
The landscape of metabolic research peptides has expanded dramatically, yet understanding retatrutide contraindications remains critically important for researchers, laboratories, and the scientific community. As an investigational triple-agonist peptide targeting GLP-1, GIP, and glucagon receptors, retatrutide has generated substantial interest—but its safety profile, contraindications, and regulatory status demand careful examination before any research application.
This comprehensive guide explores the known and emerging contraindications associated with retatrutide, regulatory considerations, safety protocols for research settings, and the critical distinctions between legitimate research-grade compounds and potentially dangerous counterfeit products circulating in unregulated markets.
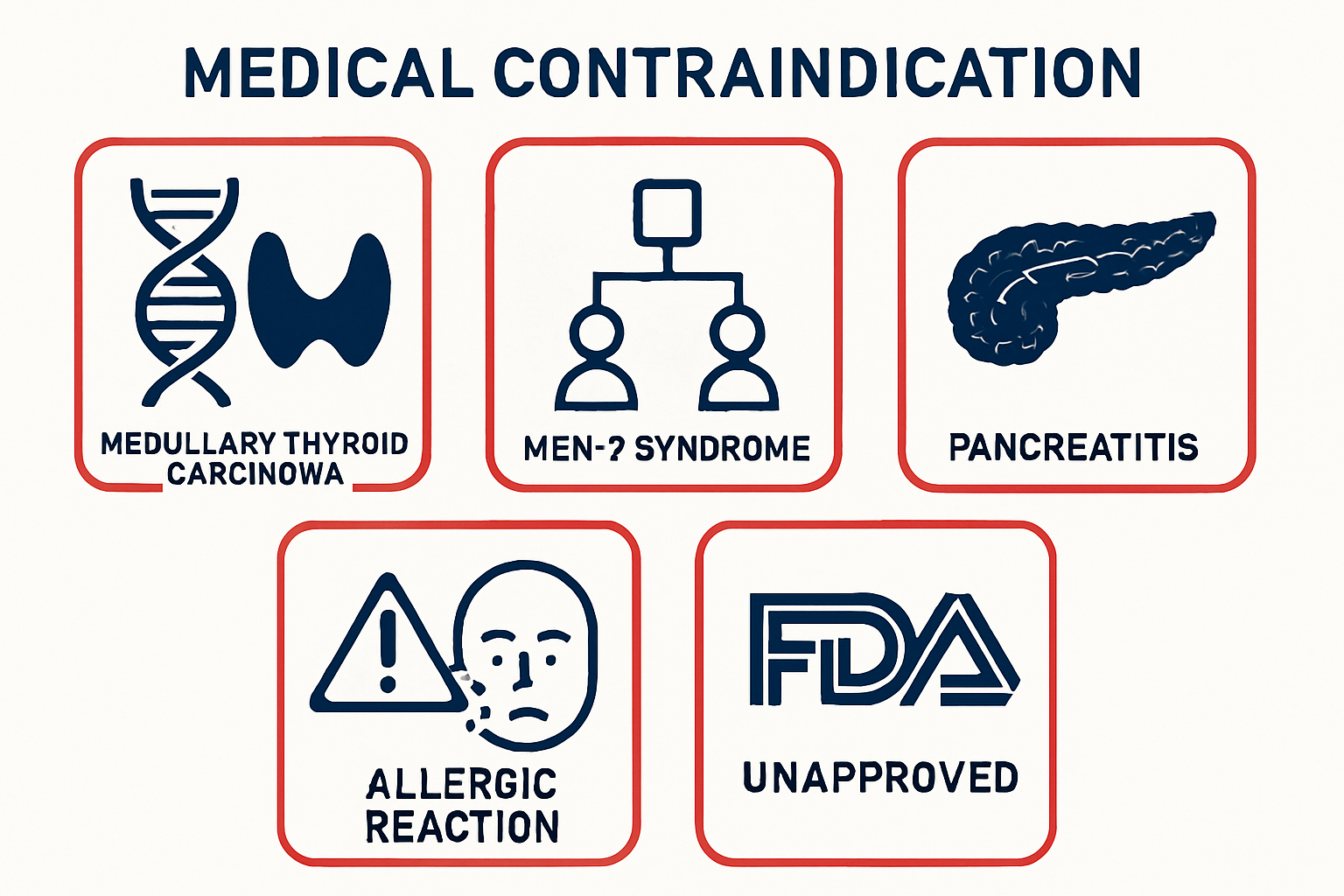
Key Takeaways
- Retatrutide is contraindicated in individuals with personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia syndrome type 2 (MEN-2)
- Pancreatitis history represents a significant contraindication consistent with other incretin-based therapeutic agents
- Retatrutide remains investigational and is not FDA-approved as of 2025, available only through authorized clinical trials
- Research-grade peptides from reputable suppliers like Peptide Pro are labeled “For Research Use Only” and must never be used for human consumption
- Counterfeit and compounded versions pose serious safety risks and are illegal under current federal regulations
Understanding Retatrutide: Mechanism and Research Context
What Is Retatrutide?
Retatrutide represents a novel class of investigational peptide therapeutics known as triple agonists. Unlike single-target compounds, retatrutide simultaneously activates three distinct metabolic pathways:
- GLP-1 (Glucagon-Like Peptide-1) receptors – Involved in glucose regulation and appetite suppression
- GIP (Glucose-Dependent Insulinotropic Polypeptide) receptors – Modulate insulin secretion and energy metabolism
- Glucagon receptors – Influence energy expenditure and metabolic rate
This triple-mechanism approach has generated significant research interest in metabolic studies, though it also introduces complexity regarding potential contraindications and safety considerations.
Current Regulatory Status
⚠️ Critical Regulatory Information:
As of 2025, retatrutide has not received FDA approval for any medical condition. The compound remains in Phase III clinical trials, with final safety and efficacy data still under evaluation. This investigational status carries several important implications:
- No approved medical use exists outside authorized clinical trials
- Contraindications are preliminary and subject to revision based on ongoing research
- Compounding is prohibited under federal law for non-approved active pharmaceutical ingredients
- Research-grade supplies must be clearly labeled for laboratory use only
Researchers and institutions should source high-purity compounds exclusively from legitimate suppliers that maintain strict quality controls and proper documentation. Peptide Pro provides research-grade peptides with comprehensive certificates of analysis and proper storage guidance for laboratory applications.
Primary Retatrutide Contraindications
Personal or Family History of Medullary Thyroid Carcinoma (MTC)
Medullary thyroid carcinoma represents one of the most significant contraindications for retatrutide and related incretin-based compounds. This rare but serious form of thyroid cancer originates from parafollicular C-cells that produce calcitonin.
Why This Matters for Retatrutide
Preclinical rodent studies with GLP-1 receptor agonists have demonstrated thyroid C-cell tumors at clinically relevant exposures. While the relevance to humans remains uncertain, the potential risk necessitates strict contraindication protocols:
- Absolute contraindication in patients with personal history of MTC
- Screening recommendations for calcitonin levels before initiating research protocols
- Immediate discontinuation if thyroid nodules or elevated calcitonin detected
- Family history assessment as part of comprehensive safety screening
🔬 Research Consideration: Laboratory studies involving retatrutide should include thyroid monitoring protocols when applicable to the research model, with particular attention to C-cell proliferation markers.
Multiple Endocrine Neoplasia Syndrome Type 2 (MEN-2)
MEN-2 is an inherited condition characterized by increased risk of multiple endocrine tumors, including medullary thyroid carcinoma, pheochromocytoma, and parathyroid adenomas.
Genetic and Familial Risk Factors
MEN-2 is caused by mutations in the RET proto-oncogene and follows an autosomal dominant inheritance pattern. Key contraindication considerations include:
| MEN-2 Subtype | Primary Features | Retatrutide Contraindication Status |
|---|---|---|
| MEN-2A | MTC, pheochromocytoma, hyperparathyroidism | Absolute contraindication |
| MEN-2B | MTC, pheochromocytoma, mucosal neuromas | Absolute contraindication |
| Familial MTC | MTC only, no other endocrine tumors | Absolute contraindication |
Researchers working with retatrutide must implement comprehensive screening protocols that identify potential genetic predispositions before compound exposure in any applicable research model.
History of Pancreatitis
Acute or chronic pancreatitis represents another critical contraindication for retatrutide, consistent with safety profiles observed across incretin-based therapeutic classes.
Mechanistic Concerns
The relationship between incretin-based compounds and pancreatitis remains an area of active investigation:
- GLP-1 receptor activation may influence pancreatic ductal secretions
- Incretin effects on pancreatic enzyme production require careful monitoring
- Individual susceptibility varies based on underlying pancreatic health
- Risk-benefit assessment must consider pancreatitis history as high-priority exclusion criterion
Warning signs that would necessitate immediate research protocol cessation include:
✋ Severe abdominal pain radiating to the back
✋ Nausea and vomiting
✋ Elevated pancreatic enzymes (lipase, amylase)
✋ Imaging evidence of pancreatic inflammation
Research protocols should exclude subjects with any history of pancreatitis and implement monitoring systems to detect early signs of pancreatic stress.
Secondary Contraindications and Precautions
Allergic Reactions and Hypersensitivity
While comprehensive allergenicity data for retatrutide remains limited due to its investigational status, cross-reactivity concerns exist for individuals with documented allergies to related compounds:
Known Hypersensitivity Risk Factors
- Previous allergic reactions to GLP-1 receptor agonists (semaglutide, liraglutide, dulaglutide)
- Documented sensitivity to GIP receptor agonists or related peptides
- History of anaphylaxis to any peptide-based compound
- Severe reactions to excipients commonly used in peptide formulations
🔍 Research Protocol Recommendation: Establish clear exclusion criteria for subjects with documented peptide allergies and maintain emergency response protocols for unexpected hypersensitivity reactions.
Renal and Hepatic Impairment
Though specific contraindications for renal or hepatic dysfunction have not been definitively established for retatrutide, precautionary principles suggest careful consideration:
Renal Considerations:
- Peptide metabolism and clearance pathways
- Potential accumulation in severe renal impairment
- Monitoring protocols for kidney function markers
- Dose adjustment considerations in research settings
Hepatic Considerations:
- Metabolic processing of triple-agonist compounds
- Potential interactions with hepatic enzyme systems
- Liver function monitoring in extended research protocols
- Exclusion criteria for severe hepatic disease
Research applications should implement comprehensive organ function monitoring, particularly in longer-duration studies or when combining retatrutide with other investigational compounds.
Pregnancy and Reproductive Considerations
Retatrutide has not been studied in pregnant individuals, and substantial data gaps exist regarding reproductive safety:
- Pregnancy category undetermined due to investigational status
- Animal reproductive toxicology data incomplete
- Lactation safety unknown
- Fertility effects require further investigation
Research protocols must exclude pregnant individuals and implement appropriate contraceptive requirements for participants of reproductive potential in applicable study designs.
Regulatory Compliance and Safety in Research Settings
FDA Position on Retatrutide Availability
The U.S. Food and Drug Administration has issued clear guidance regarding retatrutide’s regulatory status in 2025:
“Retatrutide has not been found safe and effective for any condition and cannot be legally used in pharmacy compounding under federal law.”
Implications for Research Applications
This regulatory position creates specific obligations for legitimate research activities:
- Source verification – Compounds must originate from verified research-grade suppliers
- Documentation requirements – Certificates of analysis, purity testing, chain of custody
- Labeling compliance – Clear “For Research Use Only” designations
- Storage protocols – Temperature-controlled environments with monitoring
- Handling procedures – Appropriate safety equipment and contamination prevention
Peptide Pro maintains rigorous quality standards for research-grade peptides, including retatrutide 40mg formulations, with comprehensive documentation and proper storage guidance to support legitimate laboratory research.
Warning Letters and Enforcement Actions
The FDA has actively pursued enforcement against companies illegally distributing retatrutide:
⚖️ Enforcement Focus Areas:
- Unapproved active pharmaceutical ingredients sold for human use
- Compounded preparations claiming therapeutic benefits
- Marketing of investigational compounds outside clinical trials
- Counterfeit products misrepresenting purity or composition
Researchers must ensure their peptide sources comply with all applicable regulations and maintain complete documentation of compound provenance, purity, and handling.
Dangers of Unapproved Sources and Counterfeit Products
The Counterfeit Retatrutide Problem
As research interest in retatrutide has intensified, a dangerous market for counterfeit and unapproved products has emerged:
Common Risks from Illegitimate Sources
| Risk Category | Specific Dangers | Potential Consequences |
|---|---|---|
| Incorrect Dosing | Mislabeled concentrations, inconsistent potency | Unpredictable research outcomes, safety hazards |
| Contamination | Bacterial endotoxins, heavy metals, impurities | Compromised research integrity, health risks |
| Wrong Compound | Substituted peptides, inactive ingredients | Invalid research data, wasted resources |
| No Active Ingredient | Complete absence of retatrutide | Failed experiments, false conclusions |
Identifying Legitimate Research Suppliers
Researchers should apply rigorous criteria when selecting peptide suppliers:
✅ Quality Indicators:
- Published certificates of analysis (COA) with batch-specific data
- HPLC and mass spectrometry verification
- Proper storage and handling documentation
- Clear “For Research Use Only” labeling
- Established business credentials and regulatory compliance
- Temperature-controlled shipping protocols
- Responsive customer support for technical inquiries
Peptide Pro exemplifies these quality standards, providing research-grade peptides with comprehensive documentation, same-day dispatch for orders placed before 1pm (Monday-Friday), and temperature-appropriate packaging to maintain compound integrity.
Legal and Ethical Considerations
Obtaining retatrutide from unapproved sources carries significant risks beyond compound quality:
🚫 Legal Risks:
- Violation of federal drug regulations
- Potential criminal liability for unapproved drug distribution
- Institutional compliance violations
- Loss of research credentials or funding
🚫 Ethical Concerns:
- Compromised research integrity
- Invalid scientific conclusions
- Potential harm to research subjects
- Erosion of public trust in scientific research
Legitimate research activities require complete transparency regarding compound sources, quality verification, and regulatory compliance.
Research Protocol Development and Safety Monitoring
Establishing Contraindication Screening
Research protocols involving retatrutide should implement comprehensive screening procedures:
Pre-Study Screening Elements
- Medical History Assessment
- Detailed thyroid disease history
- Pancreatic disorder screening
- Endocrine tumor family history
- Previous peptide exposure reactions
- Laboratory Baseline Testing
- Calcitonin levels
- Thyroid function panel (TSH, T3, T4)
- Pancreatic enzymes (lipase, amylase)
- Renal function (creatinine, eGFR)
- Hepatic function (ALT, AST, bilirubin)
- Imaging Considerations
- Thyroid ultrasound for high-risk subjects
- Baseline pancreatic imaging if indicated
- Documentation of normal anatomy
- Genetic Screening
- RET proto-oncogene testing for MEN-2 risk
- Family pedigree analysis
- Genetic counseling when appropriate
Ongoing Safety Monitoring
Research protocols should establish regular monitoring intervals:
Weekly Monitoring:
- Adverse event documentation
- Subject-reported symptoms
- Protocol compliance verification
Monthly Assessments:
- Thyroid palpation and symptom screening
- Pancreatic symptom evaluation
- Vital signs and basic metabolic parameters
Quarterly Evaluations:
- Comprehensive laboratory panels
- Calcitonin level trending
- Imaging studies as clinically indicated
- Protocol safety review
🔬 For laboratories conducting in vitro or animal model research, analogous monitoring protocols should assess cellular or physiological markers relevant to known contraindication pathways.
Future Directions: Pending Clinical Trial Data
Phase III Trial Outcomes
As retatrutide progresses through Phase III clinical trials in 2025, additional contraindication data will emerge:
Expected Data Points:
- Refined thyroid safety profile
- Long-term pancreatitis risk assessment
- Cardiovascular safety outcomes
- Renal and hepatic function impacts
- Drug interaction profiles
- Special population considerations
Anticipated Label Development
Upon potential FDA approval, comprehensive prescribing information will include:
- Boxed warnings for serious risks (likely thyroid C-cell tumors)
- Contraindications section with absolute exclusion criteria
- Warnings and precautions for monitored use
- Adverse reactions organized by frequency and severity
- Drug interactions with mechanistic explanations
- Use in specific populations (pregnancy, pediatrics, geriatrics, organ impairment)
Researchers should monitor FDA communications and published trial results to stay current with evolving safety information.
Comparative Contraindications: Retatrutide vs. Related Compounds

GLP-1 Receptor Agonist Class Comparisons
Understanding how retatrutide contraindications compare to established GLP-1 agonists provides valuable context:
| Contraindication | Retatrutide | Semaglutide | Liraglutide | Tirzepatide |
|---|---|---|---|---|
| Personal/Family MTC | Yes | Yes | Yes | Yes |
| MEN-2 Syndrome | Yes | Yes | Yes | Yes |
| Pancreatitis History | Yes | Precaution | Precaution | Precaution |
| Severe GI Disease | Under study | Precaution | Precaution | Precaution |
| Diabetic Retinopathy | Unknown | Precaution | No | Precaution |
Unique Considerations for Triple Agonism
Retatrutide’s triple-agonist mechanism introduces potentially unique contraindication considerations:
Glucagon Receptor Activation:
- Potential cardiovascular effects requiring monitoring
- Hepatic glucose production impacts
- Energy expenditure alterations
- Unknown interactions with metabolic disease states
Combined Receptor Effects:
- Synergistic or antagonistic pathway interactions
- Amplified risk signals across multiple systems
- Novel adverse event profiles not seen with single agonists
Research protocols should account for these mechanistic differences when establishing safety parameters and exclusion criteria.
Best Practices for Research-Grade Peptide Handling
Storage and Stability Considerations
Proper handling of research-grade retatrutide is essential for maintaining compound integrity and research validity:
Lyophilized (Powder) Form
- Storage temperature: -20°C to -80°C for long-term stability
- Humidity control: Desiccated environment to prevent moisture exposure
- Light protection: Amber pen peptides or foil-wrapped containers
- Shelf life: Typically 12-24 months when properly stored
Reconstituted Solutions
- Refrigeration: 2-8°C immediately after reconstitution
- Stability window: Use within 7-14 days (verify with supplier data)
- Sterility maintenance: Aseptic technique for all manipulations
- Aliquoting: Consider single-use portions to minimize freeze-thaw cycles
Peptide Pro’s educational resources provide comprehensive guidance on reconstitution protocols and storage best practices for maintaining research-grade peptide quality.
Documentation and Chain of Custody
Rigorous documentation supports research integrity and regulatory compliance:
📋 Essential Documentation:
- Supplier certificates of analysis
- Batch numbers and expiration dates
- Storage temperature logs
- Reconstitution records with dates and diluents
- Usage logs with dates, quantities, and applications
- Disposal records for expired or contaminated materials
Laboratory Safety Protocols
While retatrutide is a peptide rather than a highly hazardous chemical, appropriate laboratory safety practices apply:
🥽 Personal Protective Equipment:
- Laboratory coat or gown
- Nitrile gloves (peptide-compatible)
- Safety glasses or face shield for reconstitution
- Closed-toe shoes in laboratory environment
Contamination Prevention:
- Dedicated workspace for peptide handling
- Regular surface decontamination
- Proper sharps disposal for needles and syringes
- Spill response protocols and materials
Waste Disposal:
- Compliance with institutional biosafety guidelines
- Proper segregation of peptide waste
- Documentation of disposal methods
- Environmental protection considerations
Research Applications and Study Design Considerations
Appropriate Research Contexts
Retatrutide’s investigational status limits its use to specific research contexts:
Legitimate Research Applications
✅ In Vitro Studies:
- Receptor binding and activation assays
- Cellular signaling pathway investigations
- Metabolic pathway characterization
- Drug interaction screening
✅ Animal Model Research:
- Preclinical efficacy studies
- Pharmacokinetic and pharmacodynamic profiling
- Safety and toxicology assessments
- Mechanism of action elucidation
✅ Authorized Clinical Trials:
- FDA-approved investigational new drug (IND) protocols
- Institutional review board (IRB) oversight
- Informed consent procedures
- Comprehensive safety monitoring
❌ Prohibited Uses:
- Human consumption outside authorized trials
- Veterinary applications without appropriate approvals
- Compounding for therapeutic purposes
- Marketing for unapproved indications
Designing Contraindication-Aware Protocols
Research study design should incorporate contraindication knowledge:
Inclusion/Exclusion Criteria:
- Explicit exclusion of subjects with contraindicated conditions
- Screening procedures to identify at-risk populations
- Documentation of contraindication assessment
- Regular review and updates based on emerging safety data
Risk Mitigation Strategies:
- Dose escalation protocols with safety checkpoints
- Enhanced monitoring for high-risk populations
- Predetermined stopping rules for safety signals
- Data safety monitoring board oversight for clinical research
Ethical Considerations:
- Transparent disclosure of investigational status
- Clear communication of known contraindications
- Appropriate risk-benefit assessment
- Protection of vulnerable populations
Emerging Safety Signals and Pharmacovigilance
Post-Marketing Surveillance Expectations
When and if retatrutide receives regulatory approval, robust pharmacovigilance systems will monitor for safety signals:
Surveillance Mechanisms:
- FDA Adverse Event Reporting System (FAERS)
- Manufacturer-sponsored registries
- Real-world evidence collection
- Electronic health record data mining
Signal Detection Focus Areas:
- Rare adverse events not detected in clinical trials
- Long-term safety outcomes
- Special population effects
- Drug-drug interaction identification
Current Research Safety Monitoring
For ongoing research applications in 2025, proactive safety monitoring is essential:
🔍 Active Surveillance:
- Literature review for emerging safety publications
- Clinical trial database monitoring (ClinicalTrials.gov)
- FDA safety communications and warning letters
- Professional society guidance updates
Internal Reporting:
- Institutional adverse event reporting systems
- Principal investigator notification protocols
- IRB safety update submissions
- Regulatory authority reporting as required
International Regulatory Perspectives
Global Regulatory Status
Retatrutide’s regulatory status varies internationally, with implications for research activities:
| Region | Regulatory Status | Research Implications |
|---|---|---|
| United States | Investigational, not approved | Clinical trials only; research-grade for laboratory use |
| European Union | Under EMA review | Similar restrictions to US |
| United Kingdom | MHRA evaluation ongoing | Research use with appropriate approvals |
| Canada | Health Canada assessment | Investigational status |
| Australia | TGA review process | Restricted to authorized research |
Researchers conducting international collaborations must navigate multiple regulatory frameworks and ensure compliance with local requirements.
Import/Export Considerations
Cross-border movement of research-grade peptides requires careful attention to regulations:
Documentation Requirements:
- Material transfer agreements
- Import/export licenses
- Customs declarations
- End-use certifications
Compliance Obligations:
- International trafficking regulations
- Dual-use technology restrictions
- Biosecurity protocols
- Institutional approval processes
Peptide Pro provides international shipping options with appropriate documentation and compliance support for legitimate research applications worldwide.
Frequently Asked Questions About Retatrutide Contraindications
Can retatrutide be used if I have hypothyroidism?
Hypothyroidism (underactive thyroid) is distinct from the thyroid cancer contraindications for retatrutide. While medullary thyroid carcinoma represents an absolute contraindication, routine hypothyroidism managed with thyroid hormone replacement does not automatically exclude retatrutide use in research contexts. However, comprehensive thyroid assessment including calcitonin screening remains important.
Are there age-related contraindications?
Specific age-based contraindications have not been definitively established for retatrutide as of 2025, though pediatric and geriatric populations typically require special consideration in research protocols. Clinical trials have primarily enrolled adults, and safety data in children and older adults remain limited. Research protocols should establish age-appropriate inclusion criteria based on study objectives and safety considerations.
How long do contraindications persist after stopping retatrutide?
For absolute contraindications like personal history of MTC or MEN-2, the contraindication is permanent and does not change with time. These conditions represent inherent risk factors independent of retatrutide exposure duration. For other precautionary conditions, individual assessment is required based on the specific medical situation and evolving safety data.
Can genetic testing identify contraindications?
Genetic testing for RET proto-oncogene mutations can identify individuals with MEN-2 syndrome or hereditary medullary thyroid carcinoma risk, representing a valuable screening tool for research protocols. However, genetic testing is not routinely required unless family history or clinical features suggest hereditary endocrine tumor syndromes. Research protocols should establish clear criteria for when genetic screening is appropriate.
What should I do if I develop symptoms while using retatrutide in a research study?
Immediate reporting of any concerning symptoms to the research team is essential. Specific symptoms requiring urgent evaluation include:
- Lump or swelling in the neck
- Persistent hoarseness or difficulty swallowing
- Severe abdominal pain
- Persistent nausea or vomiting
- Allergic reaction symptoms (rash, difficulty breathing, swelling)
Research protocols should provide 24/7 contact information for reporting adverse events and clear guidance on when to seek emergency medical care.
Conclusion: Prioritizing Safety in Retatrutide Research
Understanding retatrutide contraindications is fundamental to conducting safe, ethical, and scientifically valid research with this investigational triple-agonist peptide. As research continues and regulatory evaluation progresses through 2025, the contraindication profile will continue to evolve based on accumulating clinical evidence.
Key Principles for Researchers
Absolute Contraindications to Remember:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2 (MEN-2)
- History of pancreatitis
- Documented hypersensitivity to related peptide compounds
Regulatory Compliance Essentials:
- Source research-grade peptides only from verified, reputable suppliers
- Maintain comprehensive documentation of compound quality and handling
- Respect the investigational status and “Research Use Only” designation
- Avoid unapproved sources, compounded products, and counterfeit materials
Safety Monitoring Imperatives:
- Implement comprehensive screening for contraindicated conditions
- Establish regular monitoring protocols for thyroid and pancreatic safety
- Maintain vigilance for emerging safety signals
- Report adverse events through appropriate channels
Next Steps for Researchers
For laboratories and institutions conducting legitimate research with retatrutide:
- Review and update protocols to incorporate current contraindication knowledge
- Verify peptide sources meet quality and regulatory standards
- Implement screening procedures to identify contraindicated conditions
- Establish monitoring systems for ongoing safety surveillance
- Stay informed about evolving safety data and regulatory guidance
Peptide Pro supports the research community with high-purity, research-grade peptides including retatrutide, comprehensive quality documentation, and expert guidance on proper handling and storage protocols. With same-day dispatch for orders placed before 1pm Monday-Friday and temperature-controlled shipping, researchers can trust that their compounds arrive in optimal condition for scientific investigation.
The Path Forward
As retatrutide advances through clinical development, the scientific community will gain increasingly refined understanding of its safety profile, contraindications, and appropriate use contexts. Researchers play a vital role in generating this knowledge while maintaining the highest standards of safety, ethics, and regulatory compliance.
The contraindications outlined in this guide reflect current understanding as of 2025, but should be viewed as evolving rather than static. Continuous engagement with emerging literature, regulatory communications, and professional guidance ensures research activities remain aligned with best practices and patient safety priorities.
Remember: Retatrutide remains strictly for research use only. It is not approved for human consumption, cannot be legally compounded, and should never be obtained from unapproved sources. Legitimate scientific research requires unwavering commitment to quality, safety, and regulatory compliance.
For questions about research-grade peptides, quality documentation, or proper handling protocols, contact the Peptide Pro team for professional support tailored to your research needs.
