
Researchers investigating novel metabolic peptides are increasingly encountering reports of retatrutide dizziness in study protocols. This triple agonist peptide, which simultaneously activates GLP-1, GIP, and glucagon receptors, has demonstrated remarkable efficacy in metabolic research—but its side effect profile requires careful documentation and understanding. For laboratories and research institutions working with this compound, comprehending the mechanisms, prevalence, and management strategies for dizziness-related observations is essential for maintaining rigorous scientific standards and ensuring proper experimental protocols.
Retatrutide dizziness represents one of the most frequently documented observations in research settings, occurring through multiple physiological pathways that researchers must account for when designing studies and interpreting results. Understanding these mechanisms not only enhances research quality but also provides valuable insights into the compound’s broader pharmacological profile.
Key Takeaways
- Retatrutide dizziness occurs in research subjects through multiple mechanisms including blood pressure changes, metabolic shifts, and central nervous system effects
- Proper documentation and monitoring protocols are essential for research integrity when observing dizziness-related phenomena
- Understanding the compound’s triple receptor agonism (GLP-1, GIP, glucagon) helps explain the physiological basis for vestibular and cardiovascular observations
- Research-grade retatrutide requires strict handling protocols and quality verification to ensure consistent, reliable study outcomes
- Laboratories should implement comprehensive safety monitoring and maintain detailed records of all observed effects
Understanding Retatrutide: Mechanism and Research Applications
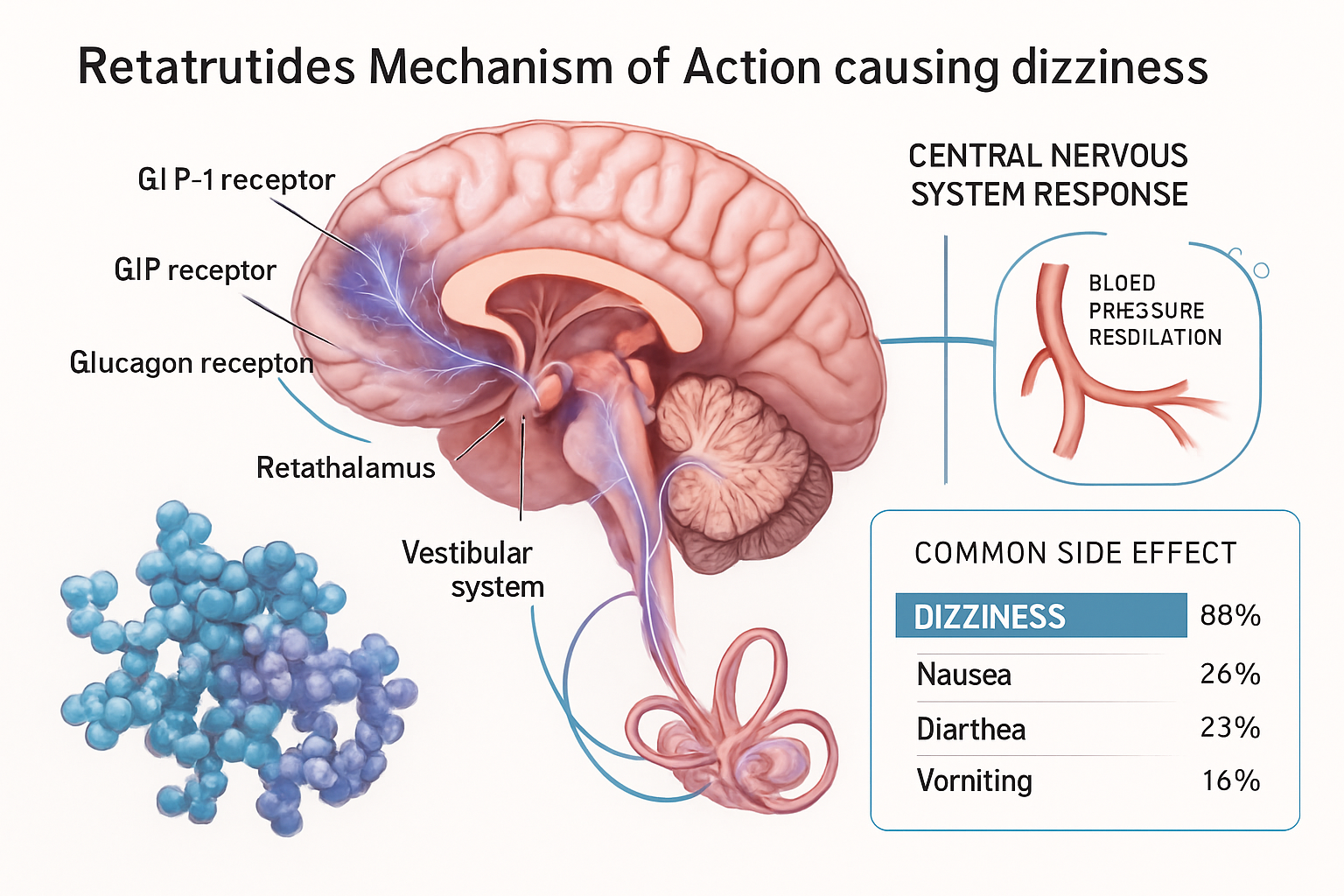
Retatrutide represents a significant advancement in peptide research, functioning as a triple receptor agonist that simultaneously targets GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors. This unique pharmacological profile distinguishes it from single or dual agonist compounds and creates a complex interaction network within metabolic pathways.
The Triple Agonist Mechanism
The compound’s three-pronged approach creates synergistic effects across multiple physiological systems:
GLP-1 Receptor Activation:
- Enhances insulin secretion in glucose-dependent manner
- Reduces gastric emptying rate
- Affects central nervous system appetite regulation
- Influences cardiovascular function
GIP Receptor Activation:
- Modulates insulin response
- Affects lipid metabolism
- Influences bone metabolism
- Contributes to energy homeostasis
Glucagon Receptor Activation:
- Increases energy expenditure
- Affects hepatic glucose production
- Influences thermogenesis
- Modulates metabolic rate
This multi-receptor activation creates a cascade of physiological responses that researchers must carefully monitor and document. Research-grade peptides require exceptional purity standards to ensure these mechanisms function consistently across experimental protocols.
Research Applications and Study Designs
Laboratories investigating retatrutide typically focus on several key research areas:
| Research Area | Primary Focus | Relevant Observations |
|---|---|---|
| Metabolic Studies | Energy balance, glucose homeostasis | Weight changes, metabolic markers |
| Cardiovascular Research | Blood pressure, heart rate variability | Hemodynamic changes, dizziness |
| Neurological Investigation | Central appetite regulation, CNS effects | Vestibular responses, cognitive markers |
| Endocrine Analysis | Hormone secretion patterns | Insulin, glucagon, incretin responses |
The complexity of retatrutide’s mechanism means that researchers must implement comprehensive monitoring protocols that capture both intended research outcomes and secondary observations like dizziness.
Retatrutide Dizziness: Mechanisms and Prevalence in Research Settings
The occurrence of retatrutide dizziness in research protocols stems from multiple interconnected physiological mechanisms. Understanding these pathways is crucial for proper experimental design and accurate data interpretation.
Primary Mechanisms Behind Retatrutide Dizziness
1. Cardiovascular and Blood Pressure Effects 💓
Retatrutide’s action on multiple receptor systems creates significant cardiovascular responses:
- Orthostatic hypotension: The compound’s influence on vascular tone and blood volume regulation can lead to position-dependent blood pressure drops
- Heart rate variability: GLP-1 receptor activation affects autonomic nervous system balance
- Peripheral vasodilation: Changes in vascular resistance may reduce cerebral perfusion pressure
- Blood volume shifts: Metabolic changes can affect fluid distribution and circulating volume
Research protocols should include regular hemodynamic monitoring to document these changes systematically.
2. Metabolic and Glycemic Fluctuations 📊
The compound’s powerful effects on glucose homeostasis can trigger dizziness through:
- Rapid glucose normalization: Subjects accustomed to elevated glucose may experience relative hypoglycemia
- Altered fuel utilization: Shifts between glucose and lipid metabolism affect cerebral energy supply
- Electrolyte changes: Metabolic shifts can influence sodium, potassium, and fluid balance
- Hormonal fluctuations: Changes in insulin, glucagon, and incretin levels affect multiple systems
3. Gastrointestinal and Absorption Effects 🔄
Delayed gastric emptying—a hallmark of GLP-1 agonism—contributes to dizziness observations:
- Nutrient absorption timing: Altered absorption patterns affect blood glucose stability
- Vagal nerve stimulation: Gastric distension and delayed emptying stimulate vagal pathways
- Dehydration risk: Reduced fluid intake combined with metabolic changes may affect hydration status
- Nausea-dizziness connection: Gastrointestinal discomfort often accompanies vestibular symptoms
4. Central Nervous System Interactions 🧠
Retatrutide’s central effects extend beyond appetite regulation:
- Hypothalamic signaling: Direct CNS effects on energy balance centers
- Vestibular system sensitivity: Potential direct or indirect effects on balance mechanisms
- Neurotransmitter modulation: Changes in dopamine, serotonin, and other signaling molecules
- Blood-brain barrier interactions: Peptide effects on CNS vascular regulation
Prevalence and Documentation in Research Studies
Current research literature suggests that dizziness-related observations occur with varying frequency depending on dosage, study design, and subject characteristics:
Frequency Patterns:
- Early-phase observations: Higher incidence during initial exposure periods
- Dose-dependent relationship: Greater frequency at higher concentrations
- Temporal patterns: Often transient, with adaptation over time
- Individual variability: Significant differences based on baseline characteristics
“Comprehensive documentation of all observed effects, including retatrutide dizziness, is essential for building a complete understanding of this compound’s pharmacological profile and ensuring research integrity across institutions.” — Research Protocol Guidelines
Risk Factors in Experimental Settings
Certain experimental conditions may increase the likelihood of observing dizziness phenomena:
✅ Higher risk scenarios:
- Rapid dose escalation protocols
- Studies involving subjects with cardiovascular history
- Combination with other metabolically active compounds
- Inadequate hydration protocols
- Insufficient acclimatization periods
✅ Lower risk scenarios:
- Gradual dose titration approaches
- Comprehensive baseline screening
- Adequate monitoring intervals
- Proper hydration maintenance
- Extended observation periods
Laboratories sourcing high-purity research peptides should ensure their suppliers provide complete documentation including Certificates of Analysis to verify compound identity and purity—factors that directly influence the consistency of observed effects.
Research Protocol Considerations for Retatrutide Dizziness
Implementing robust research protocols that account for retatrutide dizziness observations requires systematic planning and comprehensive monitoring strategies.
Pre-Study Assessment and Baseline Documentation
Establishing thorough baseline measurements creates the foundation for accurate observation and interpretation:
Essential Baseline Parameters:
📋 Cardiovascular Metrics:
- Resting blood pressure (multiple positions)
- Heart rate and variability
- Orthostatic vital signs (supine, sitting, standing)
- Electrocardiogram when appropriate
- Vascular function markers
📋 Metabolic Markers:
- Fasting glucose and insulin
- HbA1c levels
- Lipid profiles
- Electrolyte panels
- Renal and hepatic function
📋 Neurological Assessment:
- Baseline vestibular function testing
- Balance and coordination evaluation
- Subjective dizziness history
- Medication and supplement inventory
- Previous adverse event documentation
Monitoring Protocols During Research
Systematic observation schedules ensure comprehensive data collection:
Recommended Monitoring Timeline:
| Time Point | Assessments | Focus Areas |
|---|---|---|
| Pre-dose | Full vitals, subjective symptoms | Baseline confirmation |
| 30 minutes post | Blood pressure, heart rate, symptoms | Acute responses |
| 2 hours post | Metabolic markers, vitals, symptoms | Peak concentration effects |
| 4-6 hours post | Comprehensive assessment | Extended response patterns |
| 24 hours post | Full parameter review | Sustained effects |
| Weekly | Trend analysis, cumulative effects | Long-term patterns |
Standardized Documentation Methods:
Implementing consistent documentation protocols ensures data quality:
- Structured symptom questionnaires: Validated scales for dizziness severity and character
- Objective measurements: Quantifiable metrics rather than subjective impressions alone
- Temporal correlation: Precise timing of observations relative to dosing
- Environmental factors: Room temperature, position changes, concurrent activities
- Reproducibility verification: Consistent observation across multiple time points
Handling and Storage Protocols
Proper peptide handling directly impacts research reliability and observed effects. Research-grade retatrutide requires specific storage and reconstitution procedures:
Storage Requirements:
🧊 Lyophilized (Powder) Form:
- Temperature: -20°C to -80°C for long-term storage
- Protection from light and moisture
- Sealed containers with desiccant
- Minimal freeze-thaw cycles
- Documented storage duration
🧊 Reconstituted Solution:
- Temperature: 2-8°C (refrigerated)
- Protected from light
- Sterile conditions maintained
- Limited storage duration (typically 14-28 days)
- Clear labeling with reconstitution date
Reconstitution Best Practices:
- Allow temperature equilibration: Bring lyophilized peptide to room temperature before reconstitution
- Use appropriate diluent: Bacteriostatic water or specified reconstitution solution
- Gentle mixing: Avoid vigorous shaking; use gentle swirling
- Verify dissolution: Ensure complete dissolution before use
- Document concentration: Clearly label final concentration and volume
Laboratories requiring comprehensive peptide resources should establish relationships with suppliers who provide detailed handling protocols and technical support.
Quality Verification and Purity Considerations
The relationship between peptide purity and observed effects cannot be overstated:
Critical Quality Factors:
⚗️ Purity Standards:
- Minimum 98% purity for research applications
- HPLC verification documentation
- Mass spectrometry confirmation
- Endotoxin testing results
- Sterility verification
⚗️ Batch Consistency:
- Consistent synthesis methods
- Documented manufacturing protocols
- Batch-to-batch variability analysis
- Stability testing data
- Certificate of Analysis for each batch
Inconsistent purity or contamination can introduce confounding variables that complicate interpretation of observations like dizziness, making supplier selection a critical research decision.
Managing and Documenting Retatrutide Dizziness Observations
When retatrutide dizziness occurs during research protocols, systematic management and documentation strategies ensure both subject safety and data integrity.
Immediate Response Protocols
Establishing clear response procedures maintains research standards while prioritizing safety:
Tiered Response System:
Level 1 – Mild Dizziness (Subjective, No Functional Impairment):
- Document symptoms using standardized scales
- Measure vital signs immediately
- Have subject assume seated or supine position
- Provide hydration if appropriate
- Continue monitoring at increased frequency
- Document duration and resolution
Level 2 – Moderate Dizziness (Functional Impact, Stable Vitals):
- Immediate vital sign assessment
- Position subject supine with legs elevated
- Assess for orthostatic changes
- Check glucose levels if appropriate
- Extend observation period
- Consider protocol modification
- Detailed documentation of event
Level 3 – Severe Dizziness (Significant Impairment or Vital Sign Changes):
- Immediate comprehensive assessment
- Continuous vital sign monitoring
- Medical evaluation as appropriate
- Protocol suspension consideration
- Comprehensive event documentation
- Root cause analysis
- Ethics committee notification if required
Documentation Standards for Research Integrity
Comprehensive documentation serves multiple critical functions in research settings:
Essential Documentation Elements:
📝 Event Characterization:
- Precise timing relative to dosing
- Symptom onset and duration
- Severity using validated scales
- Associated symptoms (nausea, visual changes, etc.)
- Positional relationships
- Precipitating factors
- Resolution timeline
📝 Objective Measurements:
- Blood pressure (multiple positions)
- Heart rate and rhythm
- Temperature
- Glucose levels when relevant
- Respiratory rate
- Oxygen saturation if indicated
📝 Contextual Information:
- Recent food and fluid intake
- Concurrent medications or supplements
- Environmental conditions
- Activity level prior to event
- Sleep quality and duration
- Stress or anxiety factors
📝 Intervention and Outcome:
- Actions taken in response
- Subject response to interventions
- Time to symptom resolution
- Any protocol modifications
- Follow-up assessment results
Data Analysis and Pattern Recognition
Systematic analysis of dizziness observations reveals important patterns:
Analytical Approaches:
🔍 Temporal Analysis:
- Time-to-onset patterns across subjects
- Duration distributions
- Relationship to peak concentration times
- Adaptation patterns over repeated exposures
🔍 Dose-Response Relationships:
- Frequency vs. dose correlation
- Severity vs. concentration patterns
- Threshold identification
- Individual sensitivity variations
🔍 Correlation Analysis:
- Relationship to blood pressure changes
- Association with metabolic markers
- Connection to gastrointestinal effects
- Predictive baseline characteristics
🔍 Comparative Analysis:
- Differences across subject groups
- Variations by demographic factors
- Comparison to other peptide compounds
- Literature concordance assessment
Protocol Modifications and Adaptive Strategies
Research protocols may require adjustment based on dizziness observations:
Common Modification Strategies:
✏️ Dose Titration Adjustments:
- Slower escalation schedules
- Smaller increment steps
- Extended intervals between increases
- Individual optimization approaches
✏️ Administration Timing:
- Coordination with meals
- Time-of-day considerations
- Relationship to activity schedules
- Hydration protocol timing
✏️ Supportive Measures:
- Enhanced hydration protocols
- Electrolyte supplementation strategies
- Position change guidance
- Activity modification recommendations
✏️ Monitoring Enhancements:
- Increased assessment frequency
- Additional parameter measurement
- Extended observation periods
- Home monitoring implementation
Researchers working with specialized peptide compounds understand that protocol flexibility based on observed effects is a hallmark of rigorous scientific methodology.
Comparative Analysis: Retatrutide Dizziness vs. Other Metabolic Peptides
Understanding how retatrutide dizziness compares to similar observations with other metabolic peptides provides valuable context for research interpretation.
Comparison with Single and Dual Agonists
Retatrutide vs. GLP-1 Agonists:
| Characteristic | Retatrutide (Triple Agonist) | GLP-1 Agonists (Single) |
|---|---|---|
| Receptor Targets | GLP-1, GIP, Glucagon | GLP-1 only |
| Dizziness Frequency | Moderate to high | Low to moderate |
| Mechanism Complexity | Multi-pathway | Primarily GI and CNS |
| Blood Pressure Effects | More pronounced | Moderate |
| Metabolic Impact | Comprehensive | Focused on glucose/appetite |
Retatrutide vs. Tirzepatide (Dual Agonist):
Both compounds share GLP-1 and GIP agonism, but retatrutide’s additional glucagon receptor activation creates distinct differences:
- Enhanced metabolic effects: Greater energy expenditure increase
- Additional CNS pathways: Glucagon receptor CNS distribution
- Increased cardiovascular impact: Broader hemodynamic effects
- Different side effect profile: Unique combination of observations
Researchers investigating tirzepatide formulations alongside retatrutide can directly compare these profiles within controlled study designs.
Mechanistic Distinctions
Unique Aspects of Retatrutide Dizziness:
🔬 Triple Receptor Synergy: The simultaneous activation of three receptor systems creates interaction effects not present with single or dual agonists:
- Amplified metabolic shifts: Cumulative effects on energy balance
- Complex hormonal cascades: Multiple incretin and counter-regulatory responses
- Enhanced CNS penetration: Broader central nervous system effects
- Cardiovascular complexity: Additive or synergistic hemodynamic changes
🔬 Glucagon Receptor Contribution: The glucagon component specifically adds:
- Increased thermogenesis: May affect cardiovascular demand
- Enhanced lipolysis: Rapid metabolic fuel shifts
- Hepatic effects: Glucose production modulation
- Additional CNS signaling: Hypothalamic and brainstem pathways
Research Implications of Comparative Profiles
Understanding these distinctions guides research design:
Study Design Considerations:
- Control group selection: Appropriate comparator compounds
- Monitoring intensity: More comprehensive for multi-agonists
- Safety margins: Conservative approaches for novel mechanisms
- Dose escalation: Potentially slower for complex agonists
- Duration planning: Extended observation for cumulative effects
Research Safety and Quality Assurance
Maintaining the highest standards of research safety and quality assurance is paramount when working with compounds that may produce retatrutide dizziness and other physiological effects.
Supplier Selection and Verification
The foundation of reliable research begins with peptide quality:
Critical Supplier Criteria:
✓ Quality Documentation:
- Certificate of Analysis (CoA) for every batch
- HPLC purity verification (≥98%)
- Mass spectrometry confirmation
- Endotoxin testing results
- Sterility certification
- Storage condition documentation
✓ Manufacturing Standards:
- GMP-compliant facilities
- Validated synthesis protocols
- Batch-to-batch consistency data
- Stability testing documentation
- Contamination control procedures
✓ Research Support:
- Technical consultation availability
- Handling and storage guidance
- Reconstitution protocols
- Troubleshooting assistance
- Literature and reference materials
Premium research-grade peptide suppliers distinguish themselves through comprehensive quality assurance and researcher support systems.
Laboratory Handling Protocols
Proper handling maintains peptide integrity and research validity:
Standard Operating Procedures:
🔬 Receipt and Storage:
- Verify cold chain maintenance upon delivery
- Inspect packaging integrity
- Confirm product identity and batch number
- Document receipt date and condition
- Transfer immediately to appropriate storage
- Log storage location and temperature
- Implement first-in, first-out inventory management
🔬 Preparation and Use:
- Review CoA and handling instructions
- Allow temperature equilibration before opening
- Use aseptic technique throughout
- Reconstitute according to specifications
- Document reconstitution details
- Label clearly with concentration and date
- Store reconstituted solution appropriately
- Track usage and remaining volume
🔬 Quality Monitoring:
- Visual inspection before each use
- Temperature log maintenance
- Expiration date tracking
- Contamination prevention measures
- Regular inventory audits
- Deviation documentation and investigation
Regulatory and Ethical Considerations
Research involving bioactive peptides requires adherence to multiple regulatory frameworks:
Compliance Requirements:
📋 Institutional Oversight:
- Ethics committee approval for research protocols
- Institutional biosafety committee review
- Proper classification and labeling
- “Research Use Only” designation enforcement
- Subject consent procedures (when applicable)
- Adverse event reporting systems
📋 Documentation Standards:
- Comprehensive protocol documentation
- Detailed standard operating procedures
- Complete data recording systems
- Audit trail maintenance
- Secure data storage
- Retention period compliance
📋 Safety Protocols:
- Personal protective equipment requirements
- Spill response procedures
- Waste disposal protocols
- Exposure prevention measures
- Emergency response plans
Research Integrity and Transparency
Maintaining scientific integrity requires systematic approaches:
Best Practices:
Data Management:
- Real-time data recording
- Unaltered original records
- Secure backup systems
- Version control protocols
- Access restriction and audit trails
Bias Prevention:
- Blinding procedures when appropriate
- Randomization protocols
- Predefined analysis plans
- Transparent reporting of all observations
- Negative result documentation
Reproducibility:
- Detailed methodology documentation
- Batch number recording
- Environmental condition logging
- Equipment calibration records
- Statistical analysis transparency
Future Research Directions and Emerging Insights
The evolving understanding of retatrutide dizziness and related phenomena continues to shape research priorities and methodological approaches.
Emerging Research Questions
Current gaps in knowledge suggest important future research directions:
Mechanistic Investigations:
🔍 Receptor-Specific Contributions:
- Isolating individual receptor contributions to dizziness
- Mapping temporal patterns of each receptor’s effects
- Understanding synergistic vs. additive mechanisms
- Identifying receptor distribution in vestibular systems
🔍 Individual Variability:
- Genetic factors influencing susceptibility
- Baseline physiological predictors
- Metabolic phenotype relationships
- Adaptation mechanism differences
🔍 Long-Term Effects:
- Tolerance development patterns
- Sustained vs. transient observations
- Cumulative exposure effects
- Recovery timeline characterization
Technological Advances in Monitoring
Novel monitoring technologies enhance research capabilities:
Innovative Approaches:
📱 Continuous Monitoring Systems:
- Wearable blood pressure monitors
- Continuous glucose monitoring integration
- Accelerometer-based balance assessment
- Heart rate variability tracking
- Real-time symptom reporting applications
📱 Advanced Analytics:
- Machine learning pattern recognition
- Predictive modeling of susceptibility
- Multi-parameter correlation analysis
- Individual response prediction algorithms
Translational Research Implications
Understanding retatrutide’s side effect profile has broader implications:
Research Applications:
- Mechanism-based intervention development: Targeted strategies to mitigate specific pathways
- Personalized protocol optimization: Individual-specific dosing and monitoring
- Combination therapy research: Interactions with other compounds
- Formulation innovations: Modified delivery systems to reduce peak effects
Collaborative Research Opportunities
The complexity of triple agonist peptides benefits from collaborative approaches:
Multi-Institutional Studies:
- Larger sample sizes for rare observation patterns
- Diverse population representation
- Standardized protocol implementation
- Shared data repositories
- Meta-analytic capabilities
Interdisciplinary Integration:
- Cardiovascular physiology expertise
- Endocrinology specialization
- Neurology and vestibular science
- Pharmacology and pharmacokinetics
- Biostatistics and data science
Researchers seeking to contribute to this expanding knowledge base require access to high-quality research materials and collaborative networks.
Practical Recommendations for Research Laboratories
Implementing comprehensive approaches to retatrutide dizziness research requires systematic planning and execution across multiple domains.
Establishing Research Protocols
Protocol Development Checklist:
✅ Pre-Study Planning:
- Define primary and secondary research objectives
- Establish inclusion and exclusion criteria
- Design monitoring schedule and parameters
- Create standardized documentation forms
- Develop response algorithms for observations
- Obtain necessary approvals and oversight
- Arrange for emergency response capabilities
✅ Resource Allocation:
- Secure high-purity peptide supply with documentation
- Acquire necessary monitoring equipment
- Train personnel on protocols and procedures
- Establish data management systems
- Arrange for statistical analysis support
- Budget for extended observation periods
- Plan for protocol modification flexibility
✅ Quality Assurance:
- Implement equipment calibration schedules
- Establish standard operating procedures
- Create audit and oversight mechanisms
- Develop deviation reporting systems
- Plan for regular protocol reviews
- Establish data quality checks
- Implement corrective action procedures
Building Supplier Relationships
Reliable peptide sourcing is fundamental to research success:
Supplier Partnership Elements:
🤝 Initial Evaluation:
- Request sample CoAs and documentation
- Verify manufacturing and quality standards
- Assess technical support capabilities
- Evaluate delivery reliability and cold chain
- Compare pricing and minimum orders
- Review return and replacement policies
🤝 Ongoing Relationship:
- Maintain open communication channels
- Provide feedback on product quality
- Request batch consistency data
- Discuss upcoming research needs
- Coordinate delivery timing
- Report any quality concerns promptly
Laboratories throughout the UK and internationally benefit from establishing relationships with trusted peptide suppliers who understand research requirements and maintain consistent quality standards.
Training and Competency Development
Research personnel require comprehensive training:
Training Program Components:
📚 Technical Skills:
- Peptide handling and reconstitution
- Aseptic technique
- Equipment operation and calibration
- Data recording and documentation
- Safety and emergency procedures
📚 Research Methodology:
- Protocol adherence importance
- Observation and documentation standards
- Bias recognition and prevention
- Quality assurance principles
- Regulatory compliance requirements
📚 Subject Interaction:
- Professional communication
- Symptom assessment techniques
- Emergency response procedures
- Documentation best practices
- Ethical considerations
Resource Optimization
Maximizing research efficiency while maintaining quality:
Efficiency Strategies:
⚡ Standardization:
- Template development for common procedures
- Pre-printed documentation forms
- Checklist implementation
- Automated reminders and scheduling
- Shared resource calendars
⚡ Technology Integration:
- Electronic data capture systems
- Automated vital sign recording
- Cloud-based data storage
- Collaborative platforms
- Statistical software utilization
⚡ Workflow Optimization:
- Parallel processing where appropriate
- Batch preparation procedures
- Scheduled monitoring windows
- Team coordination systems
- Regular efficiency reviews
Advancing Retatrutide Research with Rigor and Safety
Understanding retatrutide dizziness represents a critical component of comprehensive research into this novel triple agonist peptide. The occurrence of dizziness through multiple physiological mechanisms—including cardiovascular effects, metabolic shifts, gastrointestinal changes, and central nervous system interactions—requires systematic documentation, analysis, and management within research protocols.
Key Principles for Research Excellence
Successful retatrutide research depends on several fundamental principles:
Quality Foundation: Beginning with high-purity, research-grade peptides from verified suppliers with comprehensive documentation ensures consistent, reliable observations across studies.
Comprehensive Monitoring: Implementing multi-parameter assessment protocols captures the full spectrum of physiological responses, enabling accurate characterization of both intended research outcomes and secondary observations like dizziness.
Rigorous Documentation: Systematic recording of all observations, using standardized scales and objective measurements, builds a robust evidence base for understanding retatrutide’s complete pharmacological profile.
Safety-First Approach: Prioritizing subject safety through established response protocols, appropriate monitoring intensity, and conservative study design maintains ethical standards while advancing scientific knowledge.
Adaptive Methodology: Remaining flexible in protocol implementation based on observed effects demonstrates scientific rigor and ensures research relevance and applicability.
Actionable Next Steps for Researchers
For laboratories planning or conducting retatrutide research:
- Establish Supplier Relationships: Partner with reputable peptide suppliers who provide complete quality documentation and technical support
- Develop Comprehensive Protocols: Create detailed standard operating procedures covering all aspects from peptide handling to adverse event management
- Implement Robust Monitoring: Design monitoring schedules that capture acute, intermediate, and long-term observations across multiple physiological systems
- Train Research Personnel: Ensure all team members understand protocols, documentation requirements, and response procedures
- Create Documentation Systems: Establish standardized forms and electronic systems for consistent, complete data capture
- Plan for Contingencies: Develop clear algorithms for managing unexpected observations and protocol modifications
- Engage in Collaboration: Connect with other research groups to share insights, standardize approaches, and contribute to collective understanding
The Path Forward
As research into retatrutide and other multi-agonist peptides continues to evolve, the systematic understanding of side effect profiles like dizziness will inform both basic science and translational applications. The complexity of triple receptor agonism demands equally sophisticated research methodologies, comprehensive monitoring, and unwavering commitment to quality and safety.
Laboratories equipped with premium research-grade peptides, robust protocols, and trained personnel are positioned to make significant contributions to this expanding field. By maintaining rigorous standards, documenting observations thoroughly, and prioritizing both scientific integrity and safety, researchers advance not only knowledge of retatrutide specifically but also broader understanding of metabolic peptide pharmacology.
The future of peptide research depends on the foundation being built today—one observation, one protocol, one carefully documented study at a time. For researchers committed to excellence in this field, understanding and properly managing retatrutide dizziness represents not a challenge to overcome but an opportunity to demonstrate the precision, rigor, and comprehensive approach that defines world-class scientific investigation.
Disclaimer: This article is intended for educational and informational purposes related to research applications only. Retatrutide and all peptides discussed are strictly for research use only and are not intended for human consumption or therapeutic application. All research involving bioactive compounds must comply with applicable institutional, local, and national regulations and receive appropriate ethical oversight. Researchers should consult with qualified professionals and obtain necessary approvals before initiating any research protocols.
