When considering the intersection of novel peptide therapeutics and maternal health, retatrutide breastfeeding safety represents a critical knowledge gap that demands careful scientific examination. As retatrutide emerges as a promising triple-agonist peptide in metabolic research, questions surrounding its compatibility with lactation have become increasingly important for researchers, healthcare professionals, and the scientific community. This comprehensive guide examines the current evidence, safety considerations, and research protocols surrounding retatrutide use during breastfeeding periods.
Key Takeaways
- No clinical data exists confirming the safety of retatrutide during breastfeeding; the compound remains strictly for research use only
- Peptide transfer into breast milk is theoretically possible but remains unstudied for retatrutide specifically
- Molecular characteristics of retatrutide suggest potential systemic effects that warrant extreme caution in lactating populations
- Research-grade peptides from reputable suppliers like PEPTIDE PRO are intended exclusively for laboratory investigation, not human consumption
- Alternative approaches and proper timing considerations are essential for any future clinical research protocols
Understanding Retatrutide: Mechanism and Research Applications
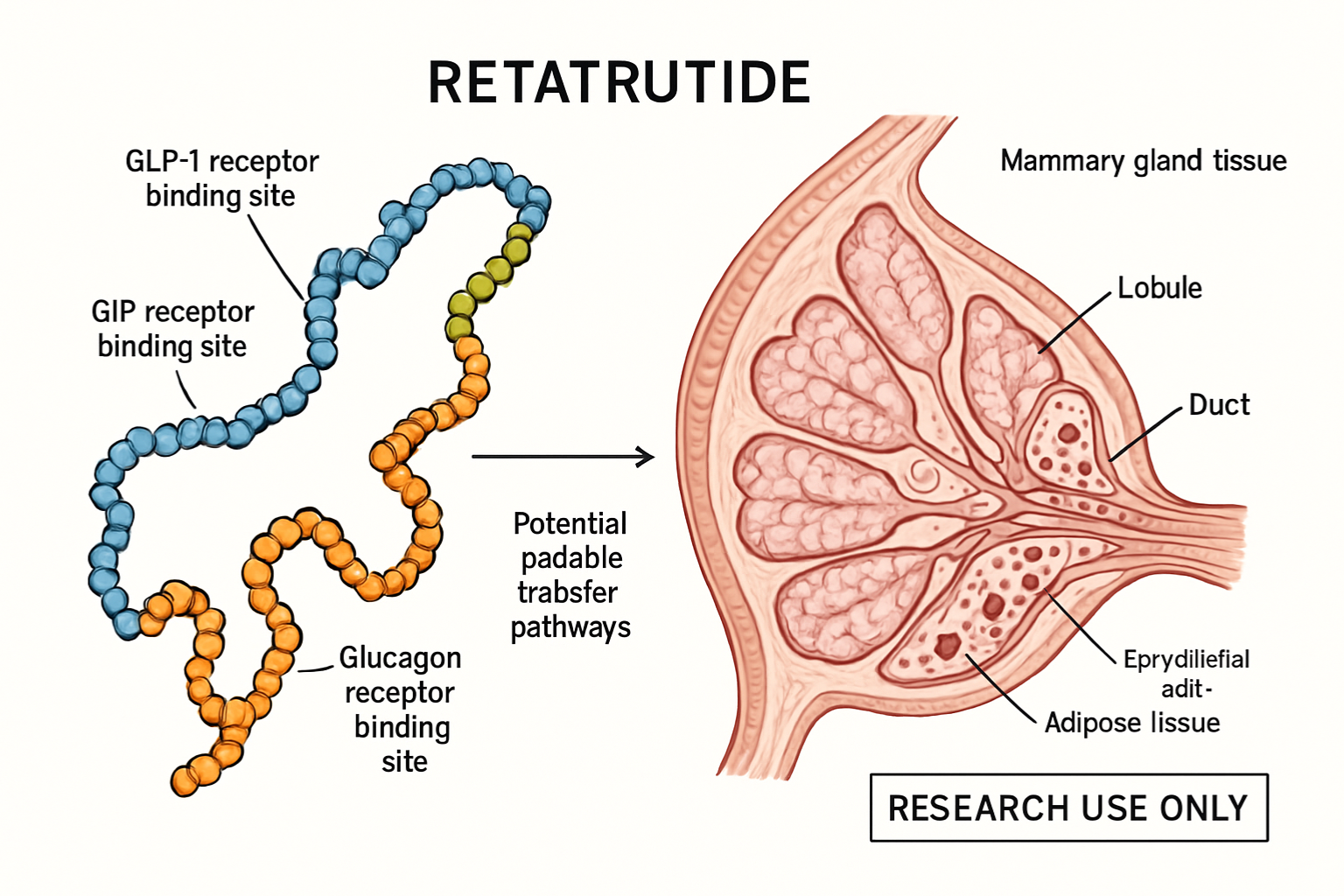
What Is Retatrutide?
Retatrutide represents an advanced class of peptide therapeutics designed as a triple receptor agonist targeting GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors simultaneously. This multi-targeted approach distinguishes it from single or dual-agonist compounds currently under investigation.
The molecular structure of retatrutide allows it to:
✅ Enhance insulin secretion through GLP-1 and GIP pathway activation
✅ Promote satiety signals via central nervous system receptor engagement
✅ Increase energy expenditure through glucagon receptor stimulation
✅ Modulate metabolic processes across multiple physiological systems
Current Research Status
As of 2025, retatrutide remains under active investigation in controlled research settings. Research-grade peptides are supplied exclusively for laboratory use, with strict protocols governing their handling and application. The compound has not received regulatory approval for clinical use in any population, including non-lactating individuals.
Important Research Context:
| Research Parameter | Status |
|---|---|
| Human clinical trials | Limited, ongoing |
| Breastfeeding safety data | None available |
| Regulatory approval | Not approved |
| Lactation transfer studies | Not conducted |
| Long-term safety profile | Under investigation |
Retatrutide Breastfeeding Safety: Current Evidence
The Critical Data Gap
The most significant concern regarding retatrutide breastfeeding compatibility is the complete absence of safety data in lactating populations. No published studies have examined:
- Transfer rates into human breast milk
- Concentration levels in lactating tissue
- Bioavailability to nursing infants
- Short-term or long-term effects on infant development
- Impact on milk production or composition
⚠️ Research Principle: The absence of evidence is not evidence of safety. Without dedicated lactation studies, retatrutide must be considered potentially hazardous during breastfeeding periods.
Theoretical Transfer Mechanisms
While specific data for retatrutide remains unavailable, understanding peptide pharmacokinetics provides insight into potential transfer pathways:
Molecular Weight Considerations
Retatrutide’s molecular structure falls within a range where some degree of transfer into breast milk is biochemically plausible. Peptides with molecular weights below 1,000 Da typically show higher transfer rates, though larger peptides can still appear in milk through various mechanisms.
Lipophilicity and Protein Binding
The compound’s lipophilic properties and protein-binding characteristics influence its distribution throughout maternal tissues, including mammary glands. Higher protein binding generally reduces free drug available for milk transfer, but doesn’t eliminate the possibility.
Active Transport Systems
Mammary epithelial cells contain various transport mechanisms that can actively move compounds from maternal circulation into milk, regardless of passive diffusion characteristics.
Comparison With Related Peptides
Examining similar peptide therapeutics offers contextual understanding, though direct extrapolation remains scientifically inappropriate:
GLP-1 Agonists:
Compounds like semaglutide and liraglutide carry explicit warnings against use during breastfeeding due to insufficient safety data. Their structural similarities to retatrutide suggest comparable caution is warranted.
Dual Agonists:
Tirzepatide, a GLP-1/GIP dual agonist, also lacks breastfeeding safety data. As retatrutide adds glucagon receptor activity to this profile, theoretical concerns are amplified rather than diminished.
Physiological Considerations for Retatrutide Breastfeeding Research
Maternal Metabolic Changes During Lactation
The lactation period involves profound metabolic adaptations that could theoretically interact with retatrutide’s mechanism of action:
Energy Balance Shifts 🔄
Lactation increases maternal energy expenditure by approximately 500 calories daily. Retatrutide’s effects on energy metabolism could potentially interfere with the delicate balance required for adequate milk production.
Glucose Homeostasis
Nursing mothers experience altered glucose metabolism to support lactose synthesis. Triple-agonist activity affecting insulin, GIP, and glucagon pathways might disrupt these carefully regulated processes.
Appetite Regulation
The satiety-promoting effects of retatrutide could theoretically reduce maternal food intake below levels necessary to support both maternal health and milk production demands.
Infant Exposure Pathways
Should retatrutide transfer into breast milk, multiple exposure scenarios warrant consideration:
- Direct ingestion through nursing
- Gastrointestinal absorption in the infant (variable for peptides)
- Systemic effects if absorption occurs
- Developmental impacts during critical growth periods
Infant Vulnerability Factors:
- 🍼 Immature metabolic enzyme systems
- 🧬 Developing endocrine regulation
- 📊 Higher body surface area to weight ratio
- ⚡ Rapid cellular division and differentiation
Milk Production Concerns
Beyond direct infant exposure, retatrutide’s metabolic effects could theoretically impact lactation itself:
Hormonal Regulation
Milk production depends on complex hormonal signaling involving prolactin, oxytocin, and metabolic hormones. Disruption of glucose and energy homeostasis could interfere with these pathways.
Nutritional Substrate Availability
Adequate maternal nutrition supports milk synthesis. Appetite suppression and altered nutrient partitioning might compromise milk composition or volume.
Research-Grade Peptides: Quality and Safety Standards
PEPTIDE PRO’s Research Commitment
When investigating compounds like retatrutide, research quality begins with peptide purity and proper handling. PEPTIDE PRO maintains rigorous standards for research-grade materials:
Quality Assurance Protocols:
✓ High-purity synthesis under controlled laboratory conditions
✓ Certificate of Analysis (COA) documentation for every batch
✓ Proper storage in temperature-controlled environments
✓ Clear labeling as “For Research Use Only”
✓ Fast UK delivery with appropriate packaging for peptide stability
Proper Research Protocols
Investigating retatrutide breastfeeding safety would require adherence to strict research ethics and methodologies:
Preclinical Requirements:
- Comprehensive animal lactation studies
- Milk transfer quantification
- Offspring developmental monitoring
- Multi-generational safety assessment
Clinical Research Standards:
- Institutional review board approval
- Informed consent with explicit risk disclosure
- Rigorous safety monitoring protocols
- Immediate intervention criteria
🔬 Research Principle: All peptide research must prioritize safety, transparency, and adherence to established scientific and ethical standards.
Alternative Research Approaches and Timing Considerations
Research Timeline Planning
For any future investigation of retatrutide in populations that may include lactating individuals, careful timeline consideration is essential:
Pre-Conception Phase
Ideally, metabolic research involving novel peptides would be conducted well before conception, allowing adequate washout periods and baseline metabolic assessment.
Postpartum Timing
If research participation is considered postpartum, waiting until after complete weaning provides the safest approach, eliminating infant exposure risks entirely.
Washout Periods
Understanding retatrutide’s half-life and elimination kinetics is crucial for determining appropriate intervals between last exposure and conception or nursing initiation.
Alternative Research Compounds
The broader research landscape includes various peptides with different safety profiles and research applications. Researchers might consider:
Established Peptides with More Data:
Compounds like BPC-157 or TB-500 have longer research histories, though they too require careful safety evaluation in special populations.
Single-Target Approaches:
Investigating individual pathway components rather than triple-agonist compounds might offer more controlled research conditions with clearer safety profiles.
Risk Mitigation Strategies
For research protocols that might inadvertently include lactating participants, robust screening and exclusion criteria are essential:
Screening Requirements:
- ✅ Pregnancy testing before enrollment
- ✅ Lactation status verification
- ✅ Contraception requirements during study periods
- ✅ Regular monitoring for pregnancy/lactation status
- ✅ Clear protocols for immediate discontinuation
Regulatory Perspectives and Research Ethics
Current Regulatory Framework
Regulatory agencies worldwide maintain conservative positions on novel therapeutics during lactation:
FDA Lactation Labeling
The U.S. Food and Drug Administration requires comprehensive lactation data including:
- Milk transfer studies
- Effects on milk production
- Infant exposure levels
- Developmental and health effects in nursing infants
EMA Requirements
The European Medicines Agency similarly demands robust safety data before any compound can be considered compatible with breastfeeding.
UK MHRA Standards
The UK Medicines and Healthcare products Regulatory Agency applies rigorous safety standards for lactating populations, recognizing the vulnerability of both mother and infant.
Ethical Research Principles
The retatrutide breastfeeding question intersects with fundamental research ethics:
Beneficence and Non-Maleficence
Research must maximize potential benefits while minimizing harm. Without safety data, exposing nursing infants to retatrutide violates the non-maleficence principle.
Informed Consent
True informed consent requires comprehensive risk disclosure. The absence of safety data makes adequate informed consent impossible for lactating participants.
Vulnerable Populations
Both lactating mothers and nursing infants qualify as vulnerable populations requiring enhanced protections in research settings.
Justice in Research
While metabolic research is important, the burden of risk must not fall disproportionately on vulnerable populations without clear potential for direct benefit.
Clinical Considerations for Healthcare Professionals
Patient Counseling Frameworks
Healthcare professionals encountering questions about retatrutide breastfeeding should employ clear, evidence-based communication:
Key Counseling Points:
- No safety data exists for retatrutide during lactation
- Research-only status means the compound is not approved for any clinical use
- Theoretical risks include infant exposure and lactation disruption
- Alternative approaches should be explored for metabolic health goals
- Timing considerations favor waiting until after complete weaning
Documentation and Reporting
Proper documentation serves both patient safety and scientific advancement:
Essential Documentation:
- 📋 Detailed medication/supplement history
- 📋 Lactation status and nursing frequency
- 📋 Any inadvertent exposures
- 📋 Monitoring parameters if exposure occurs
- 📋 Infant health and development tracking
Adverse Event Reporting:
Any suspected adverse effects from retatrutide exposure during lactation should be reported to appropriate regulatory authorities to build the safety database.
Multidisciplinary Collaboration
Optimal patient care requires coordination across specialties:
Key Collaborators:
- 🏥 Endocrinology/Metabolism specialists
- 🏥 Obstetrics and maternal-fetal medicine
- 🏥 Pediatrics and neonatology
- 🏥 Lactation consultants
- 🏥 Clinical pharmacists
- 🏥 Research ethics committees
Future Research Directions
Needed Studies for Retatrutide Breastfeeding Safety
Establishing a comprehensive safety profile for retatrutide breastfeeding would require extensive investigation:
Preclinical Research:
- Lactation transfer studies in appropriate animal models
- Dose-response relationships in milk
- Offspring developmental assessments
- Multi-generational safety evaluation
- Mechanistic studies of mammary gland effects
Clinical Research:
- Pharmacokinetic studies in postpartum women
- Milk sampling and quantification protocols
- Infant exposure modeling
- Lactation performance metrics
- Long-term developmental follow-up
Advancing Peptide Safety Science
The broader field of peptide therapeutics would benefit from:
Standardized Testing Protocols
Development of consistent methodologies for evaluating peptide transfer into breast milk across different molecular classes.
Predictive Modeling
Computational approaches to estimate lactation transfer based on molecular characteristics, reducing the need for extensive empirical testing in every case.
Registry Development
Systematic collection of exposure data when inadvertent use occurs, building real-world evidence to complement controlled studies.
Personalized Medicine Approaches
Future research may enable more nuanced risk-benefit assessments:
Biomarker Development
Identification of maternal or infant biomarkers that predict transfer rates or sensitivity to peptide exposure.
Genetic Factors
Understanding how genetic variations in metabolic pathways or transport systems influence safety profiles.
Individualized Timing
Refined protocols for determining optimal washout periods based on individual pharmacokinetic parameters.
Practical Guidance for Research Institutions

Laboratory Safety Protocols
Institutions working with research-grade peptides must maintain rigorous safety standards:
Handling Requirements:
- 🔬 Designated research-only areas
- 🔬 Proper personal protective equipment
- 🔬 Controlled access and inventory systems
- 🔬 Clear labeling and storage protocols
- 🔬 Waste disposal procedures
Personnel Training:
- Understanding of “research use only” restrictions
- Proper reconstitution and handling techniques
- Storage requirements (typically 2-8°C for reconstituted peptides)
- Emergency response procedures
- Ethical research principles
Institutional Review Board Considerations
Research protocols involving retatrutide must address lactation safety:
Required Protocol Elements:
- Clear exclusion criteria for pregnant/lactating individuals
- Pregnancy testing requirements and frequency
- Contraception mandates during study participation
- Washout period specifications before conception attempts
- Procedures for handling inadvertent pregnancy/lactation
Risk Assessment Documentation:
- Comprehensive literature review of related compounds
- Theoretical risk analysis based on mechanism of action
- Justification for inclusion/exclusion criteria
- Monitoring and intervention plans
Metabolic Health Alternatives During Lactation
Evidence-Based Approaches
For individuals seeking metabolic health optimization during lactation, established approaches offer safer alternatives:
Nutritional Interventions:
- 🥗 Balanced macronutrient distribution supporting both maternal health and milk production
- 🥗 Adequate protein intake (additional 25g daily during lactation)
- 🥗 Complex carbohydrates for sustained energy
- 🥗 Healthy fats supporting infant brain development
- 🥗 Micronutrient adequacy through diet or supplementation
Physical Activity:
🏃♀
️ Gradual return to exercise postpartum
🏃♀
️ Moderate-intensity cardiovascular activity
🏃♀
️ Resistance training for metabolic health
🏃♀
️ Flexibility and core strengthening
🏃♀
️ Appropriate energy balance to support lactation
Lifestyle Modifications:
- 😴 Prioritizing sleep (despite challenges with infant care)
- 💧 Adequate hydration
- 🧘♀️ Stress management techniques
- 📅 Regular health monitoring
- 🤝 Social support systems
Postpartum Metabolic Considerations
The postpartum period presents unique metabolic challenges and opportunities:
Physiological Changes:
- Insulin sensitivity improvements in many individuals
- Altered glucose metabolism
- Changes in lipid profiles
- Hormonal fluctuations affecting metabolism
- Gradual return to pre-pregnancy baseline
Timing Considerations:
- Immediate postpartum (0-6 weeks): Focus on recovery
- Early lactation (6 weeks-6 months): Establish nursing, gentle interventions
- Extended lactation (6+ months): Broader intervention options
- Post-weaning: Full range of metabolic interventions available
The Role of Quality Research Suppliers
PEPTIDE PRO’s Commitment to Research Excellence
Advancing scientific understanding of compounds like retatrutide requires access to high-quality research materials. PEPTIDE PRO supports the research community through:
Product Quality:
- ✨ Research-grade purity standards
- ✨ Rigorous quality control testing
- ✨ Proper storage and handling throughout supply chain
- ✨ Comprehensive product documentation
- ✨ Certificates of Analysis for verification
Researcher Support:
- 📞 Professional customer service team
- 📞 Technical guidance on handling and storage
- 📞 Fast UK delivery (same-day dispatch for orders before 1pm Mon-Fri)
- 📞 International shipping options
- 📞 Transparent pricing and secure transactions
Ethical Standards:
- ⚖️ Clear “For Research Use Only” labeling
- ⚖️ No claims regarding human consumption
- ⚖️ Responsible marketing practices
- ⚖️ Support for legitimate research applications
- ⚖️ Commitment to regulatory compliance
Building the Evidence Base
Quality research suppliers play a crucial role in advancing scientific knowledge:
Enabling Rigorous Studies
Consistent, high-purity peptides ensure research reproducibility and reliable results, essential for building the safety database needed to answer questions about retatrutide breastfeeding and other applications.
Supporting Innovation
Access to novel compounds like retatrutide enables researchers to explore new therapeutic approaches and expand scientific understanding.
Maintaining Standards
Reputable suppliers reinforce the distinction between research materials and clinical therapeutics, supporting appropriate use and regulatory frameworks.
Global Perspectives on Peptide Research Safety
International Research Standards
Different regions approach peptide research with varying frameworks:
United Kingdom:
- Strong regulatory oversight through MHRA
- Research ethics committee requirements
- Clear distinction between research and clinical use
- Robust pharmacovigilance systems
European Union:
- Harmonized standards across member states
- EMA coordination of safety data
- Stringent research ethics requirements
- Comprehensive post-market surveillance
United States:
- FDA oversight of investigational compounds
- IRB requirements for human research
- NIH guidelines for federally funded research
- State-level additional regulations
Australia and New Zealand:
- TGA (Therapeutic Goods Administration) oversight
- ANZCTR trial registration requirements
- Ethics committee approval processes
- Indigenous health research considerations
Cultural Considerations
Breastfeeding practices and attitudes vary globally, influencing research approaches:
Duration Norms:
- WHO recommends breastfeeding for 2+ years
- Cultural practices range from months to several years
- Impact on research exclusion period planning
Support Systems:
- Varying levels of lactation support and education
- Access to alternatives if research participation desired
- Community attitudes toward research participation
Monitoring and Surveillance Frameworks
Establishing Safety Databases
Comprehensive safety assessment requires systematic data collection:
Registry Components:
- Exposure circumstances (intentional vs. inadvertent)
- Timing relative to nursing (during vs. after weaning)
- Dosing information
- Maternal outcomes
- Infant outcomes
- Lactation performance metrics
Long-Term Follow-Up:
- Infant growth and development
- Metabolic health markers
- Neurodevelopmental milestones
- Reproductive health in offspring
- Multi-generational effects
Pharmacovigilance Systems
Active surveillance helps identify safety signals:
Reporting Mechanisms:
- Healthcare provider reporting channels
- Patient/participant self-reporting
- Manufacturer reporting obligations
- Regulatory agency databases
- Academic research publications
Signal Detection:
- Statistical analysis of adverse event patterns
- Comparison with background rates
- Causality assessment
- Risk communication strategies
Legal and Liability Considerations
Research Institution Responsibilities
Institutions conducting peptide research bear significant responsibilities:
Duty of Care:
- Adequate participant screening
- Comprehensive informed consent
- Appropriate safety monitoring
- Rapid response to adverse events
- Proper documentation and reporting
Liability Protections:
- Research insurance coverage
- Clear protocol adherence
- Documented decision-making processes
- Regular ethics committee review
- Compliance with regulatory requirements
Participant Protections
Research participants, particularly vulnerable populations, require robust safeguards:
Informed Consent Elements:
- Clear explanation of research vs. clinical care
- Comprehensive risk disclosure
- Right to withdraw without penalty
- Compensation for research-related injury
- Privacy and confidentiality protections
Special Protections for Lactating Individuals:
- Enhanced screening procedures
- Additional safety monitoring
- Consideration of infant welfare
- Access to lactation support
- Clear communication about unknowns
Prioritizing Safety in Retatrutide Breastfeeding Research
The intersection of retatrutide breastfeeding and scientific inquiry highlights the critical importance of evidence-based decision-making in peptide research. As of 2025, the complete absence of safety data regarding retatrutide use during lactation necessitates a cautious, conservative approach that prioritizes the wellbeing of both nursing mothers and their infants.
Key Conclusions
Current Evidence Status:
No published research examines retatrutide transfer into breast milk, infant exposure levels, or safety in nursing populations. This fundamental knowledge gap precludes any recommendation for use during lactation.
Theoretical Concerns:
Retatrutide’s mechanism as a triple receptor agonist affecting glucose metabolism, appetite regulation, and energy expenditure raises multiple theoretical concerns regarding both direct infant exposure and potential disruption of maternal lactation physiology.
Research Quality Imperative:
Investigations involving novel peptides require access to high-purity research materials handled according to rigorous protocols, ensuring reliable results that can inform future safety assessments.
Ethical Framework:
The principles of beneficence, non-maleficence, and special protection for vulnerable populations must guide all research involving lactating individuals and nursing infants.
Actionable Next Steps
For Researchers:
- Design comprehensive preclinical lactation studies
- Develop standardized milk transfer assessment protocols
- Establish long-term offspring monitoring frameworks
- Collaborate across disciplines for holistic safety evaluation
- Source research-grade peptides from reputable suppliers
For Healthcare Professionals:
- Counsel patients about the absence of safety data
- Document any inadvertent exposures thoroughly
- Report suspected adverse events to regulatory authorities
- Recommend evidence-based alternatives for metabolic health
- Coordinate multidisciplinary care for complex cases
For Research Institutions:
- Implement robust screening for pregnancy and lactation status
- Ensure IRB protocols adequately address lactation safety
- Provide comprehensive researcher training on ethical principles
- Maintain rigorous safety monitoring systems
- Support development of needed safety databases
For Lactating Individuals:
- Avoid exposure to research-only compounds like retatrutide
- Consult healthcare providers about metabolic health goals
- Pursue evidence-based approaches to postpartum wellness
- Wait until after complete weaning before considering novel interventions
- Report any inadvertent exposures to healthcare providers immediately
The Path Forward
Advancing scientific understanding of retatrutide breastfeeding safety requires sustained, systematic research efforts spanning preclinical models, clinical pharmacokinetics, and long-term surveillance. Until such evidence exists, the research and medical communities must maintain clear boundaries between investigational compounds and clinical applications, particularly in vulnerable populations.
The commitment to rigorous science, ethical research practices, and patient safety will ultimately enable evidence-based recommendations that protect maternal and infant health while advancing therapeutic innovation. Organizations like PEPTIDE PRO play a vital role in this ecosystem by providing high-quality research materials that enable the scientific investigations necessary to answer critical safety questions.
Final Reminder: Retatrutide and all research-grade peptides are strictly for laboratory research use only. They are not approved for human consumption, and no safety data supports their use during pregnancy, lactation, or any other clinical scenario. Always consult qualified healthcare professionals for medical advice and treatment decisions.
