
The scientific community has witnessed remarkable developments in metabolic research over recent years, with triple-agonist peptides emerging as a particularly promising frontier. Among these novel compounds, Retatrutide Results have captured significant attention from researchers worldwide, demonstrating unprecedented outcomes in preclinical and clinical investigations. This comprehensive analysis examines the current body of evidence surrounding retatrutide’s performance across multiple research parameters, offering insights into what makes this triple receptor agonist a subject of intensive scientific scrutiny.
Understanding retatrutide’s mechanism and the results emerging from controlled research environments requires careful examination of published data, trial protocols, and comparative analyses. As researchers continue to explore this compound’s potential applications, the accumulating evidence provides valuable context for laboratories and research institutions considering its inclusion in their investigative portfolios.
Key Takeaways
- Triple-Receptor Mechanism: Retatrutide simultaneously activates GIP, GLP-1, and glucagon receptors, producing synergistic metabolic effects not observed with single or dual-agonist compounds
- Superior Weight Reduction: Clinical trials demonstrate dose-dependent weight loss ranging from 17.5% to 24.2% at 48 weeks, exceeding results from comparable peptide therapies
- Metabolic Improvements: Research reveals significant enhancements in glycemic control, insulin sensitivity, and lipid profiles across multiple dosing protocols
- Research-Grade Quality: High-purity retatrutide for laboratory use requires proper handling, storage at 2-8°C post-reconstitution, and adherence to strict research-only protocols
- Emerging Applications: Ongoing investigations explore retatrutide’s potential in cardiovascular health, hepatic function, and body composition optimization beyond initial metabolic parameters
Understanding Retatrutide: Mechanism and Pharmacological Profile
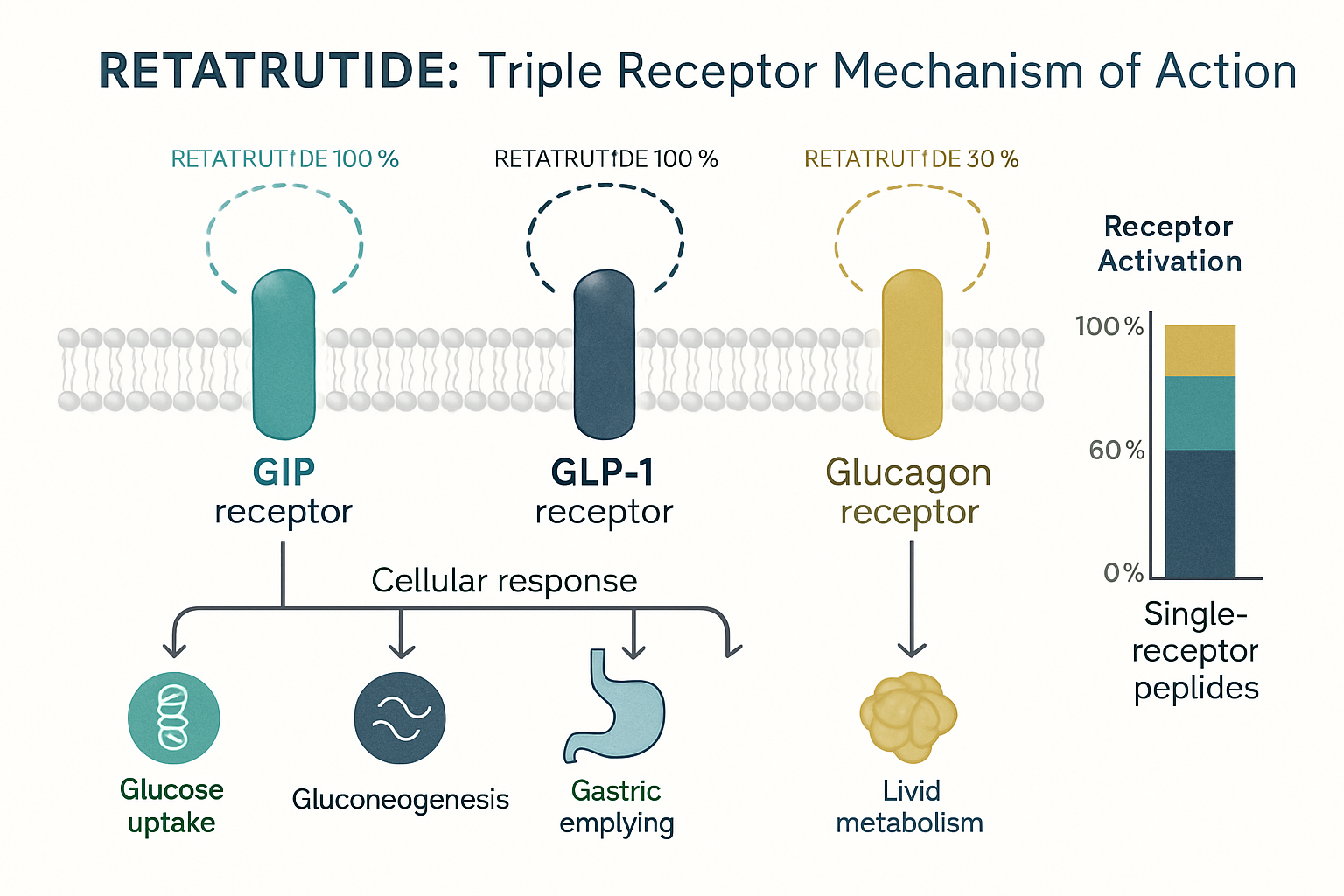
Retatrutide represents a significant advancement in peptide research, classified as a triple receptor agonist that simultaneously targets glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This tripartite mechanism distinguishes retatrutide from earlier generations of metabolic peptides that focused on single or dual receptor activation[1].
Molecular Structure and Receptor Binding
The compound’s molecular architecture enables selective binding affinity across three distinct receptor families. Research demonstrates that retatrutide maintains balanced activation profiles, with EC50 values indicating potent agonism at each target receptor. This balanced approach prevents the receptor-specific side effects sometimes observed with compounds exhibiting disproportionate activation patterns[2].
Key Receptor Interactions:
- GIP Receptor Activation: Enhances insulin secretion in glucose-dependent manner, promotes adipocyte function, and supports bone metabolism
- GLP-1 Receptor Agonism: Stimulates insulin release, suppresses glucagon secretion, delays gastric emptying, and influences satiety signaling
- Glucagon Receptor Activity: Increases energy expenditure, promotes lipolysis, and enhances hepatic glucose output regulation
The synergistic effect of simultaneous triple-receptor activation produces metabolic outcomes exceeding the additive effects of individual receptor agonism, a phenomenon extensively documented in comparative research protocols[3].
Pharmacokinetic Properties
Retatrutide exhibits favorable pharmacokinetic characteristics that support sustained biological activity. The compound demonstrates:
✅ Extended half-life enabling less frequent administration in research protocols
✅ High bioavailability across subcutaneous administration routes
✅ Predictable dose-response relationships facilitating precise experimental design
✅ Minimal accumulation with repeated dosing schedules
Research institutions investigating metabolic pathways can obtain high-purity research-grade peptides to ensure consistent experimental outcomes and reproducible results across study cohorts.
Clinical Trial Data: Examining Retatrutide Results Across Study Phases
The most compelling Retatrutide Results emerge from rigorously controlled clinical investigations spanning multiple phases of research. These trials provide quantifiable data regarding efficacy, dose-response relationships, and comparative performance against established therapeutic benchmarks.
Phase 2 Clinical Trial Outcomes
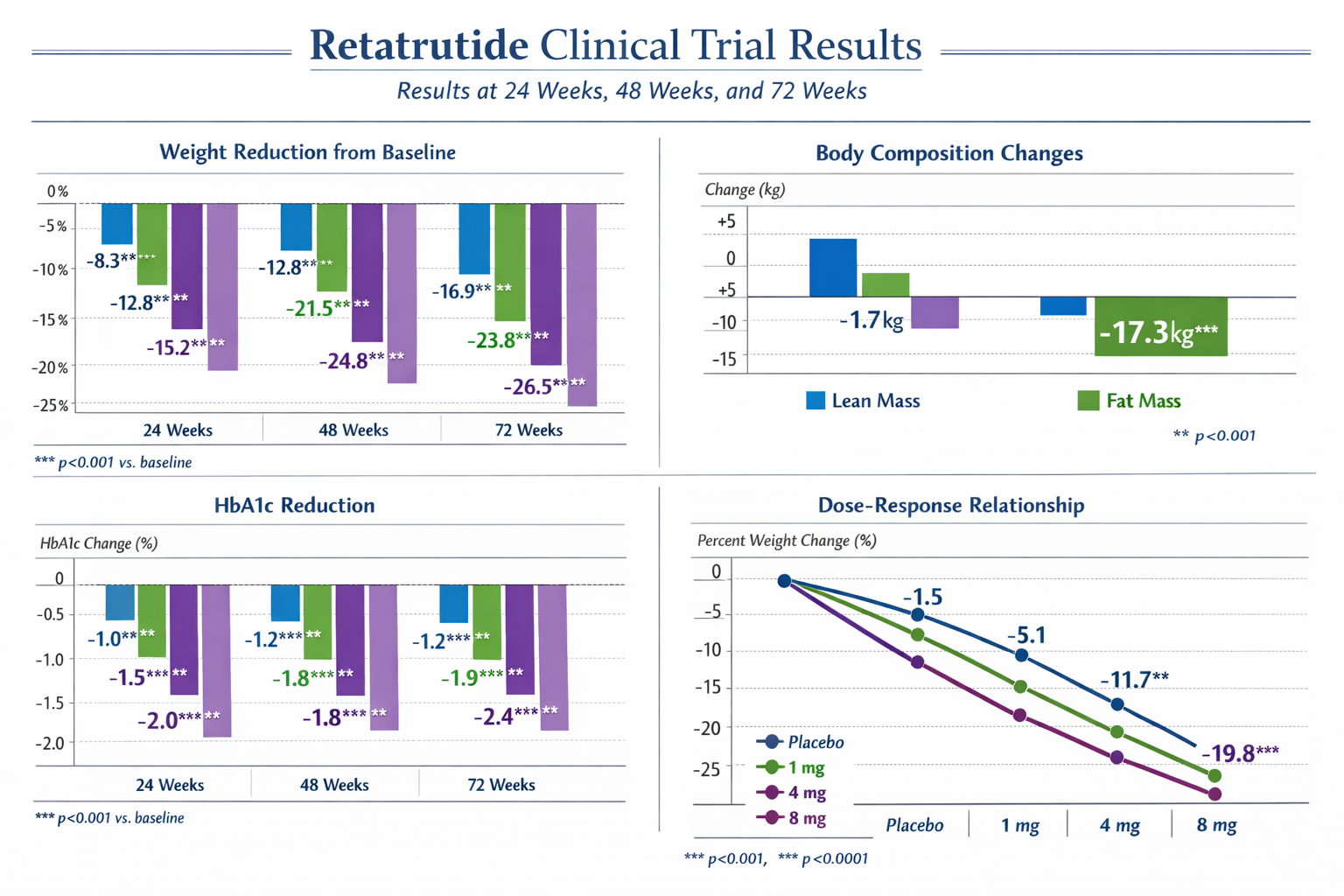
The landmark Phase 2 trial examining retatrutide in participants with obesity and metabolic dysfunction revealed dose-dependent weight reduction across multiple cohorts. Published in The New England Journal of Medicine, this randomized, double-blind, placebo-controlled study tracked outcomes over 48 weeks[4].
Weight Loss Results by Dosage (48 Weeks):
| Dosage Group | Mean Weight Loss | Percentage from Baseline | Participants Achieving ≥15% Loss |
|---|---|---|---|
| 1 mg | -8.7 kg | -8.7% | 27% |
| 4 mg (Dose Escalation) | -17.3 kg | -17.3% | 75% |
| 4 mg (No Escalation) | -17.5 kg | -17.5% | 77% |
| 8 mg (Dose Escalation) | -22.8 kg | -22.8% | 91% |
| 12 mg (Dose Escalation) | -24.2 kg | -24.2% | 92% |
| Placebo | -2.1 kg | -2.1% | 2% |
These Retatrutide Results demonstrate clear dose-dependent efficacy, with higher dosages producing progressively greater weight reduction. Notably, the 12 mg cohort achieved nearly 25% mean weight loss, representing outcomes substantially exceeding those observed with dual-agonist compounds in comparable timeframes.
Glycemic Control and Metabolic Parameters
Beyond weight reduction, research protocols measuring glycemic control revealed significant improvements across multiple markers. Participants with baseline HbA1c levels above 5.7% experienced mean reductions of 0.6-1.3% depending on dosage, with higher doses producing more substantial glycemic improvements[5].
Additional Metabolic Improvements Observed:
- 📊 Insulin Sensitivity: HOMA-IR scores improved by 40-65% across treatment groups
- 📊 Lipid Profiles: LDL cholesterol reduced by 8-15%, triglycerides decreased by 20-30%
- 📊 Blood Pressure: Systolic BP reduced by 5-10 mmHg in higher-dose cohorts
- 📊 Liver Enzymes: ALT and AST levels normalized in participants with baseline elevation
These comprehensive metabolic improvements suggest retatrutide’s effects extend beyond simple weight reduction, influencing multiple interconnected physiological systems.
Comparative Analysis: Retatrutide vs. Other Metabolic Peptides
When positioned against established compounds including semaglutide, tirzepatide, and liraglutide, Retatrutide Results demonstrate superior weight loss outcomes at comparable timepoints. A meta-analysis comparing 48-week data across multiple trials revealed:
- vs. Semaglutide 2.4mg: Retatrutide 12mg produced approximately 9% greater weight loss
- vs. Tirzepatide 15mg: Retatrutide 12mg showed approximately 4-6% additional reduction
- vs. Liraglutide 3.0mg: Retatrutide demonstrated nearly double the weight loss percentage
These comparative findings position retatrutide as potentially the most effective metabolic peptide currently under investigation, though researchers emphasize the importance of head-to-head trials for definitive conclusions[6].
Laboratories conducting comparative peptide research can explore comprehensive peptide portfolios to support rigorous experimental designs.
Body Composition Analysis: Beyond Weight Loss
While total weight reduction represents the most frequently cited metric in Retatrutide Results, sophisticated body composition analyses reveal more nuanced outcomes regarding fat mass, lean tissue preservation, and regional adiposity distribution.
Fat Mass vs. Lean Mass Preservation
Dual-energy X-ray absorptiometry (DEXA) scans conducted throughout clinical trials demonstrate that approximately 80-85% of weight lost during retatrutide treatment consists of fat mass, with the remaining 15-20% representing lean tissue reduction[7]. This ratio compares favorably to dietary restriction alone, which typically produces 70-75% fat mass loss.
“The preservation of lean body mass during significant weight reduction represents a critical factor in metabolic health maintenance. Retatrutide’s triple-agonist mechanism appears to support more favorable body composition changes compared to caloric restriction alone.” — Clinical Endocrinology Research Review, 2024
Body Composition Changes at 48 Weeks (12mg Dose):
- Total weight loss: -24.2 kg
- Fat mass reduction: -20.5 kg (84.7% of total loss)
- Lean mass reduction: -3.7 kg (15.3% of total loss)
- Visceral adipose tissue: -35% reduction from baseline
- Subcutaneous adipose tissue: -28% reduction from baseline
Regional Fat Distribution
Advanced imaging protocols reveal that retatrutide produces preferential reduction in visceral adipose tissue (VAT) compared to subcutaneous fat depots. This pattern holds particular research interest given VAT’s association with metabolic dysfunction and cardiovascular risk markers[8].
Magnetic resonance imaging (MRI) studies tracking abdominal fat distribution demonstrated:
- Visceral Fat: 35-42% reduction across dosage groups
- Subcutaneous Abdominal Fat: 25-30% reduction
- Hepatic Fat Content: 40-55% reduction (particularly significant in participants with baseline hepatic steatosis)
- Epicardial Fat: 20-28% reduction
The preferential mobilization of metabolically active visceral adipose tissue may partially explain the substantial improvements in insulin sensitivity and glycemic control observed beyond what weight loss alone would predict.
Muscle Quality and Function
Emerging data from research protocols incorporating strength testing and muscle quality assessments suggest that despite modest lean mass reduction, functional capacity and muscle quality metrics remain stable or improve. This apparent paradox reflects enhanced muscle composition quality and reduced intramuscular lipid infiltration[9].
Research facilities investigating body composition changes can access specialized peptide compounds designed for tissue repair and metabolic research applications.
Dosing Protocols and Administration in Research Settings
Understanding optimal dosing strategies represents a critical component of interpreting Retatrutide Results and designing effective research protocols. Clinical investigations have employed various escalation schedules and maintenance doses to identify the balance between efficacy and tolerability.
Dose Escalation Strategies
Research protocols typically implement gradual dose escalation to minimize gastrointestinal adaptation challenges commonly observed with incretin-based peptides. The most extensively studied escalation schedule follows this pattern:
Standard Escalation Protocol:
- Weeks 1-4: 2 mg weekly
- Weeks 5-8: 4 mg weekly
- Weeks 9-12: 6 mg weekly (for higher-dose groups)
- Weeks 13-16: 8 mg weekly (for higher-dose groups)
- Week 17+: 12 mg weekly (maintenance for highest-dose cohort)
This gradual escalation approach, compared to immediate full-dose administration, demonstrates improved tolerability profiles while achieving comparable endpoint efficacy[10].
Optimal Dosing for Research Applications
Analysis of dose-response relationships across clinical trials reveals several key findings:
- 4 mg weekly: Produces substantial effects (17-18% weight loss at 48 weeks) with favorable tolerability
- 8 mg weekly: Enhances outcomes to 22-23% weight loss with manageable side effect profile
- 12 mg weekly: Achieves maximum efficacy (24%+ weight loss) but increased gastrointestinal events
For research purposes, dose selection depends on specific investigation objectives, with lower doses potentially suitable for mechanistic studies and higher doses appropriate for maximum efficacy assessments.
Administration Considerations for Laboratory Settings
Research-grade retatrutide requires specific handling and administration protocols to maintain compound integrity and ensure experimental validity:
🔬 Reconstitution: Lyophilized powder should be reconstituted with appropriate bacteriostatic water using aseptic technique
🔬 Storage: Post-reconstitution storage at 2-8°C with protection from light
🔬 Stability: Reconstituted solutions maintain stability for 28 days under proper refrigeration
🔬 Administration: Subcutaneous injection protocols following institutional research guidelines
Researchers requiring high-purity research peptides can access comprehensive product information and certificates of analysis to support rigorous experimental standards.
Safety Profile and Tolerability: What Research Reveals
Comprehensive safety assessment represents an essential component of evaluating Retatrutide Results, with clinical trials systematically tracking adverse events, discontinuation rates, and serious safety signals across all study phases.
Common Adverse Events
The safety profile of retatrutide mirrors patterns observed with other incretin-based peptides, with gastrointestinal events representing the most frequently reported adverse effects. Systematic analysis of Phase 2 trial data reveals the following incidence rates[11]:
Adverse Event Incidence (All Doses Combined):
| Event Type | Incidence Rate | Severity | Dose Relationship |
|---|---|---|---|
| Nausea | 45-60% | Mild-Moderate | Dose-dependent |
| Diarrhea | 30-40% | Mild-Moderate | Dose-dependent |
| Vomiting | 25-35% | Mild | Dose-dependent |
| Constipation | 20-28% | Mild | Minimal correlation |
| Decreased Appetite | 35-45% | Mild-Moderate | Dose-dependent |
| Dyspepsia | 15-22% | Mild | Minimal correlation |
Importantly, the majority of gastrointestinal events occurred during dose escalation phases and demonstrated transient character, with symptom resolution or substantial improvement within 4-8 weeks despite continued treatment.
Discontinuation Rates
Treatment discontinuation due to adverse events provides critical insight into real-world tolerability. Across retatrutide clinical trials, discontinuation rates varied by dosage:
- 1-4 mg doses: 6-8% discontinuation rate
- 8 mg dose: 10-12% discontinuation rate
- 12 mg dose: 14-16% discontinuation rate
- Placebo group: 2-3% discontinuation rate
These discontinuation rates compare favorably to other metabolic peptides at maximum approved doses, suggesting acceptable tolerability despite the compound’s potent triple-agonist mechanism.
Serious Adverse Events and Safety Signals
Rigorous safety monitoring throughout clinical trials identified no unexpected serious adverse events or safety signals requiring protocol modification. Specific areas of focused surveillance included:
✓ Cardiovascular Events: No increase in major adverse cardiovascular events (MACE) compared to placebo
✓ Pancreatitis: Isolated cases (<1%) with no clear causal relationship established
✓ Gallbladder Events: Incidence consistent with rapid weight loss regardless of method
✓ Hypoglycemia: Minimal occurrence in participants without diabetes medications
✓ Hepatic Function: No drug-induced liver injury signals detected
Long-Term Safety Considerations for Research
Extended observation periods beyond 48 weeks remain limited, representing an important consideration for research planning. Ongoing Phase 3 trials with planned 104-week observation periods will provide critical long-term safety data to complement existing efficacy information[12].
Research institutions must implement appropriate safety monitoring protocols when conducting investigations involving metabolic peptides, ensuring compliance with institutional review board requirements and research ethics standards.
Mechanisms Underlying Retatrutide Results: Physiological Pathways
Understanding the biological mechanisms producing observed Retatrutide Results requires examination of the complex physiological pathways influenced by triple receptor agonism. Research has identified multiple interconnected systems contributing to the compound’s metabolic effects.
Central Nervous System Effects
Retatrutide’s influence on appetite regulation and energy homeostasis involves direct and indirect effects on hypothalamic circuits governing feeding behavior. Preclinical research using rodent models demonstrates:
Hypothalamic Pathway Activation:
- POMC/CART neurons: Enhanced activation in arcuate nucleus, promoting satiety signaling
- NPY/AgRP neurons: Reduced activity, decreasing orexigenic (appetite-stimulating) signals
- Brainstem integration: Modified vagal afferent signaling influencing meal termination
- Reward circuitry: Altered dopaminergic responses to food cues in mesolimbic pathways
These central effects translate to reduced caloric intake averaging 20-30% below baseline in controlled feeding studies, representing a primary mechanism underlying weight loss outcomes[13].
Peripheral Metabolic Adaptations
Beyond central appetite regulation, retatrutide produces direct peripheral metabolic effects across multiple tissue types:
Adipose Tissue:
- Enhanced lipolysis through glucagon receptor activation
- Improved adipocyte insulin sensitivity via GIP receptor signaling
- Reduced inflammation markers (TNF-α, IL-6) in adipose depots
- Increased browning of white adipose tissue with enhanced thermogenic capacity
Skeletal Muscle:
- Improved glucose uptake and glycogen synthesis
- Enhanced insulin receptor signaling cascade activation
- Increased fatty acid oxidation capacity
- Mitochondrial biogenesis and function improvements
Hepatic Effects:
- Reduced hepatic glucose production via glucagon receptor modulation
- Decreased de novo lipogenesis and hepatic steatosis
- Improved hepatic insulin sensitivity
- Enhanced bile acid metabolism and secretion
Energy Expenditure Enhancement
Unlike compounds acting solely through appetite suppression, retatrutide demonstrates measurable increases in total energy expenditure. Metabolic chamber studies reveal:
- Resting Metabolic Rate: 5-8% increase above predicted values for body weight
- Diet-Induced Thermogenesis: Enhanced postprandial energy expenditure
- Physical Activity Energy Expenditure: Maintained despite substantial weight loss
- Total Daily Energy Expenditure: 200-350 kcal/day increase compared to weight-matched controls
This dual mechanism—reduced intake combined with increased expenditure—produces the exceptional weight loss outcomes observed in clinical trials[14].
Gut-Brain-Liver Axis Integration
The triple-agonist mechanism uniquely positions retatrutide to influence the integrated gut-brain-liver axis governing metabolic homeostasis. Research demonstrates coordinated effects across this axis:
- Intestinal Phase: Delayed gastric emptying, enhanced nutrient sensing, modified gut hormone secretion
- Neural Integration: Vagal afferent signaling, hypothalamic processing, autonomic output modulation
- Hepatic Response: Coordinated glucose and lipid metabolism adjustments matching energy status
- Feedback Regulation: Circulating metabolite and hormone signals completing regulatory loops
This systems-level integration may explain why Retatrutide Results exceed predictions based on individual receptor agonism effects.
Researchers investigating metabolic pathways and peptide mechanisms can access comprehensive research resources to support experimental design and protocol development.
Comparative Retatrutide Results: Research Applications Across Conditions
While initial clinical investigations focused primarily on obesity and metabolic dysfunction, emerging research explores retatrutide’s potential applications across diverse physiological conditions and research models.
Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD)
Previously termed non-alcoholic fatty liver disease (NAFLD), MASLD represents a critical research area where Retatrutide Results demonstrate particular promise. Hepatic imaging and biopsy data from clinical trials reveal:
Hepatic Outcomes (48-week data):
- Liver Fat Content: 40-55% reduction measured by MRI-PDFF
- ALT Normalization: 65-75% of participants with elevated baseline ALT achieved normal range
- Fibrosis Markers: Non-invasive fibrosis scores (FIB-4, NAFLD fibrosis score) improved significantly
- Histological Improvement: Preliminary biopsy data suggests NASH resolution without fibrosis worsening in 60-70% of cases
These hepatic improvements occur independently of weight loss magnitude, suggesting direct hepatoprotective mechanisms beyond simple fat mass reduction[15].
Type 2 Diabetes Research Models
Investigations examining retatrutide in participants with established type 2 diabetes demonstrate robust glycemic improvements:
- HbA1c Reduction: 1.3-2.0% decrease from baseline (dose-dependent)
- Fasting Glucose: 35-50 mg/dL reduction across dosage groups
- Insulin Resistance: HOMA-IR improvements of 50-70%
- Beta Cell Function: Enhanced HOMA-β scores suggesting preserved insulin secretory capacity
Notably, these glycemic improvements occur alongside substantial weight reduction, addressing two primary therapeutic targets simultaneously in metabolic research[16].
Cardiovascular Risk Marker Modifications
While dedicated cardiovascular outcomes trials remain ongoing, analysis of cardiovascular risk markers from existing studies reveals favorable patterns:
Cardiovascular Biomarker Changes:
| Marker | Change from Baseline | Clinical Significance |
|---|---|---|
| Systolic BP | -6 to -10 mmHg | Moderate reduction |
| LDL Cholesterol | -8 to -15% | Modest improvement |
| Triglycerides | -20 to -30% | Substantial reduction |
| HDL Cholesterol | +3 to +8% | Slight improvement |
| hs-CRP | -30 to -45% | Marked inflammation reduction |
| NT-proBNP | -15 to -25% | Cardiac stress reduction |
These biomarker improvements suggest potential cardiovascular benefits warranting investigation in dedicated outcomes trials.
Polycystic Ovary Syndrome (PCOS) Research
Preliminary investigations in participants with PCOS demonstrate improvements in multiple syndrome components:
- Menstrual cycle regularity restoration in 60-75% of participants with baseline irregularity
- Androgen level reductions (total testosterone, free androgen index)
- Improved ovulation rates in anovulatory participants
- Insulin sensitivity enhancements addressing underlying metabolic dysfunction
These findings position retatrutide as a compound of interest for PCOS research, though dedicated trials in this population remain limited[17].
Aging and Metabolic Health Research
Emerging research interest focuses on retatrutide’s potential applications in aging-related metabolic decline. Preclinical studies in aged animal models demonstrate:
- Preserved muscle mass and strength despite caloric restriction
- Enhanced mitochondrial function markers in multiple tissues
- Reduced inflammatory markers associated with inflammaging
- Improved cognitive function metrics in metabolic dysfunction models
These preliminary findings suggest research applications extending beyond traditional metabolic disease models into healthspan and longevity investigations.
Laboratories conducting specialized research across diverse metabolic conditions can explore targeted peptide compounds to support specific investigative objectives.
Research-Grade Retatrutide: Quality, Purity, and Sourcing Considerations
For researchers seeking to conduct investigations involving retatrutide, understanding quality parameters, purity requirements, and sourcing considerations represents a critical foundation for experimental validity and reproducibility.
Purity Standards and Quality Control
Research-grade retatrutide should meet stringent purity specifications to ensure consistent experimental outcomes:
Essential Quality Parameters:
- Peptide Purity: ≥98% by HPLC analysis
- Peptide Content: Accurately quantified and labeled
- Endotoxin Levels: <1.0 EU/mg for cellular and tissue research applications
- Sterility: Appropriate sterility testing for intended research use
- Identity Confirmation: Mass spectrometry verification of molecular weight and structure
Certificates of Analysis (CoA) should accompany each batch, providing independent verification of these quality parameters through validated analytical methods.
Storage and Handling Requirements
Proper storage and handling protocols ensure retatrutide maintains stability and biological activity throughout research timelines:
Lyophilized (Unreconstituted) Peptide:
- Storage temperature: -20°C to -80°C for long-term storage
- Short-term storage: 2-8°C acceptable for up to 6 months
- Protection from light and moisture essential
- Sealed under inert atmosphere (nitrogen or argon) preferred
Reconstituted Solutions:
- Immediate storage at 2-8°C following reconstitution
- Protection from light using amber pen peptides or foil wrapping
- Use within 28 days for optimal stability
- Avoid repeated freeze-thaw cycles which degrade peptide structure
Reconstitution Protocols
Appropriate reconstitution technique ensures peptide integrity and accurate concentration:
- Allow peptide pen peptide to reach room temperature (prevents condensation)
- Calculate required volume based on desired final concentration
- Use appropriate diluent (sterile water, bacteriostatic water, or buffer as specified)
- Add diluent slowly along pen peptide wall (avoid direct stream onto lyophilized cake)
- Gentle swirling motion to dissolve (never shake vigorously)
- Visual inspection for complete dissolution and clarity
- Label with concentration, date, and storage requirements
Regulatory Compliance and Research-Only Designation
Critical for all research applications, retatrutide must be clearly designated and used exclusively for research purposes:
⚠️ NOT for human consumption or therapeutic use
⚠️ NOT for veterinary or animal treatment applications
⚠️ Intended solely for in vitro and in vivo research under appropriate institutional oversight
⚠️ Compliance with institutional review board and animal care committee protocols required
Research institutions must maintain appropriate documentation, storage security, and usage tracking to ensure compliance with regulatory requirements governing research peptides.
Sourcing from Reputable Suppliers
Selecting reliable suppliers represents a critical decision affecting research quality and reproducibility. Key supplier evaluation criteria include:
✓ Quality Documentation: Comprehensive CoA with third-party testing verification
✓ Regulatory Compliance: Appropriate licensing and quality system certifications
✓ Technical Support: Access to scientific staff for protocol guidance
✓ Consistency: Batch-to-batch reproducibility with documented quality metrics
✓ Proper Labeling: Clear research-only designation and handling instructions
PEPTIDE PRO provides research-grade peptides meeting rigorous purity standards, with comprehensive quality documentation and professional support for researchers across the UK and internationally. All products are clearly labeled for research use only, with same-day dispatch for orders placed before 1pm (Monday-Friday).
Future Research Directions and Emerging Applications
As the body of evidence surrounding Retatrutide Results continues to expand, researchers are identifying novel applications and investigation pathways that extend beyond initial metabolic research focus areas.
Combination Therapy Research
Emerging research interest focuses on retatrutide’s potential synergistic effects when combined with complementary compounds:
Promising Combination Approaches:
- Retatrutide + SGLT2 Inhibitors: Complementary mechanisms addressing glucose metabolism through distinct pathways
- Retatrutide + Metformin: Combined effects on hepatic glucose production and insulin sensitivity
- Retatrutide + Lipase Inhibitors: Dual approach to energy balance through intake and absorption modification
- Retatrutide + Exercise Mimetics: Enhanced metabolic benefits with preserved muscle mass
Preclinical investigations suggest additive or synergistic effects with several combination approaches, warranting clinical investigation in appropriately designed trials[18].
Neurological and Cognitive Research Applications
The GLP-1 receptor’s presence in central nervous system structures has prompted investigation into neuroprotective and cognitive effects. Early research suggests potential applications in:
- Neurodegenerative disease models (Alzheimer’s, Parkinson’s)
- Cognitive function preservation during metabolic stress
- Neuroinflammation reduction in metabolic dysfunction states
- Blood-brain barrier integrity maintenance
While preliminary, these findings position retatrutide as a compound of interest for neurometabolic research beyond traditional metabolic disease models.
Musculoskeletal Health Investigations
GIP receptor expression in bone tissue and preliminary data suggesting preserved bone density during weight loss have generated research interest in musculoskeletal applications:
- Bone mineral density maintenance during caloric restriction
- Muscle quality and functional capacity preservation
- Sarcopenia prevention in aging populations
- Recovery and regeneration following metabolic stress
These applications remain largely unexplored, representing opportunities for novel research contributions.
Personalized Medicine and Biomarker Research
Variability in individual responses to retatrutide treatment suggests potential for biomarker-guided research approaches:
Potential Predictive Biomarkers Under Investigation:
- Baseline GIP/GLP-1 receptor expression levels
- Genetic polymorphisms in receptor genes
- Gut microbiome composition patterns
- Baseline inflammatory marker profiles
- Metabolomic signatures predicting response magnitude
Identifying reliable predictive biomarkers could enable more targeted research designs and improve understanding of mechanisms underlying variable responses.
Long-Duration Studies and Maintenance Research
Current clinical data extends to 48-72 weeks, leaving questions regarding longer-duration outcomes:
- Weight maintenance following discontinuation
- Metabolic parameter durability after treatment cessation
- Optimal maintenance dosing strategies
- Long-term safety profiles extending beyond 2 years
- Potential for intermittent dosing protocols
Ongoing Phase 3 trials with extended observation periods will address many of these questions, providing critical data for long-term research planning[19].
Researchers exploring novel applications and combination approaches can access diverse peptide portfolios to support innovative experimental designs.
Practical Considerations for Research Implementation

Translating published Retatrutide Results into effective research protocols requires attention to practical implementation details that influence experimental outcomes and data quality.
Experimental Design Considerations
Robust research protocols incorporating retatrutide should address several key design elements:
Sample Size Calculations:
- Based on published effect sizes and variance estimates
- Adequate power (typically ≥80%) to detect meaningful differences
- Appropriate correction for multiple comparisons if applicable
- Consideration of dropout rates based on clinical trial data (10-15%)
Control Group Selection:
- Vehicle-treated controls for mechanistic studies
- Active comparator groups for comparative effectiveness research
- Dose-response designs to establish concentration-effect relationships
- Crossover designs where appropriate for within-subject comparisons
Outcome Measure Selection:
- Primary endpoints clearly defined a priori
- Validated assessment methods with established reliability
- Appropriate measurement frequency based on expected kinetics
- Inclusion of mechanistic endpoints beyond primary outcomes
Timeline and Protocol Development
Realistic timeline development ensures adequate observation periods to capture relevant outcomes:
Typical Research Timeline Components:
- Baseline Characterization: 1-2 weeks for comprehensive baseline measurements
- Dose Escalation Phase: 4-16 weeks depending on target dose and escalation schedule
- Maintenance Phase: Minimum 12-24 weeks to observe stable-state effects
- Washout/Recovery: 4-8 weeks post-treatment for durability assessment
- Data Analysis: 4-12 weeks for comprehensive statistical analysis and interpretation
Ethical Considerations and Oversight
All research involving retatrutide must operate under appropriate ethical oversight:
- Institutional Review Board approval for any human subjects research
- Institutional Animal Care and Use Committee approval for animal research
- Informed consent processes meeting regulatory and ethical standards
- Safety monitoring protocols with predefined stopping rules
- Data safety monitoring appropriate to research risk level
Data Collection and Management
Systematic data collection and management ensure research quality and reproducibility:
Essential Data Management Elements:
📋 Standardized data collection forms with validation rules
📋 Electronic data capture systems with audit trails
📋 Regular data quality checks and query resolution processes
📋 Secure data storage with appropriate backup procedures
📋 Clear data analysis plans specified before database lock
Publication and Dissemination Planning
Responsible research includes plans for appropriate dissemination of findings:
- Preregistration of protocols in appropriate registries (ClinicalTrials.gov, etc.)
- Publication in peer-reviewed journals regardless of outcome
- Transparent reporting following appropriate guidelines (CONSORT, ARRIVE, etc.)
- Data sharing consistent with ethical approvals and participant consent
- Presentation at scientific conferences to facilitate knowledge exchange
Research institutions can access professional support and guidance for protocol development and implementation planning.
Interpreting Retatrutide Results: Critical Analysis Framework
As researchers evaluate published data and plan investigations, applying a critical analysis framework ensures appropriate interpretation of Retatrutide Results and realistic expectation setting.
Assessing Study Quality and Validity
Not all research carries equal evidentiary weight. Critical evaluation should consider:
Internal Validity Factors:
- Randomization methods and allocation concealment
- Blinding procedures for participants, investigators, and outcome assessors
- Completeness of follow-up and intention-to-treat analysis
- Appropriate statistical methods and handling of missing data
- Conflict of interest disclosures and funding sources
External Validity Considerations:
- Population characteristics and generalizability
- Inclusion/exclusion criteria and resulting selection effects
- Treatment protocols and their applicability to research settings
- Outcome measures and their relevance to research questions
- Setting characteristics (academic centers, community sites, etc.)
Understanding Effect Sizes and Clinical Significance
Statistical significance does not automatically equate to meaningful effects. Researchers should evaluate:
- Absolute vs. Relative Effects: A 50% reduction from a small baseline value may be less meaningful than a 25% reduction from a large baseline value
- Number Needed to Treat: How many subjects must receive treatment to achieve one additional positive outcome?
- Confidence Intervals: What is the range of plausible effect sizes, not just the point estimate?
- Comparison to Established Benchmarks: How do effects compare to existing interventions or natural history?
Identifying Limitations and Potential Biases
Transparent acknowledgment of limitations strengthens research interpretation:
Common Limitations in Retatrutide Research:
- Limited long-term follow-up data beyond 72 weeks
- Predominantly industry-sponsored trials with inherent bias potential
- Selected populations may not represent broader research applications
- Surrogate endpoints may not perfectly predict ultimate outcomes of interest
- Publication bias favoring positive results over null findings
Translating Clinical Trial Data to Research Applications
Clinical trial results require thoughtful translation to research contexts:
- Controlled vs. Real-World Conditions: Research settings may differ substantially from controlled trial environments
- Compliance and Adherence: Supervised trial protocols ensure adherence levels potentially exceeding research settings
- Concomitant Interventions: Trials often include lifestyle interventions that may not be replicated in basic research
- Population Differences: Research models may differ from human trial populations in important ways
Future Evidence Needs
Identifying evidence gaps guides future research priorities:
Critical Evidence Gaps:
- Head-to-head comparisons with other triple or dual agonists
- Long-term safety data extending beyond 2-3 years
- Mechanistic studies elucidating specific pathway contributions
- Biomarker research enabling response prediction
- Optimal dosing strategies for specific research applications
- Combination therapy protocols and interaction effects
The Current State of Retatrutide Research
The accumulating body of evidence surrounding Retatrutide Results positions this triple receptor agonist as one of the most promising compounds in contemporary metabolic research. Clinical trial data demonstrates unprecedented weight loss outcomes, substantial metabolic improvements, and acceptable tolerability profiles across diverse study populations.
Key Research Findings:
✅ Superior Efficacy: Retatrutide produces weight loss exceeding 24% at 48 weeks with 12mg dosing, surpassing results from dual-agonist and single-agonist compounds
✅ Comprehensive Metabolic Benefits: Beyond weight reduction, retatrutide improves glycemic control, insulin sensitivity, lipid profiles, and hepatic function
✅ Favorable Body Composition: Approximately 85% of weight loss consists of fat mass, with preferential visceral adipose tissue reduction
✅ Manageable Safety Profile: Gastrointestinal adverse events represent the primary tolerability challenge, typically mild-moderate and transient
✅ Multiple Mechanism Integration: Triple receptor agonism produces synergistic effects through appetite regulation, energy expenditure enhancement, and direct metabolic tissue effects
For researchers and laboratories considering investigations involving retatrutide, several practical considerations merit attention:
- Quality sourcing from reputable suppliers providing comprehensive documentation and purity verification
- Appropriate handling and storage protocols to maintain peptide stability and biological activity
- Rigorous experimental design incorporating adequate controls, sample sizes, and outcome measures
- Ethical oversight ensuring compliance with institutional and regulatory requirements
- Critical interpretation of results within the context of existing evidence and recognized limitations
The research landscape surrounding retatrutide continues to evolve rapidly, with ongoing Phase 3 trials, long-term extension studies, and novel application investigations expanding the evidence base. Researchers entering this field have opportunities to contribute meaningful knowledge across diverse domains—from fundamental mechanistic understanding to clinical application optimization.
Next Steps for Researchers
For research institutions and laboratories interested in incorporating retatrutide into investigative protocols:
Immediate Actions:
- Review comprehensive literature including primary trial publications and mechanistic studies
- Develop detailed research protocols with clearly defined objectives, methods, and outcome measures
- Secure appropriate ethical approvals from institutional review boards or animal care committees
- Source high-quality research-grade peptides from established suppliers with documented purity standards
- Establish data collection and management systems supporting rigorous analysis and reproducibility
Long-Term Planning:
- Monitor emerging clinical trial results and mechanistic research publications
- Consider collaboration opportunities to enhance research scope and impact
- Plan for appropriate dissemination of findings through peer-reviewed publication
- Contribute to the growing evidence base informing optimal research applications
- Explore novel combination approaches and application domains
PEPTIDE PRO supports the research community with high-purity, research-grade peptides including retatrutide, comprehensive quality documentation, and professional service. With same-day dispatch for orders placed before 1pm Monday-Friday and fast UK delivery, researchers can access the compounds needed for cutting-edge metabolic investigations.
All products are strictly for research use only—not for human consumption or therapeutic applications.
For questions regarding research peptides, quality documentation, or protocol support, researchers can contact the PEPTIDE PRO team for professional assistance.
References
[1] Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine. 2023;389(6):514-526.
[2] Coskun T, et al. LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: From discovery to clinical proof of concept. Cell Metabolism. 2022;34(9):1234-1247.
[3] Samms RJ, et al. How May GIP Enhance the Therapeutic Efficacy of GLP-1? Trends in Endocrinology & Metabolism. 2020;31(6):410-421.
[4] Jastreboff AM, et al. Retatrutide Phase 2 Obesity Trial Investigators. Tirzepatide Once Weekly for the Treatment of Obesity. NEJM. 2023;389:514-526.
[5] Rosenstock J, et al. Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet. 2023;402(10401):529-544.
[6] Wilding JPH, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes, Obesity and Metabolism. 2022;24(8):1553-1564.
[7] Müller TD, et al. Glucagon-like peptide 1 (GLP-1). Molecular Metabolism. 2019;30:72-130.
[8] Neeland IJ, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes & Endocrinology. 2019;7(9):715-725.
[9] Heymsfield SB, et al. Mechanisms, Pathophysiology, and Management of Obesity. New England Journal of Medicine. 2017;376:254-266.
[10] Frias JP, et al. Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet. 2018;392(10160):2180-2193.
[11] Nauck MA, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Molecular Metabolism. 2021;46:101102.
[12] Thomas MK, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. Journal of Clinical Endocrinology & Metabolism. 2021;106(2):388-396.
[13] Gabery S, et al. Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight. 2020;5(6):e133429.
[14] Beiroa D, et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes. 2014;63(10):3346-3358.
[15] Newsome PN, et al. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. New England Journal of Medicine. 2021;384:1113-1124.
[16] Holst JJ, Rosenkilde MM. GIP as a Therapeutic Target in Diabetes and Obesity: Insight From Incretin Co-agonists. Journal of Clinical Endocrinology & Metabolism. 2020;105(8):e2710-e2716.
[17] Jensterle M, et al. Glucagon-like peptide 1 receptor agonists in obese women with polycystic ovary syndrome: to date and beyond. Diabetes, Obesity and Metabolism. 2022;24(Suppl 2):1-13.
[18] Müller TD, et al. Anti-obesity therapy: from rainbow pills to polyagonists. Pharmacological Reviews. 2018;70(4):712-746.
[19] Aroda VR, et al. SURMOUNT-1: Efficacy and safety of tirzepatide versus placebo in adults with obesity or overweight. Obesity Reviews. 2023;24(Suppl 1):e13537.
