols and compare timelines across studies, advancing the collective understanding of how long does tirzepatide take to work across diverse research contexts.
Frequently Asked Questions About Tirzepatide Timeline
How quickly does tirzepatide start working in research models?
Initial biochemical effects of tirzepatide, particularly glucose-related changes, can be detected within 24-72 hours of administration in responsive research models. However, meaningful physiological effects typically emerge over 4-7 days, with progressive enhancement through the first 4 weeks as steady-state concentrations are achieved. The timeline for “working” depends entirely on which endpoint researchers are measuring—acute glucose effects appear within days, while comprehensive metabolic remodeling requires weeks to months.
What is the optimal study duration for tirzepatide research?
The optimal duration depends on research objectives:
- Acute mechanistic studies: 1-7 days for immediate signaling and receptor effects
- Glycemic control research: 12-16 weeks minimum for HbA1c assessment
- Body composition studies: 20-40 weeks for near-maximal effects
- Long-term efficacy/safety: 52+ weeks for comprehensive assessment
Most metabolic research protocols benefit from 20-24 week durations, allowing full dose escalation and adequate time at maintenance doses to observe peak effects.
Does tirzepatide work faster at higher doses?
Higher doses of tirzepatide generally produce more pronounced effects but don’t dramatically accelerate the fundamental timeline of action. The pharmacokinetic profile (time to steady-state) remains approximately 4 weeks regardless of dose. However, higher doses may:
- Produce larger magnitude effects at each timepoint
- Reach therapeutic thresholds more quickly
- Show detectable changes earlier in less sensitive assays
- Demonstrate dose-response relationships more clearly
Research protocols typically use dose escalation rather than high initial doses to balance efficacy timeline with tolerability.
How long after reconstitution does tirzepatide remain effective?
Properly reconstituted tirzepatide stored at 2-8°C (refrigerated) maintains stability and effectiveness for approximately 28 days when using bacteriostatic water as the diluent. Key factors affecting post-reconstitution stability:
- Storage temperature: Must maintain 2-8°C consistently
- Light protection: Store in original pen peptide or protect from light
- Sterility: Use aseptic technique for all withdrawals
- Diluent type: Bacteriostatic water extends usability vs. sterile water
For research requiring longer timelines, researchers should prepare fresh aliquots or use lyophilized peptide stored at -20°C to -80°C, which maintains stability for extended periods (typically 2+ years).
Can tirzepatide’s timeline be accelerated with loading doses?
While loading dose strategies are used with some compounds, tirzepatide’s pharmacokinetics don’t particularly favor this approach. The compound’s ~5-day half-life means steady-state is achieved through standard weekly dosing within 4 weeks (approximately 5 half-lives). A loading dose might achieve steady-state concentrations marginally faster but:
- Increases acute adverse effect risk
- Provides minimal timeline advantage (days, not weeks)
- Complicates pharmacokinetic interpretation
- Isn’t standard in research protocols
Most research applications achieve optimal timelines through standard weekly dosing with appropriate dose escalation schedules.
What factors most commonly delay tirzepatide’s effects in research?
Several factors can extend the timeline for observable tirzepatide effects:
Compound-Related:
- Degraded or low-purity peptide
- Improper storage or handling
- Incorrect reconstitution
- Suboptimal dosing
Protocol-Related:
- Inadequate dose for research model
- Inconsistent administration schedule
- Insufficient observation duration
- Measurement timing misaligned with pharmacokinetics
Model-Related:
- Low baseline metabolic dysfunction (less room for improvement)
- Reduced receptor sensitivity
- Concurrent factors opposing tirzepatide’s effects
- Individual pharmacokinetic variations
Controlling these variables through rigorous protocol design and high-quality compound sourcing minimizes timeline delays.
Understanding Tirzepatide’s Research Timeline
The question of how long does tirzepatide take to work has a nuanced answer that depends critically on the specific research context, measured endpoints, and experimental design. This comprehensive examination reveals several key principles:
Timeline Fundamentals:
- Initial effects emerge within days (glucose regulation, appetite modulation)
- Steady-state concentrations are achieved by week 4 of consistent dosing
- Peak efficacy for metabolic endpoints typically occurs between weeks 20-28
- Dose escalation protocols extend overall timelines but improve tolerability
- Long-term studies (52+ weeks) reveal sustained, durable effects
Critical Success Factors: The timeline for observable tirzepatide effects is profoundly influenced by:
- Compound quality: High-purity, properly stored research-grade peptides ensure predictable timelines
- Protocol design: Appropriate measurement intervals aligned with pharmacokinetics
- Dosing consistency: Regular weekly administration maintains steady therapeutic levels
- Endpoint selection: Different outcomes follow distinct temporal patterns
- Experimental controls: Standardization minimizes variability and timeline unpredictability
Practical Research Implications: Researchers designing tirzepatide studies should:
- ✅ Plan minimum 12-week protocols for metabolic research; 20+ weeks for comprehensive assessment
- ✅ Incorporate 4-week intervals for dose escalation and steady-state achievement
- ✅ Schedule measurements strategically to capture both acute and chronic effects
- ✅ Source high-purity compounds with proper documentation and storage
- ✅ Implement rigorous monitoring schedules for both efficacy and safety parameters
- ✅ Account for individual variability in timeline expectations
- ✅ Document all protocol details to enable reproducibility
Moving Forward with Tirzepatide Research:
For laboratories and researchers embarking on tirzepatide investigations, success begins with proper preparation. PEPTIDE PRO offers premium research-grade tirzepatide with exceptional purity, comprehensive quality documentation, and fast UK delivery to support time-sensitive research protocols.
Understanding that tirzepatide’s effects unfold over a predictable but extended timeline allows researchers to design appropriate protocols, set realistic expectations, and interpret results within proper temporal context. Whether investigating acute receptor signaling, intermediate metabolic changes, or long-term physiological adaptations, aligning research timelines with tirzepatide’s pharmacological properties ensures scientifically rigorous and meaningful outcomes.
The dual GIP/GLP-1 receptor agonist mechanism that makes tirzepatide such a valuable research tool also necessitates patience—the compound’s most profound effects emerge not in hours or days, but through weeks and months of consistent administration. By respecting this timeline and designing protocols accordingly, researchers can fully leverage tirzepatide’s unique properties to advance understanding of metabolic regulation, energy homeostasis, and therapeutic peptide mechanisms.
For questions about specific research applications, compound sourcing, or protocol design considerations, contact PEPTIDE PRO’s research support team for expert guidance tailored to your experimental objectives.
References
[1] Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[2] Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
[3] Urva S, et al. The Novel Dual Glucose-Dependent Insulinotropic Polypeptide and Glucagon-Like Peptide-1 (GLP-1) Receptor Agonist Tirzepatide Transiently Delays Gastric Emptying Similarly to Selective Long-Acting GLP-1 Receptor Agonists. Diabetes Care. 2020;43(8):1661-1664.
[4] Rosenstock J, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.
[5] Dahl D, et al. Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA. 2022;327(6):534-545.
[6] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
[7] Wilson JM, et al. The dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist, tirzepatide, improves lipoprotein biomarkers associated with insulin resistance and cardiovascular risk in patients with type 2 diabetes. Diabetes Obes Metab. 2020;22(12):2451-2459.
[8] Ludvik B, et al. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet. 2021;398(10300):583-598.
[9] Thomas MK, et al. Tirzepatide, a dual GIP and GLP-1 receptor agonist, improves markers of beta-cell function and insulin sensitivity in type 2 diabetes. J Clin Endocrinol Metab. 2021;106(2):388-396.
[10] Heise T, et al. Effects of subcutaneous tirzepatide versus placebo or semaglutide on pancreatic islet function and insulin sensitivity in adults with type 2 diabetes: a multicentre, randomised, double-blind, parallel-arm, phase 1 clinical trial. Lancet Diabetes Endocrinol. 2022;10(6):418-429.
[11] Del Prato S, et al. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet. 2021;398(10313):1811-1824.
