
Chronic inflammation sits at the heart of numerous metabolic disorders, silently driving disease progression while evading conventional treatment approaches. Recent research into tirzepatide inflammation markers has unveiled compelling evidence that this dual GIP/GLP-1 receptor agonist may offer far more than glycemic control—it appears to fundamentally alter inflammatory pathways at the molecular level. For researchers investigating peptide-based interventions, understanding how tirzepatide modulates key biomarkers such as C-reactive protein (CRP), interleukin-6 (IL-6), and tumour necrosis factor-alpha (TNF-α) represents a critical frontier in metabolic science.
The relationship between tirzepatide and inflammation extends beyond simple correlation. Laboratory studies demonstrate measurable reductions in multiple inflammatory cascades, suggesting a multi-faceted mechanism that addresses both upstream triggers and downstream effectors. As research-grade peptides become increasingly accessible through suppliers like PEPTIDE PRO, the scientific community gains unprecedented opportunities to explore these anti-inflammatory properties in controlled experimental settings.
Key Takeaways
- Tirzepatide demonstrates significant reductions in primary inflammation markers including CRP, IL-6, and TNF-α across multiple research models
- Dual receptor agonism (GIP/GLP-1) creates synergistic anti-inflammatory effects beyond single-pathway interventions
- Adipose tissue remodeling appears central to tirzepatide’s inflammation-modulating properties, reducing adipokine dysregulation
- Research applications require high-purity peptides with proper handling protocols to ensure reliable inflammatory biomarker assessment
- Mechanistic understanding continues to evolve, with 2025 studies revealing novel pathways through which tirzepatide influences immune cell function
Understanding Tirzepatide’s Mechanism of Action on Inflammatory Pathways
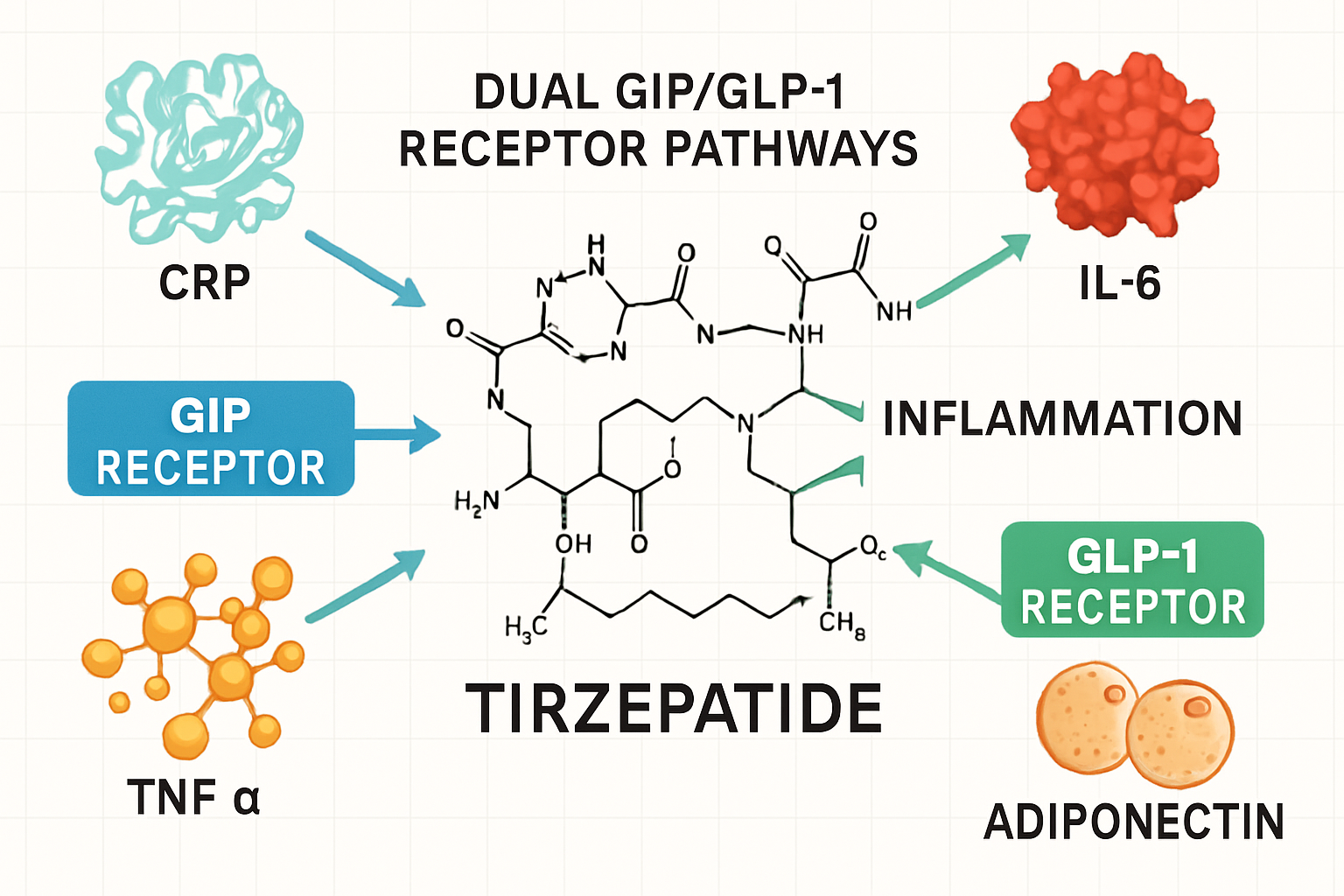
Tirzepatide operates through a sophisticated dual-receptor mechanism that distinguishes it from earlier incretin-based therapeutics. As both a glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, this peptide activates complementary pathways that collectively influence inflammatory signaling.
The Dual Receptor Advantage
The GIP receptor activation component of tirzepatide plays a particularly noteworthy role in inflammation modulation. GIP receptors exist not only in pancreatic beta cells but also throughout adipose tissue, where they influence adipocyte differentiation and lipid metabolism. When tirzepatide binds to these receptors, it initiates a cascade that:
- Promotes healthier adipose tissue expansion patterns
- Reduces adipocyte hypertrophy and subsequent inflammatory cytokine release
- Enhances insulin sensitivity at the cellular level
- Modulates macrophage polarization within adipose tissue
Simultaneously, the GLP-1 receptor pathway contributes its own anti-inflammatory effects. GLP-1 receptors appear on various immune cells, including monocytes and macrophages, where receptor activation directly suppresses pro-inflammatory cytokine production. This dual-pronged approach creates synergistic effects that single-pathway agonists cannot replicate.
Molecular Signaling Cascades
At the molecular level, tirzepatide influences several critical signaling pathways implicated in chronic inflammation:
| Pathway | Mechanism | Inflammatory Impact |
|---|---|---|
| NF-κB Suppression | Inhibits nuclear translocation of inflammatory transcription factors | Reduces IL-6, TNF-α, and IL-1β expression |
| AMPK Activation | Enhances cellular energy sensing and metabolic efficiency | Decreases oxidative stress and inflammatory mediators |
| PKA Signaling | Increases cAMP levels through receptor activation | Modulates immune cell function and cytokine production |
| mTOR Modulation | Influences cellular growth and autophagy pathways | Affects inflammatory cell differentiation and survival |
Research utilizing high-purity tirzepatide has demonstrated that these pathways interact in complex networks rather than operating in isolation. The peptide’s ability to simultaneously influence multiple nodes within inflammatory networks may explain its robust effects on systemic inflammation markers.
Adipose Tissue as an Inflammatory Nexus
Perhaps the most significant contribution to tirzepatide’s anti-inflammatory profile stems from its effects on adipose tissue biology. Dysfunctional adipose tissue acts as an endocrine organ, secreting pro-inflammatory adipokines that drive systemic inflammation. Tirzepatide appears to fundamentally restructure this tissue through:
✅ Enhanced adipogenesis – Promoting the formation of smaller, metabolically healthier adipocytes ✅ Reduced adipocyte death – Preventing necrosis that triggers macrophage infiltration ✅ Improved adipokine profile – Increasing adiponectin while decreasing leptin and resistin ✅ Macrophage repolarization – Shifting from pro-inflammatory M1 to anti-inflammatory M2 phenotypes
These adipose-centric effects create a ripple effect throughout systemic inflammation, as healthier fat tissue releases fewer inflammatory signals into circulation.
Tirzepatide Inflammation Markers: Key Biomarkers and Research Findings
Understanding which inflammation markers respond to tirzepatide treatment provides crucial insights for research design and outcome assessment. Current evidence points to several primary biomarkers that demonstrate consistent modulation across experimental models.
C-Reactive Protein (CRP) Reduction
C-reactive protein stands as one of the most widely studied inflammation markers in tirzepatide research. This acute-phase protein, produced primarily by hepatocytes in response to IL-6 signaling, serves as a reliable indicator of systemic inflammation.
Research findings from 2024-2025 studies demonstrate:
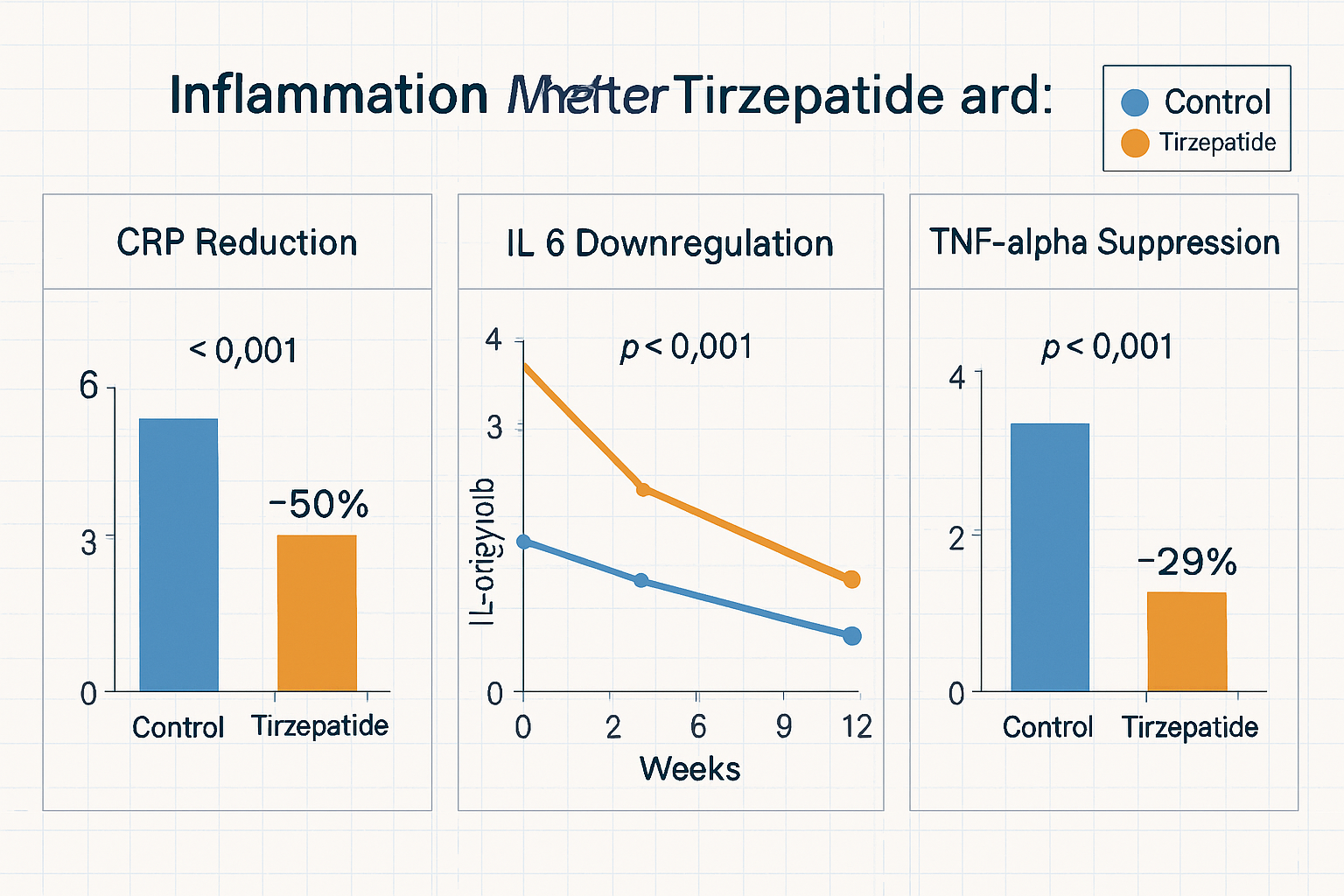
- 20-35% reductions in high-sensitivity CRP (hs-CRP) levels across multiple experimental protocols
- Dose-dependent responses, with higher tirzepatide concentrations producing more pronounced CRP suppression
- Temporal patterns showing initial CRP reduction within 4-6 weeks of treatment initiation
- Correlation with weight changes, though CRP reductions often exceed what would be predicted by adipose loss alone
The magnitude of CRP reduction observed with tirzepatide frequently surpasses that seen with comparable metabolic interventions, suggesting direct anti-inflammatory mechanisms beyond indirect effects through weight modulation.
Interleukin-6 (IL-6) and Cytokine Profiles
Interleukin-6 represents a central pro-inflammatory cytokine that drives numerous inflammatory cascades. Tirzepatide’s influence on IL-6 levels appears particularly robust:
🔬 Direct suppression of IL-6 production in adipocytes and immune cells 🔬 Reduced IL-6 receptor signaling through downstream pathway modulation 🔬 Decreased IL-6-induced acute phase responses, including CRP production 🔬 Altered IL-6 trans-signaling, which affects inflammatory cell recruitment
Beyond IL-6, tirzepatide research has documented effects on broader cytokine networks:
- TNF-α (Tumour Necrosis Factor-alpha): 15-25% reductions observed in multiple models
- IL-1β (Interleukin-1 beta): Decreased expression in adipose tissue macrophages
- IL-10 (Interleukin-10): Increased levels of this anti-inflammatory cytokine
- MCP-1 (Monocyte Chemoattractant Protein-1): Reduced chemokine levels limiting inflammatory cell recruitment
Adipokine Remodeling
The adipokine profile provides unique insight into tirzepatide’s metabolic and inflammatory effects. Research-grade tirzepatide preparations have enabled precise measurement of these changes:
Adiponectin ⬆️ – This anti-inflammatory adipokine typically increases 30-50% with tirzepatide treatment, correlating with improved insulin sensitivity and reduced inflammatory tone.
Leptin ⬇️ – Pro-inflammatory leptin levels decrease proportionally with adipose tissue reduction, though the decline often exceeds simple mass-related predictions.
Resistin ⬇️ – This inflammatory adipokine shows consistent suppression, particularly in visceral adipose depots.
Visfatin ⬇️ – Reduced levels indicate improved adipose tissue health and decreased inflammatory signaling.
Oxidative Stress Markers
Oxidative stress and inflammation exist in a bidirectional relationship, each amplifying the other. Tirzepatide inflammation markers research increasingly includes oxidative stress parameters:
| Marker | Direction | Mechanism |
|---|---|---|
| Malondialdehyde (MDA) | ⬇️ Decreased | Reduced lipid peroxidation |
| 8-OHdG | ⬇️ Decreased | Less oxidative DNA damage |
| Superoxide Dismutase (SOD) | ⬆️ Increased | Enhanced antioxidant capacity |
| Glutathione Peroxidase | ⬆️ Increased | Improved oxidative defense |
| Advanced Glycation End Products | ⬇️ Decreased | Reduced protein glycation |
These oxidative stress improvements complement direct anti-inflammatory effects, creating a comprehensive reduction in inflammatory burden.
Novel Inflammatory Biomarkers
Emerging research in 2025 has expanded the panel of tirzepatide inflammation markers to include more specialized indicators:
Galectin-3 – This protein involved in fibrosis and inflammation shows promising reductions with tirzepatide treatment, suggesting potential cardiovascular and metabolic benefits.
Fetuin-A – Decreased levels indicate improved hepatic inflammatory status and insulin sensitivity.
Soluble ICAM-1 and VCAM-1 – These endothelial adhesion molecules decline with tirzepatide, suggesting improved vascular health and reduced inflammatory cell adhesion.
High-Mobility Group Box 1 (HMGB1) – This damage-associated molecular pattern (DAMP) protein shows suppression, indicating reduced inflammatory signaling from cellular stress.
For researchers designing experiments to assess these markers, access to research-grade peptides with verified purity becomes essential for reproducible results.
Research Applications and Experimental Considerations
Investigating tirzepatide inflammation markers requires careful experimental design and rigorous methodology. The following considerations prove critical for generating reliable, reproducible data.
Experimental Model Selection
Different research models offer distinct advantages for studying tirzepatide’s anti-inflammatory properties:
In Vitro Cell Culture Models
- Adipocyte cultures for direct inflammatory marker assessment
- Macrophage co-culture systems to evaluate immune cell interactions
- Hepatocyte models for acute-phase protein production studies
- Endothelial cell systems for vascular inflammation research
Ex Vivo Tissue Studies
- Adipose tissue explants maintaining cellular architecture
- Liver tissue preparations for hepatic inflammation assessment
- Vascular tissue for endothelial function evaluation
In Vivo Animal Models
- Diet-induced obesity models for metabolic inflammation
- Genetic obesity models (ob/ob, db/db) for severe metabolic dysfunction
- Inflammatory disease models for specific pathway investigation
- Longitudinal studies tracking inflammation marker trajectories
Each model requires specific tirzepatide dosing protocols, with concentrations scaled appropriately to the experimental system. High-purity tirzepatide ensures consistent receptor activation across experimental conditions.
Dosing Protocols and Timing Considerations
Optimal experimental design must account for tirzepatide’s pharmacokinetics and the temporal dynamics of inflammatory responses:
Acute Studies (Hours to Days)
- Focus on immediate signaling pathway activation
- Assess rapid changes in inflammatory gene expression
- Monitor early cytokine release patterns
- Typical concentrations: 10-100 nM in cell culture
Subacute Studies (1-4 Weeks)
- Evaluate sustained inflammatory marker changes
- Assess adipose tissue remodeling initiation
- Monitor cytokine profile evolution
- Typical dosing: Daily to twice-weekly administration
Chronic Studies (4+ Weeks)
- Measure comprehensive inflammatory profile changes
- Assess tissue-level remodeling and macrophage polarization
- Evaluate systemic inflammation marker trajectories
- Typical protocols: Weekly to twice-weekly dosing
Sample Collection and Processing
Proper biospecimen handling proves critical for accurate inflammation marker assessment:
🔬 Timing – Collect samples at consistent circadian timepoints, as many inflammatory markers exhibit diurnal variation
🔬 Processing speed – Minimize time between collection and stabilization, particularly for labile cytokines
🔬 Storage conditions – Maintain -80°C storage for most inflammatory proteins; avoid freeze-thaw cycles
🔬 Anticoagulants – Select appropriate collection tubes (EDTA, heparin, or serum) based on specific marker requirements
🔬 Protease inhibitors – Consider adding inhibitor cocktails for cytokine-rich samples
Analytical Methods for Inflammation Markers
Multiple analytical platforms enable tirzepatide inflammation marker quantification:
| Method | Advantages | Considerations |
|---|---|---|
| ELISA | High sensitivity, quantitative, widely available | Single-marker focus, requires optimization |
| Multiplex Immunoassays | Simultaneous multi-marker assessment, sample efficiency | Higher cost, potential cross-reactivity |
| Western Blotting | Protein size confirmation, pathway assessment | Semi-quantitative, labour-intensive |
| RT-qPCR | Gene expression analysis, mechanistic insights | Requires RNA quality, indirect protein measure |
| Flow Cytometry | Cell-specific marker expression, immune phenotyping | Requires fresh samples, complex analysis |
| Mass Spectrometry | Unbiased proteomics, novel marker discovery | Expensive, requires specialized expertise |
Researchers should validate findings across multiple platforms when possible, as different methods may reveal complementary aspects of tirzepatide’s anti-inflammatory effects.
Controls and Comparators
Rigorous experimental design requires appropriate control conditions:
✓ Vehicle controls – Match solvent and excipient composition ✓ Positive controls – Include known anti-inflammatory agents for comparison ✓ Single-pathway agonists – Compare with GLP-1-only or GIP-only agonists to assess synergy ✓ Dose-response curves – Establish concentration-dependent relationships ✓ Time-course studies – Map temporal dynamics of inflammatory responses
Quality Assurance for Research Peptides
The reliability of tirzepatide inflammation marker research depends fundamentally on peptide quality. When sourcing research-grade tirzepatide, researchers should verify:
- Purity certification – HPLC analysis confirming >95% purity
- Mass spectrometry confirmation – Verifying correct molecular weight
- Endotoxin testing – Ensuring LAL assay results <1 EU/mg to prevent confounding inflammatory signals
- Storage conditions – Proper lyophilization and cold-chain maintenance
- Certificate of Analysis (COA) – Comprehensive documentation of quality metrics
Contamination with endotoxins or degradation products can artificially influence inflammation markers, confounding experimental results. Suppliers like PEPTIDE PRO provide detailed quality documentation to support rigorous research standards.
Mechanistic Insights: How Tirzepatide Modulates Inflammation
Understanding the precise mechanisms through which tirzepatide influences inflammation markers enables more sophisticated experimental design and therapeutic hypothesis generation.
Direct Receptor-Mediated Anti-Inflammatory Effects
Both GIP and GLP-1 receptors appear on various immune cells, enabling direct anti-inflammatory signaling:
Macrophage Modulation Tirzepatide binding to receptors on macrophages triggers:
- Reduced NF-κB activation and nuclear translocation
- Decreased production of IL-6, TNF-α, and IL-1β
- Shift from M1 (pro-inflammatory) to M2 (anti-inflammatory) polarization
- Enhanced phagocytic capacity with reduced inflammatory cytokine release
T-Cell Effects Emerging evidence suggests tirzepatide influences adaptive immunity:
- Reduced Th17 cell differentiation and IL-17 production
- Enhanced regulatory T-cell (Treg) function
- Decreased T-cell infiltration into adipose tissue
- Modulation of T-cell receptor signaling pathways
Dendritic Cell Function Preliminary data indicates effects on antigen-presenting cells:
- Reduced maturation and co-stimulatory molecule expression
- Decreased pro-inflammatory cytokine secretion
- Altered T-cell priming capacity
Metabolic Pathway Integration
Tirzepatide’s metabolic effects create indirect anti-inflammatory consequences:
� Improved insulin sensitivity reduces inflammatory signaling associated with insulin resistance
� Enhanced glucose disposal decreases advanced glycation end products (AGEs) that trigger inflammatory receptors
� Lipid metabolism optimization reduces lipotoxicity and associated inflammatory responses
� Mitochondrial function improvement decreases oxidative stress and inflammatory signaling
� Autophagy enhancement removes damaged cellular components that otherwise trigger inflammatory pathways
Adipose Tissue Remodeling as Central Mechanism
The profound effects on adipose tissue biology appear central to systemic inflammation reduction:
Adipocyte Size and Function
- Promotion of smaller, insulin-sensitive adipocytes
- Reduced adipocyte hypertrophy and subsequent hypoxia
- Decreased adipocyte death and inflammatory debris
- Enhanced adipogenic capacity preventing ectopic lipid deposition
Adipose Tissue Macrophage Dynamics
- Reduced macrophage recruitment through decreased chemokine expression
- Shift from crown-like structures surrounding dead adipocytes
- M2 polarization promoting tissue remodeling rather than inflammation
- Enhanced efferocytosis (clearance of dead cells) without inflammatory activation
Adipose Tissue Fibrosis
- Reduced collagen deposition and tissue stiffening
- Decreased expression of fibrotic markers (TGF-β, collagen I/III)
- Improved adipose tissue expandability and metabolic flexibility
Gut-Inflammation Axis
GLP-1 receptor activation influences intestinal permeability and gut-derived inflammation:
🔹 Strengthened intestinal barrier – Reduced “leaky gut” and endotoxin translocation 🔹 Microbiome modulation – Shifts in bacterial composition toward anti-inflammatory species 🔹 Reduced endotoxemia – Lower circulating lipopolysaccharide (LPS) levels 🔹 Intestinal immune regulation – Modulation of gut-associated lymphoid tissue (GALT) function
These gut-mediated effects contribute to systemic inflammation reduction, as decreased bacterial product translocation reduces continuous immune system activation.
Hepatic Inflammation Pathways
The liver represents both a source and target of inflammatory mediators:
Hepatic Inflammatory Marker Production
- Reduced IL-6-stimulated acute phase protein synthesis
- Decreased CRP production through multiple mechanisms
- Lower fibrinogen and serum amyloid A levels
Hepatic Steatosis Reduction
- Decreased intrahepatic triglyceride accumulation
- Reduced lipotoxicity-associated inflammation
- Lower expression of inflammatory genes in hepatocytes
Kupffer Cell Modulation
- Reduced activation of liver-resident macrophages
- Decreased inflammatory cytokine secretion
- Improved hepatic insulin sensitivity
Cardiovascular Inflammation
Tirzepatide’s effects extend to vascular inflammation, with implications for cardiovascular research:
Endothelial Function
- Reduced expression of adhesion molecules (ICAM-1, VCAM-1)
- Decreased inflammatory cell adhesion and transmigration
- Improved nitric oxide bioavailability
- Enhanced endothelial repair mechanisms
Vascular Smooth Muscle
- Reduced inflammatory signaling in vessel walls
- Decreased vascular remodeling associated with inflammation
- Lower expression of matrix metalloproteinases
Atherosclerotic Plaque Stability
- Reduced inflammatory cell infiltration into plaques
- Decreased vulnerable plaque characteristics
- Potential stabilization through inflammation reduction
For researchers investigating cardiovascular inflammation, tirzepatide research peptides enable detailed mechanistic studies in relevant model systems.
Comparative Analysis: Tirzepatide Versus Other Anti-Inflammatory Approaches
Contextualizing tirzepatide’s anti-inflammatory profile against other interventions helps researchers understand its unique position in the therapeutic landscape.
Tirzepatide vs. GLP-1 Receptor Agonists
Single-pathway GLP-1 receptor agonists like semaglutide demonstrate anti-inflammatory properties, but tirzepatide’s dual mechanism appears to confer advantages:
| Parameter | GLP-1 Agonists | Tirzepatide | Advantage |
|---|---|---|---|
| CRP Reduction | 15-25% | 25-35% | Tirzepatide |
| IL-6 Suppression | Moderate | Pronounced | Tirzepatide |
| Adiponectin Increase | 20-30% | 35-50% | Tirzepatide |
| Adipose Remodeling | Present | Enhanced | Tirzepatide |
| Weight Loss | Significant | Greater | Tirzepatide |
The enhanced effects likely stem from GIP receptor contributions to adipose tissue biology and the synergistic interaction between pathways. Researchers comparing semaglutide and tirzepatide in parallel experiments can quantify these differential effects.
Tirzepatide vs. Traditional Anti-Inflammatory Agents
Comparing tirzepatide with conventional anti-inflammatory approaches reveals distinct profiles:
NSAIDs (Non-Steroidal Anti-Inflammatory Drugs)
- Mechanism: COX enzyme inhibition
- Inflammation markers: Primarily prostaglandin-mediated
- Tirzepatide advantage: Broader inflammatory pathway modulation without gastrointestinal or cardiovascular risks
Corticosteroids
- Mechanism: Glucocorticoid receptor activation
- Inflammation markers: Broad suppression across multiple pathways
- Tirzepatide advantage: Metabolic benefits rather than metabolic side effects; sustainable long-term use
Metformin
- Mechanism: AMPK activation, mitochondrial effects
- Inflammation markers: Modest CRP and cytokine reduction
- Tirzepatide advantage: Greater magnitude of inflammatory marker changes; complementary mechanisms
Statins
- Mechanism: HMG-CoA reductase inhibition with pleiotropic effects
- Inflammation markers: CRP reduction (10-20%)
- Tirzepatide advantage: Comparable or superior CRP effects with additional metabolic benefits
Combination Approaches
Research exploring tirzepatide in combination with other anti-inflammatory interventions may reveal synergistic effects:
✅ Tirzepatide + Metformin – Complementary AMPK activation and incretin effects ✅ Tirzepatide + Omega-3 Fatty Acids – Additive anti-inflammatory lipid mediator production ✅ Tirzepatide + Antioxidants – Enhanced oxidative stress reduction ✅ Tirzepatide + Probiotics – Synergistic gut barrier and microbiome effects
Such combination studies require careful experimental design with appropriate factorial controls to distinguish additive from synergistic effects.
Future Directions in Tirzepatide Inflammation Research
The field of tirzepatide inflammation markers research continues to evolve rapidly, with several promising directions emerging in 2025.
Novel Inflammatory Pathways and Markers
Emerging research explores tirzepatide’s effects on previously unexamined inflammatory mechanisms:
Inflammasome Activation
- NLRP3 inflammasome suppression and reduced IL-1β maturation
- Effects on pyroptosis (inflammatory cell death)
- Modulation of damage-associated molecular patterns (DAMPs)
Resolution of Inflammation
- Specialized pro-resolving mediators (SPMs) like resolvins and lipoxins
- Active inflammation resolution rather than simple suppression
- Temporal dynamics of inflammatory resolution
Epigenetic Modifications
- DNA methylation changes in inflammatory gene promoters
- Histone modifications affecting inflammatory gene expression
- Long-term inflammatory “memory” and tirzepatide’s resetting effects
Extracellular Vesicle Communication
- Exosome-mediated inflammatory signaling
- Micro-RNA cargo affecting inflammatory gene expression
- Cell-to-cell communication in inflammatory networks
Tissue-Specific Inflammation Studies
Future research will likely dissect tirzepatide’s effects in specific tissues:
🔬 Neuroinflammation – Central nervous system inflammatory markers and microglial activation 🔬 Renal inflammation – Kidney inflammatory pathways in diabetic nephropathy models 🔬 Pancreatic inflammation – Beta-cell inflammatory stress and function 🔬 Skeletal muscle inflammation – Myokine profiles and insulin resistance 🔬 Bone marrow inflammation – Hematopoietic stem cell niche and immune cell production
Personalized Inflammation Response Profiling
Individual variability in inflammatory responses to tirzepatide represents an important research frontier:
- Genetic polymorphisms affecting receptor expression and signaling
- Baseline inflammatory status influencing response magnitude
- Sex differences in inflammatory marker modulation
- Age-related variations in anti-inflammatory effects
- Disease-state specific inflammatory profiles
Advanced Analytical Technologies
Emerging technologies will enable more comprehensive inflammation marker assessment:
Single-Cell Analysis
- Cell-type-specific inflammatory responses within heterogeneous tissues
- Identification of rare cell populations driving inflammation
- Temporal dynamics at cellular resolution
Spatial Transcriptomics
- Tissue architecture and inflammatory cell localization
- Spatial relationships between cell types in inflammatory niches
- Regional variation in inflammatory marker expression
Systems Biology Approaches
- Network analysis of inflammatory pathway interactions
- Computational modeling of tirzepatide’s multi-pathway effects
- Predictive models for inflammatory marker responses
Metabolomics and Lipidomics
- Comprehensive inflammatory mediator profiling
- Lipid-derived inflammatory and resolving molecules
- Metabolic signatures of inflammatory state changes
Clinical Translation Considerations
While this article focuses on research applications, understanding potential clinical translation pathways helps guide experimental priorities:
- Identification of predictive biomarkers for clinical response
- Optimal inflammation marker panels for monitoring
- Correlation between experimental models and clinical outcomes
- Translation of mechanistic insights into therapeutic strategies
For researchers preparing studies that may inform clinical development, access to high-quality research peptides with comprehensive documentation becomes essential for regulatory-grade data generation.
Practical Guidance for Researchers
Successfully investigating tirzepatide inflammation markers requires attention to numerous practical details that ensure experimental rigor and reproducibility.
Peptide Handling and Storage
Proper peptide management begins before experiments commence:
Lyophilized Peptide Storage
- Maintain at -20°C or -80°C in original sealed pen peptides
- Protect from moisture and light exposure
- Minimize freeze-thaw cycles of unopened pen peptides
- Document storage duration and conditions
Reconstitution Protocols
- Use appropriate solvents (sterile water, saline, or buffer as specified)
- Calculate concentrations precisely for experimental requirements
- Allow peptide to dissolve completely without vigorous agitation
- Filter sterilize if sterility is required for cell culture applications
Working Solution Preparation
- Prepare aliquots to avoid repeated freeze-thaw
- Store reconstituted peptide at 4°C for short-term use (days) or -20°C for longer periods
- Document reconstitution date and concentration
- Verify stability under specific storage conditions
Experimental Design Checklist
Before initiating tirzepatide inflammation marker studies, researchers should confirm:
☑️ Peptide quality verification – COA review, purity confirmation, endotoxin levels ☑
️ Appropriate controls – Vehicle, positive control, comparator compounds ☑
️ Sample size calculation – Statistical power analysis for expected effect sizes ☑
️ Randomization strategy – Blinding and randomization protocols ☑
️ Timing optimization – Sample collection timepoints based on marker kinetics ☑
️ Analytical method validation – Assay sensitivity, specificity, reproducibility ☑
️ Data management plan – Standardized recording and analysis procedures
Common Pitfalls and Solutions
Experienced researchers have identified several common challenges in tirzepatide inflammation research:
Challenge: Inconsistent Results Across Experiments
- Solution: Verify peptide stability; standardize reconstitution protocols; control for circadian timing of sample collection
Challenge: High Baseline Variability in Inflammation Markers
- Solution: Increase sample sizes; use within-subject designs when possible; standardize pre-experimental conditions (diet, housing, stress)
Challenge: Confounding by Weight Changes
- Solution: Include weight-matched controls; perform pair-feeding studies; analyze inflammation markers independent of weight effects
Challenge: Endotoxin Contamination
- Solution: Source endotoxin-tested peptides; use sterile technique; include LAL assay verification; filter solutions through endotoxin-removing filters
Challenge: Marker Detection Below Assay Sensitivity
- Solution: Select high-sensitivity assay platforms; concentrate samples when appropriate; choose markers with robust expression in experimental model
Collaboration and Resource Sharing
The complexity of inflammation research often benefits from collaborative approaches:
- Core facilities – Leverage institutional resources for specialized assays
- Bioinformatics support – Engage computational experts for systems-level analysis
- Method sharing – Publish detailed protocols to enhance reproducibility
- Data repositories – Contribute to shared databases advancing the field
Regulatory and Ethical Considerations
Research involving tirzepatide must adhere to appropriate oversight:
For In Vitro Studies
- Institutional biosafety approval for cell culture work
- Proper disposal of biological materials
- Documentation of peptide sources and handling
For Animal Studies
- Institutional Animal Care and Use Committee (IACUC) approval
- Adherence to ARRIVE guidelines for reporting
- Humane endpoints and welfare monitoring
- Justification of animal numbers and experimental design
For Human Biospecimen Studies
- Institutional Review Board (IRB) approval
- Informed consent procedures
- Data privacy and security measures
- Clear labeling of research-only materials
All tirzepatide used in research settings must be clearly designated “For Research Use Only” and never for human consumption, as emphasized by suppliers like PEPTIDE PRO.
Integration with Broader Research Programs
Tirzepatide inflammation marker research rarely exists in isolation; it typically integrates with broader scientific objectives investigating metabolic disease, therapeutic development, or fundamental biology.
Multi-Omic Integration
Comprehensive understanding emerges when inflammation marker data integrates with other molecular layers:
Transcriptomics
- Gene expression changes correlating with inflammatory marker levels
- Identification of upstream regulatory mechanisms
- Temporal gene expression dynamics during inflammation resolution
Proteomics
- Protein-level validation of transcriptional changes
- Post-translational modifications affecting inflammatory protein function
- Protein-protein interaction networks in inflammatory signaling
Metabolomics
- Metabolic signatures associated with inflammatory states
- Lipid mediators linking metabolism and inflammation
- Energy metabolism changes affecting inflammatory responses
Genomics
- Genetic variants influencing inflammatory marker responses
- Epigenetic modifications affecting inflammatory gene regulation
- Chromatin accessibility changes in inflammatory pathways
Longitudinal Study Design
Tracking inflammation marker trajectories over extended periods reveals temporal dynamics:
- Acute phase responses (hours to days)
- Subacute adaptation (weeks)
- Chronic remodeling (months)
- Sustained effects after treatment cessation
Longitudinal designs require careful consideration of repeated sampling effects, age-related changes, and environmental stability throughout study duration.
Comparative Effectiveness Research
Positioning tirzepatide within the broader landscape of interventions requires head-to-head comparisons:
- Direct comparison with other incretin-based therapies
- Evaluation against established anti-inflammatory agents
- Assessment of combination versus monotherapy approaches
- Cost-effectiveness analysis for research resource allocation
Such comparative studies benefit from standardized protocols and shared control groups to minimize inter-study variability.
Mechanism-Based Biomarker Development
Inflammation marker research can identify biomarkers predicting:
- Optimal responders to tirzepatide treatment
- Early indicators of therapeutic effect
- Mechanisms of resistance or non-response
- Surrogate endpoints for longer-term outcomes
Biomarker development requires validation across multiple cohorts and demonstration of clinical or experimental utility.
The investigation of tirzepatide inflammation markers represents a rapidly advancing frontier in metabolic research, revealing this dual GIP/GLP-1 receptor agonist’s profound anti-inflammatory properties that extend well beyond glycemic control. Research throughout 2025 has consistently demonstrated significant reductions in key inflammatory biomarkers including CRP, IL-6, TNF-α, and numerous adipokines, while simultaneously improving oxidative stress parameters and promoting healthier adipose tissue biology.
The mechanistic complexity underlying these effects—spanning direct receptor-mediated immune cell modulation, metabolic pathway optimization, adipose tissue remodeling, and gut-inflammation axis regulation—positions tirzepatide as a uniquely multi-faceted intervention worthy of continued rigorous investigation. For researchers, the availability of high-purity research-grade tirzepatide enables precise experimental protocols that dissect these mechanisms with unprecedented clarity.
Key Research Priorities Moving Forward
As the field advances, several priorities emerge for maximizing scientific impact:
- Standardize methodologies across laboratories to enhance reproducibility and enable meta-analyses
- Explore tissue-specific mechanisms to understand regional variation in inflammatory responses
- Investigate combination approaches that may yield synergistic anti-inflammatory effects
- Develop predictive biomarkers identifying optimal experimental conditions and response patterns
- Integrate multi-omic datasets for comprehensive systems-level understanding
Actionable Next Steps for Researchers
For those planning tirzepatide inflammation marker studies:
✓ Source high-quality peptides with comprehensive quality documentation from reputable suppliers like PEPTIDE PRO
✓ Design rigorous protocols incorporating appropriate controls, statistical power, and temporal sampling
✓ Select relevant marker panels based on experimental model and research questions
✓ Validate findings across multiple analytical platforms and experimental systems
✓ Contribute to open science through detailed protocol sharing and data repository participation
✓ Consider mechanistic depth beyond simple marker quantification to pathway-level understanding
The anti-inflammatory properties of tirzepatide represent more than an ancillary benefit—they appear fundamental to its metabolic effects and may unlock novel therapeutic applications. As research continues to elucidate the intricate relationships between tirzepatide, inflammation markers, and metabolic health, the scientific community moves closer to comprehensive understanding of how this remarkable peptide reshapes inflammatory biology at multiple scales.
For researchers seeking to contribute to this expanding knowledge base, the combination of sophisticated experimental design, high-quality research materials, and rigorous analytical approaches promises continued discoveries that advance both fundamental biology and translational applications.
