Administering research peptides with precision requires understanding the science behind subcutaneous delivery. When working with tirzepatide in laboratory settings, selecting the best place to inject tirzepatide directly impacts absorption rates, comfort during administration, and overall experimental outcomes. This comprehensive guide examines the anatomical considerations, site-specific advantages, and evidence-based protocols that inform optimal injection site selection for research applications.
Research-grade peptides demand meticulous attention to administration protocols. Whether conducting metabolic studies, investigating glucose regulation mechanisms, or exploring weight management pathways, understanding injection site pharmacokinetics ensures consistent, reliable results. PEPTIDE PRO supplies high-purity tirzepatide formulations specifically designed for laboratory research, accompanied by comprehensive handling guidance to support rigorous scientific investigation.
Key Takeaways
- Abdomen remains the primary injection site for tirzepatide research due to optimal subcutaneous fat distribution and consistent absorption rates
- Site rotation protocols are essential to prevent tissue complications and maintain experimental consistency across longitudinal studies
- Proper injection technique including 45-90 degree needle insertion angle and 2-inch spacing from previous sites ensures reliable peptide delivery
- Thigh and upper arm alternatives provide viable options when abdominal sites require rest or when research protocols demand varied administration locations
- Subcutaneous administration at 4-8mm depth targets the hypodermis layer for optimal peptide absorption and bioavailability
Understanding Subcutaneous Injection Fundamentals for Tirzepatide Research
The Science of Subcutaneous Peptide Delivery
Subcutaneous injection delivers research peptides into the hypodermis—the fatty tissue layer beneath the dermis and above muscle tissue. This administration route proves ideal for tirzepatide research because the subcutaneous space contains extensive capillary networks that facilitate gradual peptide absorption into systemic circulation[1].
The subcutaneous layer typically extends 4-8 millimeters beneath the skin surface, though this varies based on anatomical location and individual tissue composition. For research applications, this depth provides several distinct advantages:
Optimal Absorption Kinetics: The rich vascular supply in subcutaneous tissue enables steady peptide uptake without the rapid peaks associated with intramuscular or intravenous administration. Research protocols examining sustained GLP-1 and GIP receptor agonism benefit from this controlled release profile[2].
Reduced Tissue Trauma: Compared to deeper injection routes, subcutaneous administration minimizes tissue disruption and inflammatory responses that could confound experimental results.
Accessibility and Reproducibility: Multiple body regions offer suitable subcutaneous tissue, enabling site rotation protocols essential for long-term research studies.
Why Injection Site Selection Matters in Research Protocols
The best place to inject tirzepatide varies based on specific research objectives, but understanding site-specific characteristics ensures protocol optimization. Different anatomical locations demonstrate measurable differences in:
- Absorption rate variability: Studies indicate absorption half-life can vary by 10-15% between injection sites due to differences in blood flow and tissue composition[3]
- Tissue thickness consistency: Abdominal subcutaneous tissue typically provides more uniform depth compared to extremities
- Experimental subject comfort: Minimizing discomfort during administration supports compliance in longitudinal research designs
- Lipohypertrophy risk: Repeated injections at identical sites can cause tissue changes that alter absorption characteristics
Research-grade tirzepatide formulations require consistent administration protocols. Site selection represents a critical variable that, when standardized, enhances data reliability and experimental reproducibility.
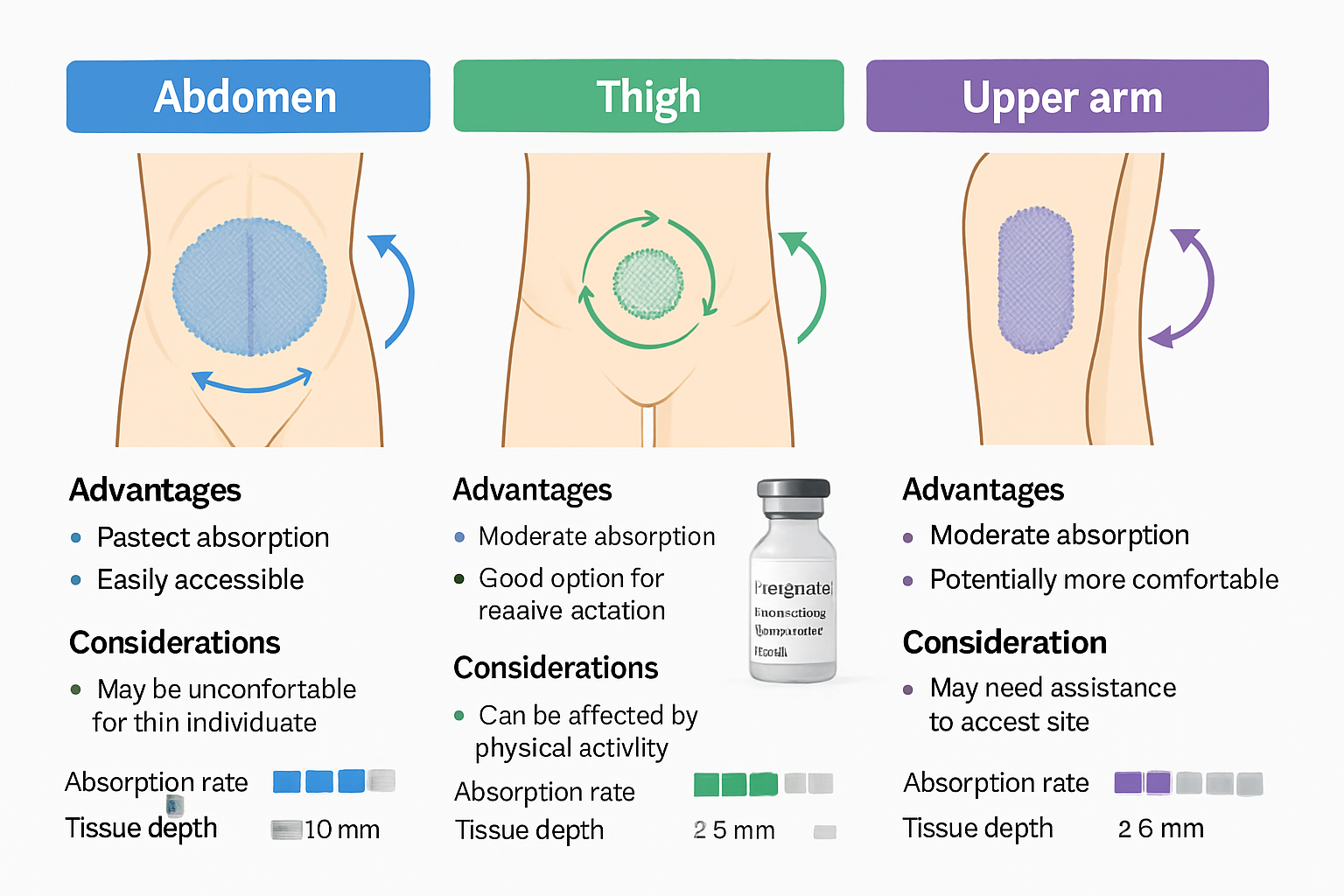
Best Place To Inject Tirzepatide: Primary Injection Sites Analyzed
Abdominal Region: The Gold Standard for Tirzepatide Administration
The abdomen consistently ranks as the best place to inject tirzepatide for research applications. This preference stems from multiple physiological and practical advantages that align with rigorous experimental protocols.
Anatomical Advantages of Abdominal Injection
The abdominal subcutaneous tissue offers superior characteristics for peptide research:
Abundant Subcutaneous Fat Distribution: The periumbilical region (around the navel) contains consistent subcutaneous tissue depth, typically 10-20mm in most research models. This abundance ensures reliable needle penetration to appropriate depth without risk of intramuscular injection[4].
Optimal Blood Flow Patterns: Abdominal tissue demonstrates moderate blood perfusion—neither too rapid (which could cause unpredictable absorption spikes) nor too slow (which might delay peptide uptake). This balanced vascular supply supports the extended-release profile characteristic of tirzepatide pharmacokinetics.
Large Surface Area for Rotation: The abdomen provides extensive territory for systematic site rotation. Researchers can establish grid patterns with multiple distinct injection points, essential for studies requiring frequent administration over extended periods.
Specific Abdominal Injection Zones
When identifying the best place to inject tirzepatide within the abdominal region, consider these optimal zones:
| Zone | Location | Advantages | Considerations |
|---|---|---|---|
| Upper Abdomen | 2-4 inches above navel | Good tissue depth, easy access | Avoid too close to ribcage |
| Lower Abdomen | 2-4 inches below navel | Excellent fat distribution | Avoid bladder region |
| Lateral Abdomen | 2-4 inches left/right of navel | Reduced sensitivity, good rotation | Ensure adequate tissue pinch |
| Oblique Regions | Diagonal quadrants from navel | Maximum rotation options | Avoid waistline pressure points |
Critical Exclusion Zones: Never inject within 2 inches of the navel itself, along the midline where tissue is thinner, or near any surgical scars that may have altered tissue architecture.
Thigh Region: Effective Alternative for Site Rotation
The anterior and lateral thigh represents the second-choice location for tirzepatide research administration, offering distinct advantages when abdominal sites require rest periods.
Optimal Thigh Injection Technique
Target Zone: The middle third of the anterior thigh (front) or lateral thigh (outer side) provides adequate subcutaneous tissue. Specifically, measure from the top of the thigh (where it meets the hip) to the knee, then identify the middle section.
Tissue Characteristics: Thigh subcutaneous tissue tends to be slightly firmer than abdominal tissue, with somewhat reduced fat layer thickness. This requires careful technique to ensure subcutaneous rather than intramuscular delivery.
Absorption Considerations: Research indicates thigh injections may demonstrate marginally slower absorption compared to abdominal sites—approximately 5-10% reduced uptake rate in comparative studies[5]. For protocols where this variation matters, researchers should maintain consistent site selection throughout the experimental period.
Advantages of Thigh Administration
- Accessibility: Easy to visualize and access during self-administration protocols
- Bilateral options: Both thighs provide independent rotation sites
- Reduced interference: Less affected by clothing pressure or movement compared to abdomen
- Suitable for lean tissue models: When abdominal subcutaneous tissue is limited, thighs often provide adequate alternatives
Upper Arm: Specialized Application Site
The posterior upper arm (back of the arm, specifically the triceps region) serves as a tertiary option for tirzepatide research administration.
Upper Arm Injection Protocols
Precise Location: The optimal zone spans the back of the upper arm, approximately halfway between the shoulder and elbow. This area requires adequate subcutaneous tissue—typically assessed through a pinch test that should yield at least 1-2 inches of tissue.
Technical Challenges: Upper arm injection presents unique considerations:
- Limited self-administration: Accessing this site without assistance proves difficult in many research models
- Reduced tissue depth: Generally contains less subcutaneous fat compared to abdomen or thighs
- Rotation limitations: Smaller surface area restricts the number of distinct injection points
Research Applications: Upper arm sites prove most valuable in protocols specifically investigating site-dependent absorption variations or when other sites are temporarily unavailable due to tissue reactions or protocol-specific restrictions.
High-purity research peptides like those supplied by PEPTIDE PRO include comprehensive administration guidance that addresses site-specific considerations for optimal experimental outcomes.
Injection Site Rotation: Essential Protocol for Long-Term Research
Why Site Rotation Prevents Experimental Confounds
Repeated injections at identical locations create tissue changes that compromise research integrity. Understanding and implementing systematic rotation protocols represents a critical component of rigorous tirzepatide research methodology.
Lipohypertrophy and Tissue Complications
Lipohypertrophy describes the abnormal accumulation of subcutaneous fat at frequently used injection sites. This condition develops through repeated tissue trauma and insulin-like growth factor stimulation, creating firm, raised areas with altered tissue architecture[6].
Research implications include:
- Altered absorption kinetics: Lipohypertrophic tissue demonstrates unpredictable peptide uptake, with studies showing 20-40% variation in absorption rates compared to normal tissue
- Experimental inconsistency: Data collected from compromised tissue sites introduces unwanted variability
- Tissue damage: Severe cases may require extended rest periods, disrupting longitudinal study timelines
Lipoatrophy, though less common with peptide research, represents the opposite condition—loss of subcutaneous fat creating depressed areas. Both conditions compromise the best place to inject tirzepatide by fundamentally altering the tissue environment.
Systematic Rotation Protocols
Implementing structured rotation schedules ensures each injection site receives adequate recovery time between administrations.
The Grid Method for Abdominal Rotation
Divide the abdominal region into a systematic grid:
- Establish quadrants: Mentally divide the abdomen into four quadrants using the navel as the central reference point
- Create injection points: Within each quadrant, identify 3-4 distinct injection sites, maintaining 2-inch minimum spacing from all previous sites
- Number the sequence: Assign numbers 1-16 (or however many sites you’ve identified) to create a rotation schedule
- Follow sequentially: Progress through numbered sites in order, returning to site #1 only after completing the full rotation
Recommended Spacing: Maintain at least 1 inch (preferably 2 inches) between any two injection sites. Never inject at the same location more frequently than once every 4 weeks in intensive research protocols.
Multi-Site Rotation Strategy
For research requiring frequent administration, incorporate multiple anatomical regions:
Week 1-2: Abdominal sites (right upper quadrant → left upper quadrant → right lower quadrant → left lower quadrant)
Week 3-4: Right thigh sites (anterior → lateral → medial aspects)
Week 5-6: Left thigh sites (anterior → lateral → medial aspects)
Week 7-8: Return to abdominal sites, using different specific points within each quadrant
This comprehensive approach provides 8-week cycles before returning to any specific injection point, minimizing tissue stress while maintaining experimental consistency.
Documentation and Site Tracking
Rigorous research demands meticulous record-keeping:
- Injection logs: Record date, time, specific anatomical location, and any observations for each administration
- Site maps: Create visual diagrams marking each injection point with corresponding dates
- Tissue assessment: Document any changes in tissue texture, appearance, or response at each site
- Rotation adherence: Track compliance with established rotation protocols to identify any deviations
Digital tools, spreadsheets, or specialized research software can streamline this documentation process, ensuring complete data capture throughout extended experimental periods.
Proper Injection Technique: Step-by-Step Protocol
Pre-Injection Preparation
Meticulous preparation ensures sterile, effective peptide administration while minimizing contamination risk and tissue trauma.
Essential Supplies Checklist
✅ Research-grade tirzepatide (properly stored and reconstituted according to manufacturer specifications) ✅ Sterile syringes with appropriate needle gauge (typically 29-31G) and length (4-8mm for subcutaneous delivery) ✅ Alcohol prep pads (70% isopropyl alcohol) ✅ Sterile gauze or cotton balls ✅ Sharps disposal container (never reuse or improperly dispose of needles) ✅ Research documentation materials (log sheets, site maps) ✅ Clean, well-lit workspace with stable surface
PEPTIDE PRO supplies include comprehensive handling instructions specific to each peptide formulation, ensuring researchers have access to manufacturer-recommended protocols.
Peptide Preparation
- Verify storage conditions: Confirm tirzepatide has been maintained at appropriate temperature (typically 2-8°C for reconstituted solutions)
- Inspect solution: Check for clarity, color consistency, and absence of particulates. Discard any solution showing cloudiness or discoloration
- Gentle mixing: If solution requires mixing, roll pen peptide gently between palms—never shake vigorously as this may denature peptide structures
- Accurate dosing: Draw prescribed research dose using aseptic technique, ensuring no air bubbles in syringe
- Temperature equilibration: Allow refrigerated solution to reach room temperature (15-20 minutes) before injection to reduce discomfort
Injection Administration Technique
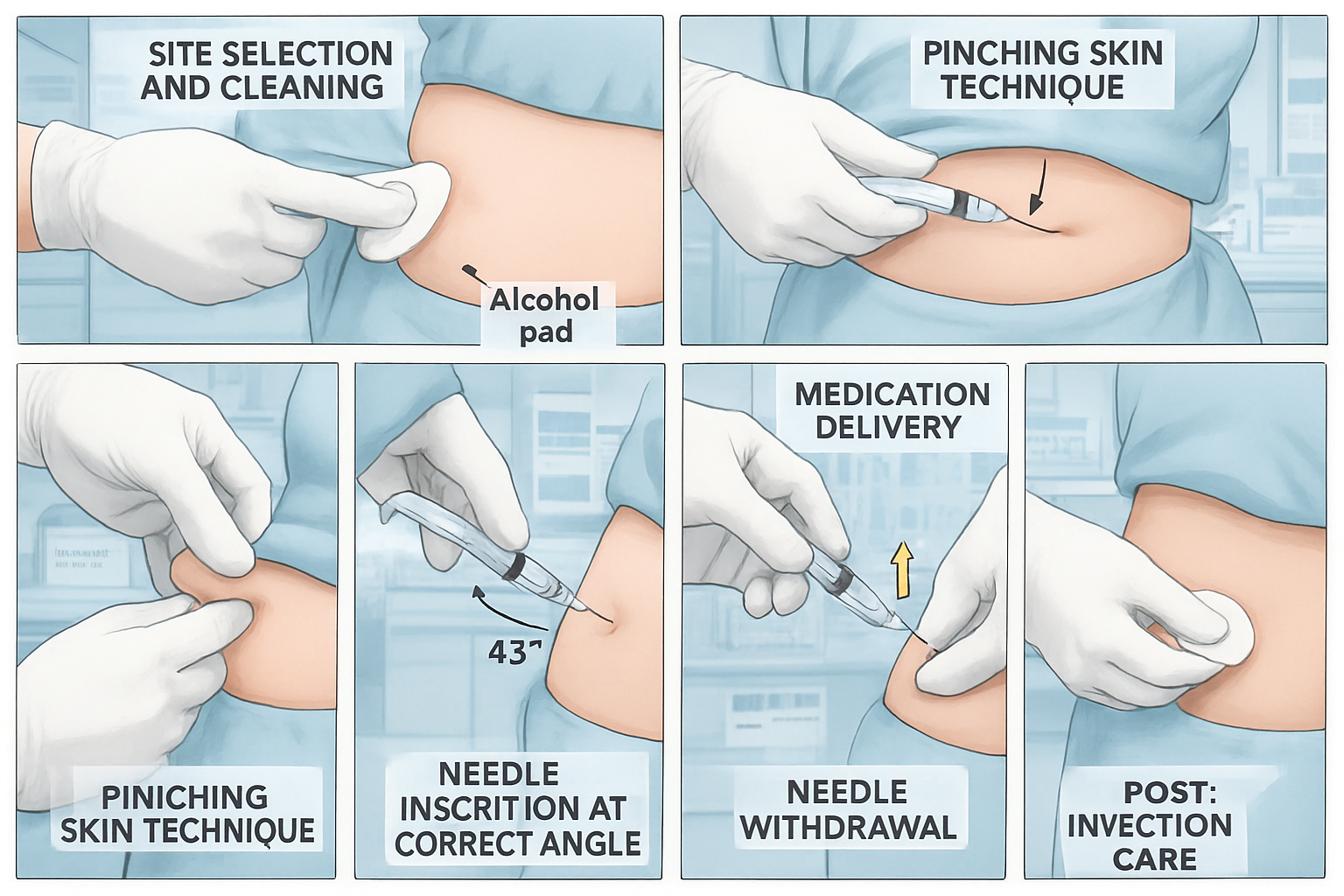
Site Preparation
Step 1: Site Selection – Choose the next location in your rotation protocol, ensuring it meets all spacing requirements and shows no signs of previous tissue reactions.
Step 2: Hygiene Protocol – Wash hands thoroughly with antibacterial soap for minimum 20 seconds. Consider using sterile gloves for enhanced contamination prevention.
Step 3: Site Cleaning – Using alcohol prep pad, clean injection site with firm circular motion, working outward from center point. Allow alcohol to air dry completely (minimum 30 seconds) before proceeding—this ensures maximum antimicrobial effect and prevents stinging.
The Injection Process
Step 4: Tissue Pinch – Using thumb and forefinger of non-dominant hand, gently pinch 1-2 inches of skin and subcutaneous tissue. This technique:
- Elevates subcutaneous layer away from underlying muscle
- Creates clear target for needle insertion
- Reduces risk of intramuscular injection
Step 5: Needle Insertion – Hold syringe like a pencil or dart with dominant hand. Insert needle at 45-90 degree angle depending on tissue thickness:
- 90 degrees: When 2+ inches of tissue can be pinched (typical for abdominal sites)
- 45 degrees: When less tissue available (may apply to lean thigh or arm sites)
Insert needle completely with smooth, swift motion—hesitation increases discomfort and tissue trauma.
Step 6: Aspiration Consideration – Current research protocols generally do not require aspiration (pulling back on plunger to check for blood) for subcutaneous injections, as major blood vessels rarely occupy subcutaneous space. However, individual research protocols may specify this step.
Step 7: Peptide Delivery – Depress plunger slowly and steadily, delivering peptide solution over 5-10 seconds. Rapid injection may cause tissue distension and discomfort.
Step 8: Needle Withdrawal – Wait 5-10 seconds after complete depression to allow tissue absorption, then withdraw needle at same angle as insertion. Release pinched skin.
Step 9: Post-Injection Care – Apply gentle pressure with sterile gauze if needed. Do NOT rub the injection site, as this may accelerate absorption beyond intended pharmacokinetic profile. A small amount of bleeding is normal; apply pressure until it stops.
Step 10: Disposal and Documentation – Immediately place used syringe in sharps container. Record injection details in research log including exact site location, time, dose, and any observations.
Troubleshooting Common Technical Issues
| Issue | Probable Cause | Solution |
|---|---|---|
| Resistance during injection | Needle in wrong tissue layer or clogged | Verify subcutaneous placement; replace needle if clogged |
| Solution leakage after withdrawal | Insufficient wait time before withdrawal | Extend post-injection wait to 10 seconds; apply pressure |
| Excessive discomfort | Cold solution, rapid injection, or improper technique | Warm solution to room temp; slow injection speed; review technique |
| Bruising at site | Capillary disruption | Normal minor occurrence; rotate to different area; apply gentle pressure |
| Tissue resistance to pinch | Lipohypertrophy or insufficient subcutaneous fat | Select alternative site; document tissue changes |
Factors Influencing Optimal Injection Site Selection
Individual Anatomical Variations
Research models demonstrate significant variability in subcutaneous tissue distribution, necessitating individualized site assessment rather than universal protocols.
Body Composition Considerations
Lean Tissue Models: Research subjects with reduced subcutaneous fat may require:
- Modified injection angles (45 degrees rather than 90 degrees)
- Shorter needle lengths (4-6mm rather than 8mm)
- Preferential use of abdominal sites where subcutaneous tissue concentrates
- Enhanced rotation protocols to prevent rapid site exhaustion
Higher Adiposity Models: Subjects with abundant subcutaneous tissue offer:
- Greater site availability and rotation options
- Reduced risk of inadvertent intramuscular injection
- Potential for standard 90-degree injection technique across all sites
- Consideration for longer needles if tissue depth exceeds standard needle length
Age-Related Tissue Changes
Research involving varied age groups should account for age-associated changes in skin elasticity, subcutaneous fat distribution, and tissue healing capacity. Older tissue models may demonstrate:
- Reduced tissue elasticity requiring gentler pinch technique
- Altered fat distribution patterns affecting optimal site selection
- Extended healing times necessitating longer rotation intervals
Research Protocol-Specific Considerations
The best place to inject tirzepatide may vary based on specific experimental objectives and study design parameters.
Frequency-Dependent Site Selection
Daily Administration Protocols: Studies requiring daily injections demand extensive rotation strategies incorporating all suitable anatomical regions. The abdominal grid method combined with bilateral thigh sites provides sufficient variety for sustainable long-term protocols.
Weekly Administration Protocols: Less frequent dosing permits more conservative rotation within preferred sites. Abdominal-only rotation may suffice for weekly schedules, though periodic site variation remains advisable.
Absorption Study Designs
Research specifically investigating site-dependent pharmacokinetic variations requires:
- Standardized administration technique across all sites
- Randomized site assignment to control for temporal variables
- Paired sampling protocols to isolate site-specific effects
- Statistical analysis accounting for within-subject variability
Environmental and Practical Factors
Temperature and Activity Considerations
Ambient Temperature: Warm environments increase peripheral blood flow, potentially accelerating subcutaneous absorption. Research conducted across varying temperature conditions should maintain consistent injection sites to minimize this confounding variable.
Physical Activity: Exercise increases blood flow to active muscle groups and adjacent tissues. Post-injection activity protocols should remain consistent, particularly when using thigh injection sites that may be affected by lower-body exercise.
Clothing and Pressure Considerations
Tight clothing or equipment creating sustained pressure over injection sites may affect absorption kinetics and tissue health. Research protocols should document:
- Typical clothing worn over injection sites
- Any equipment (belts, waistbands, straps) creating pressure points
- Modifications to eliminate pressure over active injection sites
Quality research-grade peptides from PEPTIDE PRO come with detailed guidance addressing these practical considerations, supporting researchers in developing comprehensive administration protocols.
Safety Considerations and Contraindications
Site-Specific Safety Protocols
Certain anatomical locations present elevated risks and should be avoided when determining the best place to inject tirzepatide.
Absolute Contraindications for Injection Sites
❌ Within 2 inches of the navel: Central abdominal tissue differs in structure and may have reduced subcutaneous depth
❌ Directly over bones: No adequate subcutaneous tissue; risk of periosteal injection causing significant discomfort
❌ Over joints: Movement-associated tissue stress increases complication risk
❌ Through moles, scars, or skin lesions: Altered tissue architecture affects absorption; infection risk increases
❌ Areas of active inflammation or infection: Risk of spreading infection; compromised absorption
❌ Sites of previous adverse reactions: Tissue sensitization may cause repeated or amplified reactions
❌ Regions with poor circulation: Compromised absorption and increased complication risk
❌ Areas of lipohypertrophy or lipoatrophy: Unpredictable absorption; further tissue damage risk
Recognizing and Managing Injection Site Reactions
Despite proper technique, injection site reactions occasionally occur in research settings. Systematic monitoring and appropriate response protocols ensure subject safety and data integrity.
Normal vs. Concerning Reactions
Expected Minor Reactions (generally resolve within 24-48 hours):
- Mild redness (erythema) at injection point
- Slight swelling or raised area (wheal)
- Minor tenderness to touch
- Small bruise (ecchymosis) from capillary disruption
- Temporary itching
Concerning Reactions requiring protocol modification or medical consultation:
- Persistent redness expanding beyond injection site
- Significant swelling (>2 inches diameter)
- Warmth and tenderness suggesting infection
- Drainage, pus, or abscess formation
- Severe pain disproportionate to injection
- Systemic symptoms (fever, malaise) following injection
- Allergic manifestations (hives, respiratory symptoms)
Response Protocols
For Minor Reactions:
- Document reaction characteristics (size, appearance, duration)
- Avoid that specific site for minimum 4 weeks
- Apply cold compress if swelling present (never heat, which increases absorption)
- Monitor for progression over 48-72 hours
- Continue research protocol using alternative sites
For Concerning Reactions:
- Immediately document all reaction details with photographic evidence if possible
- Discontinue injections at affected site indefinitely
- Consult medical oversight or veterinary support as appropriate to research model
- Consider protocol suspension pending reaction resolution
- Investigate potential causes (contamination, technique error, peptide degradation)
- Implement corrective measures before protocol resumption
Sterility and Contamination Prevention
Research-grade peptide administration demands rigorous aseptic technique to prevent contamination that could compromise both subject safety and experimental validity.
Multi-Dose Vial Protocols
When research protocols utilize multi-dose tirzepatide pen peptides:
Critical Sterility Practices:
- Always use new, sterile needle and syringe for each pen peptide access
- Never touch needle to any non-sterile surface
- Swab pen peptide septum with alcohol before each needle insertion
- Maintain refrigerated storage between uses
- Adhere strictly to manufacturer’s expiration dating after reconstitution
- Discard pen peptide if any contamination suspected (cloudiness, particles, discoloration)
Reconstitution Sterility: When reconstituting lyophilized peptides, use only bacteriostatic water or other manufacturer-specified diluents, maintaining aseptic technique throughout the process.
PEPTIDE PRO’s quality assurance protocols ensure all supplied peptides meet rigorous purity standards, but proper handling remains essential to maintain that quality throughout the research period.
Advanced Considerations for Tirzepatide Research Administration
Comparative Absorption Kinetics Across Injection Sites
Sophisticated research protocols may require understanding subtle pharmacokinetic differences between injection sites to optimize experimental design or interpret results accurately.
Site-Specific Absorption Data
Published research examining subcutaneous peptide delivery reveals measurable absorption variations:
Abdominal Sites:
- Baseline absorption reference (100% relative bioavailability)
- Time to peak concentration (Tmax): 8-72 hours for tirzepatide[7]
- Coefficient of variation: 15-20% (relatively consistent)
Thigh Sites:
- Relative bioavailability: 95-98% compared to abdomen
- Tmax: Potentially 10-15% longer than abdominal sites
- Coefficient of variation: 18-25% (slightly more variable)
Upper Arm Sites:
- Relative bioavailability: 90-95% compared to abdomen
- Tmax: Similar to abdominal sites but with greater individual variability
- Coefficient of variation: 20-30% (most variable)
These differences, while statistically significant in controlled studies, may have limited practical impact on many research protocols. However, studies examining precise pharmacokinetic parameters should maintain consistent injection sites throughout data collection periods.
Temperature Effects on Injection Comfort and Absorption
Solution temperature at time of injection influences both subject comfort and potentially absorption kinetics.
Cold Solution Effects:
- Increased discomfort during injection (vasoconstriction and temperature differential)
- Potential temporary reduction in local blood flow
- May slightly delay initial absorption phase
Room Temperature Solution Effects:
- Reduced injection discomfort
- Normal local blood flow maintained
- Optimal absorption kinetics
Warm Solution Effects (NOT recommended):
- Risk of peptide degradation if heated above manufacturer specifications
- Potential acceleration of absorption beyond intended profile
Best Practice: Remove tirzepatide from refrigeration 15-20 minutes before injection, allowing natural equilibration to room temperature without artificial heating.
Injection Depth Precision
While subcutaneous injection targets the hypodermis layer, precise depth control optimizes peptide delivery and minimizes complications.
Tissue Layer Anatomy
Epidermis (0-2mm): Outermost layer; too shallow for injection Dermis (2-4mm): Contains nerve endings and capillaries; intradermal injection causes significant discomfort Hypodermis/Subcutaneous (4-20mm+): Target layer for tirzepatide administration Muscle Fascia and Muscle: Below subcutaneous layer; intramuscular injection alters absorption profile
Needle Selection for Optimal Depth
| Subcutaneous Tissue Thickness | Recommended Needle Length | Injection Angle |
|---|---|---|
| <10mm (lean tissue) | 4-6mm | 45 degrees |
| 10-20mm (moderate tissue) | 6-8mm | 45-90 degrees |
| >20mm (abundant tissue) | 8-12mm | 90 degrees |
Needle Gauge Considerations:
- 29-31 gauge: Standard for peptide research; balances flow rate with minimal tissue trauma
- 27-28 gauge: Larger bore allows faster injection but increases tissue trauma
- 32+ gauge: Minimal trauma but may clog with viscous solutions; very slow injection
Special Populations and Adapted Protocols
Research involving specific populations may require modified injection site strategies.
Pediatric Research Models
Younger or smaller research subjects present unique considerations:
- Reduced subcutaneous tissue availability
- Proportionally smaller injection zones requiring closer attention to rotation
- Potential need for shorter needles (4-6mm)
- Enhanced sensitivity requiring meticulous technique
Geriatric Research Models
Older subjects may demonstrate:
- Altered subcutaneous fat distribution (often reduced in extremities, maintained or increased in abdomen)
- Reduced skin elasticity affecting pinch technique
- Thinner, more fragile skin requiring gentler handling
- Potential for delayed healing necessitating extended rotation intervals
Research Models with Mobility Limitations
Subjects with restricted mobility or dexterity may benefit from:
- Preferential use of easily accessible sites (anterior thigh, abdomen)
- Assistance protocols for hard-to-reach locations
- Adaptive equipment (injection aids, magnification devices)
- Simplified rotation schemes with clear visual guides
Optimizing Long-Term Research Protocols
Sustainability Strategies for Extended Studies
Longitudinal research spanning months or years requires thoughtful planning to maintain injection site integrity throughout the study period.
Tissue Preservation Protocols
Proactive Rotation: Begin comprehensive rotation from day one rather than waiting for tissue complications to develop. This preventive approach maintains optimal injection sites throughout the research timeline.
Site Rest Periods: Build periodic “rest weeks” into protocols where possible, allowing all tissue sites extended recovery time. For example, a 12-week study might include a mid-point week using alternative administration routes if scientifically appropriate.
Tissue Assessment Schedule: Implement regular systematic examination of all injection sites (weekly or bi-weekly), documenting any changes before they become problematic:
- Visual inspection for color changes, swelling, or texture alterations
- Palpation for firmness, lumps, or tenderness
- Comparison photographs to track subtle progressive changes
- Immediate intervention if any concerning findings emerge
Protocol Modifications Based on Tissue Response
Adaptive protocols that respond to individual tissue reactions optimize both subject welfare and data quality:
Escalating Rotation: If early tissue reactions appear, immediately expand rotation to include additional anatomical regions, reducing frequency at any single site.
Technique Refinement: Persistent reactions across multiple sites may indicate technical issues requiring:
- Review and retraining on injection technique
- Verification of needle length appropriateness
- Assessment of injection speed and pressure
- Evaluation of peptide storage and handling procedures
Temporary Protocol Suspension: Severe tissue reactions may necessitate brief protocol interruption, allowing complete healing before resumption. While potentially impacting study timelines, this approach prevents permanent tissue damage that could compromise the entire research project.
Documentation and Data Management
Comprehensive record-keeping transforms injection site management from routine procedure to valuable research data.
Essential Documentation Elements
Injection Log Components:
- Date and time of each administration
- Specific anatomical location (with reference to standardized site map)
- Dose administered
- Lot number of peptide used
- Needle gauge and length
- Any deviations from standard protocol
- Immediate observations (discomfort level, visible reactions)
- Administrator identity (if multiple personnel involved)
Follow-Up Assessment Records:
- 24-hour post-injection site evaluation
- 72-hour assessment for any delayed reactions
- Weekly comprehensive site review
- Photographic documentation of any concerning findings
Adverse Event Reporting: Establish clear criteria and procedures for documenting and reporting injection-related adverse events, ensuring appropriate oversight and intervention.
This systematic approach to the best place to inject tirzepatide not only optimizes individual injection outcomes but generates valuable methodological data that can inform future research protocols and contribute to the broader scientific understanding of peptide administration techniques.
Regulatory and Ethical Considerations for Research Use
Research-Only Designation and Compliance
All peptides supplied by PEPTIDE PRO, including tirzepatide formulations, are clearly labeled “For Research Use Only” and are not intended for human consumption or therapeutic use.
Institutional Oversight Requirements
Legitimate research involving peptide administration must operate under appropriate institutional oversight:
Institutional Review Boards (IRBs): Human research requires IRB approval with detailed protocols addressing:
- Scientific rationale and research objectives
- Participant selection criteria and informed consent procedures
- Detailed administration protocols including injection site selection
- Safety monitoring and adverse event management
- Data privacy and confidentiality protections
Institutional Animal Care and Use Committees (IACUCs): Animal research requires IACUC approval ensuring:
- Scientific justification for animal use
- Appropriate species and model selection
- Humane treatment standards and pain management
- Proper training of personnel performing injections
- Veterinary oversight and care protocols
Laboratory Safety Protocols: All peptide handling and administration must comply with:
- Institutional biosafety requirements
- Proper storage and handling of research materials
- Sharps safety and disposal regulations
- Personnel training and certification requirements
- Documentation and record-keeping standards
Ethical Administration Practices
Beyond regulatory compliance, ethical research demands commitment to best practices that prioritize subject welfare and scientific integrity.
Minimizing Discomfort: Selecting the best place to inject tirzepatide includes considering not only absorption efficiency but also subject comfort. Ethical protocols:
- Use appropriate needle gauges and lengths
- Allow solutions to reach room temperature
- Employ proper technique to minimize tissue trauma
- Provide adequate training to all personnel performing injections
- Monitor and respond to signs of discomfort or distress
Informed Decision-Making: Research involving human participants requires comprehensive informed consent addressing:
- Purpose and procedures of the research
- Potential risks including injection site reactions
- Alternative procedures if available
- Right to withdraw without penalty
- Confidentiality protections
Data Integrity: Accurate documentation of injection sites, techniques, and outcomes ensures:
- Reproducible methodology for future research
- Transparent reporting of all results including adverse events
- Scientific validity and peer review capability
- Ethical use of research subjects’ contributions
Conclusion: Implementing Evidence-Based Injection Protocols
Determining the best place to inject tirzepatide requires integrating anatomical knowledge, pharmacokinetic principles, practical technique, and individual research protocol requirements. The abdominal region consistently emerges as the optimal primary site due to abundant subcutaneous tissue, reliable absorption kinetics, and extensive rotation possibilities. However, comprehensive protocols incorporate thigh and upper arm alternatives, ensuring sustainable long-term administration without tissue compromise.
Key Implementation Principles
Systematic Rotation: Establish and adhere to structured rotation protocols from the first injection, preventing tissue complications that could compromise research integrity.
Meticulous Technique: Proper injection methodology—including site preparation, appropriate needle selection, correct insertion angle, and post-injection care—ensures consistent peptide delivery and minimizes adverse reactions.
Comprehensive Documentation: Detailed records of injection sites, techniques, and outcomes provide essential data for protocol optimization and contribute to the broader scientific understanding of peptide administration.
Adaptive Protocols: Monitor tissue response continuously and modify approaches as needed, balancing protocol consistency with subject welfare and site preservation.
Regulatory Compliance: Operate within appropriate institutional oversight frameworks, maintaining ethical standards and proper research-only use of peptides.
Next Steps for Researchers
Researchers planning tirzepatide studies should:
- Develop detailed administration protocols specifying injection sites, rotation schedules, and technique standards before initiating research
- Obtain appropriate institutional approvals through IRBs, IACUCs, or other relevant oversight bodies
- Source high-purity research-grade peptides from reputable suppliers like PEPTIDE PRO that provide comprehensive product information and handling guidance
- Train all personnel involved in peptide administration to ensure consistent, proper technique
- Establish monitoring and documentation systems to track injection sites and identify any complications early
- Plan for contingencies including alternative sites and response protocols for adverse reactions
Advancing Peptide Research Through Methodological Rigor
The quality of research outcomes depends not only on the purity of peptides used but on the precision of administration protocols. By implementing evidence-based approaches to injection site selection and technique, researchers ensure that their tirzepatide studies generate reliable, reproducible data that advances scientific understanding.
PEPTIDE PRO remains committed to supporting the research community with premium-quality peptides, comprehensive product information, and responsive customer support. For questions about specific peptide formulations, storage requirements, or administration protocols, the PEPTIDE PRO team provides expert guidance to support rigorous scientific investigation.
Ready to advance your research with high-purity tirzepatide? Explore PEPTIDE PRO’s extensive peptide catalogue featuring research-grade formulations with fast UK delivery and international shipping options. All products include detailed specifications, certificates of analysis, and comprehensive handling guidance to support your experimental protocols.
References
[1] Richter, W. F., et al. (2018). “Subcutaneous absorption of biotherapeutics: knowns and unknowns.” Drug Metabolism and Disposition, 46(11), 1663-1678.
[2] Frias, J. P., et al. (2021). “Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes.” New England Journal of Medicine, 385(6), 503-515.
[3] Gradel, A. K., et al. (2018). “Factors affecting the absorption of subcutaneously administered insulin: effect on variability.” Journal of Diabetes Research, 2018, 1-17.
[4] Gibney, M. A., et al. (2010). “Skin and subcutaneous adipose layer thickness in adults with diabetes at sites used for insulin injections: implications for needle length recommendations.” Current Medical Research and Opinion, 26(6), 1519-1530.
[5] Heise, T., et al. (2014). “Impact of injection speed and volume on perceived pain during subcutaneous injections into the abdomen and thigh: a single-centre, randomized controlled trial.” Diabetes, Obesity and Metabolism, 16(10), 971-976.
[6] Blanco, M., et al. (2013). “Prevalence and risk factors of lipohypertrophy in insulin-injecting patients with diabetes.” Diabetes & Metabolism, 39(5), 445-453.
[7] Urva, S., et al. (2021). “The novel dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 (GLP-1) receptor agonist tirzepatide transiently delays gastric emptying similarly to selective long-acting GLP-1 receptor agonists.” Diabetes Care, 44(10), 2239-2246.
