The relationship between novel therapeutic peptides and organ function has become a critical area of scientific investigation, particularly as multi-receptor agonists enter advanced research phases. Retatrutide kidney function represents one of the most important safety considerations for researchers working with this triple agonist compound, as understanding its renal impact is essential for comprehensive preclinical assessment. With retatrutide demonstrating remarkable efficacy in metabolic research models, the scientific community has intensified its focus on characterizing how this peptide interacts with kidney physiology and whether it presents any concerns for renal health.
As laboratories worldwide expand their investigations into retatrutide’s mechanisms and effects, the question of kidney safety has moved to the forefront of research protocols. This comprehensive examination explores the current understanding of retatrutide kidney function, drawing from available research data, mechanistic studies, and comparative analyses with related compounds.
Key Takeaways
- Retatrutide’s triple receptor mechanism (GIP, GLP-1, glucagon) may offer inherent renal protective properties through multiple metabolic pathways
- Current research data suggests retatrutide demonstrates a favorable kidney safety profile with potential benefits for renal function markers
- Monitoring protocols should include eGFR, serum creatinine, and urinary albumin measurements in research settings
- Comparative evidence from related incretin-based compounds indicates possible nephroprotective effects worth investigating
- Proper research practices require careful documentation of kidney function parameters throughout experimental protocols
Understanding Retatrutide’s Mechanism and Kidney Physiology
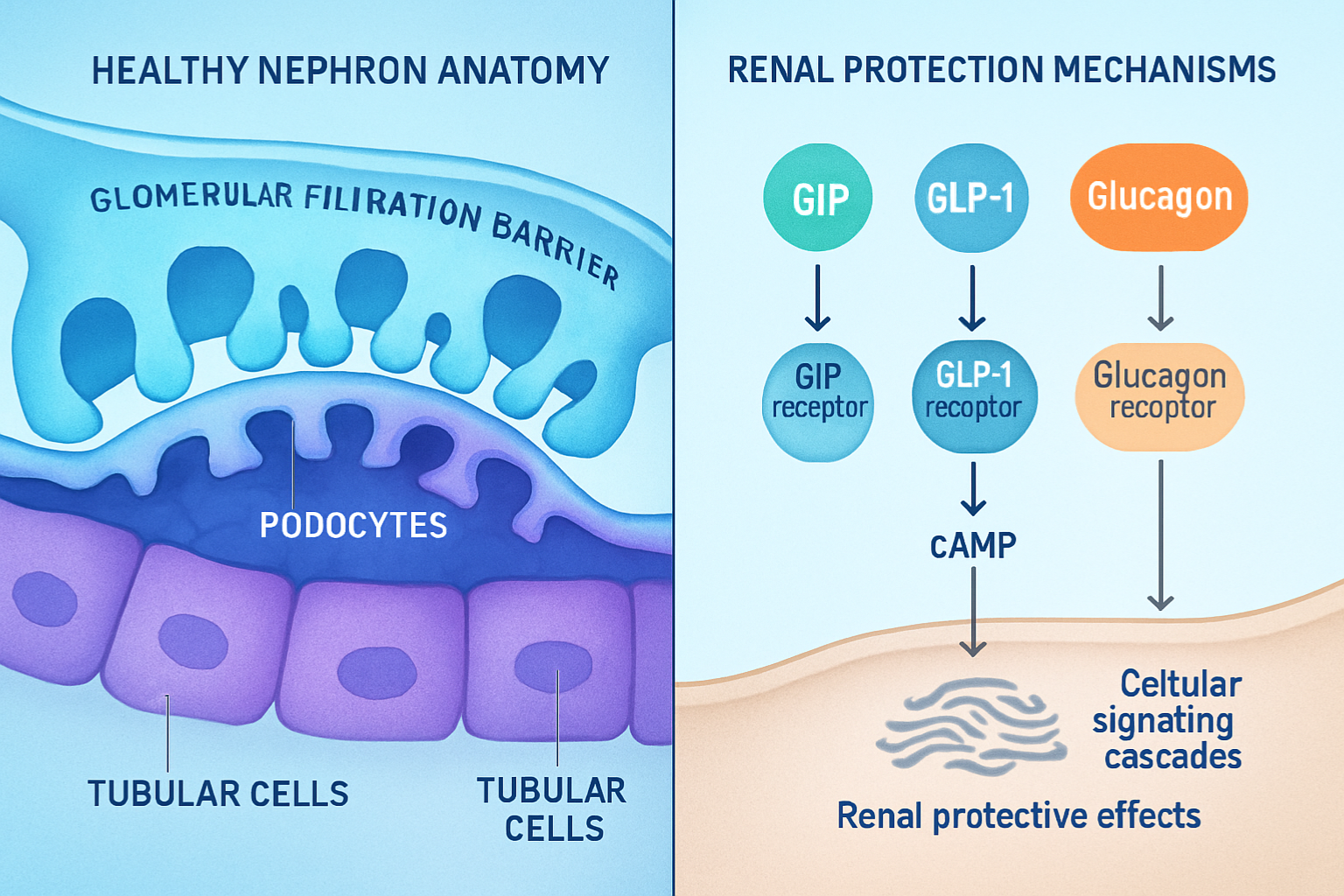
The Triple Agonist Approach
Retatrutide functions as a simultaneous agonist of three distinct receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This triple mechanism distinguishes it from earlier incretin-based compounds and creates a unique pharmacological profile that researchers must consider when evaluating retatrutide kidney function implications.
The kidney expresses receptors for all three of these pathways, making renal tissue a relevant target for retatrutide’s activity. GLP-1 receptors are present in renal vasculature, proximal tubules, and glomerular structures. GIP receptors have been identified in various kidney cell types, while glucagon receptors play roles in renal glucose handling and metabolic regulation.
Renal Expression of Target Receptors
Understanding where and how these receptors function in kidney tissue provides crucial context for interpreting retatrutide kidney function research:
GLP-1 Receptors in Kidney:
- Located in renal vasculature, influencing blood flow regulation
- Present in proximal tubular cells, affecting sodium reabsorption
- Found in glomerular endothelial cells, potentially modulating filtration
GIP Receptors in Kidney:
- Expressed in renal tubular structures
- May influence local inflammatory responses
- Potentially involved in metabolic signaling within kidney tissue
Glucagon Receptors in Kidney:
- Concentrated in proximal tubules
- Regulate gluconeogenesis in renal tissue
- Influence sodium and water handling
For researchers sourcing high-purity research peptides for kidney function studies, understanding this receptor distribution is essential for designing appropriate experimental protocols.
Research Evidence on Retatrutide Kidney Function
Clinical Trial Safety Data
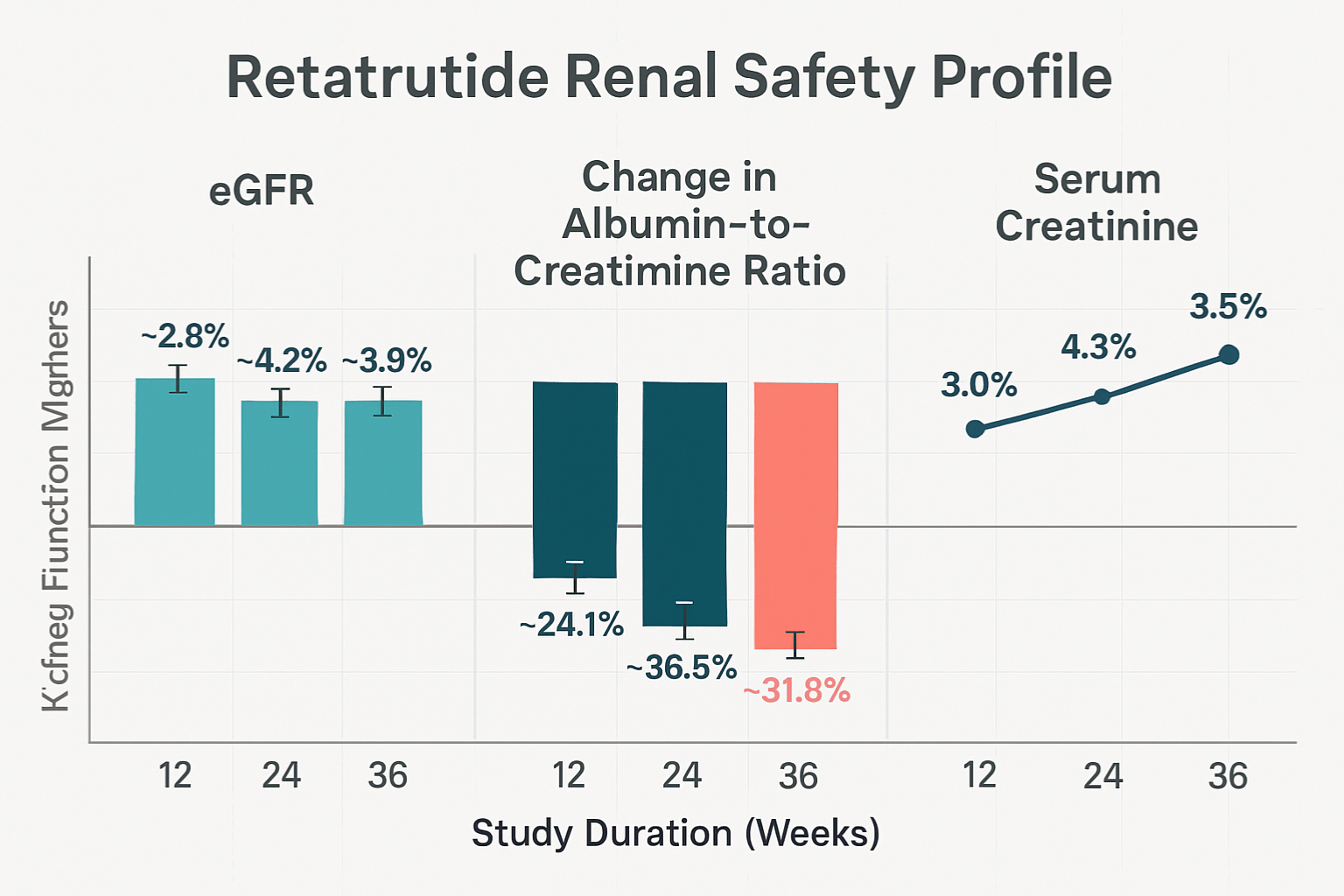
Available research data from retatrutide investigations provides valuable insights into its renal safety profile. Phase 2 clinical trials evaluating retatrutide for metabolic conditions have included comprehensive kidney function monitoring as standard safety assessments.
| Study Parameter | Baseline Assessment | Follow-up Monitoring | Key Findings |
|---|---|---|---|
| eGFR (estimated Glomerular Filtration Rate) | Measured at screening | Assessed at regular intervals | Generally stable or improved |
| Serum Creatinine | Baseline measurement | Monitored throughout study | No clinically significant elevations |
| Urinary Albumin-to-Creatinine Ratio | Initial assessment | Periodic evaluation | Trends toward improvement in some subjects |
| Blood Urea Nitrogen | Pre-treatment baseline | Regular monitoring | Remained within normal ranges |
These findings suggest that retatrutide kidney function impact appears favorable in research settings, though ongoing investigation continues to characterize long-term effects and dose-dependent relationships.
Comparative Analysis with Related Compounds
Examining kidney function data from related incretin-based compounds provides additional context for understanding retatrutide’s renal profile:
GLP-1 Receptor Agonists: Research with selective GLP-1 agonists has demonstrated potential nephroprotective effects, including reduced albuminuria, preserved glomerular filtration rate, and decreased progression of diabetic kidney disease in preclinical models. These findings suggest the GLP-1 component of retatrutide’s mechanism may contribute positively to kidney health.
Dual GIP/GLP-1 Agonists: Tirzepatide, another dual agonist compound, has shown favorable kidney function profiles in research studies, with some evidence suggesting improvements in renal parameters. This provides encouraging precedent for retatrutide’s triple agonist approach.
Researchers interested in comparative peptide studies can explore how different receptor activation patterns influence renal outcomes.
Mechanisms of Potential Renal Protection
Several biological mechanisms may explain why retatrutide kidney function data appears favorable:
🔬 Metabolic Improvements: Enhanced glucose control and insulin sensitivity reduce metabolic stress on kidney tissue, potentially protecting against hyperglycemia-induced renal damage.
🔬 Blood Pressure Effects: Modest blood pressure reductions observed with incretin-based compounds may decrease intraglomerular pressure and reduce mechanical stress on filtration structures.
🔬 Anti-inflammatory Actions: GLP-1 and GIP receptor activation may exert anti-inflammatory effects in kidney tissue, potentially reducing chronic inflammatory damage.
🔬 Oxidative Stress Reduction: Improved metabolic parameters may decrease oxidative stress in renal cells, protecting against reactive oxygen species damage.
🔬 Lipid Metabolism: Favorable effects on lipid profiles may reduce lipotoxicity in kidney tissue, particularly relevant for metabolic research models.
Monitoring Retatrutide Kidney Function in Research Settings
Essential Biomarkers and Assessment Tools
Comprehensive evaluation of retatrutide kidney function requires systematic monitoring of multiple renal parameters throughout research protocols. Establishing baseline measurements and tracking changes over time provides crucial safety data and mechanistic insights.
Primary Kidney Function Markers
Estimated Glomerular Filtration Rate (eGFR): The gold standard for assessing overall kidney function, eGFR provides an estimate of how efficiently kidneys filter blood. Research protocols should calculate eGFR using validated equations (CKD-EPI or MDRD) based on serum creatinine, age, sex, and race variables.
- Baseline assessment: Essential before initiating retatrutide research
- Monitoring frequency: Every 4-12 weeks depending on protocol duration
- Interpretation: Values >90 mL/min/1.73m² indicate normal function; declining trends warrant investigation
Serum Creatinine: This waste product measurement reflects kidney filtration capacity. Elevated levels may indicate reduced renal function, though factors like muscle mass influence results.
- Normal ranges: 0.7-1.3 mg/dL (males), 0.6-1.1 mg/dL (females)
- Monitoring: Regular assessments throughout research period
- Considerations: Acute elevations may reflect dehydration or other factors beyond direct kidney damage
Blood Urea Nitrogen (BUN): Measures nitrogen from urea, a protein breakdown product. Elevated BUN may indicate kidney dysfunction, though diet and hydration status also influence levels.
- Normal range: 7-20 mg/dL
- BUN/Creatinine ratio: Provides additional diagnostic information
- Limitations: Less specific than creatinine for kidney function
Advanced Renal Assessment Parameters
For comprehensive retatrutide kidney function research, additional markers provide deeper insights:
Urinary Albumin-to-Creatinine Ratio (UACR): This sensitive marker detects early kidney damage before eGFR changes become apparent. Albuminuria indicates glomerular barrier dysfunction.
- Normal: <30 mg/g
- Microalbuminuria: 30-300 mg/g
- Macroalbuminuria: >300 mg/g
- Research value: Early indicator of potential renal stress
Cystatin C: An alternative filtration marker less influenced by muscle mass than creatinine, providing complementary assessment of kidney function.
Urinary Biomarkers:
- Kidney Injury Molecule-1 (KIM-1): Tubular injury marker
- Neutrophil Gelatinase-Associated Lipocalin (NGAL): Early damage indicator
- N-acetyl-β-D-glucosaminidase (NAG): Tubular enzyme reflecting cellular damage
Researchers utilizing research-grade compounds should implement comprehensive monitoring protocols that include both standard and advanced biomarkers for complete safety assessment.
Recommended Monitoring Schedule
| Assessment Point | Parameters | Rationale |
|---|---|---|
| Pre-Research Screening | eGFR, serum creatinine, BUN, UACR, urinalysis | Establish baseline kidney function; identify pre-existing conditions |
| Week 2-4 | Serum creatinine, BUN | Early detection of acute changes |
| Week 8-12 | eGFR, serum creatinine, BUN, UACR | Mid-protocol comprehensive assessment |
| Week 16-24 | Full panel including advanced markers | Extended monitoring for longer protocols |
| Post-Research Follow-up | eGFR, serum creatinine, UACR | Assess any persistent changes after compound discontinuation |
Interpreting Changes in Kidney Function Markers
Understanding how to interpret retatrutide kidney function data requires consideration of multiple factors:
Expected Variations: Minor fluctuations in creatinine (±0.2 mg/dL) and eGFR (±5 mL/min/1.73m²) may occur due to hydration status, dietary protein intake, or circadian rhythms rather than true kidney function changes.
Clinically Significant Changes:
- eGFR decline >25% from baseline
- Serum creatinine increase >0.5 mg/dL
- Development of new or worsening albuminuria
- BUN elevation disproportionate to creatinine changes
Confounding Factors to Consider:
- Dehydration or volume depletion
- Concomitant medications affecting kidney function
- Dietary changes (high protein intake)
- Acute illness or infection
- Changes in muscle mass affecting creatinine production
“Comprehensive kidney function monitoring throughout research protocols provides essential safety data and contributes to our understanding of how novel peptides interact with renal physiology. Systematic assessment of multiple biomarkers offers far more insight than any single measurement.” — Research Best Practices, 2025
Potential Renal Benefits of Retatrutide
Nephroprotective Mechanisms Under Investigation
Emerging research suggests that retatrutide kidney function effects may extend beyond simple safety to potential protective benefits. Understanding these mechanisms helps researchers design studies that can properly evaluate renal outcomes.
Hemodynamic Effects
Retatrutide’s influence on systemic and renal hemodynamics may contribute to kidney protection:
Blood Pressure Modulation: Incretin-based compounds have demonstrated modest blood pressure reductions, potentially through:
- Natriuretic effects (increased sodium excretion)
- Improved endothelial function
- Reduced sympathetic nervous system activity
- Direct vasodilatory effects on renal vasculature
Intraglomerular Pressure: By influencing systemic blood pressure and potentially affecting afferent and efferent arteriolar tone, retatrutide may reduce excessive intraglomerular pressure, a key factor in progressive kidney damage.
Metabolic Protection
The metabolic improvements associated with retatrutide research may indirectly protect kidney function:
Glucose Control:
- Reduces hyperglycemia-induced oxidative stress in renal cells
- Decreases advanced glycation end-product formation
- Minimizes glucose-mediated inflammatory pathways
- Protects podocyte structure and function
Lipid Metabolism:
- Reduces circulating triglycerides and free fatty acids
- Decreases lipotoxicity in renal tubular cells
- May reduce foam cell formation in kidney tissue
- Improves overall metabolic environment
Weight Effects:
- Reduces obesity-related glomerular hyperfiltration
- Decreases mechanical stress on kidney structures
- May improve obesity-related glomerulopathy
- Reduces systemic inflammatory burden
Anti-inflammatory and Anti-fibrotic Potential
Research into GLP-1 and GIP receptor signaling has revealed anti-inflammatory properties that may benefit kidney tissue:
Inflammatory Pathway Modulation:
- Reduced NF-κB activation in renal cells
- Decreased production of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β)
- Modulation of macrophage polarization toward anti-inflammatory phenotypes
- Reduced oxidative stress through enhanced antioxidant defenses
Fibrosis Prevention:
- Potential inhibition of TGF-β signaling pathways
- Reduced extracellular matrix accumulation
- Decreased myofibroblast activation
- Protection against epithelial-to-mesenchymal transition
Researchers exploring peptide compounds with tissue-protective properties may find these mechanisms particularly relevant for comparative studies.
Research Models for Studying Renal Effects
Investigating retatrutide kidney function comprehensively requires appropriate experimental models:
In Vitro Models:
- Primary renal cell cultures (podocytes, tubular epithelial cells, mesangial cells)
- Kidney organoid systems
- Co-culture models representing glomerular structures
- High-glucose or inflammatory stress models
Preclinical Models:
- Metabolic dysfunction models with kidney involvement
- Hypertensive models with renal complications
- Age-related kidney function decline models
- Acute kidney injury models
Biomarker Studies:
- Longitudinal assessment of renal function markers
- Proteomic analysis of urinary proteins
- Metabolomic profiling of renal metabolites
- Imaging studies of kidney structure and perfusion
Safety Considerations and Risk Mitigation
Identifying Populations Requiring Enhanced Monitoring
While retatrutide kidney function data appears favorable overall, certain research contexts require particularly careful renal monitoring:
Pre-existing Kidney Dysfunction
Research involving subjects or models with baseline kidney impairment necessitates enhanced vigilance:
Mild to Moderate Renal Impairment (eGFR 30-89 mL/min/1.73m²):
- More frequent kidney function assessments
- Careful monitoring for acute changes
- Attention to volume status and hydration
- Documentation of any concurrent medications affecting kidneys
Severe Renal Impairment (eGFR <30 mL/min/1.73m²):
- May require dose adjustments in research protocols
- Intensive monitoring schedule
- Consideration of altered pharmacokinetics
- Assessment of metabolite accumulation
Dialysis-Dependent Models:
- Special considerations for timing relative to dialysis
- Modified assessment parameters
- Careful documentation of dialysis effects on measurements
Concurrent Factors Affecting Kidney Function
Researchers must account for variables that may interact with retatrutide kidney function effects:
Medications with Renal Impact:
- NSAIDs (non-steroidal anti-inflammatory drugs)
- ACE inhibitors and ARBs
- Diuretics
- Certain antibiotics
- Contrast agents
Physiological Stressors:
- Dehydration or volume depletion
- Acute illness or infection
- Extreme environmental conditions
- Intense physical activity protocols
Metabolic Factors:
- Severe hyperglycemia
- Ketoacidosis in metabolic models
- Electrolyte imbalances
- Acid-base disturbances
Hydration and Volume Status Management
Proper hydration represents a critical factor in maintaining accurate retatrutide kidney function assessments:
💧 Hydration Protocols:
- Establish consistent hydration guidelines for research subjects
- Monitor fluid intake and output when feasible
- Consider hydration status when interpreting kidney function markers
- Account for retatrutide’s potential effects on fluid balance
💧 Volume Assessment:
- Monitor for signs of volume depletion
- Track weight changes as volume indicator
- Assess for orthostatic changes suggesting volume issues
- Consider impact of concurrent medications on volume status
Gastrointestinal Effects and Renal Function
Retatrutide, like other incretin-based compounds, may cause gastrointestinal effects that could indirectly impact kidney function:
Nausea and Vomiting:
- May lead to reduced fluid intake
- Potential for volume depletion
- Risk of electrolyte disturbances
- Importance of monitoring hydration status
Diarrhea:
- Fluid and electrolyte losses
- Potential for acute kidney injury if severe
- Need for supportive care and monitoring
- Documentation of gastrointestinal symptoms
Mitigation Strategies:
- Gradual dose escalation protocols
- Supportive care for gastrointestinal symptoms
- Proactive hydration guidance
- Temporary dose reduction if symptoms severe
Laboratories sourcing research compounds should implement comprehensive protocols addressing these potential confounding factors.
Comparative Kidney Function Data: Retatrutide vs. Other Compounds
GLP-1 Receptor Agonists
Understanding how retatrutide kidney function compares to selective GLP-1 agonists provides valuable context:
Liraglutide:
- Demonstrated renal benefits in large cardiovascular outcome trials
- Reduced albuminuria in research models
- Slowed eGFR decline in diabetic kidney disease models
- Established nephroprotective profile
Semaglutide:
- Similar renal benefits to liraglutide
- Reduced risk of kidney disease progression
- Decreased albuminuria
- Favorable kidney safety profile across trials
Dulaglutide:
- Consistent kidney function preservation
- Reduced composite renal outcomes
- Lower rates of new-onset macroalbuminuria
Dual GIP/GLP-1 Agonists
Tirzepatide: As the most closely related compound to retatrutide, tirzepatide provides particularly relevant comparative data:
- Demonstrated eGFR preservation in clinical trials
- Significant reductions in urinary albumin-to-creatinine ratio
- No safety signals for acute kidney injury
- Potential for superior metabolic improvements translating to kidney benefits
The addition of glucagon receptor agonism in retatrutide represents the key differentiator, raising questions about whether this third mechanism enhances, diminishes, or neutrally affects the kidney benefits observed with dual agonism.
SGLT2 Inhibitors
While mechanistically distinct, SGLT2 inhibitors provide context for understanding kidney-protective metabolic therapies:
Mechanism Comparison:
- SGLT2 inhibitors work directly on kidney glucose reabsorption
- Retatrutide acts systemically through incretin and glucagon pathways
- Both may reduce intraglomerular pressure through different mechanisms
- Potential for complementary effects in research models
Kidney Outcomes:
- SGLT2 inhibitors show robust kidney protection data
- Reduced progression of chronic kidney disease
- Decreased risk of kidney failure
- Benefits extend beyond glucose control
Research Implications
These comparisons suggest several research priorities for retatrutide kidney function investigation:
- Head-to-head studies comparing retatrutide with selective GLP-1 agonists and dual agonists
- Mechanistic research elucidating the specific contribution of glucagon receptor agonism
- Long-term studies assessing sustained kidney function effects
- Combination protocols exploring retatrutide with other kidney-protective agents
- Biomarker research identifying predictors of renal response
Researchers conducting comparative peptide studies can contribute valuable data to this evolving understanding.
Practical Research Protocols for Kidney Function Assessment
Designing Retatrutide Studies with Renal Endpoints
Comprehensive investigation of retatrutide kidney function requires thoughtful protocol design incorporating appropriate endpoints, monitoring schedules, and analytical approaches.
Primary Renal Endpoints
Hard Kidney Outcomes:
- Progression to end-stage renal disease
- Sustained eGFR decline ≥40% from baseline
- Doubling of serum creatinine
- Initiation of renal replacement therapy
- Kidney-related mortality
Intermediate Endpoints:
- Change in eGFR over time (slope analysis)
- Change in urinary albumin-to-creatinine ratio
- Progression or regression of albuminuria categories
- Composite renal outcomes combining multiple parameters
Exploratory Endpoints:
- Novel urinary biomarkers (KIM-1, NGAL, NAG)
- Kidney imaging parameters (size, perfusion, fibrosis markers)
- Renal tubular function markers
- Proteomic or metabolomic signatures
Sample Size and Statistical Considerations
Adequately powered studies require careful calculation based on expected effect sizes:
For eGFR Endpoints:
- Consider baseline eGFR variability
- Account for expected rate of decline in control group
- Determine minimum clinically meaningful difference
- Calculate sample size with appropriate statistical power (typically 80-90%)
For Albuminuria Endpoints:
- Account for high biological variability
- Consider log-transformation for analysis
- Determine threshold for clinically meaningful change
- Plan for adequate sample size given variability
Longitudinal Analysis:
- Mixed-effects models for repeated measures
- Intention-to-treat and per-protocol analyses
- Handling of missing data
- Sensitivity analyses for robustness
Standard Operating Procedures for Kidney Function Monitoring
Establishing consistent protocols ensures reliable retatrutide kidney function data:
Sample Collection and Handling
Blood Samples:
- Fasting status: Preferably 8-12 hour fast for consistency
- Timing: Morning collection to minimize diurnal variation
- Handling: Prompt processing and separation of serum/plasma
- Storage: Appropriate temperature maintenance for biomarker stability
Urine Samples:
- First morning void preferred for UACR
- Timed collections for some advanced markers
- Proper container selection (preservatives when needed)
- Immediate refrigeration if processing delayed
Quality Control Measures
✅ Standardization:
- Use same laboratory for all measurements when possible
- Ensure assay calibration and quality control
- Document any method changes during study
- Maintain chain of custody for samples
✅ Documentation:
- Record collection time and conditions
- Note any factors affecting results (medications, hydration, illness)
- Track sample storage conditions
- Maintain detailed laboratory records
✅ Validation:
- Repeat measurements for unexpected results
- Cross-reference multiple kidney function markers
- Investigate discordant findings
- Maintain blinding when appropriate
Data Management and Analysis
Proper handling of retatrutide kidney function data ensures accurate interpretation:
Database Structure:
- Longitudinal format capturing all time points
- Linked demographic and clinical variables
- Medication and concomitant treatment tracking
- Adverse event documentation
Statistical Approaches:
- Baseline characteristics comparison
- Longitudinal mixed-effects modeling
- Time-to-event analysis for hard outcomes
- Subgroup analyses by baseline kidney function
- Adjustment for confounding variables
Visualization:
- Individual trajectory plots
- Mean change over time with confidence intervals
- Waterfall plots for individual responses
- Kaplan-Meier curves for time-to-event outcomes
Research teams utilizing high-purity peptides from reliable sources can ensure that compound quality doesn’t introduce confounding variables into kidney function assessments.
Future Directions in Retatrutide Kidney Function Research
Unanswered Questions and Research Gaps
Despite encouraging preliminary data, several important questions about retatrutide kidney function remain:
Long-Term Kidney Outcomes
Duration of Studies: Current research provides relatively short-term safety data, but kidney disease often progresses over years or decades. Key questions include:
- Does retatrutide preserve kidney function over multi-year timeframes?
- Are there delayed effects (positive or negative) that emerge with extended use?
- How does discontinuation affect kidney function trajectories?
- What is the optimal duration of treatment for maximal renal benefit?
Progression Prevention:
- Can retatrutide slow or prevent progression in established kidney disease?
- What stages of kidney dysfunction respond best?
- Are effects sustained after treatment cessation?
- How do effects compare to established nephroprotective therapies?
Mechanism-Specific Research
Glucagon Receptor Contribution: The unique aspect of retatrutide compared to dual agonists is glucagon receptor activation. Critical mechanistic questions include:
- Does glucagon receptor agonism enhance or diminish kidney protection?
- What are the direct effects of glucagon signaling on renal cells?
- How does glucagon receptor activation affect renal hemodynamics?
- Are there dose-dependent relationships between glucagon activity and kidney effects?
Receptor-Specific Effects:
- Can selective receptor blockade experiments isolate individual contributions?
- How do the three receptor pathways interact in kidney tissue?
- Are there synergistic or antagonistic effects on renal function?
- Which receptor activation is most important for kidney outcomes?
Special Populations
Advanced Kidney Disease:
- Safety and efficacy in severe renal impairment (eGFR <30 mL/min/1.73m²)
- Appropriate dosing in kidney dysfunction
- Pharmacokinetics in renal impairment
- Potential benefits versus risks in this population
Acute Kidney Injury:
- Effects in acute kidney injury models
- Potential for accelerating recovery
- Risk of worsening acute injury
- Timing considerations relative to injury
Kidney Transplant Recipients:
- Safety in immunosuppressed populations
- Interactions with transplant medications
- Effects on graft function
- Long-term graft preservation potential
Emerging Technologies and Approaches
Advanced methodologies may provide deeper insights into retatrutide kidney function mechanisms:
Omics Technologies
Proteomics:
- Urinary proteome profiling to identify early kidney damage signatures
- Kidney tissue proteomics in preclinical models
- Identification of novel biomarkers for retatrutide effects
- Pathway analysis revealing mechanistic insights
Metabolomics:
- Urinary metabolite changes reflecting kidney metabolism
- Systemic metabolic changes affecting kidney function
- Identification of metabolic signatures predicting renal response
- Understanding metabolic pathways influenced by retatrutide
Transcriptomics:
- Gene expression changes in kidney tissue
- Single-cell RNA sequencing of renal cell populations
- Identification of cell-type-specific responses
- Temporal dynamics of gene expression changes
Advanced Imaging
Functional MRI:
- Non-invasive assessment of kidney perfusion
- Measurement of glomerular filtration without contrast
- Evaluation of renal oxygenation
- Longitudinal structural and functional assessment
Molecular Imaging:
- PET tracers for receptor expression mapping
- Inflammation imaging in kidney tissue
- Fibrosis-specific imaging agents
- Real-time assessment of kidney metabolism
Computational Modeling
Systems Biology Approaches:
- Integration of multi-omics data
- Pathway modeling of retatrutide effects
- Prediction of long-term outcomes
- Identification of patient/model characteristics predicting response
Artificial Intelligence:
- Machine learning for biomarker discovery
- Prediction models for kidney outcomes
- Image analysis for automated kidney assessment
- Integration of complex multi-parameter data
Translational Research Priorities
Bridging the gap between basic research and clinical application requires focused translational studies:
🔬 Biomarker Validation:
- Prospective validation of novel kidney injury markers
- Correlation with hard clinical outcomes
- Development of point-of-care testing
- Standardization across laboratories
🔬 Mechanistic Studies:
- Detailed receptor signaling pathway analysis
- Cell-type-specific effects in kidney
- Interaction with other kidney-protective pathways
- Identification of synergistic interventions
🔬 Precision Medicine:
- Identification of genetic variants affecting response
- Biomarkers predicting kidney benefit
- Personalized dosing strategies
- Subpopulation-specific effects
Research institutions and laboratories can access research-grade retatrutide to contribute to these important investigational priorities.
Best Practices for Researchers Working with Retatrutide
Quality Assurance in Peptide Research
Ensuring reliable retatrutide kidney function data begins with high-quality research materials and rigorous protocols:
Compound Quality and Verification
Purity Standards:
- Use research-grade peptides with documented purity ≥95%
- Request certificates of analysis (COA) for each batch
- Verify identity through mass spectrometry when possible
- Ensure proper storage conditions maintained throughout supply chain
Reconstitution and Handling:
- Follow manufacturer guidelines for reconstitution
- Use appropriate sterile diluents (bacteriostatic water, sterile saline)
- Avoid vigorous shaking that may denature peptide
- Prepare fresh solutions or store according to stability data
Storage Considerations:
- Lyophilized powder: Store at -20°C or -80°C in desiccated conditions
- Reconstituted solutions: Refrigerate at 2-8°C, use within recommended timeframe
- Avoid freeze-thaw cycles
- Protect from light when indicated
Dosing Accuracy:
- Use calibrated equipment for measurement
- Account for peptide purity in dose calculations
- Document actual administered doses
- Verify concentration through appropriate assays
Researchers can ensure compound quality by sourcing from reputable suppliers with rigorous quality control, such as PEPTIDE PRO, which provides high-purity research-grade peptides with comprehensive documentation.
Ethical Considerations and Regulatory Compliance
Responsible research requires adherence to ethical principles and regulatory requirements:
Research Use Only
⚠️ Critical Reminder: Retatrutide and similar research peptides are strictly for research use only. They are not approved for human consumption, clinical use, or therapeutic application outside of properly authorized clinical trials.
Appropriate Use:
- In vitro cellular studies
- Preclinical animal models (with appropriate ethical approval)
- Authorized clinical trials with regulatory oversight
- Mechanistic and pharmacological research
Prohibited Use:
- Human self-administration
- Veterinary therapeutic use
- Sale or distribution for consumption
- Marketing for off-label purposes
Documentation and Transparency
Research Records:
- Maintain detailed laboratory notebooks
- Document all procedures, observations, and results
- Record compound lot numbers and expiration dates
- Track any deviations from protocols
Data Integrity:
- Accurate recording of all measurements
- No selective reporting of results
- Transparent acknowledgment of limitations
- Proper statistical analysis and interpretation
Publication Ethics:
- Accurate reporting of methods and results
- Disclosure of funding sources and conflicts of interest
- Appropriate authorship attribution
- Data sharing when appropriate
Collaboration and Knowledge Sharing
Advancing understanding of retatrutide kidney function benefits from collaborative research efforts:
Multi-Center Studies:
- Larger sample sizes for robust conclusions
- Diverse populations and research models
- Shared expertise and resources
- Standardized protocols across sites
Interdisciplinary Approaches:
- Collaboration between metabolic and nephrology researchers
- Integration of basic science and clinical research
- Computational and experimental partnerships
- Industry-academic collaborations
Open Science Practices:
- Preregistration of study protocols
- Sharing of data and materials when appropriate
- Publication of negative results
- Contribution to systematic reviews and meta-analyses
The Current State of Retatrutide Kidney Function Research
The evolving understanding of retatrutide kidney function presents an encouraging picture for researchers investigating this novel triple agonist compound. Available evidence suggests a favorable renal safety profile, with emerging data indicating potential nephroprotective benefits that warrant further investigation.
Summary of Key Findings
Safety Profile: Current research demonstrates that retatrutide appears safe from a kidney function perspective, with clinical trial data showing stable or improved renal parameters across multiple studies. No significant safety signals for acute kidney injury or progressive renal dysfunction have emerged from available research.
Potential Benefits: Mechanistic evidence and comparative data from related compounds suggest retatrutide may offer kidney protection through multiple pathways:
- Improved metabolic parameters reducing kidney stress
- Favorable hemodynamic effects on renal circulation
- Anti-inflammatory actions in kidney tissue
- Potential anti-fibrotic properties
Research Needs: Substantial questions remain that require focused investigation:
- Long-term kidney outcomes over years of exposure
- Effects in advanced kidney disease populations
- Specific contribution of glucagon receptor agonism
- Optimal monitoring strategies and biomarkers
- Comparative effectiveness versus other nephroprotective therapies
Actionable Next Steps for Researchers
For laboratories and research teams investigating retatrutide kidney function, several practical steps can advance the field:
- Implement Comprehensive Monitoring: Include multiple kidney function biomarkers in research protocols, extending beyond basic creatinine and eGFR to include albuminuria, novel injury markers, and advanced assessments.
- Standardize Protocols: Adopt consistent methodologies for sample collection, processing, and analysis to enable cross-study comparisons and meta-analyses.
- Investigate Mechanisms: Design studies specifically addressing the mechanisms underlying retatrutide’s renal effects, including receptor-specific contributions and cellular pathways.
- Focus on Special Populations: Conduct targeted research in models with pre-existing kidney dysfunction, acute injury, or other relevant conditions.
- Ensure Quality: Source high-purity research peptides from reputable suppliers and maintain rigorous quality control throughout research protocols.
- Share Findings: Contribute data to the scientific literature, including negative results, to build comprehensive understanding of retatrutide’s renal profile.
- Collaborate: Engage in multi-center and interdisciplinary research efforts to address complex questions requiring diverse expertise and larger sample sizes.
The Path Forward
As retatrutide research continues to expand in 2025 and beyond, kidney function assessment will remain a critical component of comprehensive safety and efficacy evaluation. The intersection of metabolic pharmacology and nephrology offers rich opportunities for discovery, with potential implications extending beyond retatrutide to inform our understanding of how multi-receptor agonism influences organ function.
The favorable preliminary data on retatrutide kidney function should not lead to complacency but rather inspire rigorous, systematic investigation that fully characterizes this compound’s renal effects across diverse contexts and timeframes. By maintaining high standards for research quality, embracing collaborative approaches, and pursuing mechanistic understanding alongside clinical outcomes, the scientific community can build a comprehensive evidence base that advances both basic knowledge and translational applications.
For researchers ready to contribute to this important area of investigation, access to high-quality research materials represents the essential foundation upon which reliable, reproducible, and meaningful research is built.
