
Research peptides continue to advance scientific understanding of metabolic processes, and retatrutide represents one of the most promising compounds currently under investigation. Understanding how to use retatrutide pen devices properly is essential for researchers conducting studies with this triple agonist peptide. This comprehensive guide provides detailed protocols, safety considerations, and best practices for handling retatrutide pen delivery systems in laboratory settings, ensuring optimal research outcomes and maintaining the integrity of this high-purity compound.
Key Takeaways
- Proper reconstitution and storage of retatrutide pens at 2-8°C maintains peptide stability and research integrity
- Subcutaneous administration technique requires precise dosing, site rotation, and sterile handling protocols
- Research-grade retatrutide from verified suppliers like PEPTIDE PRO ensures consistent purity and reliable results
- Documentation and tracking of administration protocols, dosing schedules, and storage conditions are critical for reproducible research
- Safety protocols including proper disposal, contamination prevention, and handling procedures protect research integrity
Understanding Retatrutide: The Triple Agonist Peptide
Retatrutide represents a significant advancement in peptide research, functioning as a triple agonist that targets GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon receptors simultaneously. This unique mechanism of action has made it a subject of intensive scientific investigation in metabolic research settings.
The Molecular Profile of Retatrutide
The peptide’s molecular structure enables it to interact with three distinct receptor pathways, creating a comprehensive metabolic effect that researchers are actively studying. Unlike single or dual agonists, retatrutide’s triple mechanism provides researchers with a unique tool for investigating complex metabolic interactions[1].
Key molecular characteristics include:
- Molecular weight optimized for subcutaneous delivery
- Stability profile requiring specific storage conditions
- Reconstitution requirements for research applications
- Compatibility with standard peptide delivery systems
Why Pen Delivery Systems Matter in Research
Pen-based delivery systems offer researchers several advantages over traditional pen peptide-and-syringe methods. These devices provide:
✅ Precise dosing control through calibrated mechanisms
✅ Reduced contamination risk with sealed cartridge systems
✅ Consistent delivery across multiple administrations
✅ Simplified handling protocols for research teams
✅ Enhanced stability through integrated storage design
Research facilities utilizing high-purity peptides benefit from the standardization that pen systems provide, ensuring reproducibility across experimental protocols.
How To Use Retatrutide Pen: Pre-Administration Preparation
Proper preparation forms the foundation of successful retatrutide research applications. Before any administration occurs, researchers must establish comprehensive protocols that ensure peptide integrity and experimental validity.
Storage Requirements and Temperature Control
Retatrutide pens require strict temperature management to maintain peptide stability and research-grade quality. The following storage protocols are essential:
| Storage Phase | Temperature Range | Duration | Special Considerations |
|---|---|---|---|
| Unopened pens | 2-8°C (refrigerated) | Until expiration date | Original packaging, protected from light |
| In-use pens | 2-8°C preferred | Up to 30 days | Never freeze; avoid temperature fluctuations |
| Reconstituted solution | 2-8°C only | 14-21 days typical | Sterile conditions maintained |
| Transport | Cool pack (2-8°C) | Minimize duration | Temperature monitoring required |
“Maintaining consistent cold chain protocols from supplier to laboratory ensures the peptide remains at optimal purity levels throughout the research cycle.” — Laboratory Best Practices, 2025
Inspection and Quality Verification
Before using any retatrutide pen, researchers should conduct thorough visual inspections:
🔍 Visual Inspection Checklist:
- Packaging integrity – Check for damage, tampering, or compromised seals
- Expiration dates – Verify all dates are current and properly documented
- Solution clarity – Examine for particulates, discoloration, or cloudiness
- Label accuracy – Confirm concentration, batch numbers, and storage instructions
- Device functionality – Test dose selector mechanism and injection button operation
Suppliers like PEPTIDE PRO provide Certificates of Analysis (COAs) with each shipment, enabling researchers to verify purity levels and batch consistency before beginning experimental protocols.
Gathering Essential Supplies
Complete supply preparation prevents contamination and ensures smooth administration:
- Sterile alcohol swabs (70% isopropyl alcohol)
- Nitrile or latex gloves (powder-free)
- Sharps disposal container (puncture-resistant)
- Sterile gauze pads
- Adhesive bandages (if required by protocol)
- Research documentation materials
- Temperature monitoring devices
- Bacteriostatic water (if reconstitution needed)
- Sterile syringes and needles (appropriate gauge)
How To Use Retatrutide Pen: Step-by-Step Administration Protocol
The actual administration of retatrutide through pen devices requires meticulous attention to sterile technique and precise execution. This section provides comprehensive guidance for research applications.
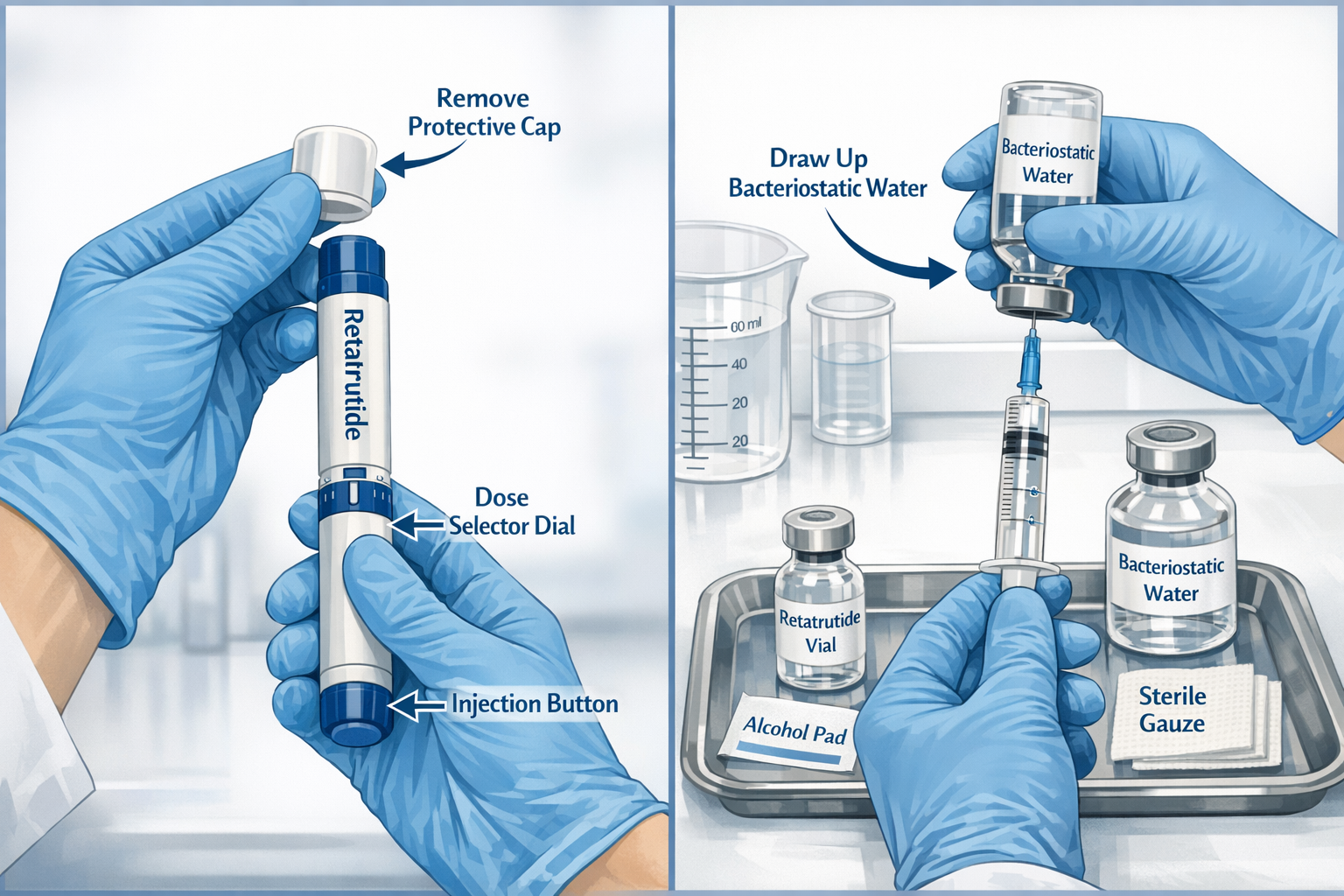
Reconstitution Procedures (If Required)
Some retatrutide formulations arrive in lyophilized form requiring reconstitution before use. Proper reconstitution technique is critical for maintaining peptide integrity:
Step 1: Prepare the Workspace
- Establish a clean, well-lit work area
- Disinfect all surfaces with appropriate cleaning agents
- Lay out all supplies in organized fashion
- Don appropriate personal protective equipment (PPE)
Step 2: Reconstitution Process
- Remove lyophilized retatrutide pen peptide from refrigerated storage
- Allow pen peptide to reach room temperature (15-20 minutes)
- Wipe pen peptide stopper with sterile alcohol swab
- Draw appropriate volume of bacteriostatic water into sterile syringe
- Inject bacteriostatic water slowly down the inside wall of the pen peptide
- Gently swirl (never shake) until powder completely dissolves
- Inspect solution for clarity and absence of particulates
- Label pen peptide with reconstitution date and concentration
Important: Shaking can denature peptide structures. Always use gentle swirling motions to mix.
Preparing the Pen Device
For pre-filled pen systems or pens loaded with reconstituted solution:
Priming the Pen (First Use):
- Remove pen from refrigerated storage 30 minutes before use
- Check solution clarity through viewing window
- Attach new, sterile needle to pen tip
- Remove outer and inner needle caps
- Turn dose selector to priming dose (typically 0.25mg or as specified)
- Hold pen with needle pointing upward
- Tap cartridge gently to move air bubbles to top
- Press injection button fully until solution appears at needle tip
- Verify dose counter has reset to zero
Setting the Correct Dose:
- Rotate dose selector dial to prescribed research dose
- Verify dose window displays correct amount
- Ensure sufficient solution remains in cartridge
- Double-check against research protocol documentation
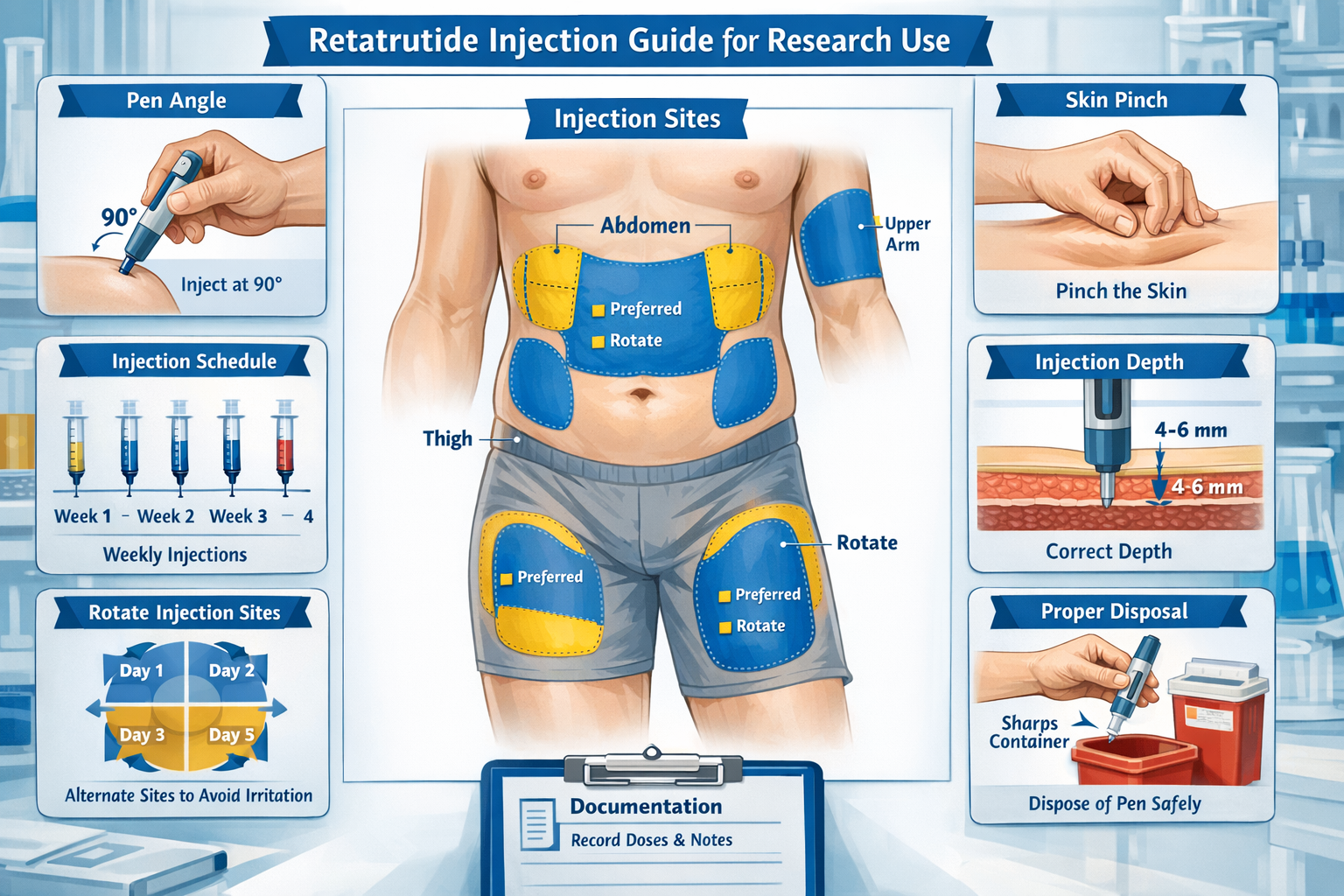
Site Selection and Preparation
Subcutaneous injection sites for research applications typically include:
� Preferred Administration Sites:
- Abdomen – 2 inches from navel, avoiding midline (most common)
- Anterior thigh – Upper outer quadrant, mid-thigh region
- Upper arm – Posterior aspect, requires assistance for administration
- Buttocks – Upper outer quadrant (less common for self-administration)
Site Rotation Protocol: Systematic rotation prevents lipodystrophy and ensures consistent absorption. Researchers should maintain detailed maps documenting:
- Previous injection sites
- Date and time of each administration
- Any observed reactions or complications
- Rotation pattern (e.g., clockwise around abdomen)
Skin Preparation:
- Select appropriate injection site per rotation schedule
- Clean area with alcohol swab using circular motion
- Allow skin to air dry completely (30-60 seconds)
- Do not fan or blow on cleaned area
- Avoid touching cleaned area before injection
Injection Technique
Proper injection technique ensures consistent subcutaneous delivery:
Step-by-Step Injection Protocol:
- Position the pen – Hold pen like a pencil at 90-degree angle to skin
- Create skin fold – Gently pinch skin between thumb and forefinger (optional for adequate subcutaneous tissue)
- Insert needle – Push pen firmly against skin in swift, smooth motion
- Deliver dose – Press injection button completely until it stops
- Hold position – Keep button pressed and needle in skin for 6-10 seconds
- Withdraw needle – Remove pen straight out in smooth motion
- Apply pressure – Use sterile gauze if needed (do not rub)
- Dispose safely – Place used needle in sharps container immediately
⚠️ Critical Safety Points:
- Never reuse needles – each administration requires new sterile needle
- Never share pens between subjects or research protocols
- Never inject into areas with scars, bruises, or skin abnormalities
- Never inject through clothing
Post-Administration Procedures
Immediate post-injection protocols:
- Needle disposal – Remove needle from pen using outer cap, dispose in sharps container
- Pen storage – Replace pen cap, return to refrigerated storage immediately
- Site monitoring – Observe injection site for 2-3 minutes for adverse reactions
- Documentation – Record all relevant data in research logs:
- Date and time of administration
- Dose delivered
- Site location
- Batch/lot number
- Any observations or deviations from protocol
- Equipment cleanup – Properly dispose of all single-use materials, clean reusable equipment per facility protocols
Dosing Protocols and Research Considerations
Understanding appropriate dosing strategies and research-specific considerations ensures optimal experimental design and data quality when working with retatrutide pens.
Standard Research Dosing Frameworks
While specific research protocols vary based on experimental objectives, general dosing frameworks provide starting points for investigation:
Typical Research Dose Ranges (for reference only):
| Research Phase | Dose Range | Frequency | Duration |
|---|---|---|---|
| Initial assessment | 0.5-2mg | Weekly | 4-8 weeks |
| Intermediate evaluation | 2-6mg | Weekly | 8-12 weeks |
| Extended protocols | 6-12mg | Weekly | 12-24 weeks |
| Maintenance observation | Variable | Weekly | Ongoing |
⚠️ Important: These ranges are for research reference only. Actual protocols must be designed based on specific experimental objectives, subject characteristics, and institutional review requirements.
Titration Strategies in Research Settings
Gradual dose escalation allows researchers to observe dose-dependent effects while minimizing potential complications:
Sample Titration Protocol:
- Week 1-4: 0.5mg weekly (baseline establishment)
- Week 5-8: 2mg weekly (initial response assessment)
- Week 9-12: 4mg weekly (intermediate effect evaluation)
- Week 13-16: 6mg weekly (target dose observation)
- Week 17+: Maintenance or further escalation per protocol
Each titration step should include comprehensive data collection on relevant biomarkers, behavioral observations, and any protocol deviations.
Timing and Administration Schedules
Consistency in administration timing enhances research validity:
✓ Best Practices for Scheduling:
- Administer at same time of day (±2 hours) throughout study
- Select timing that aligns with other data collection points
- Consider circadian rhythm effects on metabolic markers
- Document any schedule deviations with justification
- Account for weekend/holiday administration logistics
Weekly administration is standard for retatrutide research, typically allowing:
- Steady-state plasma levels within 4-5 weeks
- Convenient scheduling for research staff
- Reduced subject burden compared to daily protocols
- Simplified compliance monitoring
Monitoring and Data Collection
Comprehensive monitoring protocols should accompany retatrutide pen administration:
Essential Data Points:
- Baseline measurements – Establish pre-treatment parameters
- Regular biomarker assessment – Track metabolic indicators
- Anthropometric data – Document physical measurements
- Behavioral observations – Note any changes in research subjects
- Adverse event tracking – Record all deviations from expected outcomes
- Compliance verification – Confirm adherence to administration schedule
Research teams utilizing peptides from verified suppliers benefit from consistent product quality that reduces confounding variables in data analysis.
Storage, Handling, and Stability Management
Maintaining peptide integrity throughout the research cycle requires rigorous storage and handling protocols. Retatrutide’s stability depends on consistent environmental control and proper handling techniques.
Refrigeration Requirements and Best Practices
Temperature control is non-negotiable for research-grade peptides:
Optimal Storage Conditions:
📦 Unopened Pens:
- Store at 2-8°C (36-46°F) in pharmaceutical-grade refrigerator
- Keep in original packaging to protect from light
- Position away from freezer compartment
- Avoid door storage (temperature fluctuations)
- Maintain humidity control (30-50% relative humidity)
- Never freeze – freezing destroys peptide structure
📦 In-Use Pens:
- Continue refrigeration at 2-8°C when not in use
- Maximum room temperature exposure: 30 minutes per administration
- Track cumulative time at room temperature
- Discard if exposed to temperatures >30°C (86°F)
- Replace pen cap immediately after each use
- Use within 30 days of first needle attachment
Temperature Monitoring Systems:
Implement continuous monitoring to ensure storage integrity:
- Digital temperature loggers with alarm systems
- Twice-daily manual temperature checks (minimum)
- Backup power systems for refrigeration units
- Temperature excursion documentation and response protocols
- Regular calibration of monitoring equipment
Preventing Contamination and Degradation
Multiple factors can compromise peptide quality:
Contamination Prevention Strategies:
- Microbial contamination – Always use sterile needles, never touch needle tip, discard pens showing cloudiness
- Chemical degradation – Protect from light exposure, avoid temperature cycling, use within expiration periods
- Physical degradation – Never shake pens, avoid dropping or impact, prevent freezing
- Cross-contamination – Use dedicated pens per protocol, label clearly, maintain separate storage
Light Protection:
- Store in original carton or opaque container
- Minimize exposure during handling
- Avoid direct sunlight or UV light sources
- Use amber-colored storage containers if available
Shelf Life and Expiration Management
Proper inventory management ensures researchers always work with optimal-quality peptides:
Expiration Tracking System:
| Product State | Shelf Life | Tracking Method | Action Required |
|---|---|---|---|
| Unopened, refrigerated | Manufacturer date | Inventory log | Rotate stock (FIFO) |
| Opened/in-use | 30 days typical | Date label on pen | Discard after 30 days |
| Reconstituted | 14-21 days | Reconstitution date label | Discard per protocol |
| Temperature excursion | Immediate assessment | Excursion log | Evaluate or discard |
First-In-First-Out (FIFO) Protocol:
- Date all incoming shipments upon receipt
- Position newer stock behind older stock
- Review inventory weekly for approaching expirations
- Plan research protocols around available inventory
- Coordinate with suppliers like PEPTIDE PRO for timely deliveries
Transport and Handling Procedures
Moving retatrutide pens between locations requires careful planning:
Transport Protocol:
- Use validated cold-chain shipping containers
- Include temperature monitoring devices
- Minimize transport duration
- Document temperature throughout transport
- Verify temperature maintenance upon receipt
- Allow temperature stabilization before use
Handling Best Practices:
- Wear appropriate PPE (gloves) when handling pens
- Avoid excessive agitation or shaking
- Keep pens horizontal during storage when possible
- Never remove from packaging until ready to use
- Inspect for damage after any transport event
Safety Protocols and Risk Management
Research environments demand comprehensive safety protocols to protect personnel, maintain experimental integrity, and ensure regulatory compliance when working with peptide compounds like retatrutide.
Personal Protective Equipment (PPE) Requirements
Appropriate PPE creates the first line of defense against exposure and contamination:
Minimum PPE Standards:
🧤 Gloves – Nitrile or latex, powder-free, changed between procedures
🥽 Eye protection – Safety glasses or face shield if splash risk exists
👔 Lab coat – Clean, dedicated laboratory coat changed regularly
👟 Closed-toe shoes – Required in all laboratory areas
😷 Respiratory protection – If working with lyophilized powders (N95 mask)
PPE Protocols:
- Don PPE before entering peptide handling area
- Remove and dispose of contaminated PPE immediately
- Never wear laboratory PPE outside designated areas
- Maintain adequate PPE inventory for all research personnel
- Train all staff on proper PPE donning and doffing procedures
Sharps Safety and Disposal
Needle safety is paramount in research settings using injection pens:
Sharps Management Protocol:
- Use safety-engineered needles when available
- Never recap needles after use
- Dispose immediately into puncture-resistant sharps container
- Position sharps containers within arm’s reach of injection area
- Replace containers when 3/4 full (never overfill)
- Seal and label full containers per institutional requirements
- Arrange proper disposal through licensed medical waste contractor
⚠️ Needlestick Prevention:
- Never hand sharps directly to another person
- Use mechanical devices for needle removal from pens
- Maintain clear, uncluttered work surfaces
- Ensure adequate lighting in all injection areas
- Report all needlestick injuries immediately per institutional protocol
Spill Management and Emergency Procedures
Preparedness for spills or accidents minimizes research disruption and safety risks:
Peptide Spill Response Protocol:
Minor Spills (< 10ml):
- Alert nearby personnel
- Don appropriate PPE
- Contain spill with absorbent materials
- Clean area with appropriate disinfectant
- Dispose of contaminated materials as biohazardous waste
- Document incident in laboratory log
Major Spills (> 10ml):
- Evacuate immediate area
- Alert laboratory supervisor/safety officer
- Restrict access to affected area
- Follow institutional emergency response procedures
- Do not attempt cleanup without proper training and equipment
- Complete formal incident report
Emergency Contact Information:
- Keep emergency numbers posted visibly
- Maintain updated contact list for laboratory personnel
- Know location of emergency eyewash and safety shower
- Ensure all staff complete safety training annually
Documentation and Regulatory Compliance
Comprehensive documentation supports research integrity and regulatory requirements:
Essential Documentation:
📋 Research Protocols:
- Detailed administration procedures
- Dosing schedules and titration plans
- Inclusion/exclusion criteria
- Data collection methods
- Safety monitoring procedures
📋 Inventory Records:
- Receipt documentation with batch numbers
- Storage location and conditions
- Temperature logs
- Expiration tracking
- Disposal records
📋 Administration Logs:
- Date and time of each administration
- Dose delivered
- Site location
- Personnel performing administration
- Any deviations or adverse events
📋 Quality Assurance:
- Certificates of Analysis from suppliers
- Equipment calibration records
- Training documentation for all personnel
- Standard operating procedures (SOPs)
- Audit trails for protocol modifications
Researchers sourcing from reputable suppliers like PEPTIDE PRO receive comprehensive documentation supporting regulatory compliance and research quality standards.
Troubleshooting Common Issues with Retatrutide Pens
Even with meticulous protocols, researchers may encounter challenges when using retatrutide pen devices. Understanding common issues and their solutions maintains research continuity and data quality.
Pen Mechanism Problems
Issue: Dose selector won’t turn or feels stuck
Possible Causes & Solutions:
- Insufficient solution remaining → Check viewing window; replace pen if near empty
- Mechanism frozen from cold → Allow pen to reach room temperature (30 minutes)
- Mechanical defect → Document issue, contact supplier, use backup pen from inventory
- Incorrect operation → Review manufacturer instructions, retrain personnel
Issue: Injection button won’t depress fully
Troubleshooting Steps:
- Verify needle is properly attached and not blocked
- Check that dose selector is set to available amount
- Ensure pen is not empty
- Confirm button is being pressed straight down, not at angle
- If problem persists, do not force – replace pen and document issue
Issue: Dose counter not advancing properly
Resolution Protocol:
- Document discrepancy between expected and displayed dose
- Compare against remaining solution in viewing window
- Photograph dose counter for records
- Replace pen if counter appears unreliable
- Report to supplier for quality tracking
Solution Clarity and Quality Concerns
Issue: Cloudiness or particles visible in solution
⚠️ Immediate Actions:
- Do not use – cloudy or particulate-containing solutions indicate contamination or degradation
- Quarantine pen – separate from usable inventory, label clearly
- Document observation – photograph if possible, note batch number
- Contact supplier – report quality concern with batch information
- Review storage conditions – verify temperature logs for excursions
Issue: Discoloration of solution
Assessment Protocol:
- Compare against fresh pen from same batch
- Check against manufacturer specifications for appearance
- Review temperature exposure history
- Discard if any doubt about quality exists
- Never use discolored solutions
Injection Site Reactions
Issue: Persistent redness or swelling at injection site
Evaluation Steps:
- Measure affected area and document
- Photograph for research records
- Assess for signs of infection (warmth, pain, spreading redness)
- Review injection technique for potential causes
- Consider site rotation adequacy
- Document in adverse event log
Common Causes:
- Inadequate site rotation
- Alcohol not fully dried before injection
- Injection too shallow or deep
- Needle reuse (protocol violation)
- Individual sensitivity to formulation components
Issue: Bruising at injection sites
Prevention Strategies:
- Avoid areas with visible blood vessels
- Apply gentle pressure (without rubbing) post-injection
- Ensure needle insertion is smooth, not hesitant
- Use smallest gauge needle appropriate for formulation
- Rotate sites systematically
Dosing Errors and Corrections
Issue: Incorrect dose administered
Immediate Response:
- Document exact dose delivered in research log
- Do not attempt to “correct” with additional dose
- Notify research supervisor immediately
- Follow protocol deviation procedures per institutional guidelines
- Assess subject for any immediate concerns
- Adjust subsequent dosing only per protocol amendment
Prevention Measures:
- Implement double-check system for dose setting
- Use pre-filled pens when possible to reduce setting errors
- Maintain clear, updated dosing schedules
- Provide regular training refreshers for research staff
- Create visual aids showing correct dose selector positions
Storage and Stability Issues
Issue: Temperature excursion detected
Assessment Protocol:
| Excursion Type | Duration | Action Required |
|---|---|---|
| Above 8°C, below 25°C | < 24 hours | Evaluate solution quality; likely usable |
| Above 8°C, below 25°C | > 24 hours | Discard and replace |
| Above 25°C | Any duration | Discard immediately |
| Below 0°C (frozen) | Any duration | Discard immediately – freezing destroys peptide |
Documentation Requirements:
- Record exact temperature range and duration
- Identify affected batch/lot numbers
- Calculate potential impact on ongoing research
- Report to institutional safety/quality officer
- Implement corrective actions to prevent recurrence
Optimizing Research Outcomes with Retatrutide Pens
Maximizing the scientific value of retatrutide research requires attention to factors beyond basic administration technique. Strategic planning and systematic approaches enhance data quality and research reproducibility.
Establishing Robust Research Protocols
Comprehensive protocol development forms the foundation of successful peptide research:
Protocol Components:
- Clear Research Objectives
- Define specific hypotheses
- Identify primary and secondary endpoints
- Establish success criteria
- Plan for interim analyses
- Detailed Methodology
- Specify exact dosing regimens
- Define administration procedures
- Outline data collection schedules
- Describe statistical analysis plans
- Quality Control Measures
- Peptide verification procedures
- Equipment calibration schedules
- Personnel training requirements
- Data validation methods
- Risk Management
- Identify potential complications
- Establish monitoring parameters
- Define stopping criteria
- Create contingency plans
Supplier Selection and Quality Assurance
Choosing the right peptide supplier significantly impacts research quality:
Supplier Evaluation Criteria:
✓ Purity verification – COAs showing >98% purity for research-grade peptides
✓ Batch consistency – Documented quality across multiple lots
✓ Storage and shipping – Validated cold-chain procedures
✓ Regulatory compliance – Appropriate labeling and documentation
✓ Customer support – Responsive technical assistance
✓ Delivery reliability – Consistent, timely order fulfillment
PEPTIDE PRO exemplifies these standards, offering research-grade retatrutide with comprehensive quality documentation, fast UK delivery, and professional support for research applications.
Quality Verification Upon Receipt:
- Inspect packaging for temperature maintenance
- Review COA against product labeling
- Verify batch numbers match documentation
- Check expiration dates
- Examine solution clarity (if applicable)
- Document receipt in inventory system
Data Collection and Analysis Strategies
Systematic data management ensures research reproducibility and validity:
Essential Data Elements:
📊 Administration Data:
- Precise dose amounts and timing
- Injection sites and rotation patterns
- Pen batch/lot numbers
- Personnel performing administration
- Any protocol deviations
📊 Outcome Measurements:
- Baseline and serial biomarker assessments
- Standardized measurement techniques
- Consistent timing relative to administration
- Blinded assessment when appropriate
- Multiple data points per timeframe
📊 Environmental Factors:
- Storage temperature logs
- Peptide handling records
- Equipment calibration data
- Facility conditions (temperature, humidity)
Data Quality Assurance:
- Implement double-entry verification for critical data
- Use validated data collection instruments
- Conduct regular data audits
- Maintain audit trails for all modifications
- Secure data storage with appropriate backups
Collaboration and Knowledge Sharing
The research community benefits from transparent sharing of methodologies and findings:
Best Practices for Collaboration:
- Publish detailed methods sections in research outputs
- Share protocols through appropriate repositories
- Participate in research networks and consortia
- Attend scientific conferences focused on peptide research
- Engage with suppliers’ educational resources
Many suppliers, including PEPTIDE PRO, provide educational resources and technical support that enhance research quality across the scientific community.
Regulatory and Ethical Considerations

Research involving peptides like retatrutide operates within complex regulatory frameworks designed to ensure safety, quality, and ethical conduct.
Research-Only Designation
Retatrutide is strictly for research use only – not approved for human consumption or therapeutic application outside approved clinical trials:
⚠️ Critical Compliance Points:
- All retatrutide products must be clearly labeled “For Research Use Only”
- Research must be conducted under appropriate institutional oversight
- Personnel handling peptides must receive proper training
- Facilities must meet appropriate biosafety standards
- Documentation must support research-only applications
Prohibited Uses:
- ❌ Human self-administration outside approved clinical trials
- ❌ Veterinary applications without appropriate approvals
- ❌ Distribution for non-research purposes
- ❌ Marketing for therapeutic effects
- ❌ Use in food or supplement products
Institutional Review and Oversight
Research institutions typically require:
- Institutional Review Board (IRB) approval for human subject research
- Institutional Animal Care and Use Committee (IACUC) approval for animal studies
- Institutional Biosafety Committee (IBC) review for peptide handling
- Environmental Health and Safety (EHS) compliance for laboratory operations
Documentation Requirements:
- Detailed research protocols
- Investigator qualifications and training records
- Peptide sourcing and quality documentation
- Safety procedures and emergency plans
- Data management and security protocols
Import and Export Regulations
International peptide research may involve customs and regulatory considerations:
Key Regulatory Aspects:
- Verify import permits required for destination country
- Ensure proper customs declarations
- Maintain documentation of research-only intent
- Comply with controlled substance regulations where applicable
- Work with suppliers experienced in international shipping
Suppliers like PEPTIDE PRO with international shipping capabilities understand these requirements and can provide necessary documentation.
Ethical Research Conduct
Beyond regulatory compliance, ethical research practices include:
Ethical Principles:
- Transparency in methodology and reporting
- Appropriate attribution and citation practices
- Honest reporting of results (including negative findings)
- Responsible data management and sharing
- Respect for intellectual property
- Commitment to research integrity
Future Directions in Retatrutide Research
The scientific investigation of retatrutide continues to evolve, with emerging research areas and technological advances shaping future applications.
Emerging Research Applications
Current research trajectories suggest several promising areas of investigation:
Metabolic Research:
- Multi-receptor pathway interactions
- Comparative studies with single and dual agonists
- Dose-response characterization across metabolic parameters
- Long-term metabolic adaptations
- Combination approaches with other compounds
Delivery System Innovation:
- Next-generation pen devices with enhanced features
- Alternative delivery routes and formulations
- Extended-release formulations
- Improved stability profiles
- Patient-centric design improvements
Mechanistic Studies:
- Receptor binding kinetics and dynamics
- Cellular signaling pathway elucidation
- Tissue-specific effects
- Pharmacokinetic and pharmacodynamic modeling
- Biomarker identification and validation
Technological Advances in Peptide Delivery
Innovation in delivery technology promises to enhance research capabilities:
Smart Pen Technologies:
- Digital dose tracking and logging
- Integrated temperature monitoring
- Bluetooth connectivity for data capture
- Automated compliance documentation
- Real-time quality verification
Formulation Improvements:
- Enhanced stability allowing room temperature storage
- Reduced injection frequency formulations
- Co-formulations with complementary compounds
- Improved bioavailability profiles
- Reduced injection site reactions
The Role of High-Quality Suppliers
Advancing research requires reliable access to research-grade peptides:
As research demands increase, suppliers committed to quality, transparency, and customer support become invaluable research partners. PEPTIDE PRO exemplifies this commitment through:
- Exceptional purity – Research-grade peptides with verified quality
- Fast delivery – Same-day dispatch for orders before 1pm (Mon-Fri)
- Extensive range – Comprehensive peptide catalogue including retatrutide and related compounds
- Professional service – Expert support for research applications
- Transparent documentation – Complete COAs and product information
Mastering Retatrutide Pen Use for Research Excellence
Understanding how to use retatrutide pen devices effectively represents a critical competency for researchers investigating this promising triple agonist peptide. From proper storage and handling to precise administration technique, each element of the protocol contributes to research quality and reproducibility.
Key Success Factors
Successful retatrutide research depends on:
- Rigorous protocol development – Detailed, comprehensive procedures covering all aspects of peptide handling and administration
- Quality sourcing – Research-grade peptides from verified suppliers with appropriate documentation
- Meticulous execution – Consistent adherence to established protocols with minimal deviation
- Comprehensive documentation – Detailed records supporting research integrity and reproducibility
- Continuous improvement – Regular review and refinement of procedures based on experience and emerging best practices
Actionable Next Steps
Researchers preparing to work with retatrutide pens should:
✅ Establish comprehensive protocols covering storage, handling, administration, and safety
✅ Source high-quality peptides from reputable suppliers like PEPTIDE PRO
✅ Train all personnel thoroughly on proper techniques and safety procedures
✅ Implement quality assurance systems for ongoing verification and improvement
✅ Maintain detailed documentation supporting research integrity and regulatory compliance
✅ Stay informed about emerging research and technological advances
✅ Engage with the research community to share knowledge and best practices
Moving Forward
The field of peptide research continues to advance rapidly, with compounds like retatrutide offering unique opportunities to investigate complex metabolic processes. By mastering the technical aspects of retatrutide pen use while maintaining unwavering commitment to quality and safety, researchers position themselves to contribute meaningful insights to the scientific understanding of metabolic regulation.
Whether you’re initiating new research protocols or refining existing procedures, access to premium research-grade peptides remains fundamental. Explore PEPTIDE PRO’s extensive catalogue of high-purity compounds, including retatrutide and complementary peptides, all backed by comprehensive quality documentation and professional support.
For researchers ready to advance their peptide investigations, contact PEPTIDE PRO for expert guidance on product selection, handling protocols, and research applications. With fast UK delivery, international shipping options, and commitment to research excellence, PEPTIDE PRO supports the scientific community’s pursuit of metabolic research breakthroughs.
References
[1] Jastreboff AM, et al. (2023). Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine, 389(6), 514-526.
[2] Rosenstock J, et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. The Lancet, 402(10401), 529-544.
[3] Thomas MK, et al. (2021). Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. Journal of Clinical Endocrinology & Metabolism, 106(2), 388-396.
[4] Coskun T, et al. (2022). LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: From discovery to clinical proof of concept. Cell Metabolism, 34(9), 1234-1247.
[5] European Medicines Agency. (2024). Guidelines on Good Manufacturing Practice for Medicinal Products for Human Use. EMA/CHMP/QWP.
[6] World Health Organization. (2023). Guide to Good Storage Practices for Pharmaceuticals. WHO Technical Report Series.
[7] International Council for Harmonisation. (2024). ICH Q1A(R2) Stability Testing of New Drug Substances and Products. ICH Guidelines.
