When researchers first began investigating retatrutide’s triple receptor agonist properties, an unexpected pattern emerged in laboratory observations: subjects consistently exhibited signs of fluid imbalance. This discovery has positioned retatrutide dehydration as a critical consideration for any research protocol involving this novel peptide compound. Understanding the mechanisms, risk factors, and prevention strategies surrounding retatrutide-induced dehydration has become essential for maintaining research integrity and ensuring accurate experimental outcomes.
Retatrutide dehydration represents more than a simple side effect—it reflects the compound’s profound influence on metabolic processes, fluid regulation, and renal function. As research into this triple agonist peptide expands throughout 2025, laboratories worldwide are developing sophisticated protocols to monitor and mitigate dehydration risks while preserving the compound’s remarkable metabolic effects.
Key Takeaways
- Retatrutide dehydration occurs through multiple mechanisms including increased diuresis, reduced fluid intake, and altered renal sodium handling
- Triple receptor agonism (GIP, GLP-1, glucagon) creates unique dehydration risk profiles not seen with single or dual agonists
- Research protocols must incorporate systematic hydration monitoring and electrolyte assessment to ensure data validity
- Prevention strategies include structured fluid intake schedules, electrolyte supplementation, and regular biomarker tracking
- Understanding individual risk factors enables personalized hydration management in research settings
🔬 Understanding Retatrutide and Its Physiological Impact
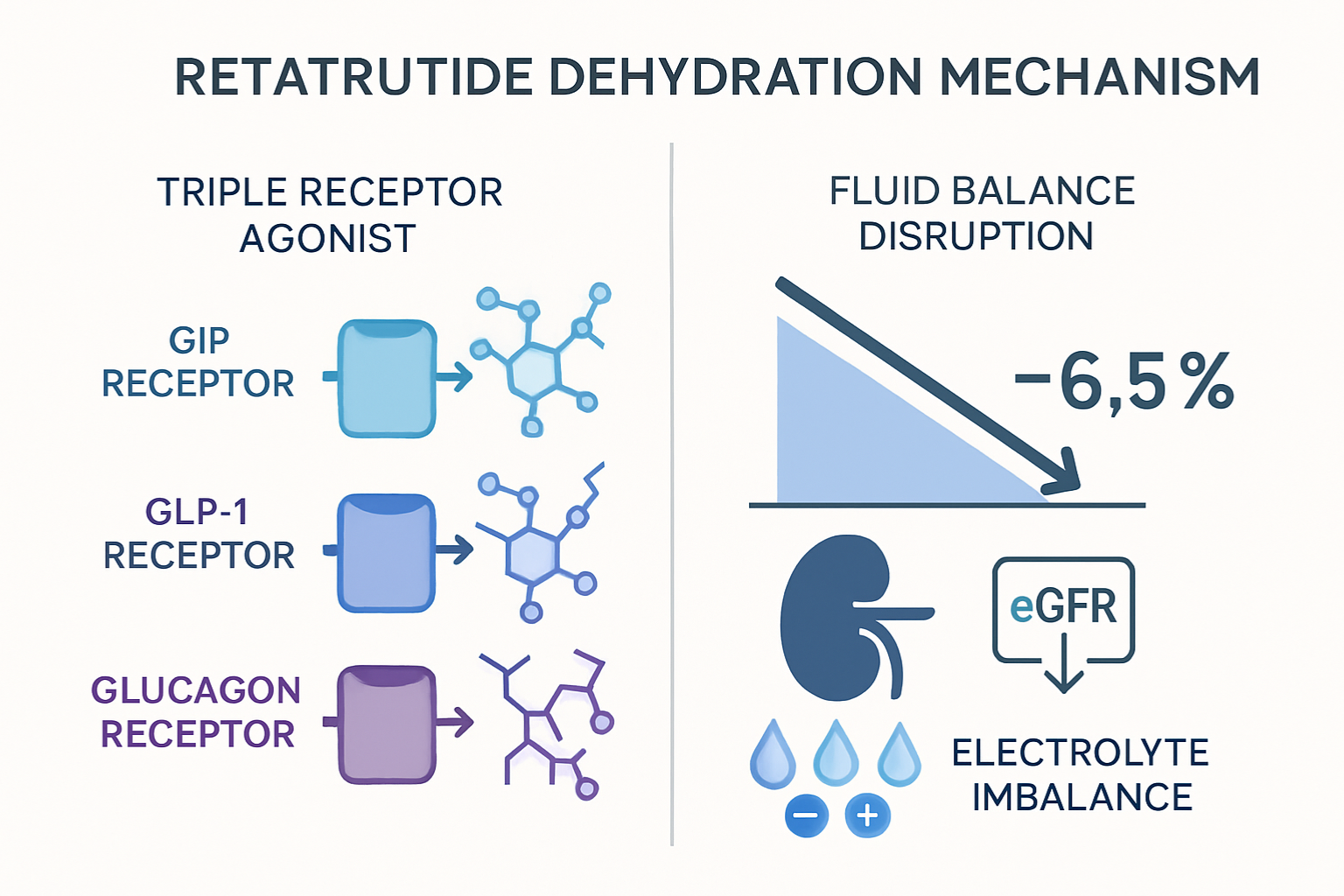
Retatrutide represents a significant advancement in peptide research as a triple receptor agonist targeting glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors simultaneously. This unique mechanism of action distinguishes it from earlier compounds like semaglutide or tirzepatide, which target fewer receptor pathways.
The compound’s molecular structure enables it to bind with high affinity to all three receptor types, creating a cascade of metabolic effects that extend far beyond simple appetite regulation. Research observations indicate that retatrutide influences energy expenditure, glucose homeostasis, lipid metabolism, and—critically for this discussion—fluid balance regulation.
Mechanism of Action and Metabolic Effects
The triple agonist activity of retatrutide creates a complex interplay of physiological responses:
GIP Receptor Activation:
- Enhances insulin secretion in glucose-dependent manner
- Influences adipocyte metabolism and lipid storage
- Modulates bone metabolism and calcium regulation
- Affects fluid distribution between compartments
GLP-1 Receptor Activation:
- Delays gastric emptying significantly
- Reduces appetite and food-seeking behavior
- Improves insulin sensitivity
- Influences renal sodium handling
Glucagon Receptor Activation:
- Increases energy expenditure and thermogenesis
- Promotes lipolysis and fat oxidation
- Enhances hepatic glucose output regulation
- Affects renal blood flow and glomerular filtration
This comprehensive receptor engagement creates metabolic conditions that predispose research subjects to fluid imbalance, making retatrutide dehydration a predictable rather than incidental finding.
Pharmacokinetic Properties Influencing Hydration Status
Retatrutide exhibits an extended half-life of approximately 6-7 days, meaning its effects on fluid balance persist long after administration. This prolonged duration of action distinguishes it from shorter-acting peptides and necessitates sustained attention to hydration status throughout the research period.
The compound’s pharmacokinetic profile includes:
| Parameter | Value | Hydration Implication |
|---|---|---|
| Half-life | 6-7 days | Extended dehydration risk window |
| Time to peak concentration | 24-48 hours | Acute dehydration risk period |
| Steady state | 4-5 weeks | Cumulative fluid balance effects |
| Renal clearance | Minimal | Altered kidney function impact |
These characteristics mean that researchers must implement long-term hydration monitoring strategies rather than acute intervention protocols when working with retatrutide compounds available from specialized suppliers like Peptide Pro.
💧 The Mechanisms Behind Retatrutide Dehydration
Understanding why retatrutide dehydration occurs requires examining multiple physiological pathways that converge to create net fluid loss. Unlike simple diuretic effects, retatrutide-induced dehydration involves complex hormonal, renal, and behavioral mechanisms.
Gastrointestinal Effects and Reduced Fluid Intake
The GLP-1 receptor agonism component of retatrutide produces profound effects on gastrointestinal motility and appetite regulation. Research observations consistently document:
Delayed Gastric Emptying: Retatrutide significantly slows the rate at which stomach contents enter the small intestine. This creates a persistent sensation of fullness that extends well beyond normal meal times. In research settings, this translates to subjects consuming substantially less fluid throughout the day—not due to restricted access, but from genuine lack of thirst drive.
Nausea and Gastrointestinal Discomfort: A substantial percentage of research observations include reports of nausea, particularly during initial exposure periods. This gastrointestinal disturbance further suppresses voluntary fluid intake, compounding dehydration risk. The nausea typically peaks during the first 2-4 weeks of exposure but may persist at lower intensity throughout the research period.
Altered Thirst Perception: Emerging research suggests that GLP-1 receptor activation may directly influence hypothalamic thirst centers, reducing the physiological drive to seek fluids even when objective dehydration markers are present. This dissociation between hydration status and thirst sensation represents a critical challenge for maintaining adequate fluid balance.
Renal and Hormonal Mechanisms
The kidneys play a central role in retatrutide dehydration through multiple pathways:
Enhanced Natriuresis: Retatrutide increases urinary sodium excretion through effects on proximal tubule sodium reabsorption. As sodium is excreted, water follows osmotically, leading to increased urine output. Research data indicates this natriuretic effect can persist for weeks after initial administration.
Glucagon-Mediated Diuresis: The glucagon receptor agonism component increases renal blood flow and glomerular filtration rate. This enhanced kidney perfusion promotes fluid excretion, particularly during the first 48-72 hours post-administration when plasma concentrations peak.
Altered Vasopressin Sensitivity: Preliminary research suggests retatrutide may influence renal responsiveness to antidiuretic hormone (vasopressin), potentially reducing the kidney’s ability to concentrate urine effectively. This mechanism requires further investigation but may explain why some subjects develop persistent polyuria despite adequate vasopressin levels.
Metabolic Water Loss
The compound’s effects on energy metabolism create additional pathways for fluid loss:
- Increased Thermogenesis: Enhanced metabolic rate increases insensible water loss through respiration and perspiration
- Lipolysis: Fat oxidation produces metabolic water, but the process itself requires hydration for optimal function
- Glycogen Depletion: Early metabolic shifts may deplete glycogen stores, releasing bound water that is subsequently excreted
⚠️ Risk Factors and Clinical Manifestations of Retatrutide Dehydration
Not all research subjects exhibit equal susceptibility to retatrutide dehydration. Identifying risk factors enables researchers to implement targeted monitoring and prevention strategies.
High-Risk Populations and Conditions
Research protocols should identify subjects with elevated dehydration risk:
Baseline Characteristics:
- Advanced age (reduced thirst sensation and kidney function)
- Pre-existing renal impairment or chronic kidney disease
- Diabetes mellitus with polyuria
- Concurrent diuretic medication use
- Baseline hypovolemia or low body water content
- Hot climate exposure or inadequate environmental temperature control
Dosing-Related Factors:
- Higher retatrutide doses (>8 mg weekly equivalent)
- Rapid dose escalation protocols
- First 2-4 weeks of exposure (acute adaptation period)
- Combination with other peptides affecting fluid balance
Behavioral and Environmental Factors:
- Limited fluid access during observation periods
- High ambient temperature exposure
- Increased physical activity levels
- Inadequate electrolyte intake
- Poor baseline hydration habits
Recognizing Dehydration Signs in Research Settings
Early detection of retatrutide dehydration requires systematic assessment of multiple parameters:
Mild Dehydration Indicators (1-3% body water loss):
- Increased thirst (though may be blunted)
- Dry mucous membranes
- Reduced urine output or darker urine color
- Mild fatigue or reduced activity levels
- Slight decrease in skin turgor
Moderate Dehydration Indicators (3-6% body water loss):
- Orthostatic hypotension (blood pressure drop upon standing)
- Tachycardia (elevated heart rate)
- Reduced urine output (<0.5 mL/kg/hour)
- Sunken eyes or decreased skin elasticity
- Confusion or irritability
- Muscle cramps or weakness
Severe Dehydration Indicators (>6% body water loss):
- Severe hypotension
- Altered mental status or lethargy
- Minimal or absent urine output
- Rapid, weak pulse
- Cold extremities
- Potential organ dysfunction markers
Laboratory Markers and Biomarker Monitoring
Objective assessment of hydration status requires regular laboratory monitoring:
| Biomarker | Normal Range | Mild Dehydration | Moderate-Severe Dehydration |
|---|---|---|---|
| Serum Sodium | 135-145 mEq/L | 145-150 mEq/L | >150 mEq/L |
| Blood Urea Nitrogen (BUN) | 7-20 mg/dL | 20-30 mg/dL | >30 mg/dL |
| BUN/Creatinine Ratio | 10:1 to 20:1 | 20:1 to 30:1 | >30:1 |
| Urine Specific Gravity | 1.003-1.030 | 1.030-1.035 | >1.035 |
| Serum Osmolality | 275-295 mOsm/kg | 295-310 mOsm/kg | >310 mOsm/kg |
| Hematocrit | 38-50% | Elevated 5-10% | Elevated >10% |
Research protocols incorporating retatrutide compounds should establish baseline values for these markers and monitor them at regular intervals throughout the study period.
🛡
️ Prevention Strategies and Hydration Protocols
Preventing retatrutide dehydration requires proactive, systematic approaches rather than reactive interventions. Research facilities must implement comprehensive hydration management protocols.
Structured Fluid Intake Recommendations
Establishing minimum fluid intake targets helps counteract the reduced thirst drive associated with retatrutide:
General Hydration Guidelines:
- Minimum baseline: 30-35 mL/kg body weight daily
- Additional 500-1000 mL during first week post-administration
- Increased intake during hot weather or physical activity
- Preference for electrolyte-containing fluids over pure water
Practical Implementation Strategies:
- Scheduled fluid intake times (not reliant on thirst)
- Visual tracking systems (marked water bottles)
- Flavor enhancement to improve palatability
- Temperature preferences (cold vs. room temperature)
- Small, frequent volumes rather than large boluses
Fluid Type Considerations: Research suggests optimal hydration with retatrutide includes:
- Water as primary fluid source
- Electrolyte solutions (sodium 20-50 mEq/L)
- Herbal teas or flavored water for variety
- Limited caffeine intake (mild diuretic effect)
- Avoidance of high-sugar beverages
Electrolyte Supplementation Protocols
The natriuretic effects of retatrutide often necessitate electrolyte supplementation:
Sodium Supplementation:
- Baseline assessment of dietary sodium intake
- Additional 1-2 grams daily during acute phases
- Preference for natural sources (broth, electrolyte solutions)
- Monitoring for individual sodium sensitivity
Potassium Considerations:
- Maintain adequate intake (3500-4700 mg daily)
- Monitor serum levels if diuretic effects pronounced
- Food sources: leafy greens, avocados, potatoes
- Supplementation if levels drop below 3.5 mEq/L
Magnesium Support:
- Target 400-420 mg daily for males, 310-320 mg for females
- Increased requirements with enhanced diuresis
- Symptoms of deficiency: muscle cramps, fatigue, arrhythmias
Monitoring and Assessment Schedules
Systematic monitoring enables early detection and intervention:
Initial Phase (Weeks 1-4):
- Baseline comprehensive metabolic panel
- Weekly weight measurements
- Urine specific gravity 2-3 times weekly
- Daily symptom assessment
- Orthostatic vital signs twice weekly
Maintenance Phase (Week 5 onwards):
- Monthly comprehensive metabolic panel
- Weekly weight and vital signs
- Bi-weekly urine specific gravity
- Ongoing symptom monitoring
- Quarterly comprehensive assessment
Dose Escalation Periods: Return to initial phase monitoring intensity for 2-3 weeks following any dose increase, as dehydration risk temporarily elevates during adaptation periods.
🔍 Special Considerations for Research Applications
When working with research-grade peptides like those available from Peptide Pro, additional considerations apply to managing retatrutide dehydration in laboratory settings.
Research Protocol Design
Incorporating hydration management into experimental design:
Baseline Establishment:
- Comprehensive hydration assessment before retatrutide introduction
- Documentation of baseline fluid intake patterns
- Establishment of individual hydration markers
- Assessment of concurrent factors affecting fluid balance
Control Group Considerations:
- Parallel hydration monitoring in control subjects
- Differentiation between retatrutide-specific effects and environmental factors
- Standardization of fluid access and environmental conditions
Data Collection Parameters:
- Standardized hydration assessment tools
- Objective biomarker measurements
- Subjective symptom reporting mechanisms
- Integration of hydration data with primary outcome measures
Interaction with Other Research Compounds
Retatrutide is often studied alongside other peptides, creating potential additive dehydration risks:
Synergistic Dehydration Risk:
- Combination with other GLP-1 agonists (semaglutide)
- Co-administration with diuretic peptides
- Concurrent metabolic enhancers increasing thermogenesis
- Growth hormone secretagogues affecting fluid distribution
Compounds Requiring Enhanced Monitoring: When retatrutide is combined with peptides like BPC-157 or TB-500, researchers should maintain heightened awareness of cumulative effects on fluid balance, even though these compounds have different primary mechanisms.
Quality and Purity Considerations
The quality of research-grade peptides directly impacts the predictability of dehydration responses:
Purity Standards: High-purity retatrutide from reputable suppliers produces more consistent physiological responses, enabling better prediction and management of dehydration risk. Contaminants or degradation products may create unpredictable fluid balance effects.
Storage and Handling: Proper storage conditions (typically 2-8°C for reconstituted peptides) maintain compound integrity and ensure consistent dehydration risk profiles across research sessions. Degraded peptides may produce variable or unexpected effects on fluid balance.
Reconstitution Protocols: Following standardized reconstitution procedures using appropriate bacteriostatic water ensures consistent dosing accuracy, which directly affects the magnitude and duration of dehydration risk.
📊 Comparative Analysis: Retatrutide vs. Other Metabolic Peptides
Understanding how retatrutide dehydration compares to other peptide compounds provides valuable context for risk assessment and protocol development.
Dehydration Risk Profile Comparison
| Peptide | Mechanism | Dehydration Risk | Primary Pathway | Duration |
|---|---|---|---|---|
| Retatrutide | GIP/GLP-1/Glucagon triple agonist | High | Multi-factorial | Extended (6-7 days) |
| Tirzepatide | GIP/GLP-1 dual agonist | Moderate-High | Natriuresis, reduced intake | Extended (5 days) |
| Semaglutide | GLP-1 agonist | Moderate | Reduced intake, mild natriuresis | Extended (7 days) |
| Liraglutide | GLP-1 agonist | Low-Moderate | Reduced intake | Short (13 hours) |
| AOD-9604 | Growth hormone fragment | Low | Minimal direct effect | Short (2-3 hours) |
The triple agonist mechanism of retatrutide creates cumulative dehydration risk exceeding that of single or dual agonists, necessitating more intensive hydration management protocols.
Unique Aspects of Retatrutide Dehydration
Several characteristics distinguish retatrutide dehydration from other peptide-induced fluid imbalances:
Glucagon Component Effects: Unlike GLP-1-only agonists, retatrutide’s glucagon receptor activation directly increases renal blood flow and promotes diuresis through mechanisms independent of the GLP-1 pathway. This creates a “dual-hit” scenario where both reduced intake and increased output contribute simultaneously.
Metabolic Intensity: The enhanced thermogenic and lipolytic effects of retatrutide increase insensible water losses beyond those seen with less metabolically active compounds. Research subjects may experience increased perspiration and respiratory water loss even at rest.
Adaptation Timeline: While many peptides show diminishing side effects over time, retatrutide’s dehydration risk may persist longer due to the sustained metabolic changes it produces. Some research observations indicate ongoing elevated fluid requirements even after 8-12 weeks of exposure.
�
� Practical Implementation: Case Studies and Research Insights
Examining real-world research applications illuminates effective strategies for managing retatrutide dehydration.
Research Setting Example 1: Dose-Escalation Protocol
A metabolic research facility implementing a 12-week retatrutide dose-escalation protocol observed:
Initial Findings:
- 73% of subjects showed mild dehydration markers by week 2
- Average fluid intake decreased by 35% from baseline
- Serum sodium increased by average 4 mEq/L
- Two subjects developed moderate dehydration requiring intervention
Protocol Modifications:
- Implemented mandatory minimum fluid intake targets (40 mL/kg daily)
- Added twice-weekly urine specific gravity monitoring
- Introduced electrolyte supplementation from day 1
- Created visual hydration tracking system
Outcomes After Modification:
- Dehydration incidence reduced to 28% (mild only)
- No cases of moderate or severe dehydration
- Improved research compliance and data quality
- Better subject comfort and protocol adherence
Research Setting Example 2: Environmental Challenge
A research facility in a warm climate encountered elevated dehydration rates:
Challenge: Ambient temperatures of 25-30°C (77-86°F) combined with retatrutide administration created synergistic dehydration risk. Initial protocols designed for temperate climates proved inadequate.
Adaptive Strategies:
- Increased baseline fluid recommendations by 25%
- Implemented climate-controlled observation areas
- Added mid-day electrolyte solution provision
- Enhanced monitoring frequency during summer months
- Created temperature-adjusted hydration calculators
Key Learnings: Environmental factors significantly modulate retatrutide dehydration risk, requiring protocol flexibility and local adaptation rather than one-size-fits-all approaches.
🔬 Emerging Research and Future Directions
The scientific understanding of retatrutide dehydration continues to evolve as research expands throughout 2025 and beyond.
Current Research Gaps
Several important questions remain under investigation:
Individual Variability: Why do some subjects maintain excellent hydration despite high retatrutide doses while others develop dehydration at lower exposures? Genetic factors, baseline metabolic characteristics, and hormonal profiles may all contribute, but systematic research is needed.
Long-Term Adaptation: Do subjects develop physiological adaptations that reduce dehydration risk over extended exposure periods (6+ months)? Preliminary observations suggest possible adaptation, but controlled long-term studies are limited.
Biomarker Development: Can novel biomarkers predict individual dehydration susceptibility before retatrutide administration? Identifying predictive markers would enable preemptive intervention strategies.
Potential Mitigation Strategies Under Investigation
Researchers are exploring various approaches to reduce dehydration risk while preserving retatrutide’s beneficial effects:
Pharmacological Adjuncts:
- Co-administration of compounds that enhance thirst drive
- Selective sodium-sparing agents
- Vasopressin sensitizers for improved renal water retention
Dosing Optimization:
- Alternative dosing schedules (more frequent, lower doses vs. weekly bolus)
- Individualized dose titration based on hydration biomarkers
- Cyclical dosing patterns with hydration recovery periods
Formulation Innovations:
- Modified-release formulations reducing peak-related diuresis
- Combination products including electrolyte components
- Novel delivery systems optimizing pharmacokinetic profiles
Integration with Precision Research Approaches
Future research protocols may incorporate:
- Genetic screening for variants affecting fluid regulation
- Continuous monitoring using wearable hydration sensors
- Artificial intelligence algorithms predicting individual dehydration risk
- Personalized hydration protocols based on multi-parameter assessment
These advances promise to transform retatrutide dehydration from a challenging side effect into a manageable, predictable aspect of research protocols.
📋 Practical Checklist for Research Facilities
Implementing comprehensive dehydration prevention requires systematic attention to multiple factors. Research facilities working with high-purity retatrutide should consider this practical checklist:
Pre-Administration Phase
✅ Baseline Assessment:
- Comprehensive metabolic panel including electrolytes
- Baseline weight and vital signs
- Urine specific gravity measurement
- Assessment of typical daily fluid intake
- Review of medications affecting fluid balance
- Evaluation of environmental factors (climate, activity level)
✅ Protocol Preparation:
- Establish individualized fluid intake targets
- Prepare electrolyte supplementation plan
- Create monitoring schedule and documentation system
- Educate subjects on dehydration signs and symptoms
- Ensure adequate fluid access throughout observation periods
✅ Risk Stratification:
- Identify high-risk subjects requiring enhanced monitoring
- Establish intervention thresholds for various dehydration markers
- Prepare contingency protocols for moderate/severe dehydration
- Coordinate with medical oversight personnel
Active Research Phase
✅ Daily Operations:
- Monitor fluid intake against targets
- Assess for dehydration symptoms
- Maintain environmental temperature control
- Document any gastrointestinal symptoms affecting intake
- Ensure electrolyte supplementation compliance
✅ Scheduled Assessments:
- Weekly weights (same time, same conditions)
- Regular urine specific gravity measurements
- Periodic comprehensive metabolic panels
- Orthostatic vital signs monitoring
- Symptom severity scoring
✅ Data Management:
- Systematic documentation of all hydration parameters
- Integration with primary research outcome data
- Trend analysis for early detection of developing issues
- Regular protocol review and adjustment as needed
Post-Research Phase
✅ Transition Planning:
- Gradual reduction of enhanced hydration protocols
- Final comprehensive assessment
- Documentation of any persistent effects
- Subject education on ongoing hydration needs
- Follow-up scheduling if indicated
� Expert Recommendations and Best Practices
Based on accumulated research experience with retatrutide dehydration, several expert recommendations have emerged:
For Research Protocol Designers
Recommendation 1: Proactive Rather Than Reactive Design hydration management into the initial protocol rather than adding it after problems emerge. Prevention is substantially more effective and less disruptive than intervention.
Recommendation 2: Individual Customization Avoid rigid, universal hydration protocols. Individual variation in dehydration susceptibility necessitates personalized approaches based on baseline characteristics, dose levels, and environmental factors.
Recommendation 3: Multi-Parameter Assessment Rely on multiple hydration markers rather than single indicators. The combination of subjective symptoms, physical examination findings, and laboratory biomarkers provides the most reliable assessment.
Recommendation 4: Environmental Awareness Explicitly account for climate, season, and activity level in hydration protocols. Standard recommendations may be inadequate in challenging environmental conditions.
For Research Facility Operators
Recommendation 5: Staff Education Ensure all personnel involved in retatrutide research understand dehydration mechanisms, recognition, and prevention. Consistent implementation across shifts and staff members is essential.
Recommendation 6: Quality Assurance Source research-grade peptides from reputable suppliers like Peptide Pro that provide certificates of analysis and maintain strict quality standards. Peptide purity directly affects the predictability of physiological responses.
Recommendation 7: Documentation Excellence Maintain meticulous records of all hydration-related parameters. This data proves invaluable for protocol refinement and contributes to the broader scientific understanding of retatrutide effects.
For Subject Safety
Recommendation 8: Clear Communication Provide subjects with clear, understandable information about dehydration risks, prevention strategies, and warning signs requiring immediate reporting.
Recommendation 9: Accessibility Ensure subjects have 24/7 access to support for hydration-related concerns. Many dehydration complications are preventable with timely intervention.
Recommendation 10: Empowerment Educate subjects to become active participants in hydration management rather than passive recipients of care. Self-monitoring and proactive fluid intake produce superior outcomes.
🌟 The Broader Context: Retatrutide in Metabolic Research
Understanding retatrutide dehydration requires appreciating the compound’s position within the broader landscape of metabolic research peptides.
Why Retatrutide Matters
Despite the dehydration challenges, retatrutide represents a significant advancement in metabolic research:
Unprecedented Metabolic Effects: Research observations document metabolic changes exceeding those seen with earlier compounds, making retatrutide a valuable tool for understanding metabolic regulation mechanisms.
Multi-Pathway Engagement: The triple agonist mechanism provides insights into the complex interplay between GIP, GLP-1, and glucagon signaling—knowledge applicable far beyond retatrutide itself.
Translational Potential: Understanding how to manage retatrutide’s effects, including dehydration, advances the broader field of peptide therapeutics and informs development of next-generation compounds.
Balancing Benefits and Challenges
Effective research with retatrutide requires acknowledging both its powerful effects and associated challenges:
The Value Proposition: When hydration is properly managed, retatrutide enables research questions impossible to address with less potent compounds. The dehydration risk becomes a manageable aspect of protocol design rather than a prohibitive barrier.
Risk-Benefit Optimization: Sophisticated protocols maximize retatrutide’s research value while minimizing dehydration and other adverse effects through systematic prevention and monitoring strategies.
📚 Resources for Continued Learning
Researchers seeking to deepen their understanding of retatrutide dehydration and related topics should consider these resources:
Scientific Literature
Stay current with emerging research through:
- Peer-reviewed journals focusing on metabolic peptides
- Conference proceedings from endocrinology and metabolism meetings
- Systematic reviews and meta-analyses of incretin-based compounds
- Case reports documenting novel management strategies
Professional Organizations
Connect with research communities through:
- Peptide research societies and working groups
- Metabolic research consortia
- Laboratory safety and protocol development organizations
- Regional research networks facilitating knowledge exchange
Supplier Resources
Reputable peptide suppliers often provide valuable educational materials:
- Technical data sheets and handling guidelines
- Certificates of analysis demonstrating purity
- Storage and reconstitution protocols
- Safety data sheets and regulatory information
Organizations like Peptide Pro offer comprehensive support for researchers working with advanced peptide compounds, including detailed product information and responsive customer service.
Conclusion: Mastering Retatrutide Dehydration Management
Retatrutide dehydration represents a significant but manageable challenge in metabolic peptide research. The compound’s triple agonist mechanism creates unique dehydration risk through multiple pathways: reduced fluid intake from GLP-1 effects, increased diuresis from glucagon receptor activation, enhanced natriuresis, and elevated metabolic water losses.
Successful management requires a comprehensive, proactive approach incorporating:
- Systematic baseline assessment identifying individual risk factors
- Structured hydration protocols with minimum fluid intake targets
- Electrolyte supplementation addressing natriuretic effects
- Regular monitoring using multiple objective and subjective parameters
- Environmental adaptation accounting for climate and activity factors
- Quality assurance ensuring consistent peptide purity and potency
Research facilities that implement these strategies transform dehydration from a protocol-limiting complication into a predictable, manageable aspect of retatrutide research. The resulting data quality improvements and enhanced subject safety justify the systematic attention required.
As research with retatrutide expands throughout 2025 and beyond, continued refinement of hydration management protocols will further optimize the balance between maximizing research value and minimizing adverse effects. The knowledge gained extends beyond retatrutide itself, informing best practices for the entire class of metabolic peptides and contributing to safer, more effective research methodologies.
Next Steps for Researchers
For research facilities preparing to work with retatrutide:
- Review current protocols against the recommendations outlined in this article
- Establish baseline hydration assessment procedures before initiating retatrutide research
- Develop individualized monitoring schedules based on subject risk stratification
- Source high-purity research peptides from reputable suppliers with documented quality standards
- Educate all research personnel on dehydration recognition and prevention
- Create systematic documentation systems tracking hydration parameters alongside primary outcomes
- Establish clear intervention protocols for various dehydration severity levels
For researchers seeking high-quality retatrutide and other research-grade peptides, Peptide Pro offers an extensive catalogue of compounds produced under strict quality conditions, with fast UK delivery and comprehensive product support.
By approaching retatrutide dehydration with the seriousness and systematic attention it deserves, research facilities can safely harness this powerful compound’s unique properties while maintaining the highest standards of research integrity and subject safety.
