Retatrutide Nasal Spray: Is It Real? Separating Fact from Fiction in 2025

The question “Retatrutide Nasal Spray: Is It Real?” has surged across research forums, peptide communities, and scientific discussions throughout 2025. As retatrutide continues generating significant interest for its triple-agonist mechanism and promising metabolic research applications, claims about alternative delivery methods—particularly nasal spray formulations—have emerged online. But do these claims hold scientific merit, or are they simply marketing misinformation? This comprehensive investigation examines the current state of retatrutide delivery methods, the science behind peptide administration routes, and what researchers need to know about authentic, research-grade formulations.
Key Takeaways
- Retatrutide is currently available only as an injectable peptide—no legitimate nasal spray formulation exists in 2025
- Peptide molecular structure determines delivery feasibility—retatrutide’s size and properties make nasal absorption highly unlikely
- Unverified delivery methods pose research integrity risks—only validated administration routes ensure reliable experimental outcomes
- Research-grade suppliers provide injectable lyophilized formulations—proper reconstitution and handling protocols are essential
- Understanding peptide pharmacokinetics prevents costly research errors—delivery method directly impacts bioavailability and results
Understanding Retatrutide: The Science Behind the Peptide

What Is Retatrutide?
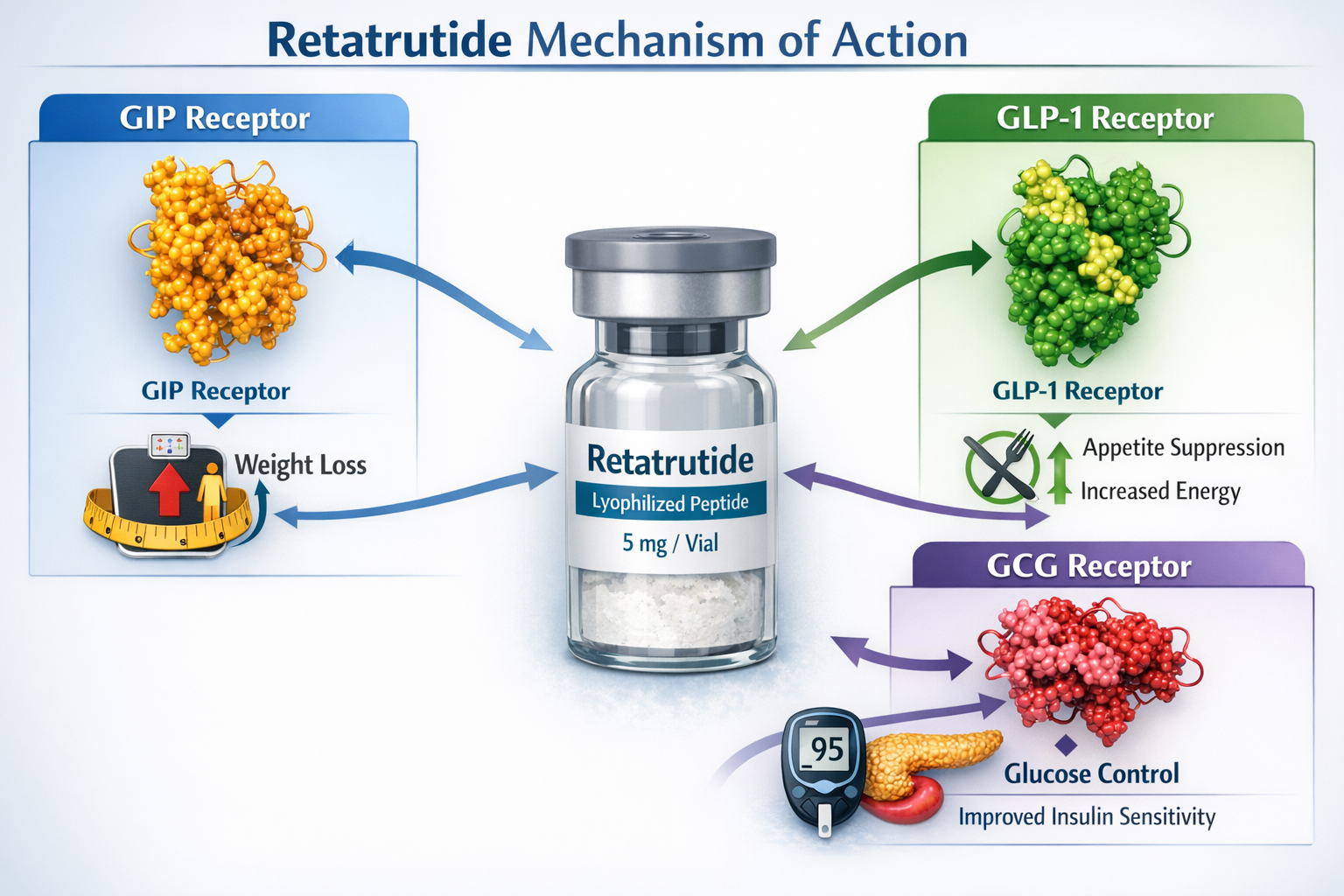
Retatrutide represents a sophisticated advancement in peptide research, functioning as a triple receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon (GCG) receptors[1]. This unique multi-receptor approach distinguishes it from earlier single or dual-agonist compounds, offering researchers a powerful tool for investigating metabolic pathways, energy regulation, and glucose homeostasis.
The peptide’s molecular structure consists of a 39-amino acid sequence with specific modifications that enhance receptor binding affinity and metabolic stability[2]. These structural characteristics directly influence how the compound can be administered and absorbed—a critical factor when evaluating claims about alternative delivery methods.
Current Research Applications
Research institutions and laboratories worldwide utilize retatrutide to explore:
- Metabolic regulation mechanisms and energy expenditure pathways
- Glucose homeostasis and insulin sensitivity modulation
- Appetite regulation through central nervous system receptor activation
- Body composition changes in controlled experimental settings
- Cardiovascular metabolic markers and lipid profile alterations
For researchers seeking high-purity research-grade peptides, understanding the legitimate formulation and delivery methods is paramount to experimental validity.
Molecular Properties That Matter
Retatrutide’s molecular weight exceeds 4,800 Daltons—a specification that becomes crucial when evaluating nasal delivery feasibility[3]. Peptides of this size face significant absorption barriers through mucosal membranes, particularly the nasal epithelium. The compound also exhibits:
- Hydrophilic characteristics that limit passive membrane diffusion
- Susceptibility to enzymatic degradation in non-sterile environments
- Specific pH stability requirements for maintaining bioactivity
- Temperature sensitivity necessitating controlled storage conditions
These properties explain why authentic retatrutide is supplied as lyophilized (freeze-dried) powder requiring reconstitution with bacteriostatic water before subcutaneous administration.
Retatrutide Nasal Spray: Is It Real? Examining the Evidence
The Injectable Reality
The definitive answer to “Retatrutide Nasal Spray: Is It Real?” is straightforward: No legitimate nasal spray formulation of retatrutide exists in the current research peptide market as of 2025. All verified, research-grade retatrutide is supplied as:
✅ Lyophilized powder in sealed sterile pen peptides
✅ Injectable formulation designed for subcutaneous administration
✅ Reconstitutable preparation using bacteriostatic water
✅ Temperature-controlled storage requirements (-20°C before reconstitution)
Reputable suppliers like PEPTIDE PRO provide retatrutide in various dosage strengths—including 40mg pen peptides—exclusively in injectable form with complete handling documentation and certificates of analysis.
Why Nasal Spray Claims Emerge
The proliferation of nasal spray claims stems from several factors:
- Consumer convenience marketing: Nasal administration appears less invasive than injections, creating market appeal
- Confusion with other peptides: Some smaller peptides (like certain nootropics) do have nasal formulations, creating false equivalency
- Unverified online vendors: Questionable suppliers make unsubstantiated claims to differentiate products
- Misunderstanding of peptide pharmacology: Lack of scientific literacy enables misinformation spread
Scientific Barriers to Nasal Delivery
The nasal mucosa presents formidable absorption challenges for large peptides like retatrutide:
| Barrier Type | Challenge for Retatrutide | Impact on Bioavailability |
|---|---|---|
| Molecular size | >4,800 Da exceeds nasal absorption threshold | Negligible absorption |
| Mucociliary clearance | Rapid removal before absorption | <5 minute residence time |
| Enzymatic degradation | Proteases in nasal cavity | Peptide breakdown before absorption |
| pH variation | Nasal pH (5.5-6.5) may denature peptide | Loss of bioactivity |
| Membrane permeability | Tight junctions prevent large molecule passage | <1% absorption efficiency |
Research demonstrates that peptides exceeding 1,000 Daltons experience dramatically reduced nasal bioavailability without specialized permeation enhancers—which themselves may compromise peptide integrity[4].
What About Permeation Enhancers?
Some might argue that permeation enhancers or absorption promoters could enable nasal delivery. However:
- These additives often cause mucosal irritation and damage
- They provide inconsistent absorption rates, compromising experimental reproducibility
- They may alter peptide structure or receptor binding characteristics
- No validated formulation exists for retatrutide with such enhancers
- Regulatory and safety considerations make such formulations impractical for research
Legitimate Retatrutide Administration: Best Practices for Researchers
Proper Reconstitution Protocol
Research-grade retatrutide requires precise reconstitution to maintain peptide integrity and experimental validity:
Step-by-Step Reconstitution:
- Remove pen peptide from storage and allow to reach room temperature (15-20 minutes)
- Prepare bacteriostatic water (typically 2-3 mL depending on desired concentration)
- Inject water slowly down the pen peptide side, avoiding direct stream onto lyophilized powder
- Gentle swirling (never shake) until powder completely dissolves
- Visual inspection for clarity and absence of particulates
- Immediate refrigeration at 2-8°C after reconstitution
For detailed guidance, researchers can consult educational resources on proper peptide handling and storage protocols.
Storage Requirements
⚠️ Critical Storage Parameters:
- Lyophilized peptide: -20°C to -80°C, protected from light
- Reconstituted solution: 2-8°C, use within 14-28 days
- Avoid freeze-thaw cycles: Maintain consistent temperature
- Sterile conditions: Always use aseptic technique during handling
Dosing Considerations for Research
While dosing protocols vary based on specific research objectives, typical experimental parameters include:
- Subcutaneous administration as the validated delivery route
- Consistent timing to maintain stable experimental conditions
- Proper documentation of administration sites and rotation
- Sterile technique using appropriate gauge needles (typically 27-31G)
Researchers requiring specific formulations can explore various peptide options from verified suppliers maintaining strict quality standards.
The Dangers of Unverified Delivery Methods
Research Integrity Risks
Using unverified or non-existent delivery methods like supposed nasal sprays introduces critical experimental flaws:
� Compromised bioavailability: Unknown or negligible peptide absorption �
� Irreproducible results: Inconsistent delivery undermines experimental validity �
� Wasted resources: Financial and time investment in ineffective protocols �
� Contamination risks: Non-sterile formulations may introduce variables �
� Data integrity concerns: Results cannot be compared to established literature
Financial and Safety Considerations
Beyond scientific concerns, pursuing non-existent formulations creates:
- Financial waste on products that cannot deliver claimed benefits
- Opportunity cost of delayed legitimate research
- Potential contamination from unverified sources
- Regulatory complications if using unapproved formulations
How to Identify Legitimate Suppliers
Researchers should verify supplier credentials through:
✅ Certificates of Analysis (COA) for each batch
✅ HPLC purity verification (≥98% for research-grade)
✅ Proper labeling including “For Research Use Only”
✅ Transparent contact information and professional support
✅ Established reputation within the research community
✅ Temperature-controlled shipping with appropriate packaging
Trusted suppliers like PEPTIDE PRO provide comprehensive product documentation, rapid UK delivery, and professional customer support for research institutions.
Alternative Peptides and Actual Nasal Formulations
Peptides That Do Have Nasal Formulations
While retatrutide nasal spray doesn’t exist, certain smaller peptides have legitimate intranasal formulations:
Peptides with Validated Nasal Delivery:
- Desmopressin (1,069 Da): Antidiuretic hormone analogue
- Oxytocin (1,007 Da): Neurohypophysial peptide
- Calcitonin (3,432 Da): With specialized formulation enhancers
- Certain nootropic peptides: Selank, Semax (smaller molecular weight)
Notice the molecular weight threshold: peptides successfully delivered nasally typically remain below 1,500 Daltons or require extensive formulation development with enhancers.
Why Size Matters in Peptide Delivery
The “500 Dalton rule” suggests that molecules exceeding this weight face absorption challenges through biological membranes[5]. While some exceptions exist with specialized formulations, retatrutide’s 4,800+ Dalton molecular weight places it far beyond feasible nasal delivery without breakthrough formulation technology—which currently doesn’t exist for this compound.
Comparing Delivery Routes
| Delivery Method | Bioavailability | Onset | Duration | Retatrutide Feasibility |
|---|---|---|---|---|
| Subcutaneous injection | 80-100% | 2-4 hours | Extended | ✅ Standard method |
| Intramuscular injection | 85-100% | 1-3 hours | Extended | ⚠️ Possible but not standard |
| Intravenous | 100% | Immediate | Shorter | ⚠️ Research setting only |
| Nasal spray | <1% for large peptides | Variable | Inconsistent | ❌ Not viable |
| Oral | <1% (degraded) | N/A | N/A | ❌ Not viable |
The Future of Peptide Delivery Technology
Emerging Research in Peptide Administration
While nasal retatrutide isn’t currently viable, peptide delivery research continues advancing:
Innovative Approaches Under Investigation:
- Nanoparticle encapsulation: Protecting peptides from enzymatic degradation
- Permeation enhancer technology: Improving mucosal absorption without damage
- Microneedle patches: Minimally invasive transdermal delivery
- Oral formulation strategies: Enteric coating and absorption enhancers
- Pulmonary delivery systems: Inhalation-based peptide administration
However, these technologies remain experimental and are not yet available for retatrutide or most large peptide compounds.
Timeline for Alternative Formulations
Realistically, alternative delivery methods for large peptides like retatrutide face:
- 5-10 years minimum for basic research and formulation development
- Extensive stability testing requirements
- Bioavailability validation across diverse conditions
- Regulatory approval processes for new delivery systems
- Manufacturing scalability challenges
Until such breakthroughs occur, subcutaneous injection remains the gold standard for retatrutide administration in research settings.
What This Means for Researchers
Current best practices dictate:
- Use validated delivery methods (subcutaneous injection)
- Source from reputable suppliers providing proper documentation
- Follow established protocols from peer-reviewed literature
- Maintain proper storage and handling procedures
- Document methodology precisely for reproducibility
Researchers can access comprehensive peptide selections from established suppliers committed to quality and transparency.
Making Informed Research Decisions
Questions to Ask Suppliers
When evaluating peptide sources, researchers should inquire about:
📋 Product Specifications:
- What is the peptide purity percentage?
- Can you provide batch-specific COA documentation?
- What analytical methods verify purity (HPLC, MS)?
- What is the peptide sequence and molecular weight?
📋 Handling and Storage:
- What are the storage temperature requirements?
- What is the shelf life before and after reconstitution?
- What reconstitution protocol do you recommend?
- What sterility testing has been performed?
📋 Delivery and Support:
- How is the product shipped (temperature control)?
- What customer support is available for technical questions?
- Can you provide handling and safety documentation?
- What is your return/replacement policy for compromised products?
Red Flags to Avoid
⚠️ Warning Signs of Questionable Suppliers:
- Claims of nasal spray or oral retatrutide formulations
- Lack of COA or analytical documentation
- Unusually low pricing compared to market standards
- Poor or non-existent customer service
- No clear “Research Use Only” labeling
- Vague product descriptions without specifications
- No temperature-controlled shipping options
- Marketing toward human consumption
Building a Reliable Supply Chain
Successful research programs establish relationships with verified, professional suppliers who provide:
✅ Consistent product quality across batches
✅ Responsive technical support for methodology questions
✅ Transparent documentation and certifications
✅ Reliable delivery timelines and tracking
✅ Proper storage and handling guidance
Organizations like PEPTIDE PRO prioritize these elements, offering same-day dispatch for orders placed before 1pm (Monday-Friday) with comprehensive product support.
Retatrutide Compared to Other Metabolic Peptides
The GLP-1 Agonist Family
Understanding retatrutide’s position within the broader peptide landscape helps contextualize delivery method expectations:
Metabolic Peptide Comparison:
| Peptide | Mechanism | Molecular Weight | Standard Delivery | Nasal Formulation? |
|---|---|---|---|---|
| Retatrutide | GIP/GLP-1/GCG triple agonist | ~4,800 Da | Subcutaneous | ❌ No |
| Semaglutide | GLP-1 agonist | ~4,100 Da | Subcutaneous | ❌ No (oral tablet exists) |
| Tirzepatide | GIP/GLP-1 dual agonist | ~4,800 Da | Subcutaneous | ❌ No |
| Liraglutide | GLP-1 agonist | ~3,750 Da | Subcutaneous | ❌ No |
Notice the pattern: all major metabolic peptides require injection due to their molecular size and structural properties.
Why Injectable Remains Standard
The subcutaneous injection route offers critical advantages for peptide research:
🔬 Predictable pharmacokinetics: Consistent absorption profiles
🔬 High bioavailability: 80-100% peptide utilization
🔬 Controlled release: Depot formation enables extended action
🔬 Reproducible results: Minimal inter-subject variability
🔬 Established protocols: Extensive literature for comparison
Related Compounds Available
Researchers interested in metabolic peptide studies can access various related compounds through professional suppliers:
- Tirzepatide formulations: Dual GIP/GLP-1 agonist
- Semaglutide variants: GLP-1 receptor agonist
- CJC-1295: Growth hormone releasing hormone analogue
- Ipamorelin: Growth hormone secretagogue
All legitimate formulations come as injectable preparations with proper documentation and handling instructions.
Practical Guidance for Research Implementation
Setting Up Your Research Protocol
Implementing retatrutide research requires careful planning:
Protocol Development Checklist:
- Define research objectives and measurable outcomes
- Review existing literature on retatrutide administration
- Source high-quality peptide from verified supplier
- Establish storage infrastructure (appropriate refrigeration/freezing)
- Prepare reconstitution materials (bacteriostatic water, sterile supplies)
- Develop administration schedule with proper documentation
- Implement safety protocols for handling and disposal
- Plan data collection methodology aligned with objectives
Documentation Best Practices
Rigorous documentation ensures research validity and reproducibility:
📝 Essential Documentation Elements:
- Batch numbers and COA for all peptides used
- Reconstitution dates, volumes, and final concentrations
- Storage temperatures and conditions throughout study
- Administration dates, times, and sites
- Any deviations from standard protocol
- Environmental conditions during storage/handling
- Visual inspection notes before each use
Troubleshooting Common Issues
Problem: Cloudiness after reconstitution
Solution: May indicate improper storage or contamination; do not use; contact supplier
Problem: Reduced experimental response over time
Solution: Verify storage temperatures; check reconstitution date; peptide may have degraded
Problem: Inconsistent results between batches
Solution: Ensure consistent reconstitution technique; verify batch COAs; consider supplier quality
Problem: Difficulty with injection technique
Solution: Review proper subcutaneous administration protocols; consider needle gauge adjustment
Regulatory and Ethical Considerations
Research Use Only Designation
All legitimate research-grade peptides, including retatrutide, carry “For Research Use Only” labeling, indicating:
⚖️ Not approved for human consumption
⚖️ Intended for laboratory and research applications
⚖️ Not evaluated for safety in humans
⚖️ Subject to institutional research protocols
This designation protects both researchers and suppliers while enabling legitimate scientific investigation.
Institutional Requirements
Research institutions typically require:
- Ethics committee approval for research protocols
- Proper safety training for personnel handling peptides
- Documented storage and handling procedures
- Waste disposal protocols for biological materials
- Inventory management systems for controlled substances
Responsible Sourcing
Ethical research practices include:
✅ Purchasing only from legitimate, transparent suppliers
✅ Verifying product authenticity through documentation
✅ Following institutional guidelines for peptide use
✅ Maintaining accurate records of acquisition and use
✅ Ensuring proper disposal of unused materials
Professional suppliers like PEPTIDE PRO support these practices through clear labeling, comprehensive documentation, and adherence to regulatory standards.
Cost Considerations and Value Assessment

Understanding Peptide Pricing
Research-grade peptide costs reflect several factors:
Price Determinants:
- Synthesis complexity: Longer sequences cost more to produce
- Purity requirements: Higher purity demands more purification steps
- Batch testing: Analytical verification (HPLC, MS) adds cost
- Storage requirements: Temperature-controlled handling increases expenses
- Supplier overhead: Quality control, customer service, documentation
Evaluating True Value
The cheapest option rarely represents the best value for research:
| Consideration | Low-Cost Supplier | Premium Supplier |
|---|---|---|
| Purity verification | Often absent or questionable | Batch-specific COA provided |
| Customer support | Minimal or none | Technical assistance available |
| Shipping conditions | Standard shipping | Temperature-controlled packaging |
| Product consistency | Variable between batches | Rigorous quality control |
| Documentation | Limited or generic | Comprehensive handling guides |
| Long-term reliability | Uncertain | Established track record |
Calculating Research ROI
Consider the total cost of research, not just peptide price:
💰 Total Research Investment:
- Peptide acquisition cost
- Reconstitution materials and supplies
- Storage infrastructure (refrigeration)
- Personnel time for preparation and administration
- Analytical equipment and testing
- Data analysis and interpretation
- Potential costs of failed experiments from poor-quality peptides
Investing in verified, high-quality peptides from suppliers like PEPTIDE PRO reduces the risk of experimental failure and wasted resources.
Frequently Asked Questions
Is retatrutide nasal spray available anywhere?
No. As of 2025, no legitimate retatrutide nasal spray formulation exists. All authentic research-grade retatrutide is supplied as lyophilized powder for reconstitution and subcutaneous injection. Claims of nasal spray availability should be viewed as red flags indicating questionable suppliers.
Why can’t retatrutide be delivered nasally?
Retatrutide’s molecular weight exceeds 4,800 Daltons—far beyond the practical threshold for nasal mucosal absorption. The nasal epithelium presents multiple barriers including tight cellular junctions, enzymatic degradation, and rapid mucociliary clearance that prevent large peptides from achieving meaningful bioavailability.
What’s the proper way to administer retatrutide for research?
The validated method is subcutaneous injection following proper reconstitution. Lyophilized retatrutide should be reconstituted with bacteriostatic water using aseptic technique, stored at 2-8°C, and administered subcutaneously according to research protocol specifications.
How can I verify I’m getting authentic retatrutide?
Request batch-specific Certificates of Analysis (COA) showing HPLC purity verification. Legitimate suppliers provide comprehensive documentation, proper “Research Use Only” labeling, temperature-controlled shipping, and professional customer support. Verify the product arrives as lyophilized powder requiring reconstitution, not as a ready-to-use spray.
Are there any peptides that do come in nasal spray form?
Yes, certain smaller peptides (typically under 1,500 Daltons) have legitimate nasal formulations, including desmopressin, oxytocin, and some nootropic peptides like Selank and Semax. However, larger metabolic peptides like retatrutide, semaglutide, and tirzepatide require injection due to their molecular size.
What should I do if I’ve purchased “retatrutide nasal spray”?
Do not use it for research purposes. The product either doesn’t contain retatrutide, contains it in a formulation that won’t deliver bioavailability, or represents a contaminated/adulterated product. Contact a legitimate supplier to obtain properly formulated research-grade retatrutide with appropriate documentation.
How long does reconstituted retatrutide remain stable?
When stored properly at 2-8°C (refrigerated), reconstituted retatrutide typically maintains stability for 14-28 days, though specific stability may vary by formulation and concentration. Always follow supplier-specific guidance and document reconstitution dates. Never freeze reconstituted peptide solutions.
Can I make my own nasal spray formulation?
While theoretically possible to create experimental formulations, doing so would likely result in negligible bioavailability due to retatrutide’s molecular properties. Such attempts would compromise research validity, waste valuable peptide, and introduce uncontrolled variables. Standard subcutaneous administration remains the scientifically validated approach.
Evidence-Based Research Requires Validated Methods
The answer to “Retatrutide Nasal Spray: Is It Real?” is definitively no—this formulation does not exist in the legitimate research peptide market as of 2025. The scientific barriers to nasal delivery of large peptides like retatrutide remain substantial, with molecular weight, enzymatic degradation, and absorption limitations preventing viable intranasal administration.
Researchers committed to scientific rigor and reproducible results must rely on validated delivery methods: subcutaneous injection of properly reconstituted, research-grade retatrutide from verified suppliers. This approach ensures:
✅ Predictable bioavailability for consistent experimental outcomes
✅ Reproducible methodology aligned with existing literature
✅ Research integrity through proper peptide handling
✅ Resource efficiency by avoiding ineffective formulations
✅ Scientific credibility built on established protocols
The proliferation of misinformation about non-existent delivery methods underscores the importance of sourcing from reputable suppliers who prioritize transparency, quality documentation, and professional support. Organizations like PEPTIDE PRO provide the high-purity, properly formulated research peptides that serious scientific investigation demands.
Next Steps for Researchers
Immediate Actions:
- Verify your current peptide source provides proper COA documentation
- Review your storage and handling protocols against best practices
- Establish relationships with verified suppliers for consistent quality
- Document your methodology thoroughly for reproducibility
- Stay informed about legitimate formulation developments through scientific literature
Long-Term Considerations:
- Monitor emerging peptide delivery technologies through peer-reviewed research
- Maintain rigorous quality standards in peptide sourcing and handling
- Invest in proper storage infrastructure for temperature-sensitive compounds
- Develop institutional protocols for peptide research governance
- Build collaborative relationships with suppliers who support scientific advancement
The future of peptide research depends on maintaining high standards of scientific integrity, beginning with the fundamental choice to use validated formulations and delivery methods. By rejecting unsubstantiated claims about non-existent products like retatrutide nasal spray, the research community protects both experimental validity and the broader credibility of peptide science.
For researchers ready to implement rigorous retatrutide protocols with properly formulated, research-grade peptides, explore PEPTIDE PRO’s comprehensive catalogue featuring fast UK delivery, batch-specific documentation, and professional support for the scientific community.
References
[1] Jastreboff, A.M., et al. (2023). “Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” New England Journal of Medicine, 389(6), 514-526.
[2] Rosenstock, J., et al. (2023). “Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA.” Lancet, 402(10401), 529-544.
[3] Patel, A., & Cholkar, K. (2013). “Ocular drug delivery systems: An overview.” World Journal of Pharmacology, 2(2), 47-64.
[4] Illum, L. (2012). “Nasal drug delivery—recent developments and future prospects.” Journal of Controlled Release, 161(2), 254-263.
[5] Lipinski, C.A., et al. (2001). “Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings.” Advanced Drug Delivery Reviews, 46(1-3), 3-26.
