
When conducting research with advanced peptides like retatrutide, understanding the proper retatrutide injection site selection is fundamental to experimental protocol design and research outcomes. This triple-agonist peptide—targeting GIP, GLP-1, and glucagon receptors—has generated significant interest within the research community, making precise administration techniques essential for laboratory investigations. Whether you’re establishing new research protocols or refining existing methodologies, mastering injection site selection ensures consistency, reliability, and reproducibility in your scientific work.
Key Takeaways
- Optimal injection sites for retatrutide research include the abdomen, anterior thigh, and posterior upper arm, with subcutaneous administration being the standard route
- Site rotation protocols are critical to prevent tissue changes and maintain experimental consistency across research trials
- Proper technique involves specific needle angles (45-90 degrees), injection depths (4-6mm into subcutaneous tissue), and sterile handling procedures
- Research-grade peptides from verified suppliers like PEPTIDE PRO ensure purity and consistency for reliable laboratory results
- Documentation standards require detailed logging of injection sites, rotation schedules, and any tissue observations throughout research protocols
Understanding Retatrutide: Research Context and Mechanism
Retatrutide represents a significant advancement in peptide research, functioning as a triple receptor agonist with activity at glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors[1]. This unique mechanism distinguishes it from single or dual-agonist compounds, making proper administration techniques particularly important for research applications.
The Importance of Injection Site Selection in Research
The retatrutide injection site directly influences several critical research parameters:
Absorption kinetics vary significantly based on tissue composition, blood flow, and subcutaneous fat thickness at different anatomical locations. Research protocols must account for these variables to ensure reproducible results across experimental trials.
Tissue integrity maintenance requires systematic rotation to prevent localized changes that could compromise data quality. Repeated injections at identical sites may alter tissue characteristics, potentially affecting absorption patterns and introducing confounding variables.
Experimental consistency depends on standardized administration techniques. Documentation of exact injection locations, tissue conditions, and any observed variations ensures research reproducibility—a cornerstone of scientific methodology.
Research-Grade Peptide Sourcing
Laboratory investigations demand the highest purity standards. PEPTIDE PRO specializes in supplying research-grade peptides with comprehensive certificates of analysis (COAs), ensuring that your retatrutide samples meet stringent quality specifications. This foundation of purity is essential before considering administration techniques.
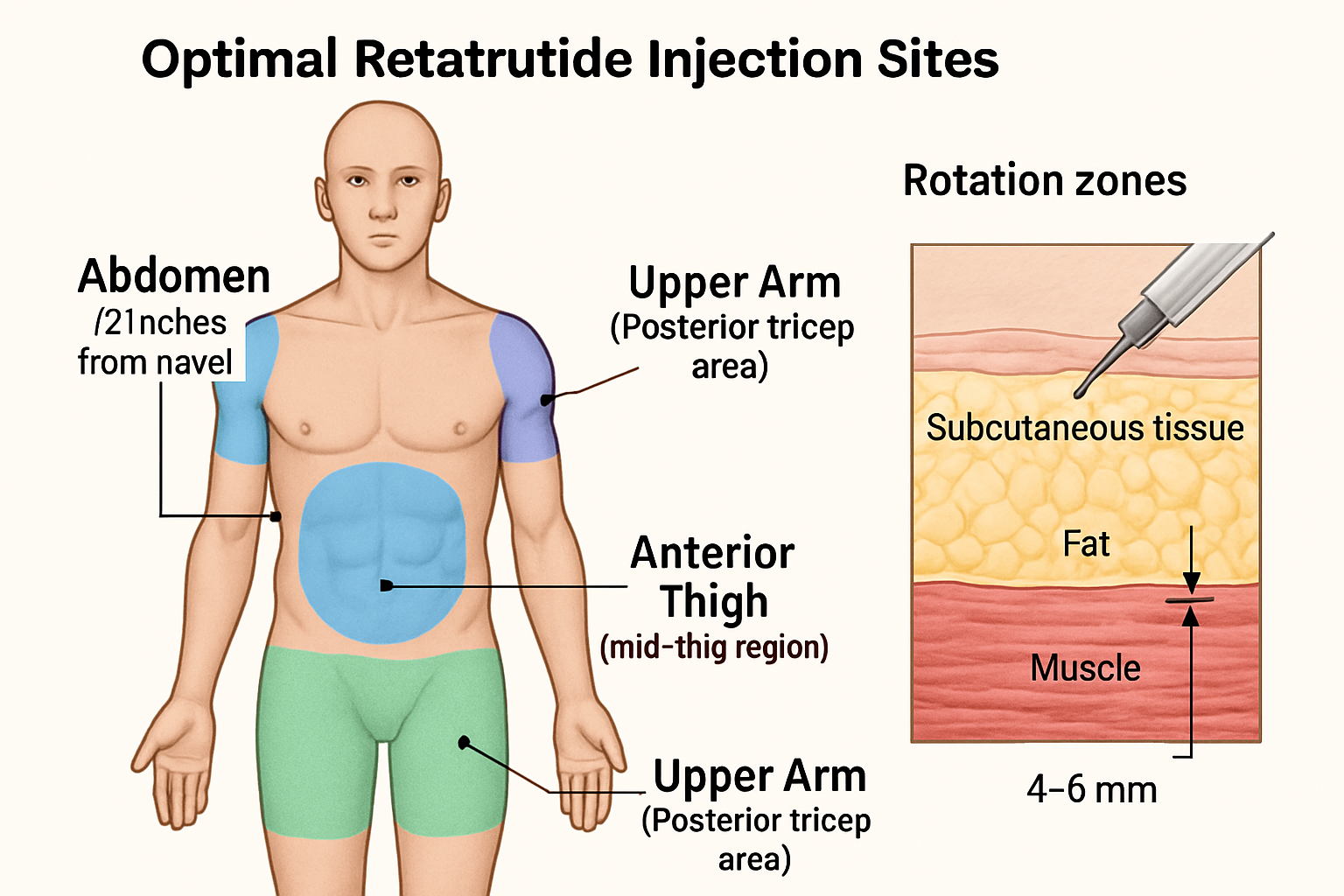
Anatomical Locations for Retatrutide Injection Sites
Understanding the anatomical options for retatrutide injection site selection enables researchers to develop robust rotation protocols and maintain experimental integrity throughout research timelines.
Primary Injection Site: Abdominal Region
The abdomen represents the most commonly utilized injection site in research protocols for several physiological reasons:
Subcutaneous tissue abundance in the abdominal region provides consistent injection depth across most research models. The area typically offers 2-4 cm of subcutaneous fat, creating a reliable target zone for peptide administration.
Optimal absorption characteristics result from the abdomen’s rich blood supply and consistent tissue composition. Research suggests this site may offer more predictable pharmacokinetic profiles compared to other locations[2].
Specific anatomical landmarks for abdominal injections include:
- 2-3 inches lateral to the umbilicus (navel) on either side
- Above the pubic bone but below the ribcage
- Avoiding the midline where tissue is thinner and more vascular
- Rotating between quadrants: upper right, upper left, lower right, lower left
⚠️ Research Note: Avoid injecting within 2 inches of the navel, previous injection sites, scars, or areas with visible bruising or tissue changes.
Secondary Injection Site: Anterior Thigh
The anterior and lateral thigh provides an excellent alternative site for rotation protocols:
Muscle-to-fat ratio in the thigh region varies considerably between research subjects, requiring careful site selection to ensure subcutaneous (not intramuscular) administration.
Specific thigh injection zones include:
- Mid-thigh region: halfway between the hip and knee
- Anterior surface: front and outer portions of the thigh
- Lateral aspects: outer thigh areas with adequate subcutaneous tissue
- Avoiding inner thigh: due to increased vascularity and sensitivity
Accessibility advantages make the thigh particularly useful for self-administration research protocols, as the site is easily visualized and reached without assistance.
Tertiary Injection Site: Posterior Upper Arm
The back of the upper arm (triceps area) serves as a third rotation option:
Tissue characteristics in this region typically provide 1-2 cm of subcutaneous fat, making it suitable for research applications though requiring more precise technique.
Anatomical specifications include:
- Triceps region: back of the upper arm
- Midpoint location: halfway between shoulder and elbow
- Adequate tissue pinch: ensuring sufficient subcutaneous fat for injection
- Assistance consideration: this site may be difficult to reach independently in some research models
Comparative Site Analysis
| Injection Site | Subcutaneous Tissue | Accessibility | Absorption Rate | Rotation Capacity |
|---|---|---|---|---|
| Abdomen | High (2-4 cm) | Excellent | Optimal | 8-12 distinct sites |
| Anterior Thigh | Moderate-High (1.5-3 cm) | Excellent | Good | 6-8 distinct sites |
| Upper Arm | Moderate (1-2 cm) | Moderate | Good | 4-6 distinct sites |
This comparative framework helps researchers design comprehensive rotation schedules that maintain tissue integrity throughout extended research protocols.
Proper Injection Technique for Retatrutide Research
Mastering the technical aspects of retatrutide injection site administration ensures experimental consistency and minimizes variables that could compromise research outcomes.
Pre-Injection Preparation Protocol
Sterile technique forms the foundation of proper peptide administration:
- Hand hygiene: Thorough washing with antimicrobial soap or use of sterile gloves
- Surface preparation: Clean, dedicated workspace with sterile field
- Material assembly: Syringe, needle, alcohol swabs, sharps container, peptide pen peptide
- Peptide reconstitution: Following manufacturer guidelines for research peptides requiring reconstitution
- Visual inspection: Examining reconstituted solution for clarity, particulates, or discoloration
Temperature considerations are critical—peptides should be brought to room temperature before administration to reduce tissue irritation and improve comfort in research models.
Injection Site Preparation
Skin preparation follows standardized clinical protocols:
- Site selection: Choose location according to rotation schedule
- Visual inspection: Examine area for bruising, redness, swelling, or previous injection marks
- Cleansing: Use alcohol swab in circular motion from center outward, covering 2-inch diameter
- Drying time: Allow 30-60 seconds for complete alcohol evaporation
� Research Tip: Never inject through wet skin—residual alcohol can cause stinging and may be carried into tissue, potentially affecting peptide stability.
Needle Selection and Angle
Needle specifications for subcutaneous retatrutide administration typically include:
- Length: 4-6 mm for most applications; 8 mm for subjects with higher subcutaneous fat
- Gauge: 29-31 gauge (higher number = thinner needle)
- Type: Insulin syringes or specialized peptide administration needles
Injection angle depends on subcutaneous tissue thickness:
- 90-degree angle: For areas with ≥2 cm subcutaneous fat (most abdominal sites)
- 45-degree angle: For areas with <2 cm subcutaneous fat (some arm sites, lean subjects)
- Pinch technique: Creating a skin fold may be necessary for thinner areas
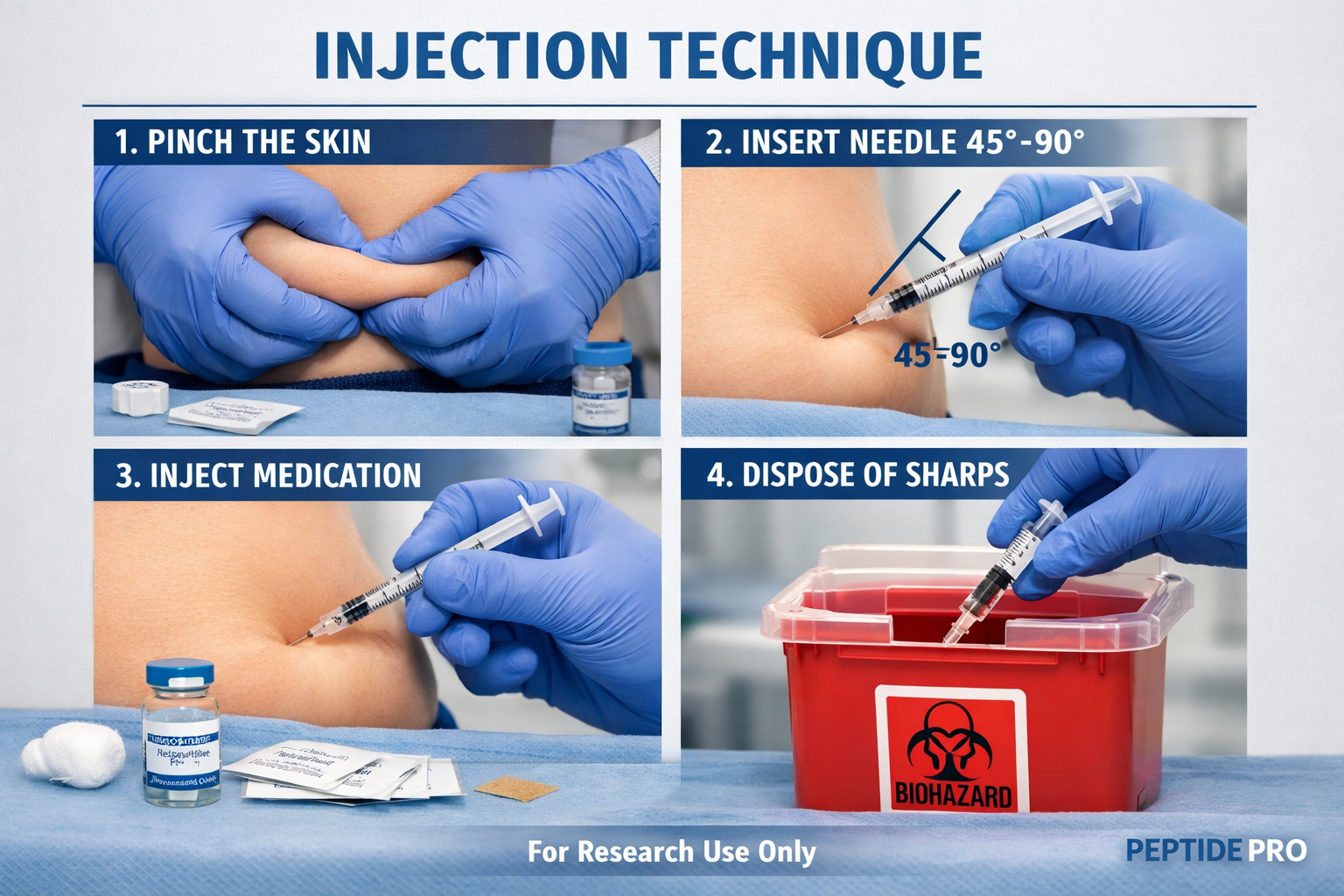
Step-by-Step Administration Technique
Systematic injection protocol for retatrutide research:
- Tissue pinch: Gently pinch skin between thumb and forefinger, creating 1-2 inch fold
- Needle insertion: Insert at appropriate angle with smooth, controlled motion
- Aspiration consideration: Generally not required for subcutaneous injections[3]
- Injection delivery: Depress plunger slowly and steadily (5-10 seconds for full dose)
- Needle withdrawal: Wait 5-10 seconds after full depression, then withdraw at same angle
- Post-injection: Release skin pinch, apply gentle pressure if needed (no rubbing)
- Disposal: Immediately place needle in sharps container
Documentation requirements for research protocols include:
- Exact anatomical location (with photographic documentation if applicable)
- Date and time of administration
- Dose volume and concentration
- Needle specifications used
- Any tissue observations or reactions
- Researcher identification
Common Technical Errors to Avoid
❌ Injection too shallow: Results in intradermal administration, causing visible wheal formation and altered absorption
❌ Injection too deep: Risks intramuscular administration, significantly changing pharmacokinetics
❌ Rapid injection: May cause tissue trauma, discomfort, and leakage after needle withdrawal
❌ Rubbing injection site: Can accelerate absorption unpredictably and cause tissue irritation
❌ Inadequate site rotation: Leads to lipohypertrophy or lipoatrophy, compromising future injections
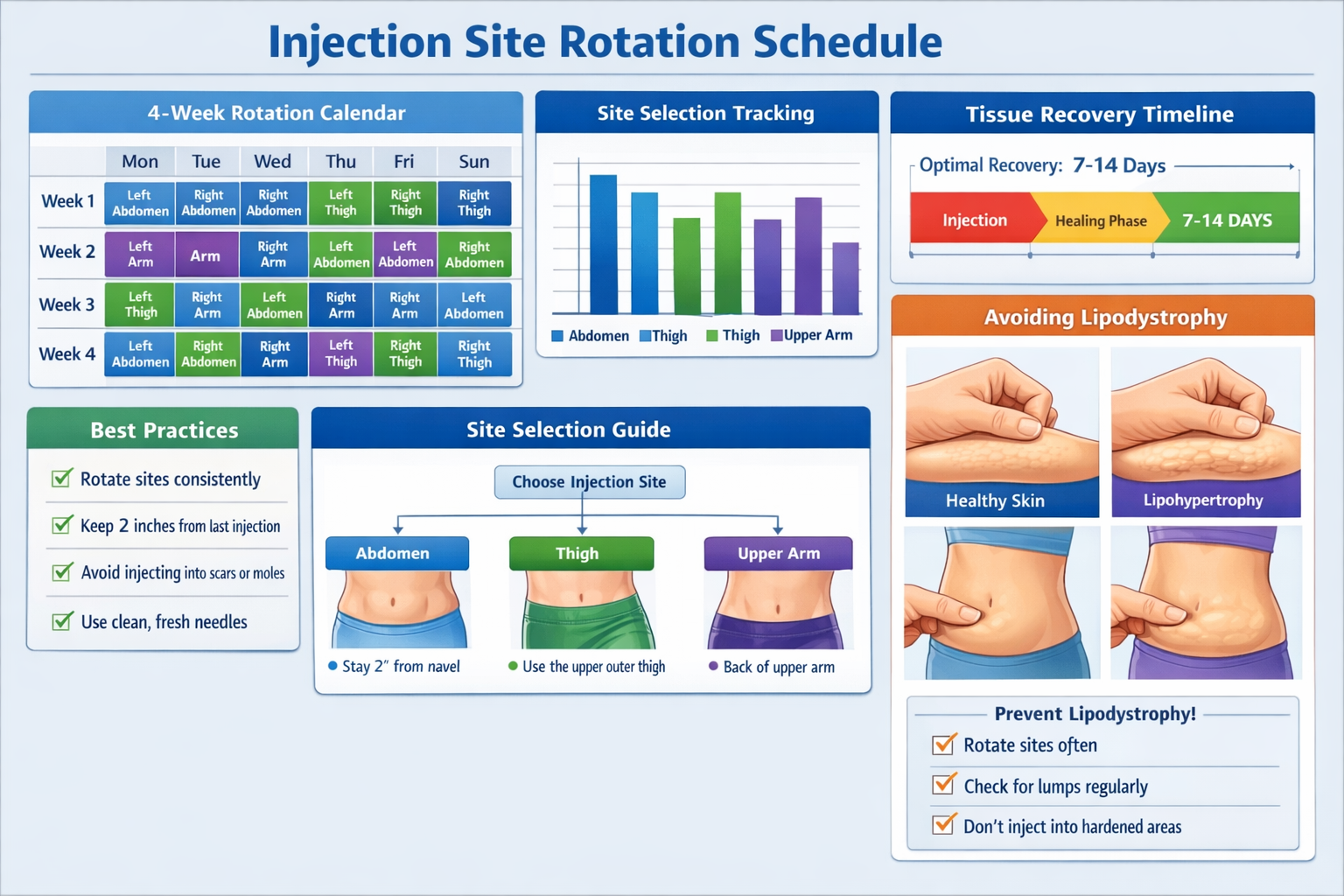
Injection Site Rotation Protocols for Extended Research
For research protocols extending beyond single-dose studies, systematic retatrutide injection site rotation becomes essential to maintaining tissue integrity and experimental consistency.
The Science Behind Site Rotation
Tissue recovery requires adequate time between injections at the same anatomical location. Repeated trauma to identical sites can cause:
- Lipohypertrophy: Fatty tissue buildup creating lumps or thickened areas
- Lipoatrophy: Fat tissue breakdown causing depressions or indentations
- Scar tissue formation: Fibrous tissue development reducing absorption capacity
- Altered vascularity: Changes in blood flow affecting pharmacokinetics
Research indicates that minimum 7-day intervals between injections at the same site help preserve tissue characteristics[4]. More conservative protocols extend this to 14-28 days depending on injection frequency.
Designing a Rotation Schedule
Weekly injection protocols (common for retatrutide research) can utilize this rotation framework:
Week 1-4 Rotation Example:
- Week 1: Right abdomen, upper quadrant
- Week 2: Left abdomen, upper quadrant
- Week 3: Right anterior thigh, mid-region
- Week 4: Left anterior thigh, mid-region
- Week 5: Right abdomen, lower quadrant
- Week 6: Left abdomen, lower quadrant
- Week 7: Right upper arm, posterior
- Week 8: Left upper arm, posterior
This eight-week cycle ensures each specific site receives at least 8 weeks recovery before reuse.
Daily or multiple weekly injection protocols require more extensive rotation:
- Subdivide each major region into multiple distinct sites (2-3 inches apart)
- Create numbered grid system for precise documentation
- Utilize all three anatomical regions (abdomen, thighs, arms) systematically
- Maintain detailed rotation logs to prevent inadvertent site repetition
Visual Mapping and Documentation
Photographic documentation provides objective records of:
- Baseline tissue appearance before protocol initiation
- Progressive tissue changes throughout research timeline
- Site-specific reactions or variations
- Long-term tissue integrity assessment
Anatomical diagrams with date-stamped markings create clear rotation records that multiple research team members can follow consistently.
Monitoring for Tissue Changes
Regular assessment of injection sites should include:
✅ Visual inspection: Color changes, swelling, bruising, lumps, or depressions
✅ Palpation: Tissue texture, temperature, firmness, or unusual formations
✅ Measurement: Subcutaneous tissue thickness using calipers (for precise research)
✅ Photographic comparison: Against baseline and previous timepoints
Intervention criteria should be established before protocol initiation, defining when site rotation adjustments or temporary cessation may be necessary.
Research Considerations and Best Practices
Optimizing retatrutide injection site selection and technique requires attention to numerous research-specific factors that influence experimental outcomes and data quality.
Standardization Across Research Subjects
Inter-subject variability in anatomy and physiology necessitates standardized protocols:
Body composition assessment helps determine optimal sites for each research subject:
- Subcutaneous fat distribution varies by genetics, sex, age, and metabolic factors
- Skinfold calipers or imaging techniques can quantify tissue thickness
- Site selection criteria should account for minimum tissue requirements
Protocol documentation must specify:
- Acceptable subcutaneous tissue thickness ranges
- Alternative site selection criteria for subjects outside normal ranges
- Standardized needle length/gauge selection based on tissue measurements
Temperature and Storage Considerations
Peptide handling before injection affects both research outcomes and injection site reactions:
Research-grade peptides from PEPTIDE PRO arrive with specific storage requirements:
- Lyophilized peptides: Store at -20°C until reconstitution
- Reconstituted solutions: Typically 2-8°C refrigeration
- Pre-injection warming: 15-30 minutes at room temperature
Cold peptide injection can cause:
- Increased tissue discomfort in research models
- Potential crystallization at injection site
- Altered absorption kinetics
- Vasoconstriction affecting distribution
Timing and Frequency Variables
Injection timing relative to other research variables should be standardized:
- Time of day: Circadian rhythms affect metabolic parameters
- Feeding status: Fasted vs. fed states influence peptide activity
- Activity level: Exercise or rest periods before/after administration
- Concurrent interventions: Other research procedures or compounds
Frequency considerations for retatrutide research protocols:
- Weekly administration is common based on peptide half-life
- More frequent dosing requires extensive rotation protocols
- Consistency in day-of-week and time-of-day reduces variability
Environmental and Sterile Technique Standards
Laboratory environment standards for peptide administration:
🔬 Dedicated clean space: Separate from general laboratory activities
🔬 Sterile field maintenance: Using sterile drapes or designated clean surfaces
🔬 Proper lighting: Adequate illumination for precise site identification
🔬 Temperature control: Comfortable ambient temperature (20-24°C)
Aseptic technique requirements extend beyond basic hand hygiene:
- Sterile gloves for all handling steps
- No-touch technique for needle and injection site
- Proper pen peptide access technique (alcohol swab, single use)
- Immediate sharps disposal without recapping
Documentation and Regulatory Compliance
Research documentation must meet institutional and regulatory standards:
📋 Protocol adherence records: Confirming each administration followed approved methods
📋 Deviation logging: Recording and explaining any protocol variations
📋 Adverse event tracking: Documenting injection site reactions or complications
📋 Chain of custody: Tracking peptide from receipt through administration
Regulatory considerations for peptide research:
- Institutional Review Board (IRB) or ethics committee approval
- Standard Operating Procedures (SOPs) for peptide handling
- Training documentation for all personnel performing injections
- Quality assurance audits and compliance verification
Troubleshooting Common Injection Site Issues
Even with meticulous technique, researchers may encounter retatrutide injection site challenges that require systematic problem-solving approaches.
Bruising and Hematoma Formation
Cause: Needle contact with small blood vessels during insertion or withdrawal.
Prevention strategies:
- Avoid areas with visible surface veins
- Use smaller gauge needles (30-31G) when possible
- Insert and withdraw at consistent angle without lateral movement
- Apply gentle pressure (not rubbing) post-injection if needed
Management protocol:
- Document location, size, and appearance photographically
- Avoid site for subsequent injections until complete resolution
- Monitor for expansion or signs of complications
- Consider coagulation assessment if bruising is excessive or frequent
Leakage After Injection
Cause: Insufficient pause before needle withdrawal or injection too shallow.
Prevention strategies:
- Wait full 10 seconds after plunger depression before withdrawing needle
- Ensure adequate subcutaneous tissue depth (minimum 4mm needle penetration)
- Inject slowly to allow tissue accommodation
- Maintain skin pinch throughout injection and brief post-injection period
Research implications:
- Leakage represents dose loss, affecting experimental accuracy
- Estimate leakage volume and document in research records
- May necessitate protocol adjustment or subject exclusion criteria
Lipodystrophy Development
Lipohypertrophy (tissue buildup) or lipoatrophy (tissue loss) indicates inadequate rotation.
Prevention strategies:
- Implement rigorous rotation schedule with minimum 7-14 day site intervals
- Vary injection depth slightly within subcutaneous range
- Avoid repeated injections within 2-inch radius
- Regular tissue assessment throughout research timeline
Management protocol:
- Permanently exclude affected sites from rotation schedule
- Document tissue changes with measurements and photography
- Consider protocol modification if multiple sites affected
- Evaluate whether tissue changes affect research outcomes
Injection Site Reactions
Local inflammation may present as redness, warmth, swelling, or tenderness.
Differential diagnosis:
- Normal tissue response: Mild, resolves within 24-48 hours
- Irritation reaction: Peptide formulation or excipient sensitivity
- Infection: Rare with proper aseptic technique, requires medical evaluation
- Allergic response: Systemic symptoms accompanying local reaction
Documentation requirements:
- Onset timing relative to injection
- Size, appearance, and progression of reaction
- Associated symptoms (systemic or local)
- Resolution timeline and any interventions
Protocol considerations:
- Establish clear criteria for normal vs. concerning reactions
- Define intervention thresholds requiring medical consultation
- Consider formulation variables if reactions are consistent
Pain or Discomfort During Injection
Excessive discomfort during administration may indicate:
- Injection too rapid (causing tissue distension)
- Peptide too cold (causing vasoconstriction)
- Intradermal injection (too shallow)
- Intramuscular injection (too deep)
- Needle dull or barbed (use new needle for each injection)
Optimization strategies:
- Ensure peptide at room temperature
- Use fresh, sharp needles
- Inject slowly and steadily
- Verify appropriate depth and angle
- Consider topical anesthetic for sensitive research models (if protocol-appropriate)
Retatrutide-Specific Research Considerations
The unique pharmacological profile of retatrutide as a triple receptor agonist introduces specific considerations for retatrutide injection site selection and administration technique.
Dose Volume and Concentration
Retatrutide research protocols typically involve varying dose ranges, affecting injection volume:
Concentration optimization:
- Higher concentrations reduce injection volume
- Smaller volumes (≤0.5 mL) generally better tolerated
- Viscosity considerations for concentrated solutions
- Reconstitution calculations must account for desired final concentration
Volume-site relationship:
- Volumes >1.0 mL may require site selection with greater subcutaneous tissue depth
- Multiple smaller injections at different sites vs. single larger volume
- Protocol consistency in volume-site matching
Reconstitution and Stability
Research peptides from PEPTIDE PRO require proper reconstitution:
Reconstitution protocol:
- Select appropriate bacteriostatic water or sterile water
- Calculate volume for desired concentration
- Add diluent slowly down pen peptide side (not directly onto peptide)
- Gentle swirling (never shaking) until fully dissolved
- Visual inspection for complete dissolution and clarity
Stability considerations:
- Reconstituted retatrutide stability varies with formulation
- Typical refrigerated stability: 14-28 days
- Freeze-thaw cycles degrade peptide structure
- Light protection may be necessary for some formulations
Pharmacokinetic Considerations
Absorption variability by injection site affects research outcomes:
Site-specific pharmacokinetics:
- Abdominal injections typically show most consistent absorption
- Thigh injections may have slightly slower absorption
- Arm injections show greater inter-subject variability
- Exercise affecting injected limb can accelerate absorption
Research design implications:
- Consistent site selection within treatment groups
- Site as potential stratification variable
- Pharmacokinetic sampling timing relative to injection site
- Cross-over designs accounting for site effects
Comparison with Other Research Peptides
Researchers familiar with other peptides may note similarities and differences:
| Peptide | Typical Dose Volume | Injection Frequency | Site Preferences |
|---|---|---|---|
| Retatrutide | 0.2-0.5 mL | Weekly | Abdomen, thigh, arm |
| Semaglutide | 0.2-0.5 mL | Weekly | Abdomen, thigh, arm |
| Tirzepatide | 0.2-0.5 mL | Weekly | Abdomen, thigh, arm |
| BPC-157 | 0.1-0.3 mL | Daily-BID | Near injury site or standard SC sites |
This comparative framework helps researchers transitioning between different peptide research protocols.
Quality Assurance and Research Integrity
Maintaining retatrutide injection site standards throughout research protocols requires systematic quality assurance measures and commitment to research integrity.
Supplier Verification and Peptide Quality
Research outcomes depend fundamentally on peptide purity and authenticity:
PEPTIDE PRO quality standards:
- ≥98% purity for research-grade peptides
- Comprehensive Certificates of Analysis (COAs) with each batch
- Third-party testing verification
- Proper storage and handling from manufacture through delivery
- Clear labeling: “For Research Use Only – Not for Human Consumption”
Researcher verification responsibilities:
- Review COA before protocol initiation
- Verify peptide identity through expected reconstitution characteristics
- Store according to manufacturer specifications
- Monitor for degradation signs (color change, precipitation, cloudiness)
- Document lot numbers in research records
Training and Competency Assessment
Personnel performing injections must demonstrate competency:
Training requirements:
- Theoretical understanding of subcutaneous injection anatomy
- Practical demonstration on training models
- Supervised performance with feedback
- Periodic competency reassessment
- Documentation of training completion
Competency indicators:
- Consistent technique across multiple administrations
- Appropriate site selection and rotation
- Proper aseptic technique maintenance
- Accurate documentation
- Appropriate response to complications
Standard Operating Procedures
Written SOPs should detail every aspect of retatrutide administration:
Essential SOP components:
- Purpose and scope: Research applications and limitations
- Responsibilities: Personnel roles and qualifications
- Materials: Specific supplies and equipment
- Procedure: Step-by-step injection protocol
- Safety: Hazard identification and mitigation
- Documentation: Required records and forms
- Quality control: Verification and validation steps
- References: Supporting scientific literature
SOP maintenance:
- Regular review and update cycle (annually minimum)
- Version control and change documentation
- Training on SOP updates
- Accessibility to all relevant personnel
Audit Trails and Data Integrity
Research documentation must support data integrity principles (ALCOA+):
- Attributable: Clear identification of who performed each injection
- Legible: Readable, permanent records
- Contemporaneous: Documented at time of performance
- Original: Primary records maintained
- Accurate: Verified and correct information
- Complete: All required data elements captured
- Consistent: Uniform documentation across protocol
- Enduring: Records preserved throughout required retention period
- Available: Accessible for review and audit
Electronic records for injection site documentation should include:
- Timestamp verification
- User authentication
- Audit trail of any modifications
- Backup and recovery systems
- Validation of electronic systems
Future Directions in Retatrutide Research
The evolving landscape of peptide research continues to refine best practices for retatrutide injection site selection and administration techniques.
Alternative Delivery Systems
Emerging technologies may influence future research protocols:
Microneedle arrays: Painless delivery through microscopic needle patches
- Potential for improved consistency
- Reduced training requirements
- Enhanced subject compliance in applicable research models
Jet injectors: Needle-free delivery using high-pressure streams
- Eliminates needle phobia variables
- Consistent depth delivery
- Higher equipment costs
Sustained-release formulations: Extended pharmacokinetic profiles
- Reduced injection frequency
- Altered site rotation requirements
- Different tissue interaction profiles
Personalized Site Selection
Precision medicine approaches may optimize injection sites based on:
- Individual body composition analysis
- Genetic factors affecting absorption
- Metabolic profiling
- Previous response patterns
Research protocols incorporating personalized site selection would require:
- Baseline tissue characterization
- Pharmacokinetic sampling
- Adaptive site assignment algorithms
- Enhanced documentation systems
Technology Integration
Digital tools enhancing injection site management:
Mobile applications for:
- Rotation schedule tracking
- Photographic documentation
- Automated site selection recommendations
- Integration with electronic research records
Imaging technologies:
- Ultrasound verification of injection depth
- Thermal imaging for tissue assessment
- 3D body mapping for precise site documentation
Regulatory Evolution
Research standards continue evolving with:
- Enhanced Good Laboratory Practice (GLP) requirements
- Standardized reporting frameworks
- International harmonization efforts
- Increased transparency expectations
Researchers utilizing premium research-grade peptides should stay informed of regulatory developments affecting peptide research protocols.
Excellence in Retatrutide Injection Site Research
Mastering retatrutide injection site selection and administration technique forms the foundation of rigorous, reproducible peptide research. From understanding the anatomical options—abdomen, anterior thigh, and posterior upper arm—to implementing systematic rotation protocols that preserve tissue integrity, every detail contributes to research quality and data reliability.
The key principles established throughout this guide emphasize:
✅ Anatomical precision: Selecting appropriate sites based on subcutaneous tissue characteristics, accessibility, and absorption properties
✅ Technical excellence: Maintaining sterile technique, proper needle angles, appropriate injection depths, and consistent administration methods
✅ Systematic rotation: Implementing evidence-based rotation schedules that allow adequate tissue recovery between injections at the same site
✅ Quality assurance: Sourcing research-grade peptides from verified suppliers, maintaining comprehensive documentation, and adhering to established protocols
✅ Continuous improvement: Monitoring injection sites for tissue changes, troubleshooting complications systematically, and adapting protocols based on emerging evidence
Actionable Next Steps for Researchers
For researchers initiating retatrutide protocols:
- Develop comprehensive SOPs detailing every aspect of injection site selection, preparation, administration, and documentation
- Source high-purity peptides from verified suppliers like PEPTIDE PRO with appropriate COAs and research-grade certifications
- Establish rotation schedules before protocol initiation, accounting for injection frequency and available anatomical sites
- Implement training programs ensuring all personnel demonstrate competency in proper injection technique
- Create documentation systems supporting data integrity principles and regulatory compliance
For ongoing research protocols:
- Conduct regular site assessments using visual inspection, palpation, and photographic documentation
- Review rotation adherence ensuring adequate intervals between injections at the same anatomical location
- Monitor for tissue changes that might compromise absorption characteristics or research outcomes
- Maintain detailed records linking injection sites to pharmacokinetic data and research endpoints
- Stay current with emerging best practices and technological advances in peptide administration
Research Resources and Support
The scientific community benefits from shared knowledge and collaborative advancement. PEPTIDE PRO’s educational resources provide additional guidance on peptide handling, reconstitution principles, and storage requirements specific to research applications.
For researchers requiring high-purity retatrutide and other research peptides, PEPTIDE PRO offers:
- Research-grade peptides with ≥98% purity
- Comprehensive COAs and product documentation
- Same-day dispatch for orders before 1pm (Mon-Fri)
- Fast UK delivery and international shipping options
- Expert customer support for research-specific inquiries
Retatrutide research continues to evolve, with ongoing investigations exploring its unique triple-agonist mechanism and potential applications. By maintaining rigorous standards for injection site selection and administration technique, researchers contribute to the growing body of knowledge surrounding this promising peptide.
The intersection of proper technique, quality materials, systematic documentation, and scientific rigor creates the foundation for meaningful research outcomes. Whether investigating metabolic pathways, receptor interactions, or pharmacokinetic profiles, excellence in retatrutide injection site management ensures that your research meets the highest standards of scientific integrity.
Remember: All peptides from PEPTIDE PRO are strictly for research use only—not for human or animal consumption. Ensure your research protocols comply with all applicable institutional, ethical, and regulatory requirements.
References
[1] Jastreboff AM, et al. (2023). Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine, 389(6), 514-526.
[2] Frid AH, et al. (2016). New Injection Recommendations for Patients with Diabetes. Diabetes & Metabolism, 42(S1), 3-18.
[3] Sisson H. (2015). Aspiration During Subcutaneous Injection: A Systematic Review. Journal of Clinical Nursing, 24(17-18), 2368-2375.
[4] Gibney MA, et al. (2010). Skin and Subcutaneous Adipose Layer Thickness in Adults with Diabetes at Sites Used for Insulin Injections: Implications for Needle Length Recommendations. Current Medical Research and Opinion, 26(6), 1519-1530.
