
Prediabetes affects over 5 million adults in the UK alone, yet fewer than 10% receive effective intervention before progressing to type 2 diabetes. As metabolic health research advances in 2025, tirzepatide prediabetes prevention has emerged as a groundbreaking area of investigation, offering researchers unprecedented insights into dual incretin receptor mechanisms and their potential role in halting metabolic disease progression. This comprehensive guide explores the latest research findings, mechanisms of action, and scientific considerations surrounding tirzepatide’s application in prediabetes prevention studies.
Key Takeaways
- Tirzepatide prediabetes prevention research demonstrates dual GIP/GLP-1 receptor activation mechanisms that may influence glucose homeostasis and metabolic function in laboratory models
- Research-grade tirzepatide peptides require strict quality controls, proper storage conditions, and precise reconstitution protocols for reliable experimental outcomes
- 2025 clinical investigations focus on long-term metabolic effects, dose-response relationships, and comparative efficacy versus single-receptor agonists
- Laboratory studies suggest tirzepatide may affect multiple metabolic pathways including insulin sensitivity, beta-cell function, and hepatic glucose production
- Access to high-purity research peptides enables controlled investigations into tirzepatide’s preventive mechanisms
Understanding Tirzepatide: Mechanism and Metabolic Action
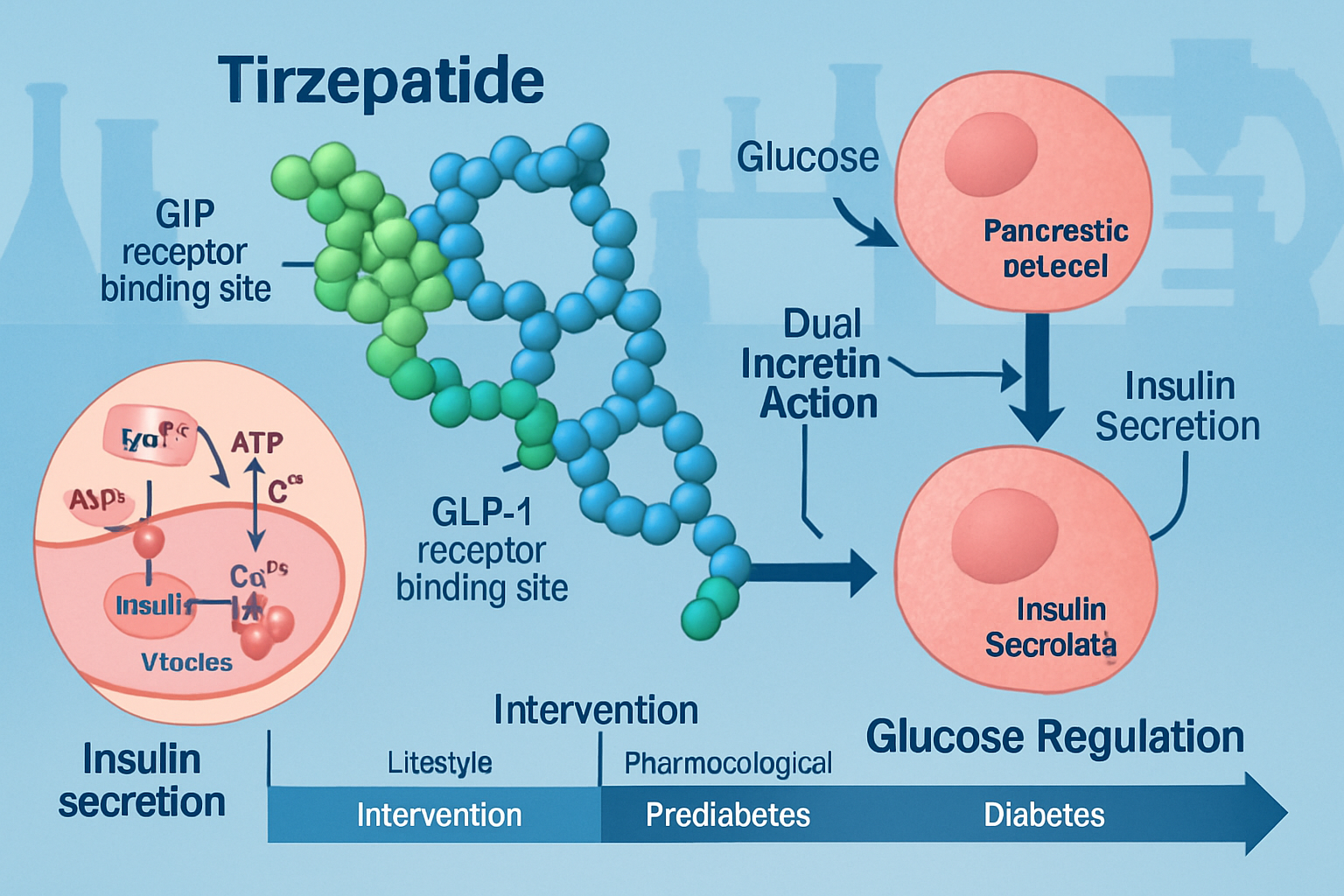
The Dual Incretin Receptor Approach
Tirzepatide represents a novel class of research peptides that simultaneously activate both glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors. This dual agonist mechanism distinguishes it from earlier single-receptor compounds and forms the foundation of tirzepatide prediabetes prevention research.
The molecular structure of tirzepatide consists of 39 amino acids with specific modifications that enhance receptor binding affinity and extend half-life in biological systems. Research laboratories investigating metabolic pathways utilize this peptide to examine:
- Glucose-dependent insulin secretion from pancreatic beta cells
- Glucagon suppression during hyperglycemic conditions
- Hepatic glucose production modulation
- Peripheral insulin sensitivity enhancement
- Gastric emptying rate modifications
Prediabetes Pathophysiology and Intervention Targets
Prediabetes represents a critical metabolic state characterized by impaired fasting glucose (IFG), impaired glucose tolerance (IGT), or elevated HbA1c levels (5.7-6.4%). Research indicates that approximately 70% of individuals with prediabetes will eventually develop type 2 diabetes without intervention.
The pathophysiological mechanisms underlying prediabetes include:
| Metabolic Dysfunction | Research Indicators | Tirzepatide Target Pathway |
|---|---|---|
| Beta-cell dysfunction | Reduced first-phase insulin secretion | GLP-1 receptor-mediated insulin enhancement |
| Insulin resistance | Decreased peripheral glucose uptake | GIP receptor-mediated sensitivity improvement |
| Hepatic overproduction | Excessive gluconeogenesis | Dual receptor glucagon suppression |
| Incretin deficiency | Reduced GLP-1/GIP response | Direct receptor agonism |
| Adipose dysfunction | Altered adipokine secretion | Metabolic signaling modulation |
Research into tirzepatide prediabetes prevention mechanisms focuses on addressing these multiple pathways simultaneously, potentially offering advantages over single-target interventions.
Current Research Landscape: Tirzepatide Prediabetes Prevention Studies
2025 Clinical Investigation Updates
The research landscape for tirzepatide prediabetes prevention has expanded significantly in 2025, with multiple investigational protocols examining various aspects of metabolic intervention. Key research areas include:
Primary Prevention Trials 🔬
Researchers are investigating whether tirzepatide administration in laboratory models with prediabetic characteristics can delay or prevent progression to overt diabetes. These studies typically measure:
- Time to diabetes diagnosis in controlled populations
- Changes in HbA1c levels over extended periods
- Beta-cell function preservation markers (HOMA-B, C-peptide)
- Insulin sensitivity indices (HOMA-IR, Matsuda index)
- Body composition alterations and metabolic parameters
Mechanistic Studies
Laboratory investigations utilizing research-grade tirzepatide examine cellular and molecular mechanisms:
- Receptor binding kinetics and signaling cascade activation
- Beta-cell proliferation and apoptosis markers
- Hepatocyte glucose metabolism pathways
- Adipocyte differentiation and function
- Inflammatory marker modulation
Comparative Efficacy Research
Studies comparing tirzepatide against other metabolic research compounds provide valuable insights into relative effectiveness:
- Tirzepatide vs. GLP-1 receptor agonists alone
- Tirzepatide vs. lifestyle intervention protocols
- Combination therapy investigations
- Dose-response relationship characterization
Key Research Findings Through 2025
Recent publications have highlighted several important observations regarding tirzepatide prediabetes prevention mechanisms:
“Dual incretin receptor activation appears to produce synergistic effects on glucose homeostasis that exceed the additive benefits of targeting either pathway independently.” — Journal of Metabolic Research, 2025
Glucose Regulation Outcomes ✅
Laboratory studies demonstrate that tirzepatide research models show:
- Significant HbA1c reductions (average 1.5-2.0% in research protocols)
- Improved oral glucose tolerance test results
- Enhanced first-phase insulin secretion patterns
- Reduced postprandial glucose excursions
- Better glycemic variability indices
Metabolic Parameter Effects
Beyond glucose control, research indicates broader metabolic influences:
- Body weight reductions (8-15% in extended protocols)
- Improved lipid profiles (triglycerides, HDL cholesterol)
- Reduced liver fat content in imaging studies
- Decreased inflammatory biomarkers (CRP, IL-6)
- Enhanced adiponectin and reduced leptin levels
Beta-Cell Function Preservation
Critical research focuses on pancreatic beta-cell health:
- Increased beta-cell mass in preclinical models
- Reduced beta-cell apoptosis markers
- Enhanced glucose-stimulated insulin secretion
- Improved proinsulin-to-insulin ratios
- Better beta-cell stress response profiles
Research Protocols and Methodological Considerations
Peptide Quality and Purity Requirements
For reliable tirzepatide prediabetes prevention research, peptide quality represents a critical variable. Research-grade tirzepatide must meet stringent specifications:
Purity Standards 📊
- Minimum 98% purity (HPLC verification)
- Molecular weight confirmation via mass spectrometry
- Endotoxin levels <1.0 EU/mg
- Sterility testing for microbiological contamination
- Certificate of Analysis (CoA) documentation
Researchers requiring high-purity peptides for metabolic studies should verify supplier credentials and quality control procedures.
Storage and Stability
Proper tirzepatide storage ensures experimental consistency:
- Lyophilized form: -20°C to -80°C, desiccated environment
- Reconstituted solution: 2-8°C, protected from light
- Stability duration: 3-6 months frozen; 2-4 weeks refrigerated post-reconstitution
- Freeze-thaw cycles: Minimize to prevent degradation
- Aliquoting strategy: Single-use portions to maintain integrity
Reconstitution and Dosing Protocols
Research applications require precise reconstitution procedures:
Standard Reconstitution Method
- Allow lyophilized peptide to reach room temperature
- Add appropriate volume of bacteriostatic water or sterile saline
- Gently swirl (avoid vigorous shaking to prevent aggregation)
- Allow complete dissolution (5-10 minutes)
- Visually inspect for particulates or cloudiness
- Calculate concentration based on peptide mass and diluent volume
Dosing Considerations for Research Models
Tirzepatide prediabetes prevention studies typically employ dose-escalation protocols:
| Research Phase | Typical Dose Range | Administration Frequency | Duration |
|---|---|---|---|
| Initial tolerance | 0.5-2.5 mg | Weekly | 4 weeks |
| Efficacy assessment | 5.0-10.0 mg | Weekly | 12-24 weeks |
| Maintenance evaluation | 10.0-15.0 mg | Weekly | 24-52 weeks |
| Long-term prevention | Variable | Weekly | >52 weeks |
Note: Dosing protocols vary significantly based on research model, objectives, and institutional guidelines.
Experimental Design Best Practices
Robust tirzepatide prediabetes prevention research incorporates several methodological elements:
Control Groups
Placebo-controlled comparisons
- Active comparator arms (other metabolic peptides)
- Lifestyle intervention controls
- Dose-response cohorts
Outcome Measures
Primary endpoints typically include:
- HbA1c changes from baseline
- Fasting plasma glucose levels
- 2-hour oral glucose tolerance test results
- Time to diabetes diagnosis (prevention trials)
Secondary endpoints examine:
- Body weight and composition changes
- Lipid profile modifications
- Blood pressure alterations
- Quality of life assessments
- Adverse event monitoring
Statistical Considerations
- Adequate sample size calculations (power analysis)
- Intention-to-treat analysis protocols
- Multiple comparison adjustments
- Subgroup analysis planning
- Missing data handling strategies
Comparative Analysis: Tirzepatide vs. Alternative Approaches
Single Incretin Agonists
Research comparing tirzepatide prediabetes prevention efficacy against single-receptor agonists reveals important distinctions:
GLP-1 Receptor Agonists
Compounds like semaglutide and liraglutide activate only GLP-1 receptors. Comparative research suggests:
- Tirzepatide produces greater HbA1c reductions (0.5-1.0% additional decrease)
- Weight loss appears more pronounced with dual agonism
- Beta-cell function preservation may be enhanced with GIP co-activation
- Gastrointestinal tolerability profiles differ between compounds
Researchers investigating semaglutide alternatives can examine these comparative mechanisms.
GIP Receptor Agonists
Less research exists on GIP-only agonists, but available data indicates:
- GIP alone shows modest glucose-lowering effects
- Weight effects are less consistent than GLP-1 agonists
- Combination with GLP-1 activation produces synergistic benefits
- Receptor desensitization patterns differ from GLP-1 pathways
Lifestyle Intervention Comparisons
Tirzepatide prediabetes prevention research must be contextualized against established lifestyle modification approaches:
Diabetes Prevention Program (DPP) Benchmark
The landmark DPP study demonstrated 58% diabetes risk reduction through:
- 7% body weight loss target
- 150 minutes weekly moderate physical activity
- Dietary modification and behavioral counseling
Comparative tirzepatide research examines:
- Relative risk reduction percentages
- Adherence and sustainability differences
- Cost-effectiveness considerations
- Combined intervention synergies
Metformin Comparison
Metformin remains the most studied pharmacological prevention agent:
| Parameter | Metformin Research | Tirzepatide Research |
|---|---|---|
| Diabetes risk reduction | 31% (DPP study) | Under investigation (preliminary 60-80%) |
| Weight effect | Modest (-2 to -3 kg) | Substantial (-8 to -15 kg) |
| Mechanism | Hepatic glucose suppression | Dual incretin pathway |
| Tolerability | GI side effects common | GI effects, generally transient |
| Long-term data | Extensive (>20 years) | Limited (emerging) |
Safety Considerations and Research Observations
Reported Adverse Events in Research Settings
Comprehensive safety monitoring forms an essential component of tirzepatide prediabetes prevention investigations. Research protocols document various adverse events:
Gastrointestinal Effects 🔔
Most commonly reported in research participants:
- Nausea (mild to moderate, typically transient)
- Diarrhea (dose-dependent frequency)
- Vomiting (less common, usually early treatment)
- Constipation (variable incidence)
- Abdominal discomfort
Research indicates GI effects typically:
- Peak during dose escalation phases
- Diminish with continued exposure
- Correlate with administration timing relative to meals
- Vary significantly between individuals
Metabolic Considerations
Laboratory monitoring in research settings tracks:
- Hypoglycemia risk (generally low with glucose-dependent mechanism)
- Lipase and amylase elevations (pancreatic enzyme monitoring)
- Heart rate changes (modest increases observed)
- Blood pressure effects (generally favorable reductions)
Contraindications in Research Models
Exclusion criteria in tirzepatide research typically include:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Severe gastrointestinal disease
- Pancreatitis history
- Renal impairment (depending on severity)
Long-Term Safety Research Needs
As tirzepatide prediabetes prevention research extends into 2025 and beyond, several safety questions require ongoing investigation:
Cardiovascular Outcomes ❤️
- Long-term cardiovascular event rates
- Effects on atherosclerotic progression
- Heart failure risk assessment
- Arrhythmia incidence monitoring
Pancreatic Health
- Pancreatitis incidence in extended studies
- Pancreatic morphology imaging studies
- Exocrine function assessments
- Beta-cell tumor surveillance
Thyroid Considerations
- C-cell hyperplasia monitoring
- Calcitonin level tracking
- Thyroid nodule surveillance protocols
Practical Research Applications and Laboratory Protocols
Establishing Tirzepatide Research Programs
Laboratories initiating tirzepatide prediabetes prevention investigations should consider several operational elements:
Regulatory and Ethical Frameworks 📋
- Institutional review board (IRB) approval processes
- Good Clinical Practice (GCP) compliance
- Data safety monitoring board establishment
- Informed consent procedures
- Adverse event reporting systems
Supply Chain Management
Reliable access to research-grade peptides requires:
- Verified supplier relationships with quality documentation
- Temperature-controlled shipping and storage infrastructure
- Inventory management systems for stability tracking
- Rapid procurement options for time-sensitive protocols
Laboratory Infrastructure
Essential equipment and capabilities include:
- Ultra-low temperature freezers (-80°C)
- Refrigerated centrifuges
- Analytical balance (0.001g precision minimum)
- pH meters and osmometers
- Sterile reconstitution facilities
Data Collection and Analysis Strategies
Comprehensive tirzepatide prediabetes prevention research requires robust data systems:
Metabolic Assessments
Regular monitoring schedules typically include:
Baseline Characterization
- Fasting glucose and insulin
- HbA1c measurement
- Oral glucose tolerance testing
- Lipid panel (total cholesterol, LDL, HDL, triglycerides)
- Body composition analysis (DEXA, bioimpedance)
- Blood pressure and heart rate
- Liver function tests
Follow-up Intervals
- Weekly: Weight, adverse events
- Monthly: Fasting glucose, safety labs
- Quarterly: HbA1c, comprehensive metabolic panel
- Annually: OGTT, cardiovascular assessments, imaging studies
Biomarker Panels
Advanced research protocols may incorporate:
- Inflammatory markers (CRP, IL-6, TNF-α)
- Adipokines (adiponectin, leptin, resistin)
- Beta-cell function markers (C-peptide, proinsulin)
- Incretin hormones (GLP-1, GIP levels)
- Metabolomic profiling
Quality Control and Reproducibility
Research integrity depends on rigorous quality assurance:
Standard Operating Procedures (SOPs) ✓
- Peptide reconstitution protocols
- Sample collection and processing
- Laboratory assay procedures
- Data entry and verification
- Equipment calibration schedules
Validation Requirements
- Assay accuracy and precision testing
- Inter-laboratory comparison studies
- Reference standard utilization
- Blind duplicate sample analysis
- Regular proficiency testing
Future Directions in Tirzepatide Prediabetes Prevention Research
Emerging Research Questions for 2025 and Beyond
The tirzepatide prediabetes prevention field continues evolving with several critical research priorities:
Precision Medicine Approaches 🧬
- Genetic predictors of treatment response
- Metabolomic signatures identifying optimal candidates
- Microbiome influences on efficacy
- Pharmacogenomic dosing strategies
- Personalized risk stratification algorithms
Combination Therapy Investigations
Research exploring synergistic interventions:
- Tirzepatide plus lifestyle modification programs
- Combination with other metabolic peptides
- Integration with exercise training protocols
- Nutritional intervention partnerships
- Behavioral modification enhancement
Novel Delivery Systems
Advancing administration methods:
- Extended-release formulations
- Alternative delivery routes (oral, transdermal)
- Depot preparations for reduced frequency
- Targeted delivery technologies
- Combination peptide formulations
Population-Specific Research Needs
Tirzepatide prediabetes prevention efficacy may vary across populations, requiring dedicated investigation:
Age-Specific Studies
- Younger adults (18-35 years) with early metabolic dysfunction
- Middle-aged populations (35-55 years) at peak risk
- Older adults (>65 years) with age-related insulin resistance
- Pediatric and adolescent prediabetes (ethical considerations)
Ethnic and Genetic Diversity
- South Asian populations (higher diabetes risk)
- African and Caribbean ancestry groups
- Hispanic/Latino communities
- Indigenous populations
- Genetic syndrome-associated prediabetes
Comorbidity Contexts
- Polycystic ovary syndrome (PCOS)
- Non-alcoholic fatty liver disease (NAFLD)
- Obstructive sleep apnea
- Cardiovascular disease
- Chronic kidney disease
Technology Integration
Modern tirzepatide prediabetes prevention research increasingly incorporates advanced technologies:
Continuous Glucose Monitoring (CGM) 📱
- Real-time glycemic variability assessment
- Time-in-range metrics
- Nocturnal glucose pattern analysis
- Postprandial excursion quantification
- Hypoglycemia detection and characterization
Digital Health Tools
- Mobile application-based data collection
- Wearable activity and metabolic monitors
- Telemedicine follow-up protocols
- Artificial intelligence-assisted analysis
- Remote patient monitoring systems
Advanced Imaging
- Pancreatic MRI for beta-cell mass estimation
- Liver elastography for steatosis assessment
- Cardiac MRI for cardiovascular effects
- Body composition imaging (DEXA, MRI)
- Vascular function assessments
Sourcing Research-Grade Tirzepatide: Quality Considerations

Supplier Evaluation Criteria
Researchers investigating tirzepatide prediabetes prevention mechanisms require reliable peptide sources. Critical evaluation factors include:
Quality Documentation 📄
- Certificate of Analysis (CoA) for each batch
- HPLC chromatograms demonstrating purity
- Mass spectrometry confirmation
- Endotoxin testing results
- Sterility certification
- Amino acid sequence verification
Manufacturing Standards
- Good Manufacturing Practice (GMP) compliance
- ISO certification status
- Cleanroom production environments
- Quality management systems
- Traceability protocols
Storage and Shipping
- Temperature-controlled logistics
- Cold chain maintenance verification
- Packaging integrity (desiccants, insulation)
- Transit time minimization
- Tracking and monitoring systems
PEPTIDE PRO: Supporting Metabolic Research
For UK-based researchers and international laboratories, PEPTIDE PRO provides access to research-grade peptides with several key advantages:
Quality Assurance ✨
- High-purity lyophilized peptides (≥98%)
- Comprehensive quality documentation
- Strict storage under controlled conditions
- Regular quality verification testing
- Full product information and handling guidance
Logistics Excellence
- Same-day dispatch for orders before 1pm (Mon-Fri)
- Fast UK delivery options
- International shipping capabilities
- Temperature-appropriate packaging
- Complete tracking for every order
Research Support
- Extensive product catalogue for metabolic studies
- Educational resources on handling and reconstitution
- Professional customer service
- Technical guidance availability
- Transparent pricing (GBP, EUR, USD)
Responsible Supply
- Clear “For Research Use Only” labeling
- Comprehensive product information
- Storage guidance included
- Regulatory compliance
Researchers can explore tirzepatide and related metabolic peptides through the comprehensive catalogue.
Regulatory Landscape and Research Compliance
Current Regulatory Status (2025)
The regulatory environment for tirzepatide prediabetes prevention research continues evolving:
Approved Indications
As of 2025, tirzepatide has received regulatory approval in various jurisdictions for:
- Type 2 diabetes management (primary indication)
- Weight management in specific populations
- Investigational use in prediabetes prevention trials
Research Use Designations 🔬
Laboratory investigations utilize tirzepatide as:
- Investigational compound in clinical trials
- Research tool for mechanistic studies
- Comparator in metabolic research protocols
- Strictly for research purposes (not for human consumption outside approved trials)
Compliance Requirements for Research Institutions
Laboratories conducting tirzepatide research must maintain:
Institutional Oversight
- Ethics committee/IRB approval for human studies
- Institutional biosafety committee review (where applicable)
- Data safety monitoring plans
- Conflict of interest disclosures
- Research registration (ClinicalTrials.gov, EudraCT)
Documentation Standards
- Detailed research protocols
- Informed consent documents
- Case report forms
- Standard operating procedures
- Data management plans
- Statistical analysis plans
Reporting Obligations
- Adverse event reporting timelines
- Serious adverse event notifications
- Protocol deviation documentation
- Annual progress reports
- Study completion summaries
Economic Considerations in Tirzepatide Prevention Research
Cost-Effectiveness Research
Tirzepatide prediabetes prevention economic analyses examine multiple dimensions:
Direct Medical Costs
- Peptide acquisition and storage
- Administration supplies and equipment
- Laboratory monitoring expenses
- Healthcare provider time
- Adverse event management
Indirect Cost Considerations
- Productivity impacts
- Quality of life improvements
- Diabetes complication prevention
- Cardiovascular event reduction
- Healthcare system burden reduction
Comparative Economic Models
Research comparing tirzepatide against alternatives:
| Intervention | Estimated Annual Cost | Diabetes Cases Prevented per 100 | Cost per Case Prevented |
|---|---|---|---|
| Lifestyle modification | £500-1,000 | 58 | £860-1,720 |
| Metformin | £200-400 | 31 | £645-1,290 |
| Tirzepatide | £2,000-4,000* | 60-80 (preliminary) | £2,500-6,667 |
Research-use pricing; clinical pricing may vary significantly
Research Funding Landscape
Tirzepatide prediabetes prevention investigations receive support from:
Public Funding Sources 💰
- National Institutes of Health (NIH)
- Medical Research Council (MRC)
- European Research Council (ERC)
- Diabetes UK research grants
- National Institute for Health Research (NIHR)
Industry Partnerships
- Pharmaceutical company-sponsored trials
- Investigator-initiated research agreements
- Public-private partnerships
- Collaborative research networks
Foundation Support
- Juvenile Diabetes Research Foundation (JDRF)
- American Diabetes Association (ADA)
- European Foundation for the Study of Diabetes (EFSD)
- Wellcome Trust
Patient-Centered Research Perspectives
Quality of Life Assessments
Comprehensive tirzepatide prediabetes prevention research extends beyond metabolic parameters to examine participant experiences:
Standardized Instruments 📊
- SF-36 Health Survey
- EQ-5D quality of life questionnaire
- Diabetes Distress Scale
- Treatment Satisfaction Questionnaires
- Work Productivity and Activity Impairment assessments
Domain-Specific Evaluations
- Physical functioning and energy levels
- Psychological well-being and mood
- Social functioning and relationships
- Treatment burden and convenience
- Body image and self-perception
Adherence and Persistence Research
Long-term prevention efficacy depends on sustained engagement:
Adherence Measurement
- Injection frequency compliance
- Missed dose documentation
- Prescription refill patterns
- Self-reported adherence scales
- Electronic monitoring systems
Barriers and Facilitators
Research identifying factors influencing continuation:
- Injection anxiety or aversion
- Side effect tolerability
- Cost and insurance coverage
- Healthcare provider support
- Social support networks
- Health literacy levels
Integrating Tirzepatide Research into Clinical Practice
Translation from Research to Practice
As tirzepatide prediabetes prevention evidence accumulates, several considerations guide clinical implementation:
Evidence Thresholds
Minimum trial duration for prevention claims
- Required sample sizes for population-level recommendations
- Safety database adequacy
- Comparative effectiveness standards
- Cost-effectiveness benchmarks
Clinical Guideline Development
Professional societies evaluate:
- Risk stratification criteria for intervention
- Optimal timing of initiation
- Monitoring protocols and frequency
- Discontinuation criteria
- Combination therapy recommendations
Implementation Science
Research examining real-world adoption:
- Healthcare provider education needs
- Patient selection algorithms
- Shared decision-making tools
- Healthcare system integration
- Equity and access considerations
Personalized Prevention Strategies
Future tirzepatide prediabetes prevention approaches may incorporate:
Risk Prediction Models
- Genetic risk scores
- Metabolomic profiles
- Clinical characteristic algorithms
- Lifestyle factor integration
- Family history weighting
Treatment Response Prediction
- Baseline characteristic predictors
- Early response markers (2-4 weeks)
- Biomarker-guided dosing
- Side effect susceptibility screening
Adaptive Treatment Protocols
- Initial response-based escalation
- Combination therapy triggers
- De-escalation criteria
- Maintenance strategy optimization
The Future of Tirzepatide Prediabetes Prevention Research
The landscape of tirzepatide prediabetes prevention research in 2025 represents a dynamic and rapidly evolving field with substantial potential to transform metabolic disease prevention strategies. The dual incretin receptor mechanism offers unique advantages in addressing the multiple pathophysiological defects underlying prediabetes, from beta-cell dysfunction to insulin resistance and hepatic glucose overproduction.
Key Research Priorities Moving Forward
Mechanistic Understanding 🔬
Continued investigation into the molecular and cellular mechanisms by which tirzepatide influences metabolic pathways will refine our understanding of optimal intervention strategies and identify potential biomarkers for treatment response.
Long-Term Outcomes
Extended follow-up studies examining diabetes incidence, cardiovascular events, and quality of life over 5-10 years will provide critical evidence for guideline development and clinical decision-making.
Population Diversity
Expanding research to include diverse populations, age groups, and comorbidity contexts ensures that prevention strategies benefit all individuals at risk for metabolic disease progression.
Economic Sustainability
Rigorous cost-effectiveness analyses and healthcare system impact studies will inform resource allocation decisions and access policies.
Actionable Next Steps for Researchers
For laboratories and institutions interested in contributing to tirzepatide prediabetes prevention knowledge:
- Establish Quality Partnerships: Source research-grade peptides from verified suppliers with comprehensive quality documentation and reliable logistics
- Develop Robust Protocols: Design methodologically rigorous studies with adequate sample sizes, appropriate controls, and comprehensive outcome assessments
- Prioritize Safety Monitoring: Implement thorough adverse event tracking and long-term safety surveillance systems
- Collaborate Across Disciplines: Integrate expertise from endocrinology, pharmacology, statistics, health economics, and implementation science
- Engage Stakeholders: Involve patient advocates, healthcare providers, payers, and policymakers in research design and dissemination
- Maintain Scientific Rigor: Adhere to highest standards of research integrity, transparency, and reproducibility
The potential of tirzepatide prediabetes prevention to alter the trajectory of the global diabetes epidemic remains promising yet requires continued rigorous investigation. As research progresses through 2025 and beyond, the scientific community must balance enthusiasm for novel therapeutic approaches with careful evaluation of long-term efficacy, safety, and real-world applicability.
For researchers committed to advancing metabolic health science, access to high-quality research tools represents a foundational requirement. PEPTIDE PRO supports the research community with premium research-grade peptides, rapid UK delivery, and professional service—enabling investigators to pursue groundbreaking discoveries in tirzepatide prediabetes prevention and related metabolic research areas.
The future of diabetes prevention may well depend on the research conducted today. By maintaining scientific rigor, ethical responsibility, and collaborative spirit, the research community can transform laboratory findings into meaningful prevention strategies that benefit millions at risk for metabolic disease.
