Insulin resistance stands as one of the most significant metabolic challenges facing modern healthcare and research communities. As scientists explore novel therapeutic approaches, tirzepatide insulin resistance research has emerged as a particularly promising area of investigation. This dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist represents a sophisticated advancement in peptide-based metabolic research, offering researchers unprecedented opportunities to examine mechanisms underlying glucose homeostasis and insulin sensitivity at the cellular level.
Understanding how tirzepatide influences insulin resistance pathways requires examining its unique dual-agonist properties, molecular mechanisms, and the broader implications for metabolic research. For laboratories and research institutions seeking high-purity research peptides, the quality and consistency of compounds like tirzepatide directly impact experimental outcomes and data reliability.
Key Takeaways
- Tirzepatide functions as a dual GIP/GLP-1 receptor agonist, offering unique mechanisms for investigating insulin resistance pathways in research models
- Insulin resistance research with tirzepatide reveals significant improvements in glucose metabolism, insulin sensitivity markers, and cellular energy utilization
- Research-grade tirzepatide quality is essential for reproducible results, requiring proper storage, handling, and reconstitution protocols
- Molecular mechanisms include enhanced insulin receptor signaling, improved beta-cell function, and reduced hepatic glucose production
- Laboratory applications span from cellular studies to metabolic pathway analysis, requiring strict adherence to research-only protocols
Understanding Insulin Resistance: The Metabolic Foundation
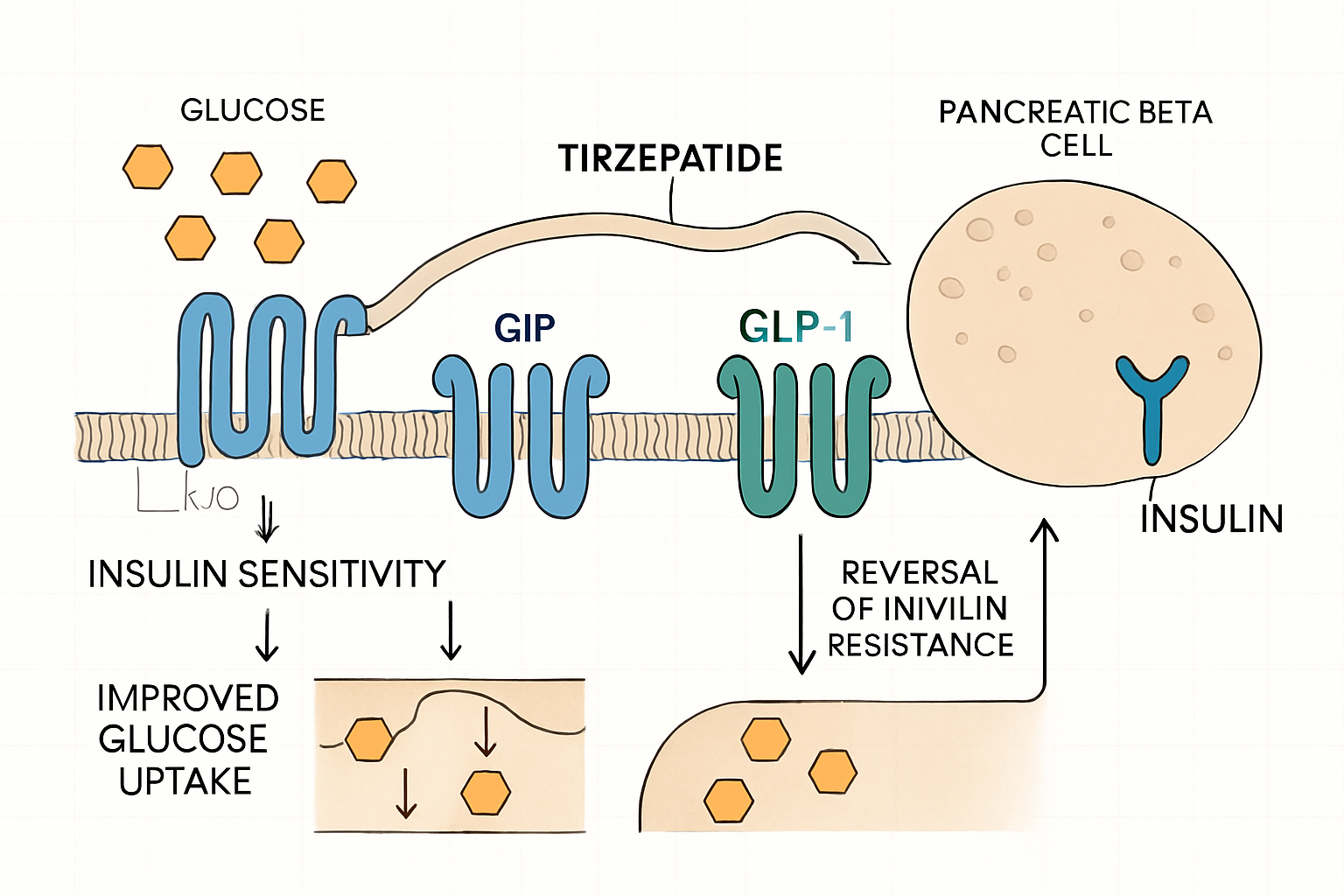
Insulin resistance represents a pathophysiological condition where cells throughout the body demonstrate diminished responsiveness to insulin signaling. This metabolic dysfunction affects multiple organ systems, including skeletal muscle, adipose tissue, and hepatic cells, creating a cascade of metabolic disturbances that researchers continue to investigate.
The Cellular Mechanisms of Insulin Resistance
At the molecular level, insulin resistance involves complex alterations in insulin receptor signaling pathways. When insulin binds to its receptor on cell surfaces, it typically initiates a phosphorylation cascade involving insulin receptor substrate (IRS) proteins, phosphoinositide 3-kinase (PI3K), and protein kinase B (Akt). In insulin-resistant states, this signaling cascade becomes impaired through various mechanisms:
- Post-receptor defects affecting IRS protein phosphorylation
- Inflammatory cytokine interference with insulin signaling
- Lipid accumulation in non-adipose tissues (lipotoxicity)
- Mitochondrial dysfunction reducing cellular energy efficiency
- Oxidative stress damaging cellular insulin machinery
Research models examining these mechanisms require precise experimental conditions and high-quality research compounds to generate reliable data.
Metabolic Consequences and Research Implications
The downstream effects of insulin resistance extend far beyond simple glucose elevation. Researchers investigating tirzepatide insulin resistance interactions observe multiple metabolic alterations:
Glucose Metabolism Disruption: Impaired glucose uptake in muscle and adipose tissue leads to sustained hyperglycemia, while hepatic glucose production remains inappropriately elevated despite high circulating insulin levels.
Lipid Metabolism Alterations: Insulin resistance promotes increased lipolysis in adipose tissue, elevating free fatty acid concentrations that further exacerbate insulin resistance through lipotoxic mechanisms.
Inflammatory Pathway Activation: Chronic low-grade inflammation characterizes insulin-resistant states, with elevated inflammatory markers including C-reactive protein, interleukin-6, and tumor necrosis factor-alpha.
Understanding these foundational mechanisms provides essential context for examining how tirzepatide influences insulin resistance pathways in controlled research settings.
Tirzepatide: Molecular Structure and Dual Receptor Mechanism
Tirzepatide represents a sophisticated peptide sequence engineered to simultaneously activate both GIP and GLP-1 receptors. This dual-agonist approach distinguishes it from single-receptor agonists and provides researchers with a unique tool for investigating incretin-based metabolic regulation.
Peptide Structure and Receptor Binding
The molecular architecture of tirzepatide consists of a 39-amino acid sequence based on the native GIP structure, with specific modifications enhancing GLP-1 receptor affinity and pharmacokinetic properties. Key structural features include:
- C20 fatty diacid moiety attached via a linker, enabling albumin binding and extended half-life
- Modified amino acid residues optimizing dual receptor activation
- Structural stability suitable for research applications when properly stored
- Specific binding domains for both GIP and GLP-1 receptors
For laboratories conducting peptide research, understanding these structural characteristics informs proper handling, storage, and experimental design protocols.
GIP and GLP-1 Receptor Activation
The dual mechanism of tirzepatide insulin resistance research involves understanding how both receptor systems contribute to metabolic regulation:
GIP Receptor Activation 🔬:
- Primarily expressed in pancreatic beta cells, adipocytes, and bone
- Stimulates insulin secretion in glucose-dependent manner
- Influences lipid metabolism and energy storage
- May enhance insulin sensitivity through adipocyte-mediated mechanisms
- Contributes to bone metabolism regulation
GLP-1 Receptor Activation 🧬:
- Widely distributed across pancreas, brain, heart, and gastrointestinal tract
- Potentiates glucose-dependent insulin secretion
- Suppresses inappropriate glucagon release
- Slows gastric emptying in physiological models
- Influences satiety signaling pathways in neural tissue
The synergistic effects of simultaneous GIP and GLP-1 receptor activation create unique research opportunities for examining insulin resistance mechanisms that single-agonist compounds cannot replicate.
Pharmacokinetic Considerations for Research Applications
Research-grade tirzepatide demonstrates specific pharmacokinetic properties relevant to experimental design:
| Parameter | Characteristics | Research Implications |
|---|---|---|
| Half-life | Extended (approximately 5 days in physiological models) | Allows for less frequent dosing in chronic studies |
| Bioavailability | High when properly reconstituted | Requires careful preparation protocols |
| Protein Binding | Extensive albumin binding | Influences tissue distribution patterns |
| Metabolism | Proteolytic degradation | Minimal metabolite interference in assays |
| Elimination | Renal and proteolytic pathways | Considerations for chronic exposure studies |
Researchers utilizing high-purity tirzepatide must account for these properties when designing experimental protocols and interpreting results.
Tirzepatide Insulin Resistance Research: Mechanisms of Action
The investigation of tirzepatide insulin resistance interactions reveals multiple molecular pathways through which this dual agonist influences glucose homeostasis and insulin sensitivity. Understanding these mechanisms provides researchers with insights into potential therapeutic targets and metabolic regulation strategies.
Enhanced Insulin Secretion and Beta-Cell Function
Tirzepatide’s glucose-dependent insulinotropic effects represent a primary mechanism influencing insulin resistance markers in research models. The compound enhances pancreatic beta-cell function through several pathways:
Direct Beta-Cell Stimulation: Both GIP and GLP-1 receptor activation on pancreatic beta cells trigger cyclic AMP (cAMP) elevation, activating protein kinase A (PKA) and exchange protein directly activated by cAMP (Epac2). These signaling cascades enhance insulin granule exocytosis in a glucose-dependent manner, preventing hypoglycemia while improving insulin availability.
Beta-Cell Preservation: Research suggests GLP-1 receptor activation may promote beta-cell survival and proliferation through anti-apoptotic pathways. Studies examining tirzepatide insulin resistance relationships observe improved beta-cell mass markers and reduced cellular stress indicators in experimental models.
Proinsulin Processing: Enhanced proinsulin-to-insulin conversion efficiency has been documented in research examining incretin effects, potentially improving insulin quality and receptor binding characteristics.
Improved Peripheral Insulin Sensitivity
Beyond insulin secretion, tirzepatide influences insulin sensitivity in peripheral tissues through multiple mechanisms:
Skeletal Muscle Glucose Uptake 💪:
- Enhanced GLUT4 translocation to cell membranes
- Improved insulin receptor substrate phosphorylation
- Increased glycogen synthesis capacity
- Enhanced mitochondrial function and oxidative metabolism
- Reduced intramyocellular lipid accumulation
Adipose Tissue Metabolism 🔄:
- Improved insulin-mediated suppression of lipolysis
- Enhanced adipocyte differentiation and healthy expansion
- Reduced inflammatory cytokine production
- Improved adiponectin secretion (insulin-sensitizing adipokine)
- Decreased ectopic lipid deposition
Hepatic Insulin Sensitivity 🏥:
- Suppressed hepatic glucose production
- Reduced gluconeogenesis enzyme expression
- Improved hepatic insulin signaling
- Decreased hepatic steatosis markers
- Enhanced glycogen storage capacity
These peripheral effects contribute significantly to the overall improvements in insulin resistance observed in tirzepatide research models.
Glucagon Regulation and Glucose Homeostasis
The GLP-1 receptor component of tirzepatide provides robust glucagon suppression, addressing the inappropriate hyperglucagonemia often observed in insulin-resistant states:
- Alpha-cell GLP-1 receptor activation directly suppresses glucagon secretion
- Paracrine insulin effects from enhanced beta-cell function further inhibit alpha cells
- Glucose-dependent mechanism prevents excessive glucagon suppression during hypoglycemia
- Reduced hepatic glucose output from combined insulin enhancement and glucagon suppression
This dual regulation of both insulin and glucagon creates a more physiologically balanced approach to glucose homeostasis in research models.
Inflammatory Pathway Modulation
Emerging research on tirzepatide insulin resistance mechanisms reveals significant anti-inflammatory properties:
Systemic Inflammation Reduction:
- Decreased circulating inflammatory cytokines (IL-6, TNF-α, CRP)
- Reduced macrophage infiltration in metabolic tissues
- Improved endothelial function and vascular inflammation markers
- Enhanced anti-inflammatory adipokine production
Cellular Stress Reduction:
- Decreased endoplasmic reticulum stress markers
- Reduced oxidative stress and reactive oxygen species production
- Improved mitochondrial function and biogenesis
- Enhanced cellular autophagy and quality control mechanisms
These anti-inflammatory effects may contribute substantially to improved insulin sensitivity independent of weight or glucose changes in research models.
Research Applications and Experimental Protocols
Investigating tirzepatide insulin resistance relationships requires rigorous experimental design, appropriate controls, and high-quality research materials. Laboratories conducting this research must implement comprehensive protocols ensuring data reliability and reproducibility.
In Vitro Research Models
Cellular research examining tirzepatide effects on insulin resistance utilizes various model systems:
Primary Cell Cultures:
- Isolated pancreatic islets for insulin secretion studies
- Primary hepatocytes for glucose production and insulin signaling analysis
- Primary adipocytes for lipolysis and insulin sensitivity measurements
- Primary myocytes for glucose uptake and glycogen synthesis studies
Established Cell Lines:
- INS-1 or MIN6 beta-cell lines for mechanistic insulin secretion research
- 3T3-L1 adipocytes for differentiation and insulin sensitivity studies
- HepG2 hepatocytes for glucose metabolism pathway analysis
- C2C12 myotubes for skeletal muscle insulin signaling research
Experimental Considerations:
- Dose-response curves establishing optimal tirzepatide concentrations
- Time-course studies examining acute versus chronic effects
- Receptor antagonist co-treatment to distinguish GIP versus GLP-1 contributions
- Glucose concentration variations to assess glucose-dependent effects
In Vivo Research Models
Animal research models provide comprehensive systems for examining tirzepatide insulin resistance interactions:
Dietary-Induced Models:
- High-fat diet protocols inducing insulin resistance
- High-fat high-sucrose combinations for metabolic syndrome models
- Western diet formulations replicating complex nutritional patterns
Genetic Models:
- Leptin-deficient models with severe insulin resistance
- Leptin receptor-deficient models for obesity research
- Tissue-specific insulin receptor knockout models
- GIP or GLP-1 receptor knockout models for mechanism studies
Measurement Parameters:
- Glucose tolerance tests (GTT) assessing glucose handling
- Insulin tolerance tests (ITT) measuring insulin sensitivity
- Hyperinsulinemic-euglycemic clamp studies (gold standard)
- HOMA-IR calculations from fasting glucose and insulin
- Tissue-specific glucose uptake using radioactive tracers
Quality Considerations for Research Peptides
The reliability of tirzepatide insulin resistance research depends critically on peptide quality. Researchers should prioritize:
Purity Standards ✨:
- Minimum 98% purity for consistent results
- HPLC verification of peptide identity
- Mass spectrometry confirmation of molecular weight
- Certificates of Analysis (COA) documentation
Storage and Handling:
- Lyophilized storage at -20°C or below
- Protection from light and moisture
- Proper reconstitution with appropriate buffers
- Aliquoting to minimize freeze-thaw cycles
Reconstitution Protocols:
- Sterile bacteriostatic water or appropriate buffer selection
- Gentle mixing avoiding foam formation
- Concentration calculations for accurate dosing
- Stability considerations post-reconstitution
Laboratories can access research-grade peptides with comprehensive quality documentation to ensure experimental consistency.
Analytical Methods and Biomarker Assessment
Comprehensive tirzepatide insulin resistance research incorporates multiple analytical approaches:
Glucose Metabolism Markers:
- Fasting glucose and insulin concentrations
- HbA1c for long-term glycemic assessment
- Glucose area under the curve (AUC) during tolerance tests
- C-peptide measurements for endogenous insulin production
Insulin Sensitivity Indices:
- HOMA-IR: [Fasting Insulin (μU/mL) × Fasting Glucose (mg/dL)] / 405
- QUICKI: 1 / [log(Fasting Insulin) + log(Fasting Glucose)]
- Matsuda Index from oral glucose tolerance test data
- Clamp-derived glucose disposal rates
Lipid Metabolism Markers:
- Triglycerides and free fatty acid concentrations
- LDL, HDL, and total cholesterol profiles
- Apolipoprotein measurements
- Tissue lipid content analysis
Inflammatory and Hormonal Markers:
- Adiponectin and leptin concentrations
- IL-6, TNF-α, and CRP measurements
- Glucagon levels during fasting and postprandial states
- GIP and GLP-1 concentrations (active and total)
Comparative Research: Tirzepatide Versus Other Metabolic Peptides
Understanding tirzepatide insulin resistance effects benefits from comparative analysis with other research peptides targeting metabolic pathways. This context helps researchers select appropriate compounds for specific experimental questions.
Single-Receptor Agonists Comparison
GLP-1 Receptor Agonists:
Research compounds like semaglutide and liraglutide provide selective GLP-1 receptor activation, offering comparison points for tirzepatide’s dual mechanism:
- Similar insulin secretion enhancement
- Comparable glucagon suppression
- Potentially less pronounced effects on insulin sensitivity
- Different weight trajectory patterns in research models
- Distinct receptor expression requirement profiles
GIP Receptor Agonists:
Pure GIP agonists reveal the specific contributions of this receptor system:
- Robust insulin secretion in non-resistant states
- Unique adipocyte metabolism effects
- Potentially attenuated effects in established insulin resistance
- Different inflammatory profile compared to GLP-1 agonists
- Bone metabolism influences not seen with GLP-1 alone
Synergistic Mechanisms of Dual Agonism
The tirzepatide insulin resistance research reveals synergistic benefits beyond simple additive effects:
Enhanced Metabolic Improvements:
- Greater insulin sensitivity enhancement than single agonists
- More substantial improvements in lipid metabolism markers
- Superior beta-cell function preservation in chronic studies
- Enhanced anti-inflammatory effects across multiple tissues
Complementary Receptor Distribution:
- GIP receptors predominant in adipose tissue and bone
- GLP-1 receptors more abundant in pancreas and brain
- Combined activation reaches broader tissue spectrum
- Synergistic effects on overlapping tissues (pancreatic islets)
Other Metabolic Research Peptides
Several additional peptides offer distinct mechanisms for insulin resistance research:
Growth Hormone Secretagogues:
Compounds like ipamorelin and CJC-1295 influence metabolism through growth hormone pathways:
- Indirect effects on insulin sensitivity through lipolysis
- Body composition changes affecting metabolic parameters
- Different receptor systems (ghrelin receptors)
- Complementary research applications examining GH-insulin interactions
Metabolic Modulators:
Peptides such as AOD9604 target specific metabolic pathways:
- Lipolytic effects without insulin secretion changes
- Targeted fat metabolism research applications
- Distinct mechanisms for comparative metabolism studies
Tissue Repair Peptides:
Compounds like BPC-157 and TB-500 offer indirect metabolic influences:
- Anti-inflammatory properties affecting insulin signaling
- Tissue healing potentially improving metabolic function
- Research applications examining inflammation-metabolism connections
This comparative framework helps researchers design comprehensive studies examining multiple aspects of insulin resistance pathology.
Experimental Design Considerations for Tirzepatide Research
Rigorous experimental design ensures reliable, reproducible results when investigating tirzepatide insulin resistance relationships. Researchers must address multiple methodological considerations to generate high-quality data.
Dose Selection and Optimization
Determining appropriate tirzepatide concentrations requires systematic dose-response analysis:
In Vitro Dosing:
- Typical concentration ranges: 0.1 nM to 1000 nM
- Physiologically relevant concentrations: 1-100 nM
- Receptor saturation curves informing optimal doses
- Time-dependent effects requiring chronic exposure studies
In Vivo Dosing:
- Weight-based calculations (μg/kg or mg/kg)
- Route of administration considerations (subcutaneous, intraperitoneal)
- Dosing frequency based on pharmacokinetic properties
- Escalation protocols for chronic studies
Dose-Response Relationships:
| Dose Range | Typical Effects | Research Applications |
|---|---|---|
| Low (0.1-1 nM in vitro) | Receptor binding, minimal functional effects | Receptor affinity studies, mechanism investigation |
| Medium (1-100 nM in vitro) | Physiological insulin secretion, signaling activation | Standard metabolic studies, pathway analysis |
| High (100-1000 nM in vitro) | Maximal receptor activation, potential non-specific effects | Receptor saturation studies, maximum effect determination |
Control Groups and Experimental Variables
Comprehensive tirzepatide insulin resistance research requires appropriate controls:
Essential Control Groups:
- Vehicle-treated controls (same reconstitution buffer)
- Insulin-sensitive baseline comparisons
- Single-receptor agonist controls (GLP-1 or GIP alone)
- Receptor antagonist co-treatment groups
- Time-matched untreated controls
Variable Standardization:
- Consistent glucose concentrations during in vitro studies
- Standardized diet compositions for in vivo models
- Age and sex matching across experimental groups
- Environmental condition control (temperature, light cycles)
- Consistent timing of measurements relative to dosing
Timeline and Duration Considerations
Tirzepatide effects on insulin resistance manifest across different timeframes:
Acute Effects (Minutes to Hours):
- Immediate insulin secretion responses
- Rapid signaling pathway activation
- Short-term glucose uptake changes
- Immediate post-receptor signaling events
Subacute Effects (Days to Weeks):
- Gene expression changes
- Protein expression alterations
- Metabolic adaptation patterns
- Inflammatory marker shifts
Chronic Effects (Weeks to Months):
- Sustained insulin sensitivity improvements
- Body composition changes
- Beta-cell mass alterations
- Long-term metabolic reprogramming
Research protocols should align duration with specific mechanistic questions being addressed.
Data Collection and Statistical Considerations
Robust statistical approaches ensure reliable conclusions:
Sample Size Calculations:
- Power analysis determining minimum group sizes
- Effect size estimates from pilot studies or literature
- Accounting for expected variability in insulin resistance models
- Adequate replication for statistical significance
Statistical Methods:
- Repeated measures ANOVA for time-course studies
- Two-way ANOVA for multiple factor analysis
- Post-hoc testing with appropriate corrections
- Non-parametric alternatives for non-normal distributions
Data Presentation:
- Mean ± SEM or SD with clear indication
- Individual data points when appropriate
- Area under curve calculations for tolerance tests
- Percent change from baseline normalization
Safety, Handling, and Regulatory Considerations
Research involving tirzepatide insulin resistance studies requires strict adherence to safety protocols, proper handling procedures, and regulatory compliance. These considerations protect researchers, ensure data quality, and maintain ethical research standards.
Laboratory Safety Protocols
Working with research peptides demands comprehensive safety measures:
Personal Protective Equipment (PPE) 🥽:
- Laboratory coats preventing skin contact
- Nitrile gloves (double-gloving for concentrated solutions)
- Safety glasses or face shields during reconstitution
- Respiratory protection if aerosolization risk exists
Handling Procedures:
- Work in designated research areas only
- Use of biological safety cabinets when appropriate
- Proper sharps disposal for needles and syringes
- Spill kits readily available with cleanup protocols
- Clear labeling of all solutions and storage containers
Waste Disposal:
- Peptide solutions treated as biohazardous waste
- Proper container labeling and segregation
- Compliance with institutional waste management protocols
- Documentation of disposal procedures
Storage and Stability Guidelines
Maintaining tirzepatide quality throughout research projects requires optimal storage:
Lyophilized Peptide Storage:
- Temperature: -20°C to -80°C for long-term storage
- Desiccated conditions preventing moisture exposure
- Light protection using amber pen peptides or foil wrapping
- Original sealed containers until ready for use
- Stability: Typically 2-3 years when properly stored
Reconstituted Solution Storage:
- Refrigeration at 2-8°C for short-term use
- Sterile conditions during preparation and storage
- Aliquoting to minimize freeze-thaw cycles
- Use within manufacturer-recommended timeframes
- Freezing at -20°C for extended storage (with stability verification)
Quality Monitoring:
- Visual inspection for precipitation or discoloration
- pH verification of reconstituted solutions
- Periodic purity re-testing for long-term studies
- Documentation of storage conditions and durations
Researchers can obtain detailed storage guidance from reputable suppliers to ensure peptide integrity throughout experiments.
Research-Only Compliance
Tirzepatide and similar compounds are strictly designated for research applications:
Regulatory Framework ⚖️:
- Clearly labeled “For Research Use Only”
- Not approved for human consumption or clinical use
- Compliance with institutional review board requirements
- Adherence to national and international research regulations
Documentation Requirements:
- Detailed experimental protocols
- Institutional approval documentation
- Material safety data sheets (MSDS)
- Chain of custody records for controlled substances
- Research outcome documentation
Ethical Considerations:
- Animal research ethics committee approval
- Adherence to 3Rs principles (Replace, Reduce, Refine)
- Humane endpoints and welfare monitoring
- Transparent reporting of all experimental outcomes
Quality Assurance and Supplier Selection
Research reliability depends on peptide source quality:
Supplier Evaluation Criteria:
- Documented purity standards (≥98% preferred)
- Comprehensive Certificates of Analysis (COA)
- HPLC and mass spectrometry verification
- Proper storage and shipping conditions
- Responsive technical support
- Transparent sourcing and synthesis methods
Verification Upon Receipt:
- COA review confirming batch purity
- Visual inspection of packaging and product
- Weight verification against stated amount
- Proper labeling with lot numbers and expiration
- Storage condition documentation
Selecting reputable research peptide suppliers ensures experimental consistency and data reliability across studies.
Future Directions in Tirzepatide Insulin Resistance Research
The field of tirzepatide insulin resistance investigation continues evolving, with emerging research questions and technological advances opening new avenues for scientific exploration.
Mechanistic Questions and Knowledge Gaps
Several fundamental questions remain regarding tirzepatide’s effects on insulin resistance:
Tissue-Specific Mechanisms:
- Relative contributions of muscle, liver, and adipose improvements
- Cell-type specific receptor expression requirements
- Tissue crosstalk and endocrine signaling pathways
- Organ-specific transcriptional changes
Temporal Dynamics:
- Sequence of metabolic improvements during treatment
- Reversibility of insulin resistance improvements upon cessation
- Long-term adaptation and tolerance development
- Optimal treatment duration for sustained benefits
Individual Variability:
- Genetic factors influencing response magnitude
- Baseline metabolic status affecting outcomes
- Sex differences in mechanism and magnitude
- Age-related response variations
Emerging Research Technologies
Advanced methodologies enable deeper investigation of tirzepatide insulin resistance mechanisms:
Omics Approaches 🧬:
- Transcriptomics: RNA sequencing revealing gene expression changes across tissues
- Proteomics: Mass spectrometry identifying protein expression and modification patterns
- Metabolomics: Comprehensive metabolite profiling showing pathway flux changes
- Lipidomics: Detailed lipid species analysis revealing membrane and signaling alterations
Advanced Imaging:
- Positron emission tomography (PET) tracking tissue-specific glucose uptake
- Magnetic resonance spectroscopy (MRS) measuring tissue lipid content
- Fluorescence imaging visualizing receptor activation in real-time
- Multi-photon microscopy examining cellular dynamics
Single-Cell Analysis:
- Single-cell RNA sequencing identifying cell-type specific responses
- Flow cytometry characterizing immune cell populations
- Spatial transcriptomics mapping tissue architecture changes
- Single-cell metabolic flux analysis
Combination Research Strategies
Investigating tirzepatide alongside other compounds reveals synergistic mechanisms:
Metabolic Peptide Combinations:
- Tirzepatide plus growth hormone secretagogues
- Combined incretin and metabolic modulator approaches
- Multi-target peptide cocktails for comprehensive metabolic regulation
Adjunctive Compound Research:
- Insulin sensitizers (metformin analogs) with tirzepatide
- Anti-inflammatory agents addressing complementary pathways
- Mitochondrial function enhancers
- Antioxidant compounds reducing oxidative stress
Lifestyle Intervention Interactions:
- Exercise training effects on tirzepatide response
- Dietary composition influences on outcomes
- Circadian rhythm optimization strategies
- Stress reduction interventions
Translational Research Opportunities
Bridging basic tirzepatide insulin resistance research to clinical applications:
Biomarker Development:
- Predictive markers identifying optimal responders
- Early response indicators guiding treatment decisions
- Mechanism-specific biomarkers distinguishing pathway contributions
- Safety monitoring parameters for long-term studies
Personalized Medicine Approaches:
- Genetic profiling informing treatment selection
- Metabolic phenotyping optimizing intervention strategies
- Precision dosing based on individual characteristics
- Combination therapy selection algorithms
Novel Delivery Systems:
- Extended-release formulations reducing dosing frequency
- Oral delivery systems improving administration routes
- Targeted delivery to specific tissues
- Combination formulations with complementary compounds
Practical Recommendations for Researchers
Conducting high-quality tirzepatide insulin resistance research requires attention to numerous practical details. These evidence-based recommendations help optimize experimental outcomes.
Experimental Design Checklist
Planning Phase ✅:
- Clear hypothesis statement with testable predictions
- Literature review identifying knowledge gaps
- Appropriate model system selection (in vitro vs. in vivo)
- Power analysis determining sample sizes
- Control group identification and justification
- Timeline development with realistic milestones
- Budget allocation for quality peptides and assays
Preparation Phase:
- Institutional approvals obtained (IACUC, biosafety, etc.)
- High-purity tirzepatide sourced with COA verification
- Reconstitution protocols established and validated
- Assay reagents and equipment calibrated
- Standard operating procedures documented
- Data management systems prepared
Execution Phase:
- Consistent timing of experiments and measurements
- Blinding implemented where appropriate
- Detailed experimental logs maintained
- Quality control samples included
- Regular equipment calibration and maintenance
- Immediate data backup and organization
Analysis Phase:
- Pre-specified statistical analysis plan followed
- Appropriate statistical tests applied
- Multiple comparison corrections implemented
- Outlier handling documented and justified
- Sensitivity analyses performed
- Results interpreted within study limitations
Common Pitfalls and Solutions
Avoiding frequent experimental errors improves research quality:
Peptide Handling Issues:
- Problem: Repeated freeze-thaw cycles degrading peptide
- Solution: Aliquot upon receipt, thaw only needed amounts
Dosing Errors:
- Problem: Inconsistent concentrations from calculation mistakes
- Solution: Double-check calculations, prepare standard curves, verify with independent measurements
Model System Limitations:
- Problem: Cell line passage number affecting insulin sensitivity
- Solution: Use consistent passage ranges, verify insulin response regularly
Temporal Confounds:
- Problem: Circadian variations affecting metabolic measurements
- Solution: Standardize measurement timing, control light cycles strictly
Statistical Underpowering:
- Problem: Insufficient sample sizes missing real effects
- Solution: Conduct power analyses, include adequate replication
Resource Optimization Strategies
Maximizing research efficiency while maintaining quality:
Peptide Usage Optimization:
- Calculate exact needs before ordering
- Utilize dose-response studies to identify minimal effective concentrations
- Share resources with collaborating laboratories when appropriate
- Store properly to maximize shelf life
Assay Selection:
- Prioritize multiplexed assays measuring multiple parameters simultaneously
- Validate high-throughput methods for large studies
- Balance cost versus information content
- Consider commercial kits versus in-house development
Collaboration Opportunities:
- Partner with laboratories having complementary expertise
- Share specialized equipment and techniques
- Coordinate studies for comprehensive mechanism investigation
- Participate in multi-center validation studies
Advancing Metabolic Research Through Tirzepatide Investigation
The investigation of tirzepatide insulin resistance mechanisms represents a dynamic and productive area of metabolic research. This dual GIP/GLP-1 receptor agonist provides researchers with a sophisticated tool for examining glucose homeostasis, insulin sensitivity, and metabolic regulation at molecular, cellular, and systemic levels.
Key Research Insights
Throughout this comprehensive examination, several critical points emerge:
Mechanistic Complexity: Tirzepatide influences insulin resistance through multiple pathways—enhanced insulin secretion, improved peripheral insulin sensitivity, glucagon suppression, anti-inflammatory effects, and metabolic tissue remodeling. This multi-faceted mechanism distinguishes it from single-target approaches and provides rich opportunities for mechanistic investigation.
Experimental Rigor Requirements: High-quality tirzepatide insulin resistance research demands meticulous attention to peptide quality, experimental design, appropriate controls, and comprehensive outcome assessment. The reliability of findings depends critically on using research-grade peptides with documented purity and proper handling throughout studies.
Translational Potential: While strictly designated for research applications, tirzepatide studies contribute valuable insights into metabolic regulation that advance scientific understanding of insulin resistance pathology and potential intervention strategies.
Actionable Next Steps for Researchers
For laboratories planning tirzepatide insulin resistance investigations:
Immediate Actions
Define Specific Research Questions: Identify precise mechanistic questions your laboratory is uniquely positioned to address, considering available models, expertise, and resources.
- Source Quality Research Materials: Obtain high-purity tirzepatide from reputable suppliers providing comprehensive documentation, ensuring experimental consistency and reproducibility.
- Develop Comprehensive Protocols: Create detailed standard operating procedures covering peptide reconstitution, storage, dosing, outcome measurements, and data analysis before initiating experiments.
- Establish Validation Studies: Conduct preliminary dose-response and time-course experiments establishing optimal conditions for your specific model system and research questions.
- Build Collaborative Networks: Connect with other researchers investigating related mechanisms, sharing methodologies, troubleshooting challenges, and coordinating complementary studies.
Long-term Strategic Considerations:
- Technology Integration: Incorporate emerging analytical methods (omics approaches, advanced imaging, single-cell analysis) to deepen mechanistic insights beyond traditional measurements.
- Comparative Framework: Design studies comparing tirzepatide with other metabolic peptides, establishing its unique contributions and identifying synergistic combination opportunities.
- Reproducibility Focus: Prioritize rigorous experimental design, appropriate statistical power, transparent reporting, and independent replication to advance reliable scientific knowledge.
- Knowledge Dissemination: Share findings through peer-reviewed publications, conference presentations, and open-access data repositories, contributing to the broader scientific community.
The Future of Metabolic Peptide Research
As research technologies advance and scientific understanding deepens, tirzepatide insulin resistance investigation will continue revealing fundamental insights into metabolic regulation. The dual-agonist approach represents an evolution in peptide-based metabolic research, demonstrating how sophisticated molecular design can create tools for examining complex physiological systems.
For researchers committed to advancing metabolic science, tirzepatide offers a powerful experimental compound—when sourced responsibly, handled properly, and investigated rigorously. The insights generated through careful tirzepatide insulin resistance research contribute to the foundational knowledge base that drives scientific progress and deepens our understanding of metabolic health and disease.
Getting Started with Your Research
Laboratories ready to initiate tirzepatide insulin resistance studies can access comprehensive support:
- Browse research-grade peptide options with detailed product specifications
- Review educational resources covering reconstitution, storage, and handling protocols
- Contact technical support for guidance on experimental design and peptide selection
- Access quality documentation including Certificates of Analysis for batch verification
The field of metabolic research continues advancing through the dedicated efforts of scientists worldwide. By maintaining rigorous standards, utilizing high-quality research materials, and pursuing mechanistic questions with creativity and precision, researchers contribute to the expanding knowledge base that defines modern metabolic science.
