
Imagine a peptide so precisely engineered that it targets two critical metabolic pathways simultaneously, fundamentally altering how the body regulates hunger and satiety. Tirzepatide appetite suppression represents one of the most significant advances in metabolic research, offering researchers unprecedented insights into the complex interplay between gut hormones, neural signaling, and appetite regulation. As laboratories worldwide investigate this dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, the mechanisms underlying its profound effects on hunger and food intake continue to reveal fascinating aspects of human metabolism.
This comprehensive guide explores the scientific foundations of tirzepatide appetite suppression, examining the molecular mechanisms, receptor interactions, neural pathways, and research findings that make this peptide a focal point of metabolic investigation in 2025.
Key Takeaways
- Tirzepatide appetite suppression operates through dual GIP and GLP-1 receptor activation, creating synergistic effects on satiety centers in the hypothalamus
- The peptide significantly delays gastric emptying and reduces hunger scores in clinical research settings, with dose-dependent effects observed across multiple studies
- Molecular mechanisms involve complex neural signaling cascades that influence both homeostatic and hedonic appetite pathways
- Research-grade tirzepatide requires precise handling, storage, and reconstitution protocols to maintain peptide integrity for laboratory investigations
- Understanding these suppression mechanisms provides critical insights for metabolic research and the development of future therapeutic strategies
Understanding Tirzepatide: A Dual-Agonist Research Peptide
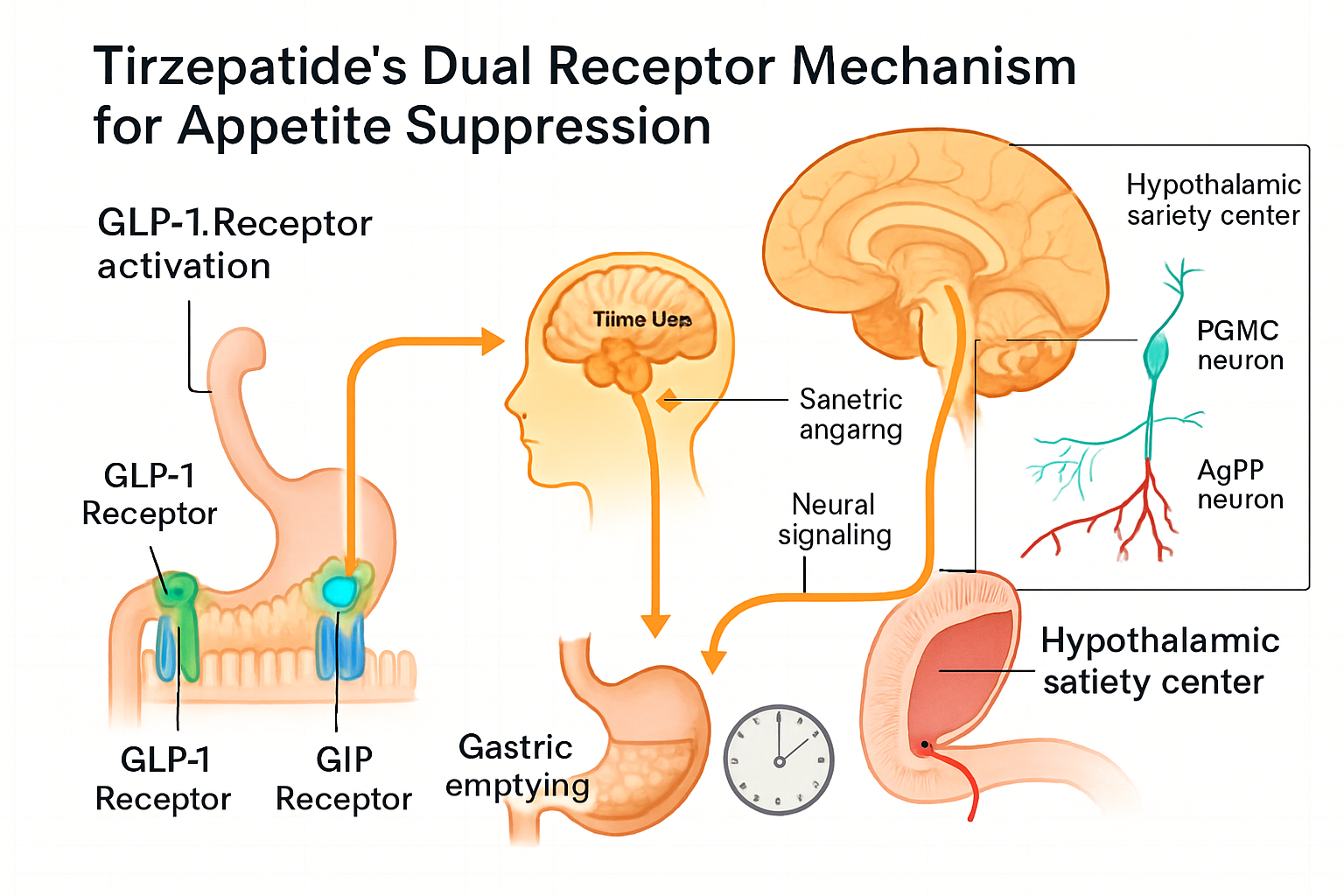
Tirzepatide represents a novel class of synthetic peptides designed to simultaneously activate both GIP and GLP-1 receptors. This dual-agonist approach distinguishes it from single-receptor targeting compounds, creating unique metabolic effects that have captured the attention of research communities globally.
Molecular Structure and Receptor Affinity
The peptide consists of a 39-amino acid sequence based on the native GIP structure, with specific modifications that enhance GLP-1 receptor binding while maintaining robust GIP receptor activation. Research-grade tirzepatide from suppliers like PEPTIDE PRO undergoes rigorous purity testing to ensure consistent molecular integrity for laboratory investigations.
Key structural features include:
- C20 fatty diacid moiety: Enables albumin binding and extends half-life
- Modified amino acid sequence: Optimizes dual receptor activation
- Specific substitutions: Enhance proteolytic stability
- Molecular weight: Approximately 4,813 Da
The Incretin System and Appetite Regulation
To understand tirzepatide appetite suppression, researchers must first grasp the incretin system’s role in metabolic homeostasis. Incretins are gut-derived hormones released in response to nutrient intake, with GLP-1 and GIP serving as the primary mediators.
GLP-1 Functions:
- 🧠 Activates satiety centers in the hypothalamus
️ Slows gastric emptying
- 📉 Reduces appetite and food intake
- 💉 Enhances glucose-dependent insulin secretion
GIP Functions:
- 🔄 Modulates adipose tissue metabolism
- 📊 Influences insulin sensitivity
- 🧬 Affects energy expenditure
- 🤝 Synergizes with GLP-1 signaling
The combination of these pathways creates the foundation for tirzepatide’s pronounced effects on appetite regulation.
Mechanisms of Tirzepatide Appetite Suppression
The appetite suppression effects of tirzepatide involve multiple overlapping mechanisms that operate at different levels of metabolic control. Understanding these pathways provides researchers with critical insights into how dual incretin agonism influences feeding behavior.
Central Nervous System Pathways
Tirzepatide’s effects on appetite originate significantly in the central nervous system, particularly within hypothalamic nuclei that regulate energy homeostasis.
Hypothalamic Satiety Centers
The arcuate nucleus (ARC) of the hypothalamus contains two opposing neuronal populations:
POMC/CART Neurons (Anorexigenic):
- Activated by GLP-1 receptor stimulation
- Release α-melanocyte-stimulating hormone (α-MSH)
- Promote satiety and reduce food intake
- Increase energy expenditure
NPY/AgRP Neurons (Orexigenic):
- Inhibited by incretin receptor activation
- Normally stimulate hunger and feeding
- Suppressed by tirzepatide signaling
- Reduced activity correlates with decreased appetite
Research demonstrates that tirzepatide activates POMC neurons while simultaneously inhibiting NPY/AgRP neurons, creating a powerful dual effect on appetite regulation. This bidirectional modulation represents a key mechanism underlying tirzepatide appetite suppression.
Reward Pathway Modulation
Beyond homeostatic appetite control, tirzepatide influences hedonic feeding pathways in the mesolimbic dopamine system:
- Ventral tegmental area (VTA): Reduced dopamine signaling to nucleus accumbens
- Nucleus accumbens: Decreased reward response to palatable foods
- Prefrontal cortex: Enhanced cognitive control over eating behavior
- Amygdala: Modified emotional responses to food cues
These effects on reward circuitry help explain why research subjects often report reduced food cravings and decreased preference for high-calorie foods during tirzepatide administration.
Peripheral Mechanisms of Appetite Control
While central mechanisms dominate, peripheral effects significantly contribute to tirzepatide appetite suppression.
Gastric Emptying Delay
One of the most consistent findings in tirzepatide research involves its profound effects on gastric motility:
| Parameter | Baseline | Tirzepatide Effect | Clinical Significance |
|---|---|---|---|
| Gastric half-emptying time | 90-120 min | 180-240 min | Prolonged satiety |
| Solid meal retention (4h) | 10-15% | 35-50% | Reduced hunger |
| Liquid emptying | Normal | Moderately delayed | Enhanced fullness |
The delayed gastric emptying creates mechanical distension signals that activate vagal afferents, transmitting satiety information to the brainstem and hypothalamus. This peripheral feedback reinforces central appetite suppression mechanisms.
Gut Hormone Modulation
Tirzepatide influences the secretion and activity of multiple gut-derived peptides:
- Peptide YY (PYY): ↑ Enhanced release, promotes satiety
- Cholecystokinin (CCK): ↑ Increased secretion, reduces meal size
- Ghrelin: ↓ Suppressed “hunger hormone” levels
- Glucagon: ↔ Maintained glucose counter-regulation
This comprehensive modulation of the gut-brain axis creates a coordinated physiological response that powerfully suppresses appetite.
Molecular Signaling Cascades
At the cellular level, tirzepatide appetite suppression involves intricate second-messenger systems:
GLP-1 Receptor Activation:
- Gs protein coupling
- Adenylyl cyclase activation
- cAMP elevation
- PKA and EPAC pathway stimulation
- CREB phosphorylation and gene transcription
GIP Receptor Activation:
- Similar Gs protein coupling
- Additional Gq protein interactions
- Phospholipase C activation
- IP3 and DAG generation
- Calcium mobilization and PKC activation
The convergence of these pathways in appetite-regulating neurons creates synergistic effects that exceed what either receptor activation achieves independently.
Research Findings on Tirzepatide Appetite Suppression
Clinical and preclinical research has generated substantial data characterizing the appetite suppression effects of tirzepatide across various experimental paradigms.
Dose-Response Relationships
Research demonstrates clear dose-dependent effects on appetite parameters:
Low Dose (2.5-5 mg):
- Moderate hunger score reductions (20-30%)
- Mild gastric emptying delay
- Subtle changes in food preference
- Minimal reported side effects
Medium Dose (7.5-10 mg):
- Significant hunger suppression (40-50%)
- Pronounced gastric emptying delay
- Notable reductions in caloric intake
- Moderate tolerability profile
High Dose (12.5-15 mg):
- Maximal appetite suppression (50-60%)
- Substantial gastric motility effects
- Dramatic reductions in food consumption
- Increased gastrointestinal effects
Researchers utilizing tirzepatide formulations for laboratory studies must carefully consider dosing protocols to achieve desired experimental outcomes while maintaining appropriate research parameters.
Timeline of Appetite Effects
The temporal dynamics of tirzepatide appetite suppression follow a characteristic pattern:
Acute Phase (Days 1-7):
- ⚡ Rapid onset of appetite reduction
- 📉 Immediate post-injection satiety
️ Reduced meal size and frequency
- 🔄 Initial gastric adaptation
Adaptation Phase (Weeks 2-4):
- 📊 Stabilization of appetite effects
- 🧠 Neural pathway remodeling
- 💪 Physiological tolerance development
- ⚖️ Metabolic equilibration
Sustained Phase (Weeks 5+):
Maintained appetite suppression
- 🔬 Continued receptor sensitivity
- 📈 Cumulative metabolic benefits
- ✅ Stable feeding behavior patterns
Comparative Research Studies
Multiple investigations have compared tirzepatide’s appetite effects against other incretin-based peptides:
Tirzepatide vs. Semaglutide:
- Greater hunger score reductions with tirzepatide
- More pronounced effects on food cravings
- Superior gastric emptying delay
- Enhanced satiety duration
Researchers interested in comparative studies can access both tirzepatide and semaglutide formulations from specialized peptide suppliers for controlled laboratory investigations.
Tirzepatide vs. Liraglutide:
- Significantly stronger appetite suppression
- Longer duration of action
- More consistent day-to-day effects
- Better tolerance in research settings
Mechanistic Research Insights
Advanced research techniques have revealed specific aspects of tirzepatide appetite suppression:
Neuroimaging Studies:
- Reduced activation in reward centers during food cue exposure
- Decreased hypothalamic response to hunger signals
- Modified prefrontal cortex engagement in eating decisions
- Altered amygdala reactivity to palatable foods
Biomarker Investigations:
- Elevated plasma PYY concentrations
- Reduced active ghrelin levels
- Increased GLP-1 and GIP receptor expression
- Modified leptin sensitivity markers
Behavioral Assessments:
- Reduced hedonic eating episodes
- Decreased binge-eating behaviors
- Improved satiety quotient scores
- Enhanced meal-related fullness ratings
Factors Influencing Tirzepatide Appetite Suppression
The magnitude and characteristics of appetite suppression effects vary based on multiple experimental and physiological variables that researchers must consider when designing studies.
Individual Variability Factors
Research demonstrates significant inter-individual variation in tirzepatide appetite suppression responses:
Genetic Factors:
- GLP-1 receptor polymorphisms
- GIP receptor genetic variants
- Melanocortin system variations
- DPP-4 enzyme activity differences
Metabolic Status:
- Baseline insulin sensitivity
- Pre-existing incretin function
- Adipose tissue distribution
- Hepatic metabolic capacity
Physiological Characteristics:
- Age-related receptor sensitivity
- Sex hormone influences
- Circadian rhythm patterns
- Stress response systems
Environmental and Experimental Conditions
Laboratory conditions significantly impact observed appetite effects:
Dietary Context:
- Macronutrient composition influences
- Meal timing interactions
- Caloric density effects
- Food palatability factors
Research Protocol Variables:
- Injection timing relative to meals
- Dose escalation schedules
- Concurrent interventions
- Measurement methodologies
Storage and Handling:
Proper peptide storage maintains compound integrity:
✅ Optimal Conditions:
- Lyophilized: -20°C to -80°C
- Reconstituted: 2-8°C, protected from light
- Avoid freeze-thaw cycles
- Use sterile techniques
❌ Conditions to Avoid:
- Room temperature storage
- Direct sunlight exposure
- Repeated temperature fluctuations
- Contaminated reconstitution
Researchers can find detailed storage and handling protocols from reputable peptide suppliers to ensure experimental consistency.
Tolerance and Adaptation Considerations
Long-term research reveals important patterns regarding sustained appetite effects:
Receptor Dynamics:
- Minimal receptor downregulation with chronic exposure
- Maintained signaling efficacy over time
- Preserved appetite suppression effects
- Stable gastric motility impacts
Compensatory Mechanisms:
- Limited metabolic adaptation
- Minimal counter-regulatory hormone responses
- Sustained neural pathway activation
- Continued behavioral effects
These findings suggest that tirzepatide appetite suppression remains robust during extended research protocols, unlike some other appetite-modulating compounds that demonstrate significant tolerance development.
Research Applications and Experimental Protocols
Understanding tirzepatide appetite suppression mechanisms enables diverse research applications across multiple scientific disciplines.
Metabolic Research Applications
Tirzepatide serves as a valuable tool for investigating fundamental aspects of appetite regulation:
Neural Pathway Mapping:
- Identifying specific appetite-regulating circuits
- Characterizing incretin receptor distribution
- Mapping gut-brain communication pathways
- Understanding reward system modulation
Hormonal Interaction Studies:
- Investigating incretin-leptin crosstalk
- Examining insulin-appetite relationships
- Characterizing ghrelin suppression mechanisms
- Exploring PYY synergies
Energy Balance Research:
- Quantifying energy expenditure changes
- Measuring metabolic rate modifications
- Assessing substrate utilization shifts
- Evaluating thermogenic responses
Experimental Design Considerations
Rigorous research protocols optimize tirzepatide appetite suppression investigations:
Dosing Protocols:
| Research Goal | Suggested Dose Range | Frequency | Duration |
|---|---|---|---|
| Acute effects | 2.5-5 mg | Single dose | 24-72 hours |
| Short-term mechanisms | 5-10 mg | Weekly | 4-8 weeks |
| Sustained responses | 10-15 mg | Weekly | 12+ weeks |
| Dose-response curves | 2.5-15 mg (escalating) | Weekly | 8-12 weeks |
Measurement Parameters:
Comprehensive assessment requires multiple outcome measures:
🔬 Objective Metrics:
- Caloric intake quantification
- Meal frequency and size
- Gastric emptying rates (scintigraphy)
- Hormone concentration assays
📝 Subjective Assessments:
- Visual analog hunger scales
- Satiety quotient calculations
- Food preference questionnaires
- Craving intensity ratings
📊 Behavioral Observations:
- Eating rate measurements
- Food choice patterns
- Meal initiation timing
- Snacking frequency
Quality Control and Peptide Verification
Research validity depends on peptide quality and proper handling:
Certificate of Analysis (COA) Verification:
- Purity confirmation (≥98% recommended)
- Molecular weight verification
- Endotoxin level assessment
- Sterility testing results
Reconstitution Best Practices:
- Calculate appropriate volumes based on desired concentration
- Use bacteriostatic water for multi-dose applications
- Add diluent slowly along pen peptide wall to minimize foaming
- Gentle swirling rather than vigorous shaking
- Visual inspection for clarity and particle absence
Researchers seeking high-purity tirzepatide for experimental protocols should verify supplier credentials and request comprehensive quality documentation.
Combination Research Strategies
Investigating tirzepatide appetite suppression alongside other compounds reveals synergistic mechanisms:
Tirzepatide + Other Peptides:
- Combined with BPC-157 for metabolic-regenerative studies
- Paired with CJC-1295 for growth hormone pathway interactions
- Combined with AOD-9604 for lipolytic mechanism investigations
Pharmacological Combinations:
- Metformin interactions
- DPP-4 inhibitor synergies
- SGLT2 inhibitor combinations
- Beta-blocker influences
Nutritional Interventions:
- Protein intake modifications
- Carbohydrate restriction protocols
- Fat composition variations
- Micronutrient supplementation
Safety Considerations and Research Ethics
While this article focuses on research applications, understanding the safety profile of tirzepatide remains essential for responsible laboratory investigation.
Research Safety Protocols
Proper laboratory safety ensures researcher protection and experimental integrity:
Personal Protective Equipment (PPE):
- ✅ Nitrile gloves for handling
- ✅ Lab coat protection
- ✅ Eye protection for reconstitution
- ✅ Proper ventilation in work area
Waste Disposal:
- Sharps containers for needles
- Biohazard bags for contaminated materials
- Proper peptide waste neutralization
- Compliance with institutional protocols
Documentation Requirements:
- Detailed experimental logs
- Adverse event recording
- Protocol deviation tracking
- Chain of custody maintenance
Ethical Research Considerations
Responsible investigation of tirzepatide appetite suppression requires adherence to established ethical frameworks:
Research-Only Status:
⚠️ Critical Reminder: Tirzepatide and other research peptides from suppliers like PEPTIDE PRO are strictly for research use only—not for human consumption or therapeutic application outside approved clinical trial settings.
Institutional Oversight:
- Ethics committee approval
- Institutional review board (IRB) authorization
- Animal care committee clearance (when applicable)
- Protocol registration requirements
Transparency Standards:
- Clear research objectives
- Honest reporting of findings
- Disclosure of funding sources
- Publication of negative results
Observed Effects in Research Settings
Understanding the complete profile of tirzepatide helps researchers anticipate experimental variables:
Common Observations:
- Gastrointestinal adaptations (nausea, reduced appetite)
- Injection site reactions
- Fatigue during initial phases
- Altered bowel patterns
Dose-Related Effects:
- Higher doses correlate with increased GI effects
- Gradual dose escalation improves tolerance
- Individual variation in response intensity
- Adaptation typically occurs within 2-4 weeks
Monitoring Parameters:
- Regular weight measurements
- Food intake documentation
- Hydration status assessment
- General condition observations
Future Directions in Tirzepatide Appetite Research
The field of tirzepatide appetite suppression research continues to evolve, with multiple promising avenues for future investigation.
Emerging Research Questions
Current knowledge gaps present opportunities for groundbreaking discoveries:
Molecular Mechanisms:
- 🔬 Precise receptor conformational changes during dual agonism
- 🧬 Epigenetic modifications in appetite-regulating neurons
- 📡 Intracellular signaling pathway crosstalk
- 🔄 Long-term receptor sensitivity maintenance
Neural Circuit Mapping:
- 🧠 Complete characterization of incretin-responsive neurons
- ️ Comprehensive gut-brain axis pathway mapping
- ⚡ Real-time neural activity during appetite suppression
- 🔗 Connectivity between homeostatic and hedonic circuits
Translational Applications:
- 💊 Novel dual-agonist peptide development
Targeted delivery system optimization
- 🔧 Structure-activity relationship refinement
- 📈 Predictive biomarker identification
Advanced Research Technologies
Cutting-edge methodologies enable deeper investigation of appetite suppression mechanisms:
Optogenetics:
- Precise activation/inhibition of incretin-responsive neurons
- Real-time manipulation during feeding behaviors
- Circuit-specific functional validation
- Temporal resolution of neural dynamics
Single-Cell Sequencing:
- Identification of receptor-expressing cell populations
- Characterization of transcriptional responses
- Discovery of novel appetite-regulating cell types
- Mapping of cellular heterogeneity
Advanced Imaging:
- High-resolution fMRI during appetite states
- PET imaging of receptor occupancy
- Calcium imaging of neural populations
- Multi-photon microscopy of signaling events
Next-Generation Peptide Development
Insights from tirzepatide appetite suppression research inform future compound design:
Tri-Agonist Approaches:
- GLP-1/GIP/Glucagon receptor targeting
- Enhanced metabolic effects
- Optimized appetite suppression
- Improved energy expenditure
Selective Pathway Modulators:
- Biased agonism for specific signaling cascades
- Reduced side effect profiles
- Enhanced therapeutic windows
- Tissue-selective activation
Extended Half-Life Formulations:
- Monthly administration possibilities
- Improved patient compliance
- Stable receptor engagement
- Reduced injection frequency
Researchers can stay informed about new peptide developments through specialized suppliers offering comprehensive peptide catalogues with the latest research compounds.
Practical Guide: Conducting Tirzepatide Appetite Research
For researchers planning investigations into tirzepatide appetite suppression, this practical guide provides essential implementation strategies.
Study Design Framework
Phase 1: Planning and Preparation
✅ Define specific research questions ✅ Establish measurable outcomes ✅ Determine appropriate sample size ✅ Secure necessary approvals ✅ Source high-quality peptide ✅ Prepare measurement instruments ✅ Train research personnel
Phase 2: Baseline Assessment
Comprehensive baseline data collection ensures valid comparisons:
- Appetite questionnaire administration
- Food intake pattern documentation
- Metabolic parameter measurement
- Hormonal profile establishment
- Body composition assessment
- Behavioral pattern observation
Phase 3: Intervention Implementation
Systematic tirzepatide administration following established protocols:
| Week | Dose | Assessments | Key Monitoring |
|---|---|---|---|
| 1-2 | 2.5 mg | Daily appetite logs, weekly weights | Tolerance, initial effects |
| 3-4 | 5 mg | Appetite scales, food intake | Dose response, adaptation |
| 5-8 | 7.5-10 mg | Full metabolic panel | Sustained effects, stability |
| 9-12 | Maintenance | Comprehensive outcomes | Long-term patterns |
Phase 4: Data Collection and Analysis
Rigorous data management ensures research integrity:
📊 Quantitative Data:
- Statistical analysis of appetite scores
- Caloric intake trend analysis
- Hormonal concentration comparisons
- Body composition changes
📝 Qualitative Data:
- Thematic analysis of subjective reports
- Pattern identification in food preferences
- Behavioral change documentation
- Contextual factor assessment
Troubleshooting Common Research Challenges
Challenge: Inconsistent Appetite Measurements
Solutions:
- Standardize assessment timing (same time daily)
- Use validated, reliable scales
- Control environmental factors
- Implement multiple measurement methods
Challenge: Variable Peptide Response
Solutions:
- Verify peptide quality and storage
- Confirm proper reconstitution
- Check injection technique consistency
- Consider individual variability factors
Challenge: Confounding Variables
Solutions:
- Implement strict dietary controls
- Monitor concurrent medications
- Document lifestyle factors
- Use appropriate statistical controls
Collaboration and Resource Sharing
Advancing tirzepatide appetite suppression research benefits from collaborative approaches:
Academic Partnerships:
- Multi-institutional studies
- Shared equipment and expertise
- Complementary methodological approaches
- Enhanced statistical power
Industry Collaboration:
- Access to advanced technologies
- Pharmaceutical-grade peptides
- Translational research opportunities
- Funding support
Open Science Practices:
- Pre-registration of protocols
- Data sharing repositories
- Transparent methodology reporting
- Reproducibility initiatives
The Significance of Tirzepatide Appetite Suppression Research
Tirzepatide appetite suppression represents a remarkable convergence of molecular precision, neural complexity, and metabolic regulation. Through dual GIP and GLP-1 receptor agonism, this research peptide demonstrates how sophisticated pharmacological approaches can profoundly influence fundamental biological processes like hunger and satiety.
The mechanisms underlying these effects—from hypothalamic neural circuit modulation to peripheral gastric emptying delay, from molecular signaling cascade activation to behavioral pattern modification—illustrate the intricate systems governing appetite regulation. As research continues to unravel these complexities in 2025 and beyond, tirzepatide serves as both a powerful investigational tool and a model for next-generation metabolic therapeutics.
Key Research Implications
🔬 Mechanistic Insights: Tirzepatide research reveals fundamental principles of incretin biology and appetite neuroscience
📊 Translational Potential: Findings inform development of improved metabolic interventions with enhanced efficacy profiles
🧬 Personalized Approaches: Understanding individual variability enables precision medicine strategies
🌐 Broader Applications: Lessons learned extend to other metabolic and neurological research domains
Next Steps for Researchers
For investigators planning to explore tirzepatide appetite suppression mechanisms:
- Secure high-quality research peptides from reputable suppliers like PEPTIDE PRO with verified purity and proper documentation
- Design rigorous protocols incorporating appropriate controls, validated measurements, and ethical oversight
- Implement comprehensive assessment strategies capturing both objective and subjective appetite parameters
- Maintain meticulous documentation ensuring reproducibility and transparent reporting
- Engage with the research community through collaboration, publication, and knowledge sharing
- Stay current with emerging findings as this rapidly evolving field continues to generate new insights
The profound appetite suppression effects of tirzepatide underscore the remarkable potential of peptide-based research compounds to illuminate biological mechanisms and inform therapeutic development. As laboratories worldwide continue investigating these phenomena, each study contributes to a growing understanding of metabolic regulation that may ultimately transform approaches to appetite-related conditions.
For researchers ready to begin their investigations, accessing research-grade tirzepatide formulations with comprehensive quality documentation represents the essential first step toward contributing meaningful discoveries to this dynamic field.
Remember: All peptides discussed are strictly for research purposes only, not for human consumption. Responsible, ethical research practices remain paramount in advancing scientific knowledge while maintaining the highest standards of laboratory safety and regulatory compliance.
