
The landscape of weight management and Type 2 diabetes treatment in the UK has transformed dramatically with the emergence of GLP-1 receptor agonists. Mounjaro on the NHS: Current Position & Criteria represents one of the most frequently searched topics among patients, healthcare professionals, and researchers seeking clarity on access to tirzepatide—the active pharmaceutical ingredient in Mounjaro. As NHS England continues to navigate unprecedented demand for weight-loss medications alongside budgetary constraints, understanding the current position and eligibility criteria for Mounjaro has never been more critical for those exploring treatment options.
This comprehensive guide examines the regulatory status, clinical criteria, prescription pathways, and practical considerations surrounding Mounjaro availability through the National Health Service in 2025, providing evidence-based insights for informed decision-making.
Key Takeaways
- Mounjaro (tirzepatide) is licensed in the UK for Type 2 diabetes treatment, but NHS availability remains highly restricted based on specific clinical criteria and local formulary decisions
- Eligibility requires meeting stringent BMI thresholds (typically 35+ with comorbidities or 40+ without) alongside diabetes diagnosis and previous treatment failures
- Access pathways vary significantly across England, Scotland, Wales, and Northern Ireland, with specialist referrals and tier 3 weight management services often required
- Private prescription remains the primary route for most patients seeking tirzepatide, as NHS provision faces capacity and funding limitations
- Research-grade peptides including tirzepatide analogues are available through specialist suppliers for laboratory and scientific investigation purposes only
Understanding Mounjaro: Mechanism and Clinical Applications
Mounjaro (tirzepatide) represents a breakthrough in metabolic disease management as the first dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist approved for clinical use. This novel mechanism differentiates tirzepatide from single-pathway GLP-1 agonists like semaglutide, offering enhanced metabolic benefits through complementary receptor activation[1].
The Science Behind Tirzepatide
The dual-agonist mechanism works by:
✅ Enhancing insulin secretion in a glucose-dependent manner, reducing hypoglycaemia risk
✅ Suppressing glucagon release when blood glucose is elevated
✅ Slowing gastric emptying to promote satiety and reduce caloric intake
✅ Acting on central appetite regulation through hypothalamic pathways
✅ Improving insulin sensitivity in peripheral tissues
Clinical trial data from the SURPASS programme demonstrated that tirzepatide achieved superior glycaemic control and weight reduction compared to existing therapies, with participants losing an average of 15-22% of body weight depending on dosage[2]. These remarkable outcomes have driven significant patient and clinician interest in accessing Mounjaro through NHS pathways.
Licensed Indications in the UK
As of 2025, the Medicines and Healthcare products Regulatory Agency (MHRA) has licensed Mounjaro specifically for:
- Type 2 diabetes mellitus in adults as an adjunct to diet and exercise
- Use as monotherapy when metformin is contraindicated or not tolerated
- Combination therapy with other glucose-lowering medications
Importantly, while Mounjaro demonstrates substantial weight-loss efficacy, its UK licence does not currently extend to obesity treatment as a standalone indication—a crucial distinction that impacts NHS prescribing decisions and Mounjaro on the NHS: Current Position & Criteria discussions.
For researchers investigating peptide mechanisms and metabolic pathways, PEPTIDE PRO supplies research-grade compounds under strict quality controls for laboratory applications only.
Current NHS Position on Mounjaro Availability
The Mounjaro on the NHS: Current Position & Criteria landscape in 2025 reflects a complex interplay between clinical evidence, health economics, capacity constraints, and regulatory frameworks. Understanding this position requires examining multiple authoritative sources and regional variations.
NICE Guidance and Technology Appraisals
The National Institute for Health and Care Excellence (NICE) conducts rigorous health technology assessments to determine whether treatments represent cost-effective use of NHS resources. As of early 2025, NICE guidance on tirzepatide includes:
For Type 2 Diabetes: NICE has issued positive guidance for tirzepatide use within specific parameters, recognizing its superior efficacy in glycaemic control and weight management compared to existing therapies. However, this guidance includes significant caveats regarding:
- Position in treatment pathway (typically third-line or later)
- Requirement for inadequate control with dual therapy
- BMI thresholds that must be met
- Regular monitoring and review protocols
- Discontinuation criteria if targets aren’t achieved
For Weight Management: NICE has not yet issued comprehensive guidance supporting tirzepatide for obesity treatment outside diabetes indications, creating a significant barrier to NHS access for patients seeking weight loss as the primary therapeutic goal[3].
NHS England Commissioning Decisions
Beyond NICE guidance, NHS England’s commissioning framework determines practical availability. Key considerations include:
📊 Budget Impact: The substantial cost of GLP-1 receptor agonists (£150-250 per patient per month) creates significant budgetary pressure
📊 Capacity Constraints: Specialist diabetes and weight management services face overwhelming demand
📊 Supply Chain: Pharmaceutical supply limitations have periodically restricted availability
📊 Equity Concerns: Geographic variation in access raises fairness questions
Many Integrated Care Boards (ICBs) have implemented restrictive formularies that prioritize older, less expensive diabetes medications before considering newer agents like Mounjaro, regardless of NICE guidance.
Regional Variations Across the UK
England: Access depends heavily on local ICB formulary decisions, with significant postcode lottery effects. Some areas require exhaustive trials of alternative therapies before considering tirzepatide.
Scotland: The Scottish Medicines Consortium (SMC) conducts independent assessments. Access through NHS Scotland follows similar restrictive patterns with emphasis on cost-effectiveness.
Wales: All Wales Medicines Strategy Group (AWMSG) provides guidance, though implementation varies by health board.
Northern Ireland: Department of Health commissioning decisions create another distinct access landscape.
This fragmented approach means Mounjaro on the NHS: Current Position & Criteria can vary dramatically based on geographic location, creating frustration for patients and clinicians alike.
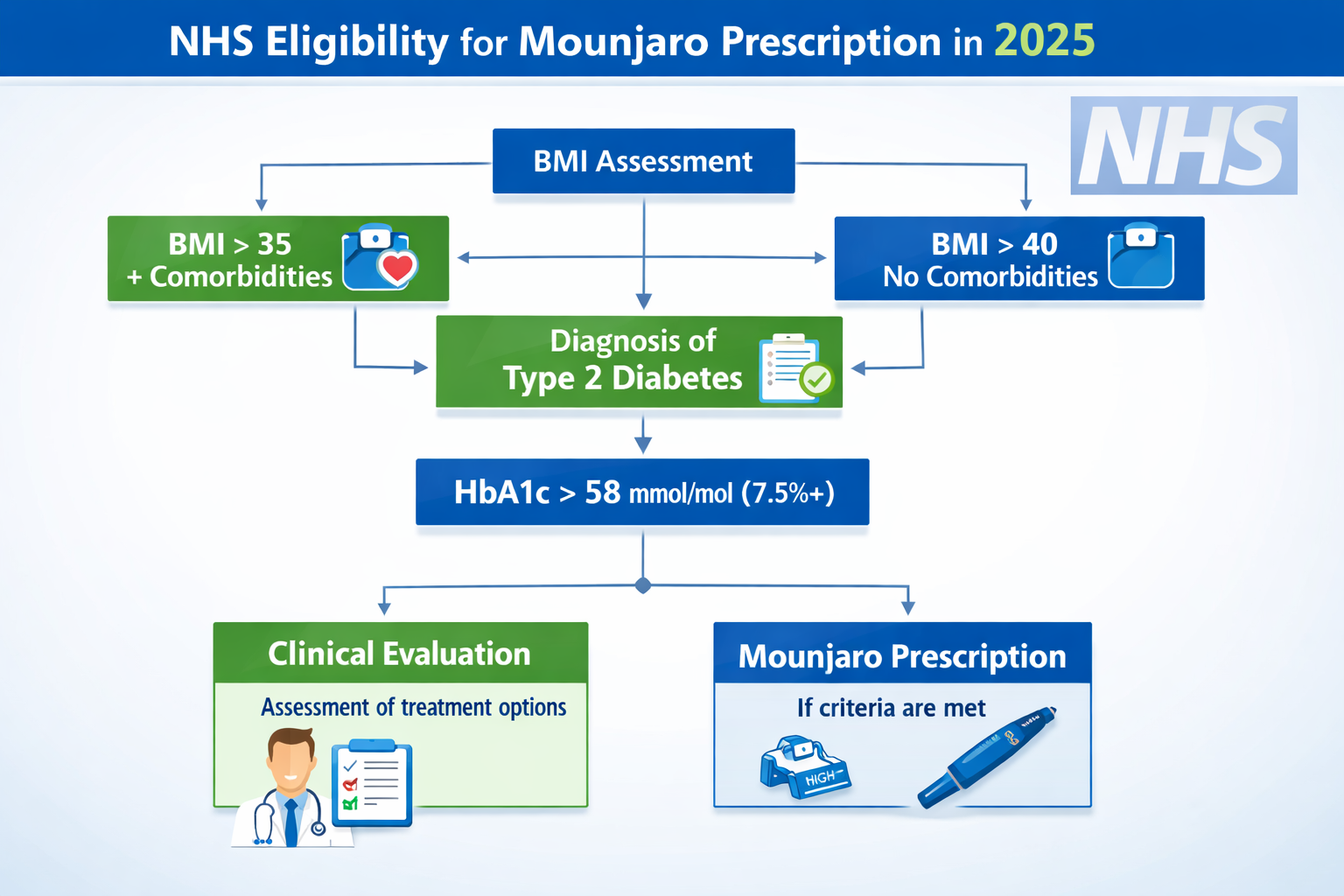
Detailed Eligibility Criteria for NHS Mounjaro Prescription
Understanding the specific clinical criteria that determine NHS eligibility for Mounjaro represents essential knowledge for patients considering this treatment pathway. While criteria may vary by region, common requirements include:
Primary Diagnosis Requirements
Type 2 Diabetes Mellitus
Patients must have a confirmed diagnosis of Type 2 diabetes with documented evidence of:
- HbA1c levels above target range (typically ≥58 mmol/mol or 7.5%) despite existing therapy
- Duration of diabetes diagnosis (some ICBs require minimum disease duration)
- Absence of Type 1 diabetes or secondary diabetes forms
- C-peptide levels confirming endogenous insulin production (in borderline cases)
Previous Treatment Failures
Most NHS pathways require documented trials of:
- Metformin at maximum tolerated dose
- At least one additional oral hypoglycaemic agent (sulfonylurea, SGLT2 inhibitor, DPP-4 inhibitor, or thiazolidinedione)
- Adequate trial duration (typically 3-6 months per agent)
- Documented inadequate response with HbA1c remaining above target
Some commissioning groups require trial of basal insulin before considering GLP-1 receptor agonists, adding further treatment steps.
Body Mass Index (BMI) Thresholds
BMI requirements represent a critical component of Mounjaro on the NHS: Current Position & Criteria:
| BMI Category | Additional Requirements | Typical NHS Stance |
|---|---|---|
| ≥40 kg/m² | None beyond diabetes diagnosis | May be considered after treatment failures |
| 35-39.9 kg/m² | Obesity-related comorbidities required | Considered with documented complications |
| 30-34.9 kg/m² | Multiple significant comorbidities | Rarely approved; exceptional cases only |
| <30 kg/m² | Not applicable | Not commissioned for tirzepatide |
Obesity-related comorbidities that may support eligibility include:
- Hypertension requiring medication
- Obstructive sleep apnoea
- Non-alcoholic fatty liver disease (NAFLD/NASH)
- Osteoarthritis significantly impacting mobility
- Cardiovascular disease
- Polycystic ovary syndrome (PCOS)
Specialist Assessment Requirements
Most NHS pathways require assessment and initiation by specialist services:
🏥 Tier 3 Weight Management Services: Multidisciplinary programmes providing intensive lifestyle intervention alongside medical management
🏥 Specialist Diabetes Clinics: Consultant endocrinologist or diabetologist assessment
🏥 Obesity Medicine Specialists: Where available, dedicated obesity medicine consultants
GP-initiated prescribing of Mounjaro remains uncommon, with most ICBs requiring specialist recommendation before approval. This creates significant access barriers, as waiting times for specialist appointments can extend 6-12 months in many regions.
Exclusion Criteria
Patients are typically excluded from NHS Mounjaro prescription if they have:
- Type 1 diabetes or latent autoimmune diabetes (LADA)
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2 (MEN2)
- Severe gastroparesis or gastrointestinal disorders
- Pregnancy, breastfeeding, or planning pregnancy
- Severe renal impairment (eGFR <30 mL/min in some protocols)
- History of pancreatitis (relative contraindication requiring specialist assessment)
- Active eating disorders
- Inability to engage with monitoring requirements
For those exploring the scientific basis of peptide therapeutics, educational resources on peptide handling and research applications provide valuable context for understanding these medications’ mechanisms.
The NHS Prescription Pathway: Step-by-Step Process
Navigating the NHS system to obtain Mounjaro requires understanding a multi-stage process that can span many months. This section outlines the typical pathway patients encounter when seeking Mounjaro on the NHS: Current Position & Criteria approval.
Stage 1: Primary Care Assessment
Initial GP Consultation
The journey typically begins with a GP appointment where:
- Diabetes control is reviewed with recent HbA1c results
- Current medication regimen is assessed
- Weight and BMI are documented
- Comorbidities are catalogued
- Previous treatment responses are evaluated
Optimization of Existing Therapy
Before specialist referral, GPs often:
- Maximize metformin dosing if tolerated
- Add or optimize second-line agents
- Ensure adherence to current medications
- Provide lifestyle intervention support
- Address barriers to glycaemic control (medication timing, dietary patterns, etc.)
This optimization phase typically requires 3-6 months of documented attempts before progression to next stages.
Stage 2: Specialist Referral
Referral Criteria Verification
GP practices verify that patients meet local ICB criteria before submitting referrals to:
- Tier 3 weight management services
- Specialist diabetes clinics
- Obesity medicine services (where available)
Waiting Times
Current NHS waiting times for these services vary dramatically:
- Tier 3 services: 6-18 months in many areas
- Diabetes specialist clinics: 3-12 months
- Obesity medicine: 12-24 months (very limited availability)
Some regions have suspended new referrals to these services entirely due to capacity constraints, creating complete access barriers regardless of clinical need.
Stage 3: Specialist Assessment
Comprehensive Evaluation
Specialist appointments typically include:
✓ Detailed medical history review
✓ Medication history and previous treatment responses
✓ Physical examination including anthropometric measurements
✓ Blood tests (HbA1c, lipids, liver function, renal function, thyroid function)
✓ Assessment of complications (retinopathy, nephropathy, neuropathy, cardiovascular disease)
✓ Psychological assessment and readiness for intervention
✓ Dietary and activity pattern evaluation
Treatment Discussion
Specialists discuss:
- Available treatment options within local formulary
- Relative benefits and risks of different agents
- Injection technique and administration
- Side effect management
- Monitoring requirements
- Treatment goals and discontinuation criteria
Funding Application
If tirzepatide is deemed appropriate, specialists must:
- Complete individual funding request (IFR) forms in many ICBs
- Provide clinical justification against local criteria
- Document previous treatment failures
- Submit to ICB pharmacy or medicines management teams
Stage 4: ICB Review and Approval
Funding Decision Timeline
ICB review processes typically take:
- Standard applications: 4-8 weeks
- Exceptional funding requests: 8-12 weeks
- Appeals of declined applications: Additional 6-12 weeks
Approval Rates
Approval rates vary significantly but many ICBs report:
- 30-50% approval rate for tirzepatide requests
- Higher approval rates when all criteria are comprehensively documented
- Lower approval rates for weight management primary indications
Common Decline Reasons:
❌ Insufficient documentation of previous treatment trials
❌ BMI below threshold
❌ Inadequate specialist assessment
❌ Alternative cheaper options not exhausted
❌ Lack of engagement with lifestyle interventions
Stage 5: Prescription Initiation and Monitoring
Starting Treatment
Upon approval:
- Initial prescription issued (typically 1-3 months supply)
- Injection training provided
- Side effect management plan discussed
- Follow-up appointments scheduled
Ongoing Monitoring Requirements
NHS protocols typically mandate:
- Monthly reviews for first 3 months
- Quarterly HbA1c testing
- Weight and BMI tracking at each visit
- Renal function monitoring (6-12 monthly)
- Assessment of side effects and tolerability
- Review of treatment goals and continuation criteria
Discontinuation Criteria
Treatment may be discontinued if:
- HbA1c reduction <5.5 mmol/mol (0.5%) at 6 months
- Weight loss <3% at 6 months (when weight loss is treatment goal)
- Intolerable side effects develop
- Patient becomes pregnant
- Adherence issues emerge
- Patient preference changes
This complex, multi-stage pathway explains why many patients seeking Mounjaro on the NHS: Current Position & Criteria ultimately pursue private prescription routes instead.
Private Prescription Alternatives and Considerations
Given the substantial barriers to NHS access, private prescription has become the predominant route for patients seeking tirzepatide in the UK. Understanding this pathway, its costs, and considerations is essential for comprehensive treatment planning.
Private Healthcare Pathways
Private Endocrinology/Obesity Medicine Clinics
Specialist private clinics offer:
- Rapid appointment availability (typically 1-4 weeks)
- Comprehensive metabolic assessment
- Prescription of tirzepatide where clinically appropriate
- Ongoing monitoring and dose titration
- Integration with private pathology services
Initial consultation costs: £150-350
Follow-up appointments: £80-200
Monitoring blood tests: £50-150 per panel
Online Prescription Services
Regulated online medical services have emerged offering:
- Remote consultations via video or questionnaire
- Medical assessment by GMC-registered prescribers
- Direct medication supply to patient’s address
- Lower overhead costs than traditional private clinics
Typical costs: £120-200 per month including consultation and medication
⚠️ Important considerations: Ensure services are regulated by the Care Quality Commission (CQC) and prescribers are GMC-registered. Avoid unregulated suppliers.
Medication Costs
Mounjaro Pricing (2025)
Private prescription costs for branded Mounjaro:
| Dosage | Monthly Cost | Annual Cost |
|---|---|---|
| 2.5 mg starting dose | £150-180 | £1,800-2,160 |
| 5 mg | £180-220 | £2,160-2,640 |
| 7.5 mg | £200-240 | £2,400-2,880 |
| 10 mg | £220-260 | £2,640-3,120 |
| 12.5 mg | £240-280 | £2,880-3,360 |
| 15 mg (maximum) | £250-300 | £3,000-3,600 |
These costs represent substantial financial commitment, particularly as treatment duration typically extends 12-24 months or longer for sustained weight management.
Compounded Tirzepatide
Some private providers offer compounded tirzepatide at lower costs (£80-150 monthly), though this raises important considerations:
- Compounded medications lack the regulatory oversight of licensed products
- Quality, purity, and potency may vary
- MHRA has issued warnings about unlicensed compounded GLP-1 products[4]
- Insurance and legal protections may differ
Insurance Coverage
Private Medical Insurance (PMI)
Most UK private medical insurance policies exclude coverage for:
- Weight management treatments
- Medications for obesity
- Long-term medication costs for chronic conditions
Some policies may cover:
- Initial specialist consultations
- Diagnostic tests and monitoring
- Treatment of obesity-related complications
Always verify coverage with insurers before assuming costs will be reimbursed.
Hybrid NHS-Private Models
Some patients adopt hybrid approaches:
✓ NHS GP for ongoing diabetes management and monitoring
✓ Private specialist for tirzepatide prescription
✓ NHS pathology services for blood test monitoring
✓ Private pharmacy for medication supply
This model can reduce overall costs while maintaining continuity of care, though coordination between NHS and private providers requires careful management.
For researchers and laboratories investigating metabolic peptides and their mechanisms, research-grade peptide supplies provide high-purity compounds for controlled scientific study, distinct from clinical therapeutic use.
Research Peptides: Scientific Investigation and Laboratory Use

While clinical access to Mounjaro remains restricted, the scientific community continues investigating tirzepatide and related peptide compounds through rigorous laboratory research. Understanding the distinction between clinical therapeutics and research-grade materials is essential.
Research-Grade Tirzepatide and GLP-1 Analogues
PEPTIDE PRO supplies research-grade peptides to laboratories, universities, and research institutions across the UK and internationally. These materials are:
🔬 Manufactured to high purity standards for consistent experimental results
🔬 Clearly labelled “For Research Use Only” to prevent misuse
🔬 Accompanied by Certificates of Analysis (COAs) documenting purity and composition
🔬 Stored under controlled conditions to maintain stability
🔬 Supplied with handling and reconstitution guidance for laboratory protocols
Legitimate Research Applications
Research-grade peptides serve critical roles in:
Mechanistic Studies
Understanding receptor binding, signaling cascades, and metabolic effects at cellular and molecular levels.
Pharmacokinetic Research
Investigating absorption, distribution, metabolism, and excretion profiles in experimental models.
Comparative Pharmacology
Evaluating different peptide structures, modifications, and formulations for therapeutic potential.
Assay Development
Creating and validating analytical methods for peptide detection and quantification.
Educational Purposes
Training researchers in peptide handling, reconstitution, and experimental protocols.
Regulatory Framework for Research Peptides
Research peptides exist within a distinct regulatory framework:
- Not licensed for human consumption or therapeutic use
- Exempt from medicines regulations when clearly labelled for research only
- Subject to laboratory safety protocols and institutional oversight
- Require appropriate facilities and training for handling
Researchers must maintain clear documentation of:
✓ Intended research applications
✓ Institutional ethical approval where applicable
✓ Appropriate storage and handling procedures
✓ Waste disposal protocols
✓ Security measures preventing diversion
Accessing Research-Grade Peptides
Legitimate research peptide suppliers like PEPTIDE PRO implement verification procedures:
- Institutional affiliation confirmation for bulk orders
- Research purpose documentation
- Compliance with export regulations for international orders
- Age verification (18+ only)
- Clear terms prohibiting human consumption
Fast UK Delivery: Orders placed before 1pm Monday-Friday are dispatched same day in temperature-appropriate packaging with full tracking.
Extensive Catalogue: Beyond tirzepatide analogues, researchers can access semaglutide, BPC-157, TB-500, and numerous other research peptides for diverse experimental applications.
Ethical Considerations
The research community maintains strict ethical standards:
❗ Never represent research peptides as clinical medications
❗ Never supply to individuals for self-administration
❗ Always maintain clear labelling and documentation
❗ Report suspected misuse to appropriate authorities
❗ Educate about risks of unregulated peptide use
These safeguards protect both public health and the integrity of legitimate scientific research.
Comparing Mounjaro with Alternative Therapies
Understanding Mounjaro on the NHS: Current Position & Criteria requires context of alternative treatments available through NHS pathways. This comparison helps patients and clinicians make informed decisions.
GLP-1 Receptor Agonists Available on NHS
Semaglutide (Ozempic, Wegovy)
The most comparable alternative to Mounjaro:
- Mechanism: Single GLP-1 receptor agonist (vs. dual GIP/GLP-1 for tirzepatide)
- Efficacy: 10-15% weight loss in clinical trials (vs. 15-22% for tirzepatide)
- Dosing: Weekly injection
- NHS availability: More established, though still restricted
- Cost: Similar to Mounjaro (£150-250 monthly)
Liraglutide (Victoza, Saxenda)
Older GLP-1 agonist:
- Mechanism: GLP-1 receptor agonist
- Efficacy: 5-8% weight loss
- Dosing: Daily injection
- NHS availability: More widely available for diabetes; restricted for weight management
- Cost: £150-200 monthly
Dulaglutide (Trulicity)
Weekly GLP-1 agonist:
- Mechanism: GLP-1 receptor agonist
- Efficacy: Moderate weight loss (3-5%)
- Dosing: Weekly injection
- NHS availability: Reasonably available for diabetes management
- Cost: £120-180 monthly
Exenatide (Byetta, Bydureon)
First-generation GLP-1 agonist:
- Mechanism: GLP-1 receptor agonist
- Efficacy: Modest weight loss (2-4%)
- Dosing: Twice daily or weekly depending on formulation
- NHS availability: Widely available as established therapy
- Cost: £80-120 monthly
Other Diabetes Medications
SGLT2 Inhibitors (empagliflozin, dapagliflozin, canagliflozin)
- Mechanism: Increase glucose excretion in urine
- Weight loss: 2-3 kg average
- NHS availability: Good, particularly for patients with cardiovascular or renal disease
- Cost: £30-50 monthly
DPP-4 Inhibitors (sitagliptin, linagliptin, saxagliptin)
- Mechanism: Increase incretin levels
- Weight effect: Weight neutral
- NHS availability: Excellent
- Cost: £30-40 monthly
Insulin Therapy
- Various formulations (basal, bolus, premixed)
- Weight effect: Often causes weight gain
- NHS availability: Excellent
- Cost: £20-60 monthly
Weight Management Medications
Orlistat
- Mechanism: Lipase inhibitor reducing fat absorption
- Weight loss: 3-5% average
- NHS availability: Available but limited by tolerability
- Cost: £30-50 monthly (£20 NHS prescription)
Naltrexone-Bupropion (Mysimba)
- Mechanism: Combination affecting appetite and reward pathways
- Weight loss: 5-8% average
- NHS availability: Not routinely commissioned
- Cost: £150-200 monthly privately
Bariatric Surgery
For patients with BMI ≥40 or ≥35 with comorbidities who meet criteria:
Procedures Available:
- Gastric bypass
- Sleeve gastrectomy
- Gastric band
Efficacy: 25-35% total body weight loss
NHS Availability: Available but with extensive waiting lists (12-36 months)
Cost: Fully funded when criteria met; £8,000-15,000 privately
Comparative Effectiveness Summary
| Treatment | Weight Loss | HbA1c Reduction | NHS Access | Monthly Cost |
|---|---|---|---|---|
| Tirzepatide (Mounjaro) | 15-22% | 1.5-2.5% | Very restricted | £200-300 |
| Semaglutide | 10-15% | 1.5-2.0% | Restricted | £180-250 |
| Liraglutide | 5-8% | 1.0-1.5% | Moderate | £150-200 |
| Dulaglutide | 3-5% | 1.0-1.5% | Good | £120-180 |
| SGLT2 inhibitors | 2-3 kg | 0.5-1.0% | Excellent | £30-50 |
| Bariatric surgery | 25-35% | 2.0-3.0% | Moderate (long waits) | £0 NHS |
This comparison demonstrates why Mounjaro generates such interest despite access barriers—its superior efficacy in both glycaemic control and weight management makes it highly attractive for appropriate patients.
Future Outlook: Anticipated Changes to NHS Access
The landscape of Mounjaro on the NHS: Current Position & Criteria continues evolving. Several factors may influence future access:
Regulatory Developments
Expanded Licensing
The MHRA is reviewing tirzepatide for:
- Obesity treatment as standalone indication (beyond diabetes)
- Cardiovascular risk reduction (following ongoing trials)
- Broader patient populations
Approval for obesity treatment would significantly impact NHS commissioning decisions and NICE guidance.
NICE Guidance Updates
Technology Appraisal Reviews
NICE conducts periodic reviews incorporating:
- New clinical trial data
- Real-world effectiveness evidence
- Updated health economic modeling
- Price negotiations with manufacturers
Anticipated Timeline: Updated guidance expected 2025-2026 incorporating:
- SURMOUNT obesity trial data
- Long-term cardiovascular outcome studies
- Cost-effectiveness reassessments
NHS Capacity Expansion
Tier 3 Service Investment
NHS England has committed funding to expand:
- Weight management service capacity
- Specialist diabetes clinics
- Integrated care pathways
However, workforce shortages (specialist nurses, dietitians, endocrinologists) limit rapid expansion regardless of funding.
Pharmaceutical Pricing
Patent Expiry and Biosimilars
Tirzepatide patent protection extends into the 2030s, meaning:
- No generic/biosimilar competition imminent
- Pricing remains controlled by manufacturer
- NHS negotiating position limited
Volume-Based Pricing Agreements
NHS England may negotiate:
- Reduced per-unit costs with increased volume commitments
- Outcome-based pricing models
- Risk-sharing arrangements
Such agreements could improve cost-effectiveness profiles and expand access.
Political and Policy Factors
Government Priorities
The obesity and diabetes epidemic represents:
- £27 billion annual NHS cost (diabetes alone)[5]
- Significant workforce productivity impacts
- Major health inequality driver
Political pressure to address these issues may drive:
✓ Increased investment in prevention and treatment
✓ Prioritization of cost-effective interventions
✓ Integration of pharmaceutical and lifestyle approaches
Public Health Strategies
Comprehensive approaches combining:
- Medication access for appropriate patients
- Population-level prevention initiatives
- Environmental and policy interventions
- Health education and behaviour change support
Technological Innovations
Digital Health Integration
Remote monitoring, telemedicine, and digital therapeutics may:
- Reduce specialist appointment requirements
- Enable broader geographic access
- Improve treatment adherence and outcomes
- Lower overall programme costs
Artificial Intelligence
AI-driven tools for:
- Patient selection and risk stratification
- Personalized treatment optimization
- Predictive modeling of treatment response
- Efficient resource allocation
Realistic Expectations for 2025-2027
Likely Scenarios:
📈 Gradual expansion of NHS access for diabetes patients meeting strict criteria
📈 Continued restrictions for weight management without diabetes diagnosis
📈 Persistent regional variation in access and approval rates
📈 Growing private prescription market as primary access route
📈 Increased focus on treatment discontinuation and long-term sustainability
Unlikely Scenarios:
📉 Unrestricted NHS access for all eligible patients
📉 Removal of BMI thresholds or comorbidity requirements
📉 GP-initiated prescribing without specialist input
📉 Significant price reductions enabling mass rollout
For those interested in the evolving science of metabolic peptides, staying informed through educational resources on peptide research and mechanisms provides valuable context for understanding these therapeutic developments.
Patient Perspectives and Real-World Experiences
Understanding Mounjaro on the NHS: Current Position & Criteria from patient perspectives provides crucial context beyond clinical and regulatory frameworks.
Common Patient Experiences
Successful NHS Access
Patients who successfully navigate NHS pathways typically report:
✅ Lengthy process (12-24 months from initial GP discussion to first prescription)
✅ Multiple appointments with various healthcare professionals
✅ Extensive documentation requirements and form completion
✅ Requirement to “fail” multiple other treatments before approval
✅ Excellent support once approved with regular monitoring and specialist input
“After 18 months of trying different medications and attending tier 3 services, I was finally approved for Mounjaro. The results have been life-changing—I’ve lost 42 pounds and my HbA1c has dropped from 78 to 52. The wait was frustrating, but the NHS support has been excellent.”
— Patient testimonial (anonymized)
NHS Application Declined
Patients whose applications are declined report:
❌ Frustration and disappointment after lengthy process
❌ Lack of clear explanation for decline decisions
❌ Difficulty accessing appeals processes
❌ Feeling of postcode lottery when comparing to others’ experiences
❌ Financial barriers to private alternatives
“My specialist recommended Mounjaro and submitted all the paperwork, but the ICB declined without clear reasoning. I can’t afford £250 monthly for private prescription, so I’m stuck with medications that aren’t working well. It feels incredibly unfair.”
— Patient testimonial (anonymized)
Private Prescription Route
Patients accessing tirzepatide privately report:
💷 Significant financial burden but worthwhile investment for many
💷 Faster access (weeks rather than months/years)
💷 More flexible prescribing based on clinical judgment
💷 Concerns about long-term affordability and treatment sustainability
💷 Variable quality of private provider support and monitoring
Healthcare Professional Perspectives
GPs’ Views
Primary care physicians report:
- Difficulty navigating complex local formularies and criteria
- Frustration with restrictive commissioning decisions
- Time-consuming referral and documentation processes
- Concern about patient expectations vs. realistic access
- Need for clearer guidance and pathways
Specialist Perspectives
Endocrinologists and obesity medicine specialists note:
- Clinical evidence strongly supports tirzepatide use
- Funding restrictions prevent optimal patient care
- Administrative burden of individual funding requests
- Ethical challenges of unequal access
- Need for sustainable, equitable commissioning models
Support and Advocacy Resources
Patient Organizations
Several organizations provide support and advocacy:
- Diabetes UK: Information, support groups, policy advocacy
- Obesity UK: Patient support and healthcare professional resources
- National Obesity Forum: Clinical guidance and patient information
Online Communities
Social media and forum communities offer:
- Peer support and experience sharing
- Practical advice on navigating NHS systems
- Information about private prescription options
- Emotional support during treatment journeys
⚠️ Caution: Online communities sometimes promote unregulated sources or inappropriate use. Always verify information with qualified healthcare professionals.
Safety Considerations and Side Effect Management
Comprehensive understanding of Mounjaro on the NHS: Current Position & Criteria must include safety profiles and side effect management, whether accessing through NHS or private routes.
Common Side Effects
Gastrointestinal Effects (most frequent):
- Nausea (30-40% of patients, typically mild-moderate)
- Diarrhea (20-30%)
- Vomiting (10-20%)
- Constipation (10-15%)
- Abdominal pain (10-15%)
- Dyspepsia (10-15%)
Management strategies:
✓ Start at lowest dose and titrate gradually
✓ Take with food or adjust meal timing
✓ Stay well hydrated
✓ Avoid high-fat, spicy, or large meals
✓ Use anti-nausea medications if needed (domperidone, metoclopramide)
Injection Site Reactions:
- Redness, swelling, or itching at injection site (5-10%)
- Usually mild and self-limiting
Management: Rotate injection sites, apply cold compress, ensure proper injection technique
Serious Adverse Effects (Rare)
Pancreatitis
- Incidence: <1%
- Symptoms: Severe persistent abdominal pain, nausea, vomiting
- Action: Discontinue immediately and seek urgent medical attention
Gallbladder Disease
- Rapid weight loss increases gallstone risk
- Symptoms: Right upper quadrant pain, nausea
- Monitoring: Clinical vigilance; imaging if symptomatic
Hypoglycemia
- Risk increased when combined with sulfonylureas or insulin
- Symptoms: Sweating, tremor, confusion, dizziness
- Prevention: Dose reduction of concurrent medications as needed
Thyroid Tumors
- Theoretical risk based on animal studies
- Contraindicated in patients with personal/family history of medullary thyroid cancer or MEN2
- Monitoring: Clinical assessment; calcitonin testing not routinely recommended
Renal Impairment
- Dehydration from GI side effects can worsen renal function
- Monitoring: Regular renal function tests, especially in at-risk patients
Contraindications and Precautions
Absolute Contraindications:
🚫 Personal or family history of medullary thyroid carcinoma
🚫 Multiple endocrine neoplasia syndrome type 2
🚫 Hypersensitivity to tirzepatide or excipients
🚫 Pregnancy or breastfeeding
Relative Contraindications/Cautions:
⚠️ History of pancreatitis
⚠️ Severe gastroparesis
⚠️ Inflammatory bowel disease
⚠️ Severe renal impairment
⚠️ Diabetic retinopathy (rapid glucose reduction may temporarily worsen)
Drug Interactions
Medications Requiring Dose Adjustment:
- Insulin: Reduce dose to prevent hypoglycemia
- Sulfonylureas: Reduce dose to prevent hypoglycemia
- Oral medications: Delayed gastric emptying may affect absorption (take 1 hour before tirzepatide)
- Warfarin: Monitor INR more frequently during initiation
Monitoring Requirements
Baseline Assessments:
✓ HbA1c, fasting glucose
✓ Renal function (eGFR, creatinine)
✓ Liver function tests
✓ Lipid profile
✓ Thyroid function
✓ Blood pressure
✓ Weight and BMI
✓ Retinal screening (diabetic patients)
Ongoing Monitoring:
- Monthly for first 3 months: Weight, side effects, tolerability
- Quarterly: HbA1c, weight, blood pressure
- 6-12 monthly: Renal function, liver function, lipids
- As needed: Retinal screening, cardiovascular assessment
Long-Term Safety Considerations
Duration of Treatment
Clinical trials have demonstrated safety up to 2+ years. Long-term safety beyond this remains under investigation through:
- Extension studies
- Real-world evidence registries
- Post-marketing surveillance
Weight Regain After Discontinuation
Studies show significant weight regain (50-80% of lost weight) within 12 months of stopping GLP-1 receptor agonists[6]. This raises questions about:
- Optimal treatment duration
- Strategies for maintaining weight loss
- Cost-effectiveness of indefinite treatment
- Psychological impacts of weight regain
Cardiovascular Outcomes
Ongoing trials (SURPASS-CVOT) are investigating:
- Major adverse cardiovascular event reduction
- Heart failure outcomes
- Stroke prevention
- All-cause mortality
Results expected 2025-2026 will significantly inform clinical practice and commissioning decisions.
For researchers investigating peptide safety profiles and mechanisms, high-purity research peptides enable controlled laboratory studies under appropriate safety protocols.
Practical Guidance for Patients Considering Mounjaro
For individuals exploring Mounjaro on the NHS: Current Position & Criteria, this practical guidance provides actionable steps.
Step 1: Assess Your Eligibility
Self-Assessment Checklist:
Do you have:
️ Confirmed Type 2 diabetes diagnosis? ☑
️ HbA1c above target despite current medications? ☑
️ BMI ≥35 (with comorbidities) or ≥40? ☑
️ Tried metformin and at least one other diabetes medication? ☑
️ Documented inadequate response to current treatments? ☑
️ No contraindications (thyroid cancer history, MEN2, pregnancy)? ☑
️ Ability to attend regular monitoring appointments? ☑
️ Commitment to lifestyle modifications alongside medication?
If you answered “yes” to most questions, you may meet basic eligibility criteria, though local ICB requirements may be more stringent.
Step 2: Prepare for GP Discussion
Documentation to Gather:
📋 Recent HbA1c results (last 6-12 months)
📋 Current medication list with doses and duration
📋 Record of previous medications tried and responses
📋 Weight history
📋 List of comorbidities and complications
📋 Lifestyle modification attempts (diet programmes, exercise, etc.)
Questions to Ask Your GP:
- “Do I meet the local ICB criteria for GLP-1 receptor agonist prescription?”
- “What additional treatments do I need to try before specialist referral?”
- “What is the typical waiting time for tier 3/specialist diabetes services in this area?”
- “Can you optimize my current medications while I’m waiting?”
- “What documentation do I need to support a specialist referral?”
Step 3: Optimize Current Management
While navigating NHS pathways:
✅ Maximize current medication adherence and dosing
✅ Engage with lifestyle interventions (diet, physical activity, sleep, stress management)
✅ Document your efforts (food diaries, activity logs, weight tracking)
✅ Address barriers to glycaemic control (medication timing, dietary patterns, etc.)
✅ Attend all appointments and monitoring sessions
This demonstrates commitment to treatment and strengthens specialist referral applications.
Step 4: Navigate Specialist Services
Preparing for Specialist Appointment:
- Bring all documentation (test results, medication history, weight records)
- Prepare questions about treatment options
- Be honest about lifestyle, adherence, challenges
- Discuss realistic goals and expectations
- Ask about local approval rates and timelines
During Assessment:
- Provide comprehensive medical history
- Discuss previous treatment experiences thoroughly
- Express commitment to monitoring and lifestyle changes
- Ask about alternative options if Mounjaro isn’t approved
- Request written summary of recommendations
Step 5: Consider Alternative Pathways
If NHS Access Delayed or Declined:
Option 1: Appeal Process
- Request detailed decline reasoning
- Work with specialist to address gaps in application
- Submit formal appeal with additional documentation
- Seek patient advocacy support if needed
Option 2: Private Prescription
- Research regulated private providers (CQC-registered)
- Compare costs and service offerings
- Verify prescriber credentials (GMC-registered)
- Ensure ongoing monitoring included
- Budget for long-term treatment costs
Option 3: Alternative NHS Medications
- Discuss semaglutide, liraglutide, or dulaglutide availability
- Consider SGLT2 inhibitors or other options
- Optimize combination therapy
- Reassess Mounjaro eligibility in future
Option 4: Bariatric Surgery Pathway
- Discuss surgical options if BMI and criteria met
- Begin referral process (lengthy waiting times)
- Attend pre-surgical assessments
- Consider as definitive treatment option
Step 6: Manage Treatment Effectively
If Mounjaro Prescribed:
✓ Attend injection training and practice proper technique
✓ Start at lowest dose (2.5 mg) and titrate as directed
✓ Monitor and report side effects promptly
✓ Maintain lifestyle modifications (medication is adjunct, not replacement)
✓ Attend all monitoring appointments
✓ Track progress (weight, HbA1c, symptoms, quality of life)
✓ Communicate with healthcare team about challenges or concerns
Step 7: Plan for Long-Term Management
Sustainability Considerations:
- Discuss treatment duration expectations with specialists
- Develop maintenance strategies for weight and glycaemic control
- Address psychological aspects of weight management
- Plan for potential treatment discontinuation
- Build sustainable lifestyle habits independent of medication
Financial Planning for Private Prescription
Budgeting Framework:
| Cost Component | Monthly | Annual |
|---|---|---|
| Medication | £200-300 | £2,400-3,600 |
| Private consultations | £50-100 | £600-1,200 |
| Monitoring blood tests | £30-50 | £360-600 |
| Total | £280-450 | £3,360-5,400 |
Cost Reduction Strategies:
💰 Use NHS GP for monitoring where possible
💰 Request NHS pathology for blood tests
💰 Compare private provider pricing
💰 Consider payment plans if offered
💰 Budget for 12-24 month treatment duration minimum
Frequently Asked Questions
Can I get Mounjaro on the NHS for weight loss alone (without diabetes)?
Currently, Mounjaro on the NHS: Current Position & Criteria requires Type 2 diabetes diagnosis in virtually all ICBs. Weight management as sole indication is not routinely commissioned, even when BMI thresholds are met. NICE has not issued positive guidance for obesity treatment without diabetes, creating a significant barrier to NHS access for this indication.
Exceptional funding requests may be considered in rare cases with:
- Extreme obesity (BMI >50)
- Multiple severe comorbidities
- Failed bariatric surgery
- Specialist recommendation with strong clinical justification
However, approval rates for such requests remain very low.
How long does the NHS approval process typically take?
The complete pathway from initial GP discussion to first Mounjaro prescription typically spans:
- Minimum: 6-9 months (with existing specialist relationship and rapid approval)
- Typical: 12-18 months (including referral waits and approval processes)
- Maximum: 24+ months (in areas with suspended referrals or multiple appeal cycles)
Factors affecting timeline:
⏱️ Local specialist service waiting times
⏱️ ICB review process efficiency
⏱️ Completeness of initial application
⏱️ Need for appeals or additional documentation
What happens if I don’t lose enough weight on Mounjaro?
NHS protocols typically include discontinuation criteria:
- <3% weight loss at 6 months (when weight loss is treatment goal)
- <5.5 mmol/mol HbA1c reduction at 6 months (for diabetes management)
If targets aren’t met:
- Specialist reviews adherence, dosing, lifestyle factors
- Considers dose escalation if not at maximum
- Assesses barriers to effectiveness
- May discontinue if inadequate response confirmed
- Discusses alternative treatment options
Some patients respond better to alternative GLP-1 agonists, so switching may be considered before complete discontinuation.
Can I switch from private to NHS prescription later?
Potentially, though challenges exist:
Favorable Factors:
✓ Demonstrated effectiveness (weight loss, HbA1c improvement)
✓ Good tolerability and adherence
✓ Meeting NHS eligibility criteria
✓ Specialist support for transition
Challenges:
❌ Some ICBs refuse to fund medications initiated privately
❌ May require discontinuation and trial of NHS-preferred alternatives first
❌ Documentation requirements may be extensive
❌ No guarantee of approval despite private success
Recommended Approach:
- Discuss NHS pathway with GP before starting private treatment
- Maintain thorough documentation of treatment and response
- Work with specialist willing to support NHS funding application
- Be prepared for potential treatment interruption during transition
Are there patient assistance programs for Mounjaro?
UK-Specific Programmes:
Eli Lilly (Mounjaro manufacturer) does not currently offer patient assistance programs in the UK comparable to US programmes. However:
- Some private clinics negotiate bulk pricing discounts
- Payment plan options may be available through providers
- Charitable organizations occasionally provide limited support for exceptional cases
NHS Access Remains Primary Route for cost-free access, though with significant eligibility restrictions.
What’s the difference between research peptides and prescription Mounjaro?
Critical distinctions exist:
Prescription Mounjaro:
✓ Licensed medicine regulated by MHRA
✓ Manufactured to pharmaceutical GMP standards
✓ Prescribed by qualified medical professionals
✓ Intended for human therapeutic use
✓ Quality, purity, and safety rigorously tested
✓ Accompanied by medical supervision and monitoring
Research Peptides (from suppliers like PEPTIDE PRO):
✓ For laboratory and scientific research only
✓ Not licensed for human consumption
✓ Manufactured for research applications
✓ Supplied with “For Research Use Only” labelling
✓ No medical supervision or therapeutic guidance
✓ Intended for controlled experimental settings
⚠️ CRITICAL WARNING: Research peptides should NEVER be used for self-administration or therapeutic purposes. Doing so:
- Violates medicines regulations
- Carries serious health risks
- Lacks medical supervision and monitoring
- May involve unknown purity or composition
- Could result in severe adverse effects
Always obtain therapeutic peptides through legitimate medical channels with appropriate prescribing and monitoring.
Navigating Mounjaro Access in 2025
The landscape of Mounjaro on the NHS: Current Position & Criteria in 2025 reflects the complex intersection of clinical innovation, health economics, regulatory frameworks, and healthcare capacity constraints. While tirzepatide represents a significant therapeutic advance for Type 2 diabetes and weight management, NHS access remains highly restricted, creating challenges for patients and clinicians alike.
Key Points Summary
Current NHS Position:
- Mounjaro is licensed for Type 2 diabetes treatment in the UK
- NHS commissioning is highly restrictive with stringent eligibility criteria
- Access requires diabetes diagnosis, BMI thresholds, previous treatment failures, and specialist assessment
- Significant regional variation creates postcode lottery effects
- Lengthy approval processes (typically 12-24 months) deter many patients
Eligibility Criteria:
- Type 2 diabetes with inadequate glycaemic control (HbA1c ≥58 mmol/mol typically)
- BMI ≥35 with comorbidities or ≥40 without
- Documented trials of metformin plus additional oral agents
- Specialist assessment and recommendation
- Ability to engage with monitoring and lifestyle interventions
Alternative Pathways:
- Private prescription offers faster access but significant costs (£2,400-3,600 annually)
- Alternative NHS medications (semaglutide, liraglutide, SGLT2 inhibitors) may be more accessible
- Bariatric surgery remains option for appropriate candidates
- Research peptides serve legitimate scientific purposes but never for self-administration
Future Outlook:
- Gradual expansion of NHS access likely for diabetes patients
- Continued restrictions for weight management without diabetes
- Updated NICE guidance expected 2025-2026
- Price negotiations and capacity expansion may improve access
- Digital health integration could streamline pathways
Actionable Next Steps
For Patients Seeking NHS Access:
- Assess eligibility against local ICB criteria
- Optimize current diabetes management and document efforts
- Discuss with GP about specialist referral pathways
- Prepare comprehensive documentation of treatment history
- Engage with lifestyle interventions while navigating approval process
- Set realistic timeline expectations (12-24 months typical)
- Consider alternatives if NHS access delayed or declined
For Those Considering Private Prescription:
- Research regulated providers (CQC-registered, GMC prescribers)
- Budget for long-term costs (minimum 12-24 months)
- Verify monitoring and support included in service
- Maintain NHS GP relationship for ongoing care coordination
- Document treatment response thoroughly
- Plan for sustainability and potential NHS transition
For Healthcare Professionals:
- Familiarize with local ICB criteria and formularies
- Optimize existing therapies before specialist referral
- Prepare comprehensive referral documentation
- Set realistic patient expectations about timelines and approval likelihood
- Advocate for equitable access through professional organizations
- Stay informed about evolving guidance and commissioning decisions
For Researchers:
- Source research peptides from reputable suppliers like PEPTIDE PRO
- Maintain clear documentation of research purposes
- Follow appropriate handling protocols and safety procedures
- Never represent research materials as therapeutic products
- Contribute to evidence base through rigorous scientific investigation
- Educate about risks of unregulated peptide use
While Mounjaro on the NHS: Current Position & Criteria presents significant access challenges in 2025, understanding the regulatory landscape, eligibility requirements, and alternative pathways empowers patients and clinicians to navigate these complexities effectively. The balance between clinical innovation, cost-effectiveness, and equitable access will continue evolving as evidence accumulates, capacity expands, and policy frameworks adapt.
For those unable to access Mounjaro through NHS pathways, numerous alternative evidence-based treatments exist—both pharmaceutical and surgical—that can achieve meaningful improvements in glycaemic control and weight management. The key is working collaboratively with qualified healthcare professionals to identify the most appropriate, sustainable, and accessible treatment approach for individual circumstances.
The future likely holds expanded access as health economic evidence strengthens, capacity increases, and political prioritization of obesity and diabetes management grows. Until then, informed decision-making, realistic expectations, and comprehensive treatment planning remain essential for navigating the current landscape.
For scientific and research communities, continued investigation of peptide mechanisms, efficacy, and safety through rigorous laboratory studies will inform future therapeutic developments. PEPTIDE PRO remains committed to supplying high-purity research-grade peptides to support this vital scientific work, maintaining clear distinctions between research applications and clinical therapeutics.
References
[1] Frias JP, et al. (2021). “Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes.” New England Journal of Medicine, 385(6):503-515.
[2] Jastreboff AM, et al. (2022). “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine, 387(3):205-216.
[3] National Institute for Health and Care Excellence (NICE). (2024). “Type 2 diabetes in adults: management.” NICE guideline [NG28]. Updated December 2024.
[4] Medicines and Healthcare products Regulatory Agency (MHRA). (2024). “Patient safety alert: Compounded GLP-1 receptor agonists.” MHRA Publication, March 2024.
[5] Diabetes UK. (2024). “The Cost of Diabetes Report 2024.” Diabetes UK Publications.
[6] Wilding JPH, et al. (2022). “Weight regain and cardiometabolic effects after withdrawal of semaglutide.” Diabetes, Obesity and Metabolism, 24(8):1553-1564.
