Imagine a single peptide compound capable of simultaneously influencing three critical appetite-regulating hormones—a breakthrough that represents one of the most significant advances in metabolic research in recent years. Retatrutide appetite hormones interaction is revolutionizing our understanding of how multi-receptor agonists can modulate hunger, satiety, and energy balance through coordinated hormonal pathways. This triple-action mechanism targeting GLP-1, GIP, and glucagon receptors simultaneously offers researchers unprecedented insights into the complex interplay between appetite regulation and metabolic function.
As laboratories worldwide investigate novel approaches to metabolic disorders, retatrutide has emerged as a compelling research compound demonstrating how strategic hormone receptor activation can produce synergistic effects far exceeding single-pathway interventions. The sophisticated interaction between retatrutide and appetite hormones provides a unique window into understanding how the body’s natural regulatory systems can be influenced through targeted peptide therapy.
Key Takeaways
- Triple Receptor Activation: Retatrutide uniquely engages GLP-1, GIP, and glucagon receptors simultaneously, creating coordinated appetite hormone modulation that differs fundamentally from single-agonist approaches
- Synergistic Hormonal Effects: The compound’s interaction with multiple appetite-regulating pathways produces amplified satiety signals and enhanced metabolic responses beyond what individual receptor activation achieves
- Research-Grade Applications: High-purity retatrutide formulations enable controlled laboratory investigations into appetite hormone mechanisms, receptor binding dynamics, and metabolic pathway interactions
- Complex Regulatory Networks: Understanding retatrutide’s influence on appetite hormones reveals intricate feedback loops involving gut peptides, pancreatic hormones, and central nervous system signaling
- Advanced Peptide Science: The development of triple agonists like retatrutide represents cutting-edge research in peptide pharmacology and hormone receptor biology
Understanding Appetite Hormones and Their Regulatory Functions
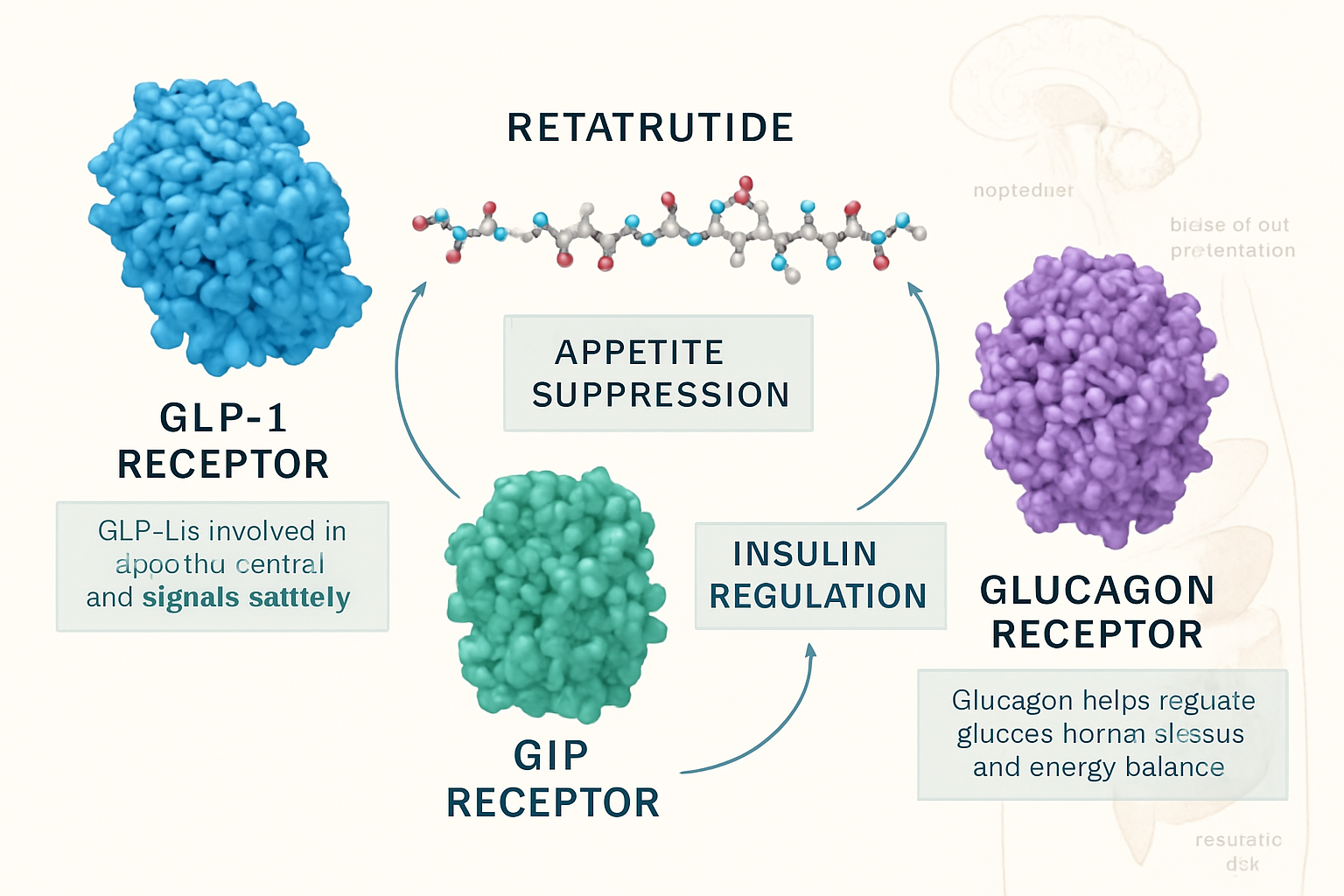
The human appetite regulation system operates through an extraordinarily complex network of hormonal signals, neural pathways, and metabolic feedback mechanisms. At the foundation of this system lie several key hormones that communicate between the gastrointestinal tract, pancreas, adipose tissue, and the central nervous system to coordinate hunger, satiety, and energy homeostasis.
The Primary Appetite-Regulating Hormones
Glucagon-Like Peptide-1 (GLP-1) stands as one of the most extensively studied incretin hormones, secreted primarily by L-cells in the intestinal epithelium in response to nutrient intake. This hormone exerts powerful effects on appetite suppression through multiple mechanisms:
- Direct action on hypothalamic appetite centers, particularly the arcuate nucleus
- Delayed gastric emptying, prolonging feelings of fullness
- Enhanced glucose-dependent insulin secretion
- Reduced glucagon release during hyperglycemic states
Glucose-Dependent Insulinotropic Polypeptide (GIP), formerly known as gastric inhibitory polypeptide, represents another crucial incretin hormone secreted by K-cells in the proximal small intestine. While historically recognized primarily for its insulinotropic effects, research has revealed GIP’s significant role in:
- Nutrient sensing and metabolic coordination
- Adipose tissue metabolism and fat storage regulation
- Potential central nervous system effects on feeding behavior
- Synergistic interaction with GLP-1 pathways
Glucagon itself, produced by pancreatic alpha cells, traditionally understood as a counter-regulatory hormone to insulin, plays a more nuanced role in appetite regulation than previously recognized:
- Promotion of hepatic glucose production during fasting states
- Stimulation of lipolysis and fat oxidation
- Potential appetite-suppressive effects through central mechanisms
- Energy expenditure enhancement through thermogenic pathways
Hormonal Integration and Feedback Systems
The appetite regulatory system doesn’t function through isolated hormonal signals but rather through integrated networks where multiple hormones interact, amplify, or modulate each other’s effects. This integration occurs at several levels:
Peripheral Integration: At the gut level, nutrient detection triggers coordinated release of multiple hormones. A mixed meal stimulates simultaneous secretion of GLP-1, GIP, cholecystokinin (CCK), and peptide YY (PYY), creating a hormonal “signature” that communicates meal composition and volume to the brain.
Central Processing: The hypothalamus, particularly the arcuate nucleus, serves as the primary integration center for appetite signals. Here, hormones interact with neuropeptide systems including:
- POMC/CART neurons (appetite-suppressing)
- NPY/AgRP neurons (appetite-stimulating)
- Leptin and insulin signaling pathways
- Ghrelin receptor activation
Metabolic Feedback: Blood glucose levels, fatty acid availability, and amino acid concentrations provide additional input that modulates hormonal sensitivity and receptor expression, creating dynamic adaptation to nutritional status.
Retatrutide’s Mechanism: Triple Receptor Agonism Explained
Retatrutide appetite hormones interaction represents a paradigm shift in peptide-based metabolic research. Unlike traditional single-target compounds, retatrutide functions as a triple agonist, simultaneously activating GLP-1, GIP, and glucagon receptors with carefully calibrated potency at each target.
Molecular Structure and Receptor Binding
Retatrutide’s molecular architecture has been specifically engineered to achieve balanced activation across three distinct receptor types—a remarkable feat of peptide design. The compound’s structure incorporates:
- Modified peptide backbone: Engineered for enhanced stability and prolonged half-life compared to native hormones
- Specific binding domains: Regions optimized for selective interaction with GLP-1R, GIPR, and GCGR
- Pharmacokinetic enhancements: Structural modifications that extend duration of action and improve bioavailability
- Receptor selectivity balance: Carefully calibrated to avoid excessive activation of any single pathway while maintaining therapeutic efficacy across all three
The binding affinity profile demonstrates sophisticated selectivity, with retatrutide showing robust activation of all three target receptors while minimizing off-target effects. This balanced agonism creates a unique pharmacological profile distinct from dual agonists or single-target compounds.
Coordinated Appetite Hormone Modulation
When retatrutide engages its three target receptors, it initiates a cascade of coordinated hormonal responses that collectively influence appetite regulation:
GLP-1 Receptor Activation produces the well-characterized incretin effects:
- Hypothalamic appetite suppression through POMC neuron activation
- Gastric motility reduction and delayed emptying
- Enhanced satiety signal transmission
- Reduced food-seeking behavior in research models
GIP Receptor Activation contributes complementary metabolic effects:
- Insulin secretion enhancement in glucose-dependent manner
- Potential adipose tissue remodeling effects
- Synergistic amplification of GLP-1 pathway effects
- Possible direct central nervous system appetite modulation
Glucagon Receptor Activation adds unique metabolic benefits:
- Increased energy expenditure through thermogenic activation
- Enhanced lipolysis and fat oxidation
- Hepatic glucose output modulation
- Potential appetite-suppressive effects through distinct neural pathways
Synergistic Effects Beyond Single-Pathway Activation
The most compelling aspect of retatrutide appetite hormones interaction lies in the synergistic effects produced by simultaneous multi-receptor activation. Research observations suggest that triple agonism creates outcomes that exceed the simple additive effects of individual pathway activation:
Amplified Satiety Signaling: When GLP-1 and GIP pathways activate simultaneously, they appear to create reinforcing signals that produce more robust and sustained appetite suppression than either alone. The addition of glucagon receptor activation may further enhance this effect through complementary neural circuits.
Enhanced Metabolic Efficiency: The combination of insulin secretion enhancement (GLP-1/GIP), glucagon’s lipolytic effects, and coordinated nutrient partitioning creates a metabolic environment optimized for energy utilization rather than storage.
Sustained Hormonal Response: Unlike native hormones with short half-lives, retatrutide’s extended pharmacokinetics maintain receptor activation over prolonged periods, potentially creating more stable appetite regulation without the peaks and troughs associated with meal-triggered hormone release.
Adaptive Receptor Dynamics: Preliminary research suggests that balanced multi-receptor activation may reduce compensatory upregulation or downregulation that sometimes limits single-agonist efficacy over time.
For researchers interested in investigating these mechanisms, high-purity retatrutide formulations provide the quality necessary for controlled laboratory studies examining receptor binding dynamics and hormonal pathway interactions.
The Science Behind Retatrutide and Appetite Hormone Pathways
Understanding how retatrutide appetite hormones interaction manifests at the molecular, cellular, and systemic levels requires examination of the specific pathways through which this triple agonist exerts its effects.
GLP-1 Pathway Activation and Appetite Suppression
The GLP-1 receptor pathway represents perhaps the most thoroughly characterized mechanism through which retatrutide influences appetite. When retatrutide binds to GLP-1 receptors, it initiates a signaling cascade that includes:
Intracellular Signaling:
- G-protein coupled receptor activation (Gs pathway)
- Cyclic AMP (cAMP) elevation
- Protein kinase A (PKA) activation
- Downstream transcription factor modulation
Central Nervous System Effects: The GLP-1 receptors in key brain regions respond to retatrutide activation with measurable changes in neural activity:
- Arcuate nucleus: Activation of POMC/CART neurons that produce appetite-suppressing neuropeptides
- Paraventricular nucleus: Enhanced satiety signal processing
- Nucleus tractus solitarius: Integration of peripheral satiety signals
- Area postrema: Direct sensing of circulating peptide levels
Peripheral Gastrointestinal Effects:
- Gastric smooth muscle relaxation reducing emptying rate
- Pyloric sphincter tone modulation
- Intestinal motility coordination
- Nutrient absorption timing optimization
GIP Pathway Contribution to Metabolic Regulation
While GIP’s role in appetite regulation has been historically less emphasized than GLP-1, emerging research reveals significant contributions to metabolic homeostasis and potentially to appetite control:
Pancreatic Effects:
- Beta cell stimulation and insulin secretion enhancement
- Glucose-dependent mechanism preventing hypoglycemia
- Potential beta cell preservation and proliferation signals
- Coordination with GLP-1 effects for amplified insulin response
Adipose Tissue Interactions: GIP receptors are abundantly expressed in adipose tissue, where activation influences:
- Lipid uptake and storage during fed states
- Adipokine secretion patterns
- Adipose tissue remodeling and browning potential
- Insulin sensitivity in fat cells
Central Appetite Effects: Recent research suggests GIP may exert direct central nervous system effects:
- Potential hypothalamic receptor activation
- Interaction with reward pathways
- Modulation of food preference and palatability responses
- Synergistic enhancement of GLP-1 satiety signals
Glucagon Receptor Activation and Energy Balance
The glucagon component of retatrutide’s triple mechanism adds a unique dimension focused on energy expenditure rather than solely appetite suppression:
Hepatic Metabolic Effects:
- Glycogenolysis stimulation during fasting states
- Gluconeogenesis pathway activation
- Ketone body production enhancement
- Hepatic fat oxidation promotion
Thermogenic Activation: Glucagon receptor activation influences energy expenditure through:
- Brown adipose tissue activation
- Uncoupling protein expression enhancement
- Metabolic rate elevation
- Heat production increase
Lipolytic Effects:
- Hormone-sensitive lipase activation in adipocytes
- Free fatty acid mobilization from stored triglycerides
- Substrate provision for oxidative metabolism
- Coordination with insulin effects for metabolic balance
Integrated Hormonal Response Patterns
When examining retatrutide appetite hormones interaction comprehensively, the integrated response pattern reveals sophisticated coordination:
| Hormone Pathway | Primary Effect | Appetite Impact | Metabolic Contribution | Timeframe |
|---|---|---|---|---|
| GLP-1 Activation | Satiety enhancement | Direct appetite suppression | Insulin secretion, gastric delay | Minutes to hours |
| GIP Activation | Metabolic coordination | Indirect through glucose regulation | Nutrient partitioning, insulin amplification | Minutes to hours |
| Glucagon Activation | Energy expenditure | Potential central suppression | Fat oxidation, thermogenesis | Hours to sustained |
| Combined Effect | Synergistic regulation | Amplified, sustained suppression | Optimized energy balance | Sustained over days |
This integrated pattern creates a hormonal environment fundamentally different from fasting, feeding, or single-hormone manipulation, representing a novel metabolic state that researchers are actively investigating.
Laboratories conducting research on these complex hormonal interactions can access research-grade peptide compounds specifically designed for controlled experimental protocols examining appetite regulation mechanisms.
Comparative Analysis: Retatrutide vs. Single and Dual Agonists
To fully appreciate the unique profile of retatrutide appetite hormones modulation, comparison with other peptide-based approaches provides valuable context.
Single Agonist Approaches
GLP-1 Receptor Agonists (e.g., liraglutide, semaglutide):
- Established mechanism focusing primarily on incretin pathway
- Well-characterized appetite suppression through central and peripheral mechanisms
- Proven efficacy but limited to single-pathway effects
- Potential for compensatory mechanisms over time
- Extensively studied safety and efficacy profiles
GIP Receptor Agonists:
- Less developed as standalone appetite modulators
- Primary focus on glucose regulation and insulin secretion
- Limited direct appetite suppression in isolation
- Potential metabolic benefits through adipose tissue effects
- Research ongoing regarding optimal applications
Glucagon Receptor Agonists:
- Primarily investigated for metabolic effects rather than appetite
- Energy expenditure enhancement as primary benefit
- Potential concerns regarding glucose elevation
- Limited appetite suppression as monotherapy
- Interesting thermogenic properties
Dual Agonist Developments
GLP-1/GIP Dual Agonists (e.g., tirzepatide):
- Synergistic combination of two incretin pathways
- Enhanced efficacy compared to GLP-1 alone in research models
- Improved metabolic outcomes beyond single agonists
- Balanced insulin/glucose regulation
- Represents significant advancement over monotherapy
GLP-1/Glucagon Dual Agonists:
- Combination of appetite suppression with energy expenditure
- Potential for enhanced fat loss through dual mechanisms
- Glucose effects require careful balance
- Less developed than GLP-1/GIP combinations
- Interesting metabolic profile in research settings
The Triple Agonist Advantage
Retatrutide’s triple mechanism theoretically offers several potential advantages over dual or single agonists:
Comprehensive Pathway Coverage: By engaging all three major metabolic hormone receptors simultaneously, retatrutide creates a more complete hormonal intervention that addresses:
- Appetite suppression (GLP-1 dominant)
- Metabolic optimization (GIP contribution)
- Energy expenditure (glucagon component)
- Synergistic amplification across pathways
Potential for Enhanced Efficacy: Research observations suggest that triple agonism may produce:
- Greater magnitude of appetite reduction
- More substantial metabolic improvements
- Enhanced fat oxidation and energy balance
- Potentially superior outcomes in metabolic research models
Balanced Hormonal Milieu: Rather than creating extreme activation of a single pathway, the balanced triple approach may:
- Reduce compensatory counter-regulation
- Minimize receptor desensitization
- Create more physiologically balanced metabolic state
- Potentially improve sustainability of effects
Mechanistic Redundancy: With three pathways contributing to similar outcomes, the approach offers:
- Continued efficacy if one pathway becomes less responsive
- Multiple mechanisms supporting appetite regulation
- Reduced dependence on single receptor sensitivity
- Potentially more robust long-term effects
Considerations and Research Questions
Despite theoretical advantages, important research questions remain regarding retatrutide appetite hormones modulation:
- Optimal Receptor Balance: What ratio of GLP-1:GIP:glucagon activation produces ideal outcomes?
- Individual Variability: How do differences in receptor expression affect response?
- Temporal Dynamics: Do the three pathways maintain synchronized effects over time?
- Safety Profile: Does multi-pathway activation create unique considerations?
- Mechanistic Interactions: Are there unexpected synergies or antagonisms between pathways?
These questions drive ongoing research in laboratories utilizing high-quality research peptides to investigate complex hormonal interactions and receptor pharmacology.
Research Applications and Laboratory Considerations
For researchers investigating retatrutide appetite hormones mechanisms, several methodological considerations and experimental approaches merit attention.
In Vitro Research Models
Receptor Binding Studies: Laboratory investigations of retatrutide’s receptor interactions employ several approaches:
- Competitive binding assays: Measuring retatrutide’s affinity for GLP-1R, GIPR, and GCGR using radiolabeled or fluorescent ligands
- Functional activation studies: Assessing cAMP production, calcium mobilization, or other second messenger responses
- Receptor selectivity profiling: Screening against related receptors to confirm specificity
- Structure-activity relationship studies: Examining how molecular modifications affect binding and activation
Cell Culture Systems: Various cell lines enable investigation of specific pathway components:
- CHO or HEK293 cells transfected with individual receptors for isolated pathway study
- Pancreatic beta cell lines (MIN6, INS-1) for insulin secretion research
- Adipocyte cultures for metabolic response investigation
- Neuronal cell lines for central mechanism exploration
Ex Vivo Tissue Studies
Pancreatic Islet Preparations: Isolated islets provide valuable models for studying:
- Glucose-stimulated insulin secretion in response to retatrutide
- Coordination between GLP-1 and GIP pathway activation
- Glucagon secretion modulation
- Islet cell viability and function over time
Hypothalamic Tissue Sections: Brain slice preparations enable investigation of:
- Neuronal activation patterns in appetite-regulating nuclei
- Electrophysiological responses to retatrutide exposure
- Neuropeptide expression changes
- Receptor localization and distribution
Gastrointestinal Tissue Studies: Intestinal preparations allow examination of:
- Effects on gastric smooth muscle contractility
- Enteroendocrine cell hormone secretion
- Gut motility pattern changes
- Local tissue responses to peptide exposure
In Vivo Research Models
Metabolic Phenotyping: Comprehensive assessment of retatrutide’s effects requires multiple measurement approaches:
- Food intake monitoring: Automated systems tracking meal patterns, frequency, and volume
- Body composition analysis: MRI, DEXA, or other methods assessing fat and lean mass changes
- Energy expenditure measurement: Indirect calorimetry in metabolic chambers
- Glucose homeostasis testing: Glucose tolerance tests, insulin sensitivity assessments
- Hormonal profiling: Serial measurements of insulin, glucagon, GLP-1, GIP, and other relevant hormones
Behavioral Assessments: Understanding appetite hormone effects extends beyond simple food intake:
- Meal microstructure analysis: Examining meal size, duration, and eating rate
- Food preference tests: Assessing macronutrient selection and palatability responses
- Motivated feeding paradigms: Investigating effort expended to obtain food
- Satiety assessment: Measuring duration of appetite suppression following meals
Neurobiological Investigations: Central mechanisms require specialized approaches:
- c-Fos immunohistochemistry: Mapping neuronal activation in appetite-regulating brain regions
- Neuropeptide expression analysis: Quantifying POMC, AgRP, NPY, and other relevant peptides
- Receptor autoradiography: Localizing GLP-1R, GIPR, and GCGR in brain tissue
- Electrophysiological recordings: Measuring neuronal firing rates in response to retatrutide
Quality Considerations for Research Applications
When conducting research on retatrutide appetite hormones interactions, peptide quality becomes paramount:
Purity Requirements:
- ≥98% purity for reliable, reproducible results
- Minimal contamination with related sequences or degradation products
- Verification through HPLC and mass spectrometry
- Batch-to-batch consistency for longitudinal studies
Storage and Handling:
- Lyophilized storage at -20°C or -80°C for long-term stability
- Reconstitution in appropriate sterile buffer (often bacteriostatic water or saline)
- Aliquoting to avoid freeze-thaw cycles
- Refrigerated storage (2-8°C) of reconstituted peptide with prompt use
Dosing Considerations: Research protocols must account for:
- Species-specific differences in receptor sensitivity
- Pharmacokinetic variations affecting optimal dosing frequency
- Route of administration effects on bioavailability
- Dose-response relationships for each receptor pathway
Researchers requiring high-purity retatrutide for controlled experimental protocols can access research-grade formulations specifically manufactured for laboratory applications with appropriate quality documentation.
Hormonal Feedback Loops and Regulatory Networks
The interaction between retatrutide appetite hormones and the body’s endogenous regulatory systems creates complex feedback dynamics that researchers must consider when designing experiments and interpreting results.
Homeostatic Compensation Mechanisms
Ghrelin Counter-Regulation: When appetite suppression occurs through GLP-1, GIP, and glucagon pathway activation, the body may respond with compensatory changes:
- Increased ghrelin secretion from gastric fundus
- Enhanced ghrelin receptor sensitivity in hypothalamus
- Upregulation of orexigenic neuropeptide systems
- Potential adaptation reducing long-term efficacy
Leptin Sensitivity Modulation: Retatrutide’s effects may interact with leptin signaling:
- Changes in adipose tissue mass affecting leptin levels
- Potential alterations in hypothalamic leptin receptor sensitivity
- Interaction between GLP-1 and leptin pathways in POMC neurons
- Synergistic or antagonistic effects on appetite regulation
Metabolic Rate Adaptation: Energy expenditure changes triggered by glucagon activation may face compensatory responses:
- Thyroid hormone axis adjustments
- Sympathetic nervous system activity modulation
- Mitochondrial efficiency adaptations
- Potential metabolic rate reduction over time
Receptor Dynamics and Desensitization
Receptor Expression Changes: Chronic activation of GLP-1R, GIPR, and GCGR may influence receptor levels:
- Downregulation in response to sustained agonism
- Internalization and recycling dynamics
- Transcriptional regulation of receptor genes
- Tissue-specific differences in adaptive responses
Signaling Pathway Modifications: Downstream signaling components may adapt to chronic stimulation:
- G-protein coupling efficiency changes
- Second messenger system sensitivity adjustments
- Phosphorylation state alterations affecting signal transduction
- Compensatory pathway activation
Cross-Talk Between Pathways: The three receptor systems don’t function in isolation:
- Shared intracellular signaling components
- Potential heterodimer formation between receptors
- Coordinated transcriptional responses
- Synergistic or antagonistic interactions at cellular level
Temporal Dynamics of Hormonal Responses
Understanding retatrutide appetite hormones effects requires consideration of time-dependent changes:
Acute Phase (Hours to Days):
- Initial receptor activation and signaling cascade initiation
- Immediate appetite suppression and metabolic shifts
- Maximal hormonal responses before adaptation begins
- Establishment of new homeostatic set points
Adaptation Phase (Days to Weeks):
- Compensatory mechanism activation
- Receptor expression and sensitivity adjustments
- Metabolic rate adaptations
- Potential efficacy plateau or reduction
Chronic Phase (Weeks to Months):
- Stabilization of new metabolic equilibrium
- Sustained vs. diminished effects on appetite
- Long-term receptor dynamics
- Potential for tolerance development
Integration with Other Metabolic Signals
Retatrutide’s effects occur within a complex metabolic environment including:
Nutrient Sensing Pathways:
- AMPK activation in response to energy status
- mTOR signaling reflecting amino acid availability
- Fatty acid sensing through GPR40 and other receptors
- Glucose sensing mechanisms in pancreas and brain
Circadian Rhythm Influences:
- Time-of-day variations in hormone sensitivity
- Circadian regulation of receptor expression
- Feeding-fasting cycle coordination
- Sleep-wake cycle interactions with metabolic hormones
Stress and Cortisol Effects:
- Glucocorticoid influences on appetite regulation
- Stress-induced eating behavior modulation
- HPA axis interactions with metabolic pathways
- Potential interference with retatrutide effects
Researchers investigating these complex regulatory networks benefit from comprehensive peptide resources that support controlled experimental designs examining multi-factorial hormonal interactions.
Clinical Research Insights and Translational Considerations
While this article focuses on research applications, understanding the translational context of retatrutide appetite hormones research provides valuable perspective for laboratory investigators.
Observed Effects in Research Models
Appetite and Food Intake: Research observations in appropriate models have documented:
- Significant reductions in food intake across multiple species
- Dose-dependent appetite suppression correlating with receptor occupancy
- Sustained effects over extended observation periods
- Changes in meal patterns including reduced meal size and frequency
Body Composition Changes: Metabolic research has revealed:
- Substantial reductions in total body weight in research subjects
- Preferential loss of adipose tissue vs. lean mass
- Improvements in body composition metrics
- Fat distribution changes with reduced visceral adiposity
Metabolic Parameters: Comprehensive metabolic assessments show:
- Improved glucose tolerance and insulin sensitivity
- Favorable lipid profile changes
- Enhanced markers of metabolic health
- Coordination between weight loss and metabolic improvements
Mechanistic Insights from Research Programs
Receptor Contribution Analysis: Studies attempting to dissect individual receptor contributions suggest:
- GLP-1 pathway appears dominant for appetite suppression
- GIP activation enhances metabolic efficiency
- Glucagon component contributes significantly to energy expenditure
- Synergistic effects exceed predictions from individual pathway studies
Dose-Response Relationships: Careful dose-ranging studies reveal:
- Non-linear relationships between dose and effect
- Potential threshold effects for certain outcomes
- Receptor-specific dose requirements for optimal activation
- Therapeutic window considerations for balanced triple agonism
Duration of Action: Pharmacokinetic and pharmacodynamic research demonstrates:
- Extended half-life enabling less frequent administration
- Sustained receptor occupancy over days
- Prolonged hormonal effects outlasting plasma concentrations
- Potential for depot formulations or modified-release approaches
Translational Challenges and Opportunities
Species Differences: Researchers must consider:
- Variations in receptor expression patterns across species
- Differences in metabolic regulation between rodents and primates
- Scaling factors for dose translation
- Species-specific compensatory mechanisms
Individual Variability: Research reveals substantial variation in responses:
- Genetic polymorphisms affecting receptor function
- Baseline metabolic status influencing efficacy
- Age, sex, and hormonal status effects
- Previous metabolic history impact on response
Long-Term Sustainability: Critical questions for ongoing research include:
- Maintenance of effects over extended periods
- Strategies to prevent tolerance development
- Optimal dosing regimens for sustained efficacy
- Combination approaches to enhance durability
Safety and Tolerability Research
Gastrointestinal Effects: Research models commonly show:
- Nausea and reduced appetite as expected on-target effects
- Delayed gastric emptying contributing to tolerability issues
- Dose-dependent GI side effects
- Adaptation over time with continued exposure
Metabolic Safety Signals: Comprehensive safety assessments monitor:
- Glucose regulation including hypoglycemia risk
- Pancreatic effects and enzyme elevations
- Cardiovascular parameters
- Hepatic and renal function markers
Long-Term Monitoring: Extended research protocols examine:
- Chronic exposure effects on organ systems
- Potential for receptor-mediated tissue changes
- Reversibility of effects upon discontinuation
- Durability of metabolic improvements
Laboratories conducting translational research on these aspects require access to consistently high-quality research peptides with full documentation supporting experimental protocols and regulatory compliance.
Future Directions in Retatrutide Appetite Hormone Research
The field of retatrutide appetite hormones investigation continues to evolve rapidly, with several promising research directions emerging.
Advanced Mechanistic Studies
Receptor Structural Biology: Cutting-edge research employing:
- Cryo-electron microscopy revealing receptor-ligand complex structures
- Molecular dynamics simulations predicting binding interactions
- Structure-guided design of next-generation agonists
- Understanding of receptor activation conformational changes
Single-Cell Transcriptomics: Modern genomic approaches enabling:
- Cell-type-specific responses to retatrutide in heterogeneous tissues
- Identification of novel responding cell populations
- Transcriptional signatures predicting efficacy
- Pathway analysis revealing unexpected mechanisms
Optogenetics and Chemogenetics: Neuroscience tools allowing:
- Precise activation or inhibition of specific neuronal populations
- Causal testing of hypothesized appetite circuits
- Temporal control of pathway activation
- Circuit mapping of retatrutide’s central effects
Optimization and Next-Generation Compounds
Receptor Balance Refinement: Research exploring:
- Optimal ratios of GLP-1:GIP:glucagon activation
- Context-dependent receptor balance requirements
- Personalized agonist profiles based on individual receptor expression
- Tissue-selective activation strategies
Pharmacokinetic Enhancements: Development efforts focused on:
- Extended half-life formulations for reduced dosing frequency
- Oral bioavailability improvements
- Targeted delivery to specific tissues
- Controlled-release systems for stable plasma levels
Combination Approaches: Investigating synergies with:
- Other metabolic peptides (amylin analogs, leptin, etc.)
- Small molecule metabolic modulators
- Lifestyle interventions in research models
- Sequential or concurrent multi-peptide protocols
Biomarker Development and Precision Research
Predictive Biomarkers: Identifying factors that predict response:
- Genetic variants in receptor genes or downstream pathways
- Baseline hormonal profiles indicating likely efficacy
- Metabolomic signatures correlating with outcomes
- Imaging biomarkers of target tissue engagement
Pharmacodynamic Markers: Developing measures of target engagement:
- Circulating biomarkers reflecting pathway activation
- Imaging approaches visualizing receptor occupancy
- Functional assessments of hormonal responses
- Real-time monitoring of metabolic effects
Response Monitoring: Creating tools for tracking efficacy:
- Continuous glucose monitoring integration
- Wearable technology for activity and metabolic rate
- Advanced body composition imaging
- Multi-omic profiling of treatment response
Expanding Research Applications
Metabolic Disease Models: Investigating retatrutide in:
- Obesity research models with varying etiologies
- Type 2 diabetes models examining glucose regulation
- Metabolic syndrome representations
- Fatty liver disease models
Neurobiological Research: Exploring central mechanisms:
- Reward pathway interactions and food reward processing
- Cognitive effects of appetite hormone modulation
- Mood and behavior correlations with metabolic changes
- Neuroprotective potential of incretin signaling
Aging and Longevity Research: Examining potential roles in:
- Metabolic optimization in aging models
- Healthspan extension through metabolic regulation
- Age-related metabolic dysfunction prevention
- Interaction with longevity pathways (AMPK, sirtuins, etc.)
Technological Innovations in Research Methods
Advanced Imaging Techniques:
- PET ligands for receptor occupancy visualization
- Functional MRI examining brain activation patterns
- Metabolic imaging tracking substrate utilization
- High-resolution tissue imaging revealing cellular effects
Computational Modeling:
- Systems biology approaches integrating multi-level data
- Predictive models of hormonal network dynamics
- Machine learning identifying response patterns
- Virtual screening for next-generation compounds
Organ-on-Chip Systems:
- Microfluidic platforms modeling multi-organ interactions
- Human tissue-based models reducing species translation issues
- High-throughput screening capabilities
- Personalized medicine approaches using patient-derived cells
Researchers pursuing these advanced investigations require access to premium research-grade peptides with rigorous quality control supporting sophisticated experimental protocols and cutting-edge methodologies.
Practical Considerations for Researchers Working with Retatrutide
For laboratories investigating retatrutide appetite hormones mechanisms, several practical aspects merit careful attention to ensure reliable, reproducible results.
Peptide Handling and Storage Best Practices
Receipt and Initial Storage: Upon receiving lyophilized retatrutide:
✅ Immediate Actions:
- Inspect packaging for temperature excursions during shipping
- Verify peptide quantity and purity documentation
- Store immediately at -20°C to -80°C in original sealed pen peptide
- Record receipt date and storage location in laboratory inventory
- Review Certificate of Analysis for purity, mass spec, and HPLC data
❄️ Long-Term Storage:
- Maintain lyophilized peptide at -20°C or colder
- Protect from light exposure using amber pen peptides or foil wrapping
- Minimize freeze-thaw cycles by never refreezing reconstituted aliquots
- Use desiccant in storage area to prevent moisture exposure
- Monitor freezer temperature with calibrated recording devices
Reconstitution Protocols: Proper reconstitution ensures peptide stability and activity:
- Allow temperature equilibration: Remove pen peptide from freezer and allow to reach room temperature before opening (prevents condensation)
- Use appropriate solvent: Bacteriostatic water, sterile saline, or specified buffer depending on experimental requirements
- Calculate concentration carefully: Determine target concentration based on experimental needs and dosing calculations
- Gentle mixing: Add solvent slowly down pen peptide wall, swirl gently—avoid vigorous shaking that may denature peptide
- Visual inspection: Ensure complete dissolution; solution should be clear without particulates
- Aliquot immediately: Divide into single-use aliquots to avoid repeated freeze-thaw
- Label comprehensively: Include peptide identity, concentration, reconstitution date, and expiration
Post-Reconstitution Handling:
- Store reconstituted aliquots at 2-8°C for short-term use (days to weeks depending on stability data)
- For longer storage, freeze aliquots at -20°C or -80°C (though this may reduce activity)
- Thaw frozen aliquots at 4°C, never at room temperature or with heat
- Use thawed aliquots within specified timeframe (typically 24-48 hours)
- Never refreeze previously thawed material
Experimental Design Considerations
Dose Selection: Choosing appropriate retatrutide doses requires consideration of:
- Receptor activation profiles: Different doses may preferentially activate different receptors
- Species scaling: Allometric scaling factors when translating between species
- Route of administration: Bioavailability varies significantly (subcutaneous, intraperitoneal, intravenous)
- Experimental endpoint: Acute vs. chronic studies may require different dosing strategies
- Literature review: Published research provides starting points for dose-ranging studies
Control Groups: Robust experimental designs include:
- Vehicle control: Identical administration of reconstitution buffer without peptide
- Positive controls: Established compounds (GLP-1 agonists) for comparison
- Pair-fed controls: Animals receiving same caloric intake as treated group to separate appetite effects from secondary metabolic changes
- Receptor-specific controls: Single or dual agonists to dissect pathway contributions
Timing Considerations:
- Circadian effects: Time of day influences hormonal sensitivity and metabolic responses
- Fed vs. fasted state: Nutrient status dramatically affects incretin responses
- Acute vs. chronic: Single-dose studies reveal immediate effects; chronic protocols show adaptations
- Washout periods: Sufficient time between doses in crossover designs to prevent carryover effects
Data Collection and Analysis
Appetite and Food Intake Measurements: Rigorous quantification requires:
- Automated feeding systems: Eliminate observer bias and enable continuous monitoring
- Meal pattern analysis: Beyond total intake, examine meal size, frequency, duration
- Food spillage accounting: Separate consumed food from wasted/scattered food
- Palatability controls: Ensure appetite changes aren’t due to taste aversion
Hormonal Measurements: Accurate hormone quantification demands:
- Appropriate sample collection: Plasma vs. serum, protease inhibitors, rapid processing
- Validated assays: Use established ELISA, RIA, or mass spec methods with known cross-reactivity
- Sample timing: Consider hormone half-lives and pulsatile secretion patterns
- Multiple timepoints: Single measurements may miss peak responses or temporal dynamics
Statistical Approaches: Proper analysis of retatrutide appetite hormones research data includes:
- Sufficient sample sizes: Power calculations based on expected effect sizes and variability
- Appropriate statistical tests: Account for repeated measures, multiple comparisons, non-normal distributions
- Outlier handling: Pre-specified criteria for data exclusion
- Reporting standards: Complete transparency regarding all measured outcomes, not just significant findings
Quality Assurance and Reproducibility
Documentation: Maintain comprehensive records including:
- Peptide batch numbers and purity certificates
- Exact reconstitution procedures and concentrations
- Environmental conditions during experiments
- Any deviations from planned protocols
- Complete raw data with metadata
Validation Experiments: Periodically verify:
- Peptide activity using standard bioassays
- Equipment calibration (scales, pipettes, analytical instruments)
- Consistency of results across batches
- Reproducibility by independent researchers
Collaboration and Transparency:
- Share detailed protocols with research community
- Deposit data in appropriate repositories
- Report negative and null results to prevent publication bias
- Engage in open scientific dialogue about methods and findings
Researchers committed to rigorous, reproducible science can partner with suppliers dedicated to quality and transparency who provide comprehensive documentation and support for research applications.
Regulatory and Ethical Considerations in Peptide Research
Conducting research on retatrutide appetite hormones requires adherence to established regulatory frameworks and ethical principles governing peptide research.
Research-Only Status and Compliance
Clear Labeling Requirements: All retatrutide materials for research must be:
- Clearly labeled “For Research Use Only”
- Never marketed or represented as suitable for human consumption
- Accompanied by documentation specifying research-only status
- Stored and handled separately from any approved substances
Regulatory Framework: Researchers must understand:
- Local and national regulations governing research peptides
- Institutional biosafety and chemical safety requirements
- Import/export regulations for international shipments
- Record-keeping requirements for controlled substances (if applicable)
Institutional Oversight: Appropriate research requires:
- Institutional Review Board (IRB) approval for any human-related research
- Institutional Animal Care and Use Committee (IACUC) approval for animal studies
- Biosafety committee review if applicable
- Environmental health and safety compliance
Ethical Research Practices
Animal Welfare Considerations: Research involving animals must adhere to:
- 3Rs Principles: Replacement (use alternatives when possible), Reduction (minimize animal numbers), Refinement (minimize suffering)
- Humane endpoints: Pre-specified criteria for early termination if distress occurs
- Appropriate housing and care: Environmental enrichment, social housing when appropriate, veterinary oversight
- Justification: Clear scientific rationale for animal use with expected knowledge gain
Scientific Integrity: Ethical research demands:
- Honest reporting of all results including negative findings
- Proper attribution and avoidance of plagiarism
- Transparent disclosure of conflicts of interest
- Responsible authorship practices
- Data sharing and reproducibility support
Responsible Communication: When discussing research findings:
- Avoid overstating implications or clinical relevance
- Clearly distinguish research findings from approved therapies
- Communicate limitations and uncertainties
- Prevent misinterpretation by non-scientific audiences
Supply Chain Responsibility
Vendor Selection: Choose peptide suppliers based on:
- Quality documentation: Comprehensive Certificates of Analysis with purity verification
- Regulatory compliance: Proper labeling and research-only positioning
- Transparency: Clear communication about manufacturing and testing processes
- Reputation: Established track record in research community
- Support: Technical assistance and customer service for research applications
Verification Practices: Responsible researchers should:
- Review CoA documentation for each batch
- Consider independent verification testing for critical experiments
- Report quality concerns to suppliers
- Maintain records of all peptide sources and batches used
Secure Handling: Institutional requirements typically include:
- Restricted access to research peptides
- Inventory tracking systems
- Proper disposal of expired or unused materials
- Prevention of diversion to non-research purposes
Researchers can ensure compliance by partnering with responsible suppliers who prioritize proper labeling, quality documentation, and adherence to research-only frameworks.
Conclusion: The Future of Appetite Hormone Research
The investigation of retatrutide appetite hormones represents a fascinating frontier in metabolic research, offering unprecedented insights into how coordinated multi-receptor activation can influence complex physiological systems. This triple agonist approach—simultaneously engaging GLP-1, GIP, and glucagon receptors—has revealed the sophisticated interplay between appetite regulation, energy homeostasis, and metabolic function.
Key Insights from Current Research
The research landscape surrounding retatrutide has illuminated several critical concepts:
Synergistic Hormonal Integration: The coordinated activation of three distinct receptor pathways creates effects that exceed simple additive predictions, demonstrating how the body’s regulatory systems function through integrated networks rather than isolated pathways.
Multi-Level Regulation: Appetite control operates simultaneously at peripheral (gastrointestinal, pancreatic) and central (hypothalamic, reward circuit) levels, with retatrutide’s triple mechanism engaging this multi-level system comprehensively.
Dynamic Adaptation: The body’s homeostatic mechanisms respond to sustained hormonal modulation through complex feedback loops, receptor dynamics, and compensatory adjustments—understanding these adaptations remains crucial for optimizing research protocols.
Translational Complexity: While research models provide valuable mechanistic insights, species differences, individual variability, and context-dependent effects require careful consideration when interpreting findings and designing experiments.
Advancing the Research Agenda
Future investigations into retatrutide appetite hormones will benefit from:
🔬 Advanced Technologies: Employing cutting-edge tools including single-cell genomics, advanced imaging, computational modeling, and organ-on-chip systems to dissect mechanisms with unprecedented resolution.
🧬 Precision Approaches: Developing biomarkers, genetic profiling, and personalized strategies that account for individual variability in receptor expression and hormonal responses.
🔄 Integrative Frameworks: Moving beyond reductionist single-pathway studies toward systems biology approaches that capture the full complexity of metabolic regulation.
📊 Rigorous Methodology: Maintaining highest standards for experimental design, statistical analysis, reproducibility, and transparent reporting to advance the field reliably.
Practical Next Steps for Researchers
For laboratories planning to investigate retatrutide’s effects on appetite hormones:
1. Establish Robust Protocols
- Develop standardized operating procedures for peptide handling, storage, and reconstitution
- Design comprehensive experimental protocols with appropriate controls and statistical power
- Implement quality assurance measures ensuring reproducibility
2. Secure High-Quality Materials
- Source research-grade retatrutide from reputable suppliers with comprehensive quality documentation
- Verify purity and identity through certificates of analysis
- Maintain detailed records of all materials used in experiments
3. Build Collaborative Networks
- Engage with other researchers investigating appetite hormones and metabolic peptides
- Share protocols, findings, and methodological insights
- Participate in scientific discourse advancing the field collectively
4. Maintain Ethical Standards
- Ensure all research complies with institutional and regulatory requirements
- Adhere to highest standards of animal welfare and scientific integrity
- Communicate findings responsibly and transparently
Accessing Research-Grade Retatrutide
Researchers ready to begin investigations into retatrutide appetite hormones mechanisms require access to high-purity, well-characterized peptide materials. PEPTIDE PRO provides research-grade retatrutide and related compounds specifically manufactured for laboratory applications, with:
- Exceptional Purity: Research-grade peptides produced under strict quality conditions with comprehensive testing
- Fast Delivery: Same-day dispatch for orders placed before 1pm (Mon-Fri) with reliable UK and international shipping
- Complete Documentation: Full Certificates of Analysis, storage guidance, and technical support
- Research-Only Commitment: Clear labeling and positioning strictly for research applications
The investigation of appetite hormone mechanisms through compounds like retatrutide continues to reveal the remarkable complexity and elegance of metabolic regulation. As research techniques advance and our understanding deepens, the insights gained will undoubtedly contribute to fundamental knowledge about how the body maintains energy balance and regulates feeding behavior.
For researchers committed to advancing this important field, maintaining rigorous scientific standards, utilizing high-quality materials, and approaching these complex systems with appropriate sophistication will ensure continued progress in understanding the intricate dance of retatrutide appetite hormones and metabolic regulation.
