
The emergence of novel multi-receptor agonist peptides has revolutionised metabolic research, yet with innovation comes the critical responsibility to understand potential safety concerns. Retatrutide pancreatitis risk has become a focal point for researchers investigating this triple-agonist compound, particularly as clinical trials expand and laboratory studies intensify. As a GLP-1/GIP/glucagon receptor agonist, retatrutide demonstrates remarkable efficacy in metabolic research models—but does this potency come with elevated pancreatic safety considerations? Understanding the relationship between retatrutide and pancreatic inflammation is essential for researchers designing protocols, interpreting data, and advancing the field responsibly.
This comprehensive analysis examines the current evidence surrounding retatrutide pancreatitis risk, exploring mechanistic pathways, clinical trial data, comparative safety profiles, and practical considerations for research applications. Whether you’re conducting metabolic studies or evaluating peptide safety profiles, this resource provides the professional insights needed to navigate this complex topic.
Key Takeaways
- Retatrutide pancreatitis risk appears low based on current clinical trial data, with incidence rates comparable to or lower than other incretin-based peptides
- Multi-receptor agonism (GLP-1/GIP/glucagon) presents unique mechanistic considerations that differ from single-receptor compounds
- Research-grade retatrutide requires proper handling, storage, and quality verification to ensure experimental validity and safety assessment accuracy
- Monitoring protocols and risk mitigation strategies are essential components of responsible retatrutide research design
- Individual variability and pre-existing pancreatic conditions represent important confounding factors in safety evaluations
Understanding Retatrutide: Mechanism and Pancreatic Considerations
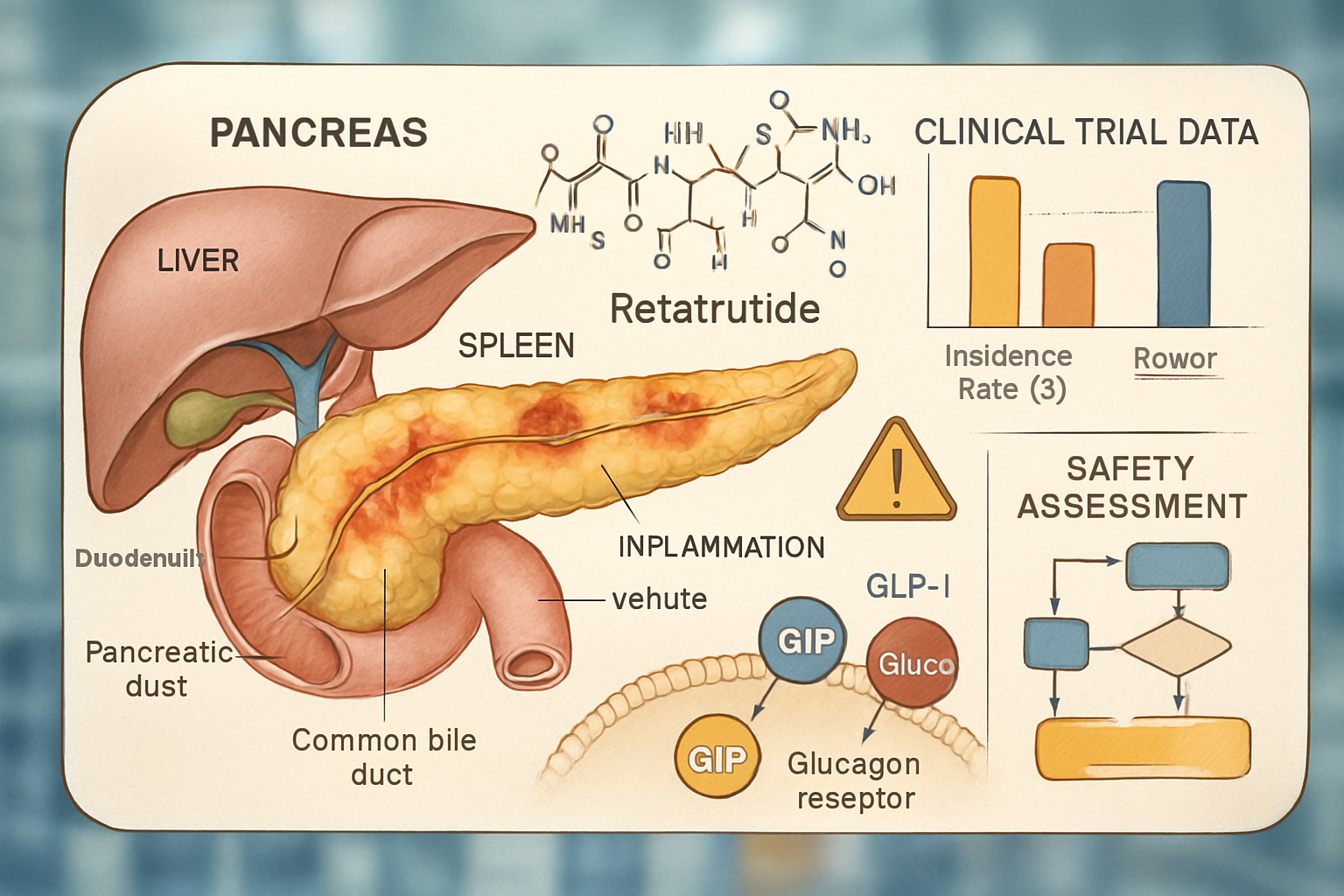
What Makes Retatrutide Unique?
Retatrutide (LY3437943) represents a significant advancement in peptide therapeutics research, functioning as a triple-agonist targeting GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors simultaneously. This multi-receptor approach distinguishes it from earlier incretin-based compounds like semaglutide or tirzepatide, which target one or two receptors respectively.
The compound’s mechanism involves:
✅ GLP-1 receptor activation – Enhances insulin secretion, suppresses glucagon release, delays gastric emptying
✅ GIP receptor activation – Amplifies insulin response, influences lipid metabolism
✅ Glucagon receptor activation – Increases energy expenditure, promotes lipolysis
This triple mechanism creates potent metabolic effects in research models, but also raises questions about cumulative pancreatic stimulation and the retatrutide pancreatitis risk profile.
Pancreatic Physiology and Incretin Signalling
The pancreas plays dual roles as both an endocrine and exocrine organ. Understanding how retatrutide interacts with pancreatic tissue requires examining:
| Pancreatic Component | Function | Incretin Receptor Expression |
|---|---|---|
| Beta cells | Insulin production | High GLP-1, moderate GIP |
| Alpha cells | Glucagon secretion | Moderate GLP-1, glucagon receptors |
| Acinar cells | Digestive enzyme production | Low to moderate GLP-1 |
| Ductal cells | Bicarbonate secretion | Variable receptor expression |
Incretin receptor activation, particularly GLP-1 stimulation, influences pancreatic cell proliferation, enzyme secretion, and inflammatory pathways—all factors relevant to retatrutide pancreatitis risk assessment. Research suggests that chronic GLP-1 receptor stimulation may affect pancreatic ductal pressure and enzyme activation patterns, though the clinical significance remains under investigation.
For researchers working with high-purity research peptides, understanding these mechanistic foundations is crucial for experimental design and data interpretation.
Clinical Evidence: Retatrutide Pancreatitis Risk in Trials
Phase 2 Clinical Trial Safety Data
The most comprehensive safety data for retatrutide comes from the landmark Phase 2 trial published in The New England Journal of Medicine (2023), which evaluated 338 participants across multiple dosing regimens over 48 weeks.
Key findings regarding pancreatitis:
- Zero confirmed cases of acute pancreatitis were reported in the retatrutide treatment groups
- Lipase elevations (>3× upper limit of normal) occurred in 2.1% of participants receiving retatrutide versus 0% in placebo
- Asymptomatic lipase increases resolved without intervention in all cases
- No correlation was found between lipase elevation and gastrointestinal symptoms
Research Note: Elevated lipase levels do not automatically indicate pancreatitis. Clinical pancreatitis requires both biochemical markers AND characteristic symptoms (severe abdominal pain, nausea, vomiting).
Comparative Safety: Retatrutide vs Other Incretin Peptides
How does retatrutide pancreatitis risk compare to established compounds? Examining pooled safety data provides context:
Pancreatitis Incidence Rates (per 1000 patient-years):
- Retatrutide: 0-2 (limited data, ongoing trials)
- Semaglutide: 2-4 (extensive clinical trial data)
- Tirzepatide: 1-3 (dual GLP-1/GIP agonist)
- Liraglutide: 3-5 (established GLP-1 agonist)
- Background population: 4-8 (general metabolic disorder population)
These preliminary comparisons suggest retatrutide’s safety profile may be comparable or potentially favourable relative to established incretin-based therapies, though longer-term data collection remains essential.
Ongoing Phase 3 Trials and Safety Monitoring
As of 2025, multiple Phase 3 trials are actively recruiting and monitoring participants receiving retatrutide:
- TRIUMPH-1: Obesity management (target n=1,200)
- TRIUMPH-2: Type 2 diabetes (target n=800)
- TRIUMPH-3: Cardiovascular outcomes (target n=2,500)
These trials incorporate rigorous pancreatic safety monitoring protocols including:
📊 Baseline and periodic lipase/amylase measurements
📊 Structured adverse event reporting for abdominal symptoms
📊 Imaging studies when clinically indicated
📊 Independent safety monitoring boards reviewing blinded data
Researchers following these developments can access updated safety information as trial results are published throughout 2025-2027.
Mechanistic Pathways: Why Pancreatitis Concerns Exist
Historical Context: GLP-1 Agonists and Pancreatic Safety
The concern about retatrutide pancreatitis risk doesn’t emerge in isolation—it reflects broader historical questions about incretin-based therapies. Early post-marketing surveillance of exenatide (the first GLP-1 agonist) identified case reports of pancreatitis, triggering extensive investigation.
What we’ve learned from two decades of incretin research:
- Case reports ≠ causation: Initial pancreatitis reports often involved patients with multiple risk factors
- Meta-analyses show reassurance: Large pooled analyses demonstrate no significant increase in pancreatitis risk
- Mechanistic studies are inconclusive: Animal models show variable pancreatic effects depending on species, dose, and duration
- Regulatory agencies maintain vigilance: FDA and EMA require ongoing safety monitoring but haven’t restricted use
Theoretical Mechanisms of Peptide-Induced Pancreatitis
Several biological pathways have been proposed to explain potential pancreatic effects of incretin agonists:
🔬 Proposed Mechanisms:
- Increased pancreatic duct pressure: GLP-1 may reduce sphincter of Oddi relaxation, potentially increasing ductal pressure
- Acinar cell proliferation: Some animal studies show increased pancreatic mass with chronic GLP-1 exposure
- Enzyme activation: Premature activation of digestive enzymes within the pancreas could trigger inflammation
- Inflammatory mediator modulation: Incretin receptors may influence cytokine production and inflammatory cascades
⚖️ Counter-Evidence:
- Human pancreatic tissue shows minimal GLP-1 receptor expression in acinar cells
- Proliferative effects in rodents don’t consistently translate to primates
- Anti-inflammatory properties of GLP-1 may actually protect against pancreatitis
- Clinical trial data doesn’t support theoretical concerns
For retatrutide pancreatitis risk specifically, the addition of glucagon receptor agonism adds complexity. Glucagon traditionally increases pancreatic enzyme secretion, but in the context of simultaneous GLP-1/GIP activation, the net effect remains under investigation.
Researchers sourcing compounds from reputable peptide suppliers can ensure experimental consistency when investigating these mechanistic questions.
Risk Factors and Individual Variability
Pre-Existing Conditions That Elevate Risk
Not all research subjects or experimental models carry equal retatrutide pancreatitis risk. Identifying high-risk characteristics is essential for protocol design and safety monitoring:
⚠️ Established Pancreatitis Risk Factors:
| Risk Factor | Relative Risk Increase | Research Considerations |
|---|---|---|
| Prior pancreatitis history | 10-20× | Exclusion criterion in most trials |
| Gallstone disease | 3-5× | Imaging screening recommended |
| Hypertriglyceridemia (>500 mg/dL) | 4-8× | Lipid monitoring essential |
| Chronic alcohol use | 3-6× | Detailed exposure history needed |
| Certain medications | 2-4× | Drug interaction assessment |
| Genetic predispositions | Variable | Emerging research area |
Research protocols should incorporate comprehensive screening for these factors, particularly when evaluating long-term safety outcomes.
Dose-Dependent Considerations
Does retatrutide pancreatitis risk increase with higher doses? Clinical trial data provides insights:
Retatrutide Dosing Tiers (Phase 2 Trial):
- 4 mg weekly: Lowest therapeutic dose, minimal adverse events
- 8 mg weekly: Moderate efficacy dose, well-tolerated
- 12 mg weekly: High efficacy dose, increased GI side effects but no pancreatitis
Importantly, no dose-dependent pattern of pancreatitis or severe pancreatic adverse events emerged across these ranges. However, gastrointestinal side effects (nausea, vomiting, diarrhoea) did show dose-dependency, which could theoretically complicate clinical assessment of abdominal symptoms.
Genetic and Metabolic Variability
Emerging research highlights individual variability in incretin response and metabolism:
🧬 Pharmacogenomic factors affecting receptor sensitivity
🧬 Metabolic enzyme polymorphisms influencing peptide clearance
🧬 Inflammatory response genes modulating pancreatic susceptibility
As research advances, personalised risk assessment may become possible, allowing more refined evaluation of retatrutide pancreatitis risk for specific populations.
Practical Research Considerations and Safety Protocols
Quality Assurance in Research-Grade Retatrutide
The validity of any safety assessment depends fundamentally on compound quality and purity. When investigating retatrutide pancreatitis risk or any aspect of this peptide’s profile, researchers must ensure:
✅ Verified purity (≥98% by HPLC)
✅ Proper storage conditions (-20°C to -80°C for lyophilised powder)
✅ Appropriate reconstitution (sterile bacteriostatic water, gentle mixing)
✅ Documented chain of custody (certificates of analysis, batch tracking)
PEPTIDE PRO supplies research-grade retatrutide with comprehensive documentation, ensuring experimental consistency and data reliability. All compounds are clearly labelled “For Research Use Only” and include detailed handling guidance.
Monitoring Protocols for Pancreatic Safety
Responsible research design incorporates systematic safety monitoring. Recommended protocols for studies involving retatrutide include:
📋 Baseline Assessment:
- Complete medical history focusing on pancreatic disorders
- Lipase and amylase measurements
- Liver function tests (elevated transaminases may confound interpretation)
- Lipid panel (triglycerides particularly important)
- Abdominal imaging if indicated by history
📋 Ongoing Monitoring:
- Periodic lipase measurements (frequency based on study duration and risk profile)
- Structured symptom questionnaires assessing abdominal pain, nausea, vomiting
- Rapid response protocols for acute symptoms
- Documentation of all gastrointestinal adverse events
📋 Endpoint Evaluation:
- Comprehensive safety data analysis
- Comparison of pancreatic enzyme trends across treatment groups
- Adjudication of potential pancreatitis cases by independent reviewers
Reconstitution and Handling Best Practices
Proper peptide handling ensures both experimental validity and accurate safety assessment. For retatrutide research:
Step-by-Step Protocol:
- Allow pen peptide to reach room temperature (15-20 minutes from freezer)
- Use appropriate bacteriostatic water volume (typically 1-2 mL for 10-40 mg pen peptides)
- Inject water slowly along pen peptide wall (avoid direct stream onto peptide)
- Gentle swirling (never shake vigorously)
- Visual inspection (solution should be clear, no particulates)
- Immediate refrigeration (2-8°C, use within 28 days)
Researchers can find detailed reconstitution guidance and technical support through professional peptide suppliers.
Interpreting Adverse Event Data: Lipase Elevations vs Clinical Pancreatitis
Understanding Asymptomatic Enzyme Elevations
A critical distinction in evaluating retatrutide pancreatitis risk is differentiating between:
Asymptomatic lipase elevation ➜ Biochemical finding without clinical significance
Clinical pancreatitis ➜ Syndrome requiring elevated enzymes PLUS characteristic symptoms
Research shows that asymptomatic lipase elevations occur relatively frequently with incretin-based peptides (2-5% of subjects) but rarely progress to clinical pancreatitis (<0.1%). Potential explanations include:
- Subclinical pancreatic stimulation without inflammation
- Laboratory variability and measurement error
- Concurrent medications or conditions affecting enzyme levels
- Enhanced pancreatic secretion as part of normal peptide pharmacology
Diagnostic Criteria for Acute Pancreatitis (Requires 2 of 3):
- Characteristic abdominal pain (severe, persistent, often radiating to back)
- Serum lipase or amylase >3× upper limit of normal
- Imaging findings consistent with pancreatitis (CT, MRI, ultrasound)
Researchers must avoid over-interpreting isolated enzyme elevations without clinical correlation.
Case Adjudication in Clinical Trials
Rigorous trials employ independent adjudication committees to review potential pancreatitis cases. This process involves:
🔍 Blinded review of all relevant clinical data
🔍 Application of standardised diagnostic criteria
🔍 Assessment of alternative explanations
🔍 Causality evaluation (temporal relationship, biological plausibility)
🔍 Classification of certainty level (definite, probable, possible, unlikely)
This systematic approach prevents both under-reporting (missing genuine cases) and over-reporting (attributing unrelated events to the intervention) of retatrutide pancreatitis risk.
Comparative Analysis: Retatrutide vs Established Peptides

Safety Profile Comparison Table
Understanding retatrutide pancreatitis risk requires context from established compounds:
| Peptide | Receptor Targets | Pancreatitis Reports | Lipase Elevation Rate | Years of Data |
|---|---|---|---|---|
| Retatrutide | GLP-1/GIP/Glucagon | Minimal (ongoing trials) | ~2% | 2-3 years |
| Tirzepatide | GLP-1/GIP | Very low | 2-3% | 4-5 years |
| Semaglutide | GLP-1 | Low | 1-2% | 8+ years |
| Liraglutide | GLP-1 | Low | 2-4% | 15+ years |
| Exenatide | GLP-1 | Low (early concerns) | 3-5% | 18+ years |
Key Insights:
- All incretin-based peptides show low absolute pancreatitis risk
- Lipase elevations are common across the class but rarely clinically significant
- Longer-term data for established compounds provides reassurance
- Multi-receptor agonism (tirzepatide, retatrutide) doesn’t appear to increase risk
Lessons from Tirzepatide Safety Experience
Tirzepatide, as a dual GLP-1/GIP agonist, provides the closest comparator for retatrutide pancreatitis risk assessment. The SURPASS clinical trial programme (>10,000 participants) demonstrated:
✓ Pancreatitis incidence: 0.13% (comparable to comparator groups)
✓ No dose-dependent increase in pancreatic adverse events
✓ Asymptomatic lipase elevations resolved without intervention
✓ No signal for chronic pancreatic pathology on extended follow-up
This experience suggests that adding additional receptor targets (as retatrutide does with glucagon) may not inherently increase pancreatic risk, though continued monitoring remains essential.
Researchers interested in comparative studies can access both tirzepatide and retatrutide through specialised research suppliers.
Regulatory Perspectives and Guidelines
FDA and EMA Position on Incretin Safety
Regulatory agencies have extensively reviewed incretin-based therapies and pancreatic safety:
FDA Position (Updated 2025):
- No restriction on GLP-1 agonist use based on pancreatitis concerns
- Requires pancreatitis warning in prescribing information
- Mandates post-marketing surveillance and periodic safety updates
- Recommends discontinuation if pancreatitis is suspected
EMA Position (Updated 2025):
- Similar stance to FDA with ongoing pharmacovigilance
- Emphasis on patient selection and risk factor assessment
- Requirement for Risk Management Plans in marketing authorisations
For retatrutide pancreatitis risk specifically, regulatory review is ongoing as Phase 3 trials progress. Researchers should anticipate similar safety monitoring requirements and labelling considerations.
Research Ethics and Informed Consent
Studies involving retatrutide must address pancreatic safety in ethics applications and consent processes:
Essential Elements:
📄 Clear explanation of theoretical pancreatitis risk
📄 Description of monitoring protocols and safety measures
📄 Instructions for recognising and reporting symptoms
📄 Exclusion criteria for high-risk individuals
📄 Withdrawal criteria and medical response procedures
Transparency about retatrutide pancreatitis risk, even when evidence suggests low incidence, maintains research integrity and participant trust.
Future Research Directions and Knowledge Gaps
Unanswered Questions About Retatrutide Pancreatitis Risk
Despite encouraging preliminary data, several research gaps remain:
🔬 Outstanding Research Questions:
- Long-term safety (>2 years): Does chronic exposure alter pancreatic risk profile?
- Paediatric populations: How does developing pancreatic tissue respond?
- Genetic susceptibility: Can we identify high-risk individuals prospectively?
- Mechanistic clarity: What are the precise pancreatic effects of triple agonism?
- Combination therapies: Does retatrutide interact with other medications affecting pancreatic risk?
- Real-world evidence: Will post-marketing data confirm clinical trial findings?
Emerging Technologies for Safety Assessment
Advanced research methodologies may provide deeper insights into retatrutide pancreatitis risk:
Novel Approaches:
- Advanced imaging: Magnetic resonance cholangiopancreatography (MRCP) for subclinical changes
- Biomarker development: More specific markers than lipase/amylase
- Organoid models: Human pancreatic organoids for mechanistic studies
- Pharmacogenomic screening: Identifying genetic risk modifiers
- Real-world data analytics: Large database studies tracking long-term outcomes
Researchers pursuing these innovative approaches require access to high-quality research compounds with verified purity and consistency.
Integration with Broader Metabolic Research
Understanding retatrutide pancreatitis risk contributes to larger questions about:
- Optimal multi-receptor targeting strategies
- Balancing efficacy and safety in peptide therapeutics
- Personalised medicine approaches to metabolic disorders
- Long-term consequences of chronic incretin receptor stimulation
These broader research directions will shape the next generation of metabolic peptide development.
Practical Recommendations for Researchers
Study Design Considerations
When designing research protocols involving retatrutide:
✅ DO:
- Implement comprehensive baseline pancreatic assessment
- Establish clear adverse event definitions and reporting procedures
- Include appropriate comparator groups (placebo, active controls)
- Power studies adequately for safety endpoint detection
- Plan for independent safety monitoring and adjudication
- Document all protocol deviations and safety concerns
❌ AVOID:
- Enrolling subjects with prior pancreatitis history without strong justification
- Relying solely on lipase measurements without clinical correlation
- Under-reporting minor gastrointestinal symptoms
- Making causality assumptions without thorough investigation
- Ignoring potential confounding factors (medications, alcohol, gallstones)
Data Interpretation Guidelines
When analysing safety data related to retatrutide pancreatitis risk:
Statistical Considerations:
- Use appropriate denominators (patient-years of exposure, not just participant numbers)
- Calculate confidence intervals for rare events
- Perform sensitivity analyses excluding uncertain cases
- Compare to background population rates in similar cohorts
- Avoid over-interpreting small numbers or isolated events
Clinical Considerations:
- Distinguish asymptomatic enzyme elevations from clinical pancreatitis
- Consider alternative diagnoses for abdominal symptoms
- Evaluate temporal relationships (onset, duration, resolution)
- Assess biological plausibility based on known mechanisms
- Consult gastroenterology expertise for complex cases
Collaboration and Knowledge Sharing
Advancing understanding of retatrutide pancreatitis risk requires collaborative approaches:
🤝 Participate in research consortia sharing safety data
🤝 Publish negative findings (absence of pancreatitis is valuable information)
🤝 Engage with regulatory agencies through scientific advice procedures
🤝 Contribute to systematic reviews and meta-analyses
🤝 Maintain transparency in adverse event reporting
The research community benefits when individual studies contribute to collective knowledge.
Storage, Handling and Quality Control
Optimal Storage Conditions for Research Retatrutide
Proper storage is critical for maintaining peptide integrity and ensuring valid safety assessments:
Lyophilised (Powder) Form:
- Temperature: -20°C to -80°C
- Protection: Light-protected, desiccated environment
- Stability: 12-24 months when stored properly
- Handling: Minimise freeze-thaw cycles
Reconstituted Solution:
- Temperature: 2-8°C (refrigerated)
- Duration: Use within 28 days
- Container: Sterile glass pen peptide, protected from light
- Inspection: Check for clarity, particulates before each use
Researchers should document storage conditions meticulously, as degraded peptides may produce unreliable safety data and confound retatrutide pancreatitis risk assessments.
Certificates of Analysis and Batch Testing
Professional research requires verified compound quality. Essential documentation includes:
📊 HPLC purity analysis (≥98% target)
📊 Mass spectrometry confirmation (molecular weight verification)
📊 Bacterial endotoxin testing (LAL assay)
📊 Peptide content quantification (accurate dosing)
📊 Storage and handling history (chain of custody)
PEPTIDE PRO provides comprehensive Certificates of Analysis with every research peptide order, ensuring transparency and experimental reproducibility.
Reconstitution Protocols and Sterility
Maintaining sterility during reconstitution prevents confounding variables:
Best Practices:
- Work in clean, controlled environment (ideally laminar flow hood)
- Use sterile bacteriostatic water (0.9% benzyl alcohol)
- Employ aseptic technique throughout
- Use appropriate needle gauge (avoid excessive turbulence)
- Allow complete dissolution before use
- Label reconstituted pen peptides with date, concentration, batch number
Contaminated preparations may trigger inflammatory responses that confound pancreatic safety assessments.
Evidence-Based Perspective on Retatrutide Pancreatitis Risk
After comprehensive examination of available evidence, clinical trial data, mechanistic understanding, and comparative analysis, several conclusions emerge regarding retatrutide pancreatitis risk:
Summary of Key Findings
Current Evidence Suggests Low Risk:
- Phase 2 trial data shows zero confirmed pancreatitis cases across 338 participants
- Asymptomatic lipase elevations (~2%) occurred without clinical sequelae
- Safety profile appears comparable to established incretin-based peptides
- No dose-dependent pattern of pancreatic adverse events identified
Mechanistic Considerations:
- Theoretical concerns about incretin-induced pancreatitis have not been substantiated by clinical evidence
- Triple receptor agonism (GLP-1/GIP/glucagon) does not appear to amplify pancreatic risk
- Anti-inflammatory properties of incretin signalling may provide protective effects
Knowledge Gaps Remain:
- Long-term safety data (>2 years) is still accumulating
- Rare events require larger populations and extended follow-up to detect
- Individual susceptibility factors need further characterisation
- Real-world evidence will provide important validation
Actionable Recommendations for Researchers
For those conducting research involving retatrutide:
- Implement comprehensive safety monitoring including baseline and periodic pancreatic enzyme measurements
- Screen for established risk factors and consider exclusion criteria for high-risk individuals
- Ensure compound quality through verified suppliers with complete documentation
- Distinguish biochemical findings from clinical events using standardised diagnostic criteria
- Contribute to collective knowledge through transparent reporting of all safety data
- Stay current with emerging evidence as Phase 3 trials report results throughout 2025-2027
The Path Forward
Understanding retatrutide pancreatitis risk remains an evolving area of investigation. The current evidence provides reassurance while maintaining appropriate vigilance. As the research community continues to generate data, our understanding will become increasingly refined, enabling more precise risk-benefit assessments and potentially identifying specific populations where extra caution is warranted.
For researchers committed to advancing metabolic science responsibly, partnering with professional peptide suppliers ensures access to high-purity compounds, comprehensive documentation, and expert support—essential foundations for generating reliable, reproducible safety data.
The future of metabolic peptide research is promising, and retatrutide represents an exciting frontier. By maintaining rigorous safety standards, transparent reporting, and collaborative knowledge-sharing, the research community can unlock this compound’s potential while safeguarding participant welfare and scientific integrity.
Ready to advance your retatrutide research with confidence? Explore PEPTIDE PRO’s research-grade peptides including verified retatrutide, comprehensive certificates of analysis, and expert technical support. All products strictly for research use only—delivered with the quality and transparency your studies demand.
