
Researchers investigating novel metabolic peptides have turned their attention to an emerging concern: retatrutide fatigue. As this triple-agonist peptide continues to advance through clinical development, understanding its complete side effect profile—including potential fatigue-related responses—has become essential for laboratories and research institutions conducting metabolic studies. While gastrointestinal effects have dominated early safety discussions, the question of whether retatrutide fatigue represents a significant research consideration demands thorough examination.
This comprehensive guide explores the current state of knowledge regarding retatrutide fatigue, examining available clinical data, potential mechanisms, and research considerations for laboratories working with this compound.
Key Takeaways
- Limited Direct Evidence: Current published clinical trial data does not extensively document fatigue as a primary adverse event in retatrutide research, though comprehensive Phase 3 results are still emerging
- Mechanism Considerations: Retatrutide’s triple-agonist activity on GLP-1, GIP, and glucagon receptors may theoretically influence energy metabolism through multiple pathways
- Research Context Matters: Proper peptide handling, reconstitution protocols, and storage conditions are essential for maintaining compound integrity in laboratory settings
- Individual Variability: Research models may exhibit different responses to retatrutide based on metabolic baseline, dosing protocols, and concurrent factors
- Quality Sourcing: Working with high-purity research-grade peptides ensures consistent experimental results and reduces confounding variables
Understanding Retatrutide: The Triple-Agonist Peptide
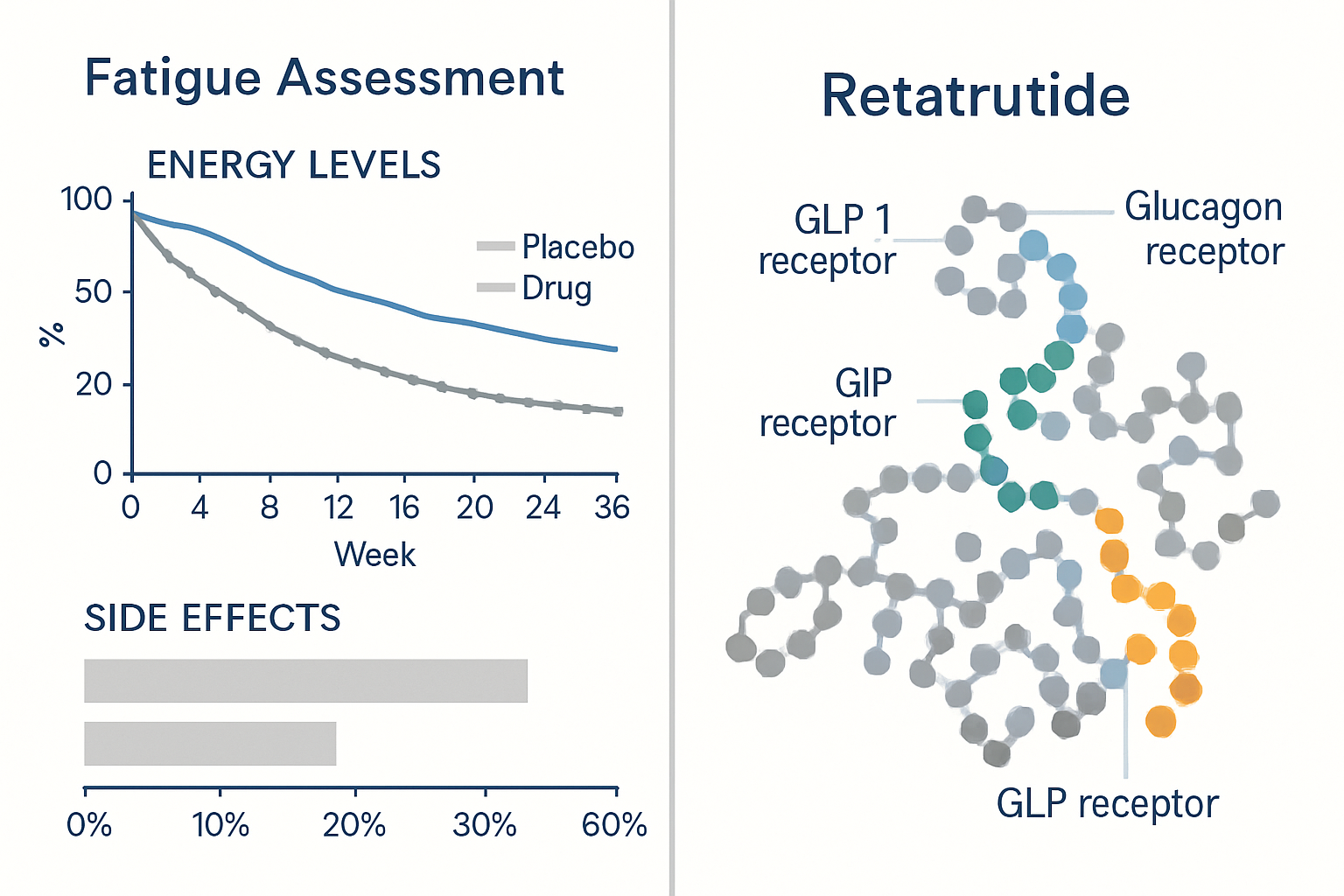
What Is Retatrutide?
Retatrutide represents a novel class of metabolic peptide designed to simultaneously activate three distinct receptor pathways: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This triple-agonist mechanism distinguishes it from earlier single or dual-agonist compounds in metabolic research.
Key Characteristics:
| Property | Details |
|---|---|
| Mechanism | Triple receptor agonist (GLP-1/GIP/Glucagon) |
| Primary Research Focus | Metabolic regulation, weight management studies |
| Development Stage | Phase 3 clinical trials (as of 2025) |
| Administration | Subcutaneous injection protocols |
| Storage Requirements | Lyophilized form: cool, dry conditions; Reconstituted: 2-8°C |
Mechanism of Action and Metabolic Effects
The triple-agonist activity of retatrutide creates a complex metabolic profile:
🔬 GLP-1 Receptor Activation: Influences satiety signaling, glucose regulation, and gastric motility
🔬 GIP Receptor Activation: Modulates insulin secretion, lipid metabolism, and energy storage
🔬 Glucagon Receptor Activation: Affects energy expenditure, hepatic glucose production, and fat oxidation
This multi-pathway approach theoretically provides more comprehensive metabolic effects than single-target compounds, but it also introduces additional complexity when evaluating potential side effects like fatigue.
Retatrutide Fatigue: Current Research Evidence
What Clinical Data Reveals
As of 2025, published clinical trial data for retatrutide has primarily focused on efficacy outcomes (weight reduction, metabolic parameters) and the most common adverse events—predominantly gastrointestinal in nature. Retatrutide fatigue has not been prominently featured in available trial summaries, though this does not definitively rule out its occurrence.
Documented Side Effect Profile:
✅ Most Common: Nausea, diarrhea, vomiting, constipation (mild to moderate severity)
✅ Discontinuation Rates: 6-16% of participants in early trials discontinued due to adverse events
❓ Fatigue Reporting: Not specifically highlighted in current published summaries
⚠️ Important Limitation: Complete adverse event data from Phase 3 trials may not yet be publicly available, and comprehensive safety profiles typically emerge as research programs mature.
Why Fatigue Data May Be Limited
Several factors explain the current gap in retatrutide fatigue documentation:
- Reporting Thresholds: Clinical trials often prioritize adverse events that occur above specific frequency thresholds or severity levels
- Subjective Nature: Fatigue represents a subjective symptom that may be influenced by multiple confounding factors
- Publication Timeline: Detailed safety analyses typically follow primary efficacy publications
- Research Focus: Early-stage trials emphasize dose-finding and primary endpoints rather than comprehensive side effect profiling
For researchers requiring detailed adverse event information, consulting primary sources such as ClinicalTrials.gov registries or FDA submission documents provides the most comprehensive data.
Potential Mechanisms Behind Retatrutide Fatigue
Metabolic Pathway Considerations
Even without extensive clinical documentation, understanding the theoretical mechanisms by which retatrutide fatigue might occur provides valuable research context:
1. Caloric Restriction Effects
Retatrutide’s potent effects on satiety and food intake may lead to significant caloric reduction. In metabolic research models, rapid caloric deficits can produce:
- Reduced available energy substrates
- Metabolic adaptation responses
- Temporary energy balance disruption
- Compensatory fatigue signals
2. Glucagon Receptor Activation
The glucagon component of retatrutide’s mechanism increases energy expenditure and fat oxidation. While beneficial for metabolic outcomes, this may theoretically contribute to:
- Increased basal metabolic rate demands
- Altered fuel utilization patterns
- Potential temporary energy perception changes
3. Gastrointestinal Side Effects
The well-documented GI effects of retatrutide could indirectly contribute to fatigue through:
- Nutrient Absorption: Altered absorption patterns affecting micronutrient availability
- Dehydration: Diarrhea or vomiting leading to fluid and electrolyte imbalances
- Sleep Disruption: Nausea or discomfort affecting sleep quality
- Reduced Intake: Decreased food consumption affecting energy availability
4. Hormonal Adaptations
Multi-receptor agonism may trigger complex hormonal responses:
- Insulin sensitivity changes
- Thyroid function adaptations
- Cortisol response patterns
- Growth hormone interactions
Comparing to Related Compounds
Examining fatigue profiles of related metabolic peptides provides additional context:
| Compound | Mechanism | Fatigue Reporting |
|---|---|---|
| Semaglutide | GLP-1 agonist | Occasionally reported; typically mild and transient |
| Tirzepatide | GLP-1/GIP dual agonist | Not prominently featured in major trials |
| Retatrutide | GLP-1/GIP/Glucagon triple agonist | Limited current documentation |
Researchers working with semaglutide or tirzepatide may observe that fatigue, when present, often correlates with the initial adjustment period or aggressive dosing protocols.
Research Considerations for Retatrutide Fatigue
Laboratory Protocol Factors
When investigating potential retatrutide fatigue in research settings, several methodological considerations warrant attention:
Dosing Protocols
📊 Escalation Schedules: Gradual dose titration versus rapid initiation may influence adaptation responses
📊 Frequency: Weekly versus more frequent administration patterns
📊 Concentration: Proper reconstitution ensuring accurate dosing
Quality and Purity Variables
Research outcomes depend fundamentally on compound integrity. Working with research-grade peptides from reputable suppliers ensures:
- ✅ Consistent purity levels (typically >98%)
- ✅ Proper storage and handling throughout supply chain
- ✅ Accurate Certificate of Analysis documentation
- ✅ Minimal degradation or contamination
Storage Best Practices:
| Form | Storage Conditions | Stability Considerations |
|---|---|---|
| Lyophilized (unopened) | Cool, dry place away from light | Extended stability when properly stored |
| Reconstituted | Refrigerated 2-8°C | Use promptly; minimize freeze-thaw cycles |
| In-use | Maintain cold chain | Protect from temperature excursions |
Baseline Assessments
Comprehensive research protocols should establish baseline measurements before retatrutide administration:
- Energy expenditure metrics
- Subjective fatigue scales (if applicable to research model)
- Metabolic rate assessments
- Nutritional intake monitoring
- Sleep pattern documentation
- Electrolyte and hydration status
Confounding Variables
Distinguishing retatrutide fatigue from other factors requires careful experimental design:
🔍 Dietary Changes: Caloric restriction itself may produce fatigue independent of peptide effects
🔍 Hydration Status: GI side effects leading to fluid imbalance
🔍 Concurrent Interventions: Other experimental variables affecting energy levels
🔍 Circadian Factors: Timing of administration relative to activity cycles
🔍 Environmental Conditions: Temperature, humidity, and housing affecting metabolic demands
Managing Potential Fatigue in Research Settings
Optimization Strategies
For researchers observing potential fatigue-related responses in retatrutide studies, several optimization approaches merit consideration:
1. Dose Titration Protocols
Implementing gradual dose escalation may allow better physiological adaptation:
- Start with lower initial doses
- Extend time between dose increases
- Monitor response at each titration step
- Document threshold doses for any fatigue emergence
2. Nutritional Optimization
Ensuring adequate nutrient availability despite reduced intake:
- Monitor macronutrient composition
- Assess micronutrient sufficiency
- Evaluate protein intake adequacy
- Consider supplementation protocols where appropriate
3. Hydration and Electrolyte Management
Proactive fluid and electrolyte monitoring:
- Regular hydration status assessment
- Electrolyte panel monitoring
- Supplementation protocols if deficiencies emerge
- Adjustment for GI fluid losses
4. Administration Timing
Strategic timing of peptide administration:
- Evening dosing to minimize daytime impact
- Coordination with feeding schedules
- Consideration of activity cycle patterns
5. Quality Control Measures
Ensuring compound integrity throughout research:
- Source from verified suppliers with quality certifications
- Follow proper reconstitution protocols
- Maintain cold chain integrity
- Document storage conditions
- Regular potency verification
Monitoring and Documentation
Comprehensive data collection enables better understanding of retatrutide fatigue patterns:
Recommended Monitoring Schedule:
| Timepoint | Assessments |
|---|---|
| Baseline | Complete metabolic panel, energy metrics, activity levels |
| Post-Initiation (Days 1-7) | Daily fatigue scoring, GI symptoms, intake monitoring |
| Weekly | Weight, metabolic parameters, comprehensive symptom assessment |
| Monthly | Full panel including hormonal markers, nutrient status |
| End of Study | Comprehensive analysis with pre/post comparisons |
Comparing Retatrutide to Other Metabolic Peptides
Side Effect Profile Context
Understanding retatrutide fatigue requires context within the broader metabolic peptide landscape:
GLP-1 Agonists
Single-mechanism compounds like semaglutide primarily affect:
- Satiety and food intake
- Gastric emptying
- Glucose regulation
Fatigue Profile: Generally minimal; when present, often correlates with rapid weight loss or GI effects
Dual Agonists (GLP-1/GIP)
Compounds like tirzepatide add GIP receptor activity:
- Enhanced metabolic effects
- Potentially improved tolerability
- Synergistic weight management outcomes
Fatigue Profile: Not prominently reported in major clinical trials
Triple Agonists (Retatrutide)
Adding glucagon receptor activation introduces:
- Increased energy expenditure
- Enhanced fat oxidation
- More complex metabolic effects
Theoretical Fatigue Considerations: Greater metabolic complexity may theoretically increase fatigue potential, though current evidence does not strongly support this
Research Applications Across Peptide Classes
Laboratories studying metabolic regulation may work with various peptide compounds. PEPTIDE PRO supplies research-grade versions of multiple metabolic peptides, including:
- AOD9604 for fat metabolism studies
- CJC-1295 for growth hormone research
- BPC-157 for tissue repair investigations
- Ipamorelin for growth hormone secretion studies
Each compound presents unique considerations for research design and side effect profiling.
Future Research Directions
Emerging Data Needs
As retatrutide advances through late-stage development, several research questions regarding fatigue warrant investigation:
🔬 Incidence Quantification: Precise frequency data across different populations and dosing regimens
🔬 Severity Assessment: Standardized grading of fatigue intensity when present
🔬 Time Course Analysis: Onset patterns, duration, and resolution timelines
🔬 Dose-Response Relationships: Correlation between retatrutide dose and fatigue occurrence
🔬 Mechanistic Studies: Physiological pathways underlying any fatigue responses
🔬 Mitigation Strategies: Evidence-based approaches to prevent or manage fatigue
🔬 Comparative Analysis: Head-to-head comparisons with other metabolic peptides
Phase 3 Trial Implications
Ongoing Phase 3 programs for retatrutide (as of 2025) will provide:
- Larger sample sizes for rare adverse event detection
- Longer exposure durations for chronic effect assessment
- Diverse population representation
- Comprehensive safety monitoring protocols
- Standardized adverse event reporting
Researchers should monitor publications from these trials for updated safety information, including detailed retatrutide fatigue data if it emerges as a significant finding.
Practical Guidance for Research Institutions
Sourcing Quality Research Peptides
The foundation of reliable research outcomes begins with compound quality. When sourcing retatrutide or related peptides:
✅ Verify Supplier Credentials: Look for established suppliers with quality certifications and transparent practices
✅ Request Documentation: Certificate of Analysis (CoA), purity verification, and storage history
✅ Assess Shipping Protocols: Temperature-controlled packaging and rapid delivery options
✅ Evaluate Customer Support: Technical expertise and responsive communication
✅ Review Labeling: Clear “For Research Use Only” designation and proper handling instructions
PEPTIDE PRO provides research-grade peptides with:
- 🏆 High purity standards (>98% typical)
- 🏆 Same-day dispatch for orders before 1pm (Mon-Fri)
- 🏆 Temperature-appropriate packaging
- 🏆 Comprehensive product documentation
- 🏆 Expert customer support for research applications
Establishing Research Protocols
Comprehensive protocols for retatrutide research should include:
Pre-Study Planning
- Literature review of current safety data
- Protocol design incorporating fatigue monitoring
- Baseline assessment procedures
- Defined intervention and control conditions
- Statistical power calculations
During Study Execution
- Standardized administration procedures
- Consistent monitoring schedules
- Real-time adverse event documentation
- Quality control checkpoints
- Data integrity measures
Post-Study Analysis
- Comprehensive safety analysis
- Fatigue incidence and severity quantification
- Correlation analyses with other variables
- Comparison to literature benchmarks
- Publication and data sharing
Regulatory and Ethical Considerations
Research institutions must maintain strict compliance:
⚖️ Research Use Only: All peptides must be clearly designated and used exclusively for laboratory research
⚖️ Proper Documentation: Maintain complete records of sourcing, storage, and usage
⚖️ Safety Protocols: Appropriate handling procedures and safety equipment
⚖️ Institutional Oversight: Compliance with institutional review and safety committees
⚖️ Transparent Reporting: Accurate documentation of all findings, including adverse observations
Frequently Asked Questions About Retatrutide Fatigue
Is fatigue a common side effect of retatrutide?
Based on currently available published clinical trial data, fatigue has not been prominently reported as a common adverse event in retatrutide research. The most frequently documented side effects are gastrointestinal in nature (nausea, diarrhea, vomiting). However, comprehensive Phase 3 safety data is still emerging, and complete adverse event profiles may provide additional information.
How does retatrutide fatigue compare to other metabolic peptides?
Current evidence does not suggest that retatrutide produces substantially different fatigue profiles compared to related compounds like semaglutide or tirzepatide. When fatigue occurs with metabolic peptides, it typically correlates with the initial adjustment period, rapid weight loss, or secondary factors like dehydration rather than direct peptide effects.
What mechanisms might cause retatrutide fatigue?
Theoretical mechanisms include: caloric restriction effects from reduced food intake, increased energy expenditure from glucagon receptor activation, indirect effects from GI side effects (dehydration, nutrient malabsorption), and complex hormonal adaptations. However, these remain theoretical considerations pending comprehensive clinical data.
Can proper dosing protocols reduce retatrutide fatigue?
Gradual dose titration, rather than rapid initiation at higher doses, may theoretically improve tolerance and reduce potential fatigue responses by allowing better physiological adaptation. Research protocols incorporating stepwise dose escalation often demonstrate improved overall tolerability.
Where can researchers obtain high-quality retatrutide for studies?
Researchers should source retatrutide from established suppliers specializing in research-grade peptides. PEPTIDE PRO offers research peptides with verified purity, proper storage conditions, and comprehensive documentation—essential factors for reliable research outcomes.
What monitoring should be included in retatrutide research protocols?
Comprehensive protocols should include baseline and ongoing assessments of: energy levels and fatigue scales, metabolic parameters, nutritional intake, hydration and electrolyte status, GI symptoms, sleep quality, and activity levels. This multi-dimensional approach enables better understanding of any fatigue-related responses.
The Current State of Retatrutide Fatigue Research
The question of retatrutide fatigue represents an evolving area of investigation as this promising triple-agonist peptide advances through clinical development. While current published data does not extensively document fatigue as a prominent adverse event, the complexity of retatrutide’s multi-pathway mechanism and the emerging nature of comprehensive safety data warrant continued attention from the research community.
Key Points for Researchers
Evidence Status: Current published clinical trial summaries have not highlighted fatigue as a major concern, though complete Phase 3 adverse event data continues to emerge.
Theoretical Considerations: Retatrutide’s effects on energy metabolism, food intake, and metabolic rate provide plausible mechanisms by which fatigue could theoretically occur, even if not prominently documented in current literature.
Research Quality: Reliable investigation of retatrutide fatigue requires high-purity compounds, proper storage and handling, comprehensive monitoring protocols, and careful attention to confounding variables.
Comparative Context: Retatrutide does not appear to present substantially different fatigue concerns compared to related metabolic peptides based on available evidence.
Future Directions: Ongoing Phase 3 trials and post-marketing surveillance will provide more comprehensive safety data, including detailed information on fatigue incidence, severity, and management if it emerges as a significant finding.
Next Steps for Research Institutions
For laboratories and research institutions investigating retatrutide or planning studies involving this compound:
- Stay Current: Monitor emerging publications from Phase 3 trials and regulatory submissions for updated safety information
- Design Comprehensive Protocols: Incorporate multi-dimensional monitoring that can detect and characterize fatigue responses if they occur
- Source Quality Compounds: Work with established suppliers of research-grade peptides to ensure compound integrity and experimental reliability
- Document Thoroughly: Maintain detailed records of all observations, including any fatigue-related findings, to contribute to the growing knowledge base
- Consider Context: Evaluate any fatigue observations within the broader context of metabolic changes, GI effects, and individual variability
- Implement Best Practices: Follow proper reconstitution, storage, and handling protocols to minimize variables that could confound results
The research community’s understanding of retatrutide fatigue will continue to evolve as more comprehensive data becomes available. By maintaining rigorous research standards, careful observation, and thorough documentation, researchers contribute to the collective knowledge that will ultimately define the complete safety and tolerability profile of this innovative metabolic peptide.
For research institutions seeking high-purity retatrutide and related compounds, PEPTIDE PRO provides research-grade peptides with same-day dispatch, comprehensive documentation, and expert support—essential foundations for reliable scientific investigation.
