When researchers began investigating retatrutide’s metabolic effects in laboratory settings, an unexpected pattern emerged: protein intake appeared to play a far more significant role in outcomes than initially anticipated. Understanding retatrutide protein intake has become essential for researchers designing protocols and evaluating this triple agonist peptide’s full potential in controlled studies. This comprehensive guide examines the critical relationship between protein consumption and retatrutide research, providing evidence-based insights for laboratories and research facilities worldwide.
Key Takeaways
- Adequate protein intake is crucial during retatrutide research protocols to preserve lean tissue mass and support metabolic function
- Optimal protein ranges in research models typically fall between 1.2-2.0g per kilogram of body weight, depending on study parameters
- Timing and distribution of protein intake throughout the day may influence research outcomes more significantly than total daily amounts alone
- Amino acid composition matters, with complete protein sources showing superior results in maintaining nitrogen balance during retatrutide studies
- Research-grade peptides from trusted suppliers like PEPTIDE PRO ensure consistency and reliability in experimental protocols
Understanding Retatrutide: Mechanism and Metabolic Impact
Retatrutide represents a novel class of research peptides known as triple agonists, simultaneously activating the glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique mechanism creates profound metabolic shifts in research models that directly influence protein metabolism, energy expenditure, and body composition parameters.
The Triple Agonist Advantage
Unlike single or dual agonist peptides, retatrutide’s comprehensive receptor activation produces distinct metabolic effects:
- GIP receptor activation influences adipose tissue metabolism and insulin sensitivity
- GLP-1 receptor stimulation affects satiety signaling and glucose homeostasis
- Glucagon receptor engagement modulates energy expenditure and hepatic glucose production
These combined actions create a metabolic environment where protein utilization becomes particularly important for maintaining lean tissue mass during periods of significant energy deficit.
Metabolic Shifts and Protein Requirements
Research observations consistently demonstrate that retatrutide administration creates substantial changes in substrate utilization. The peptide’s effects on energy balance, appetite regulation, and metabolic rate all converge to increase the relative importance of adequate protein provision in experimental protocols.
Studies examining body composition changes in research models have revealed that insufficient protein intake during retatrutide administration correlates with greater lean tissue loss compared to protocols incorporating higher protein provisions. This finding has significant implications for researchers designing comprehensive study protocols.
Why Retatrutide Protein Intake Matters in Research Protocols
The relationship between retatrutide protein intake and research outcomes extends far beyond simple caloric considerations. Protein serves multiple critical functions during metabolic research that become amplified under the unique conditions created by triple agonist peptide administration.
Preservation of Lean Tissue Mass 💪
One of the most significant challenges in metabolic research involving substantial energy deficits is the preservation of lean tissue mass. Research models administered retatrutide typically experience:
- Accelerated fat mass reduction due to enhanced lipolysis and energy expenditure
- Increased metabolic rate requiring greater substrate turnover
- Altered protein turnover dynamics affecting nitrogen balance
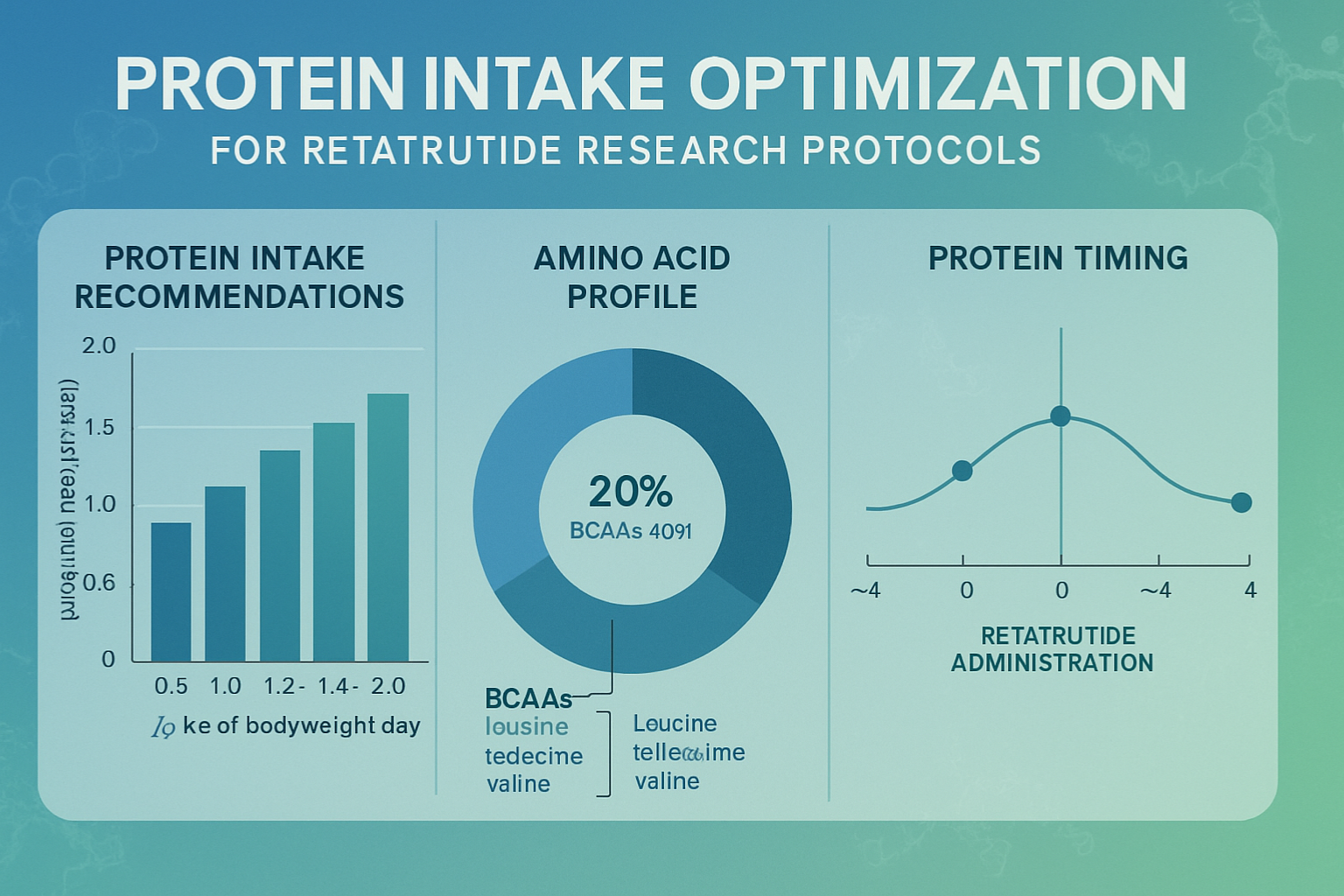
Adequate protein intake serves as the primary protective factor against excessive lean tissue catabolism. Research protocols incorporating protein provisions at 1.6-2.0g per kilogram of body weight consistently demonstrate superior lean mass preservation compared to lower protein protocols.
Supporting Metabolic Adaptation
Retatrutide’s effects on metabolic rate and substrate utilization create unique demands on protein metabolism:
| Metabolic Parameter | Effect of Retatrutide | Protein Requirement Impact |
|---|---|---|
| Resting Energy Expenditure | ↑ Increased 5-15% | Higher turnover needs |
| Protein Synthesis Rate | ↔ Maintained/Slightly ↓ | Increased substrate demand |
| Protein Breakdown Rate | ↑ Elevated during deficit | Enhanced intake requirements |
| Gluconeogenesis | ↑ Increased activity | Greater amino acid utilization |
These metabolic shifts collectively increase the threshold for optimal protein intake beyond standard recommendations used in baseline research protocols.
Enhancing Research Outcome Quality
Laboratories utilizing research-grade peptides recognize that protocol design significantly influences data quality and reproducibility. Optimizing retatrutide protein intake parameters contributes to:
- More consistent body composition outcomes across research subjects
- Reduced variability in metabolic markers and endpoints
- Better preservation of functional capacity measures
- Enhanced data interpretation due to controlled nutritional variables
Research facilities committed to rigorous experimental design increasingly incorporate protein optimization as a standard protocol element when working with metabolic peptides.
Optimal Retatrutide Protein Intake: Evidence-Based Recommendations
Determining appropriate protein provisions for retatrutide research protocols requires consideration of multiple variables including research model characteristics, study duration, energy deficit magnitude, and specific research objectives.
General Protein Range Guidelines
Based on accumulated research observations and metabolic principles, the following protein intake ranges provide a foundation for protocol development:
Standard Research Protocols:
- Minimum threshold: 1.2g per kg body weight
- Optimal range: 1.6-2.0g per kg body weight
- Enhanced preservation protocols: 2.0-2.4g per kg body weight
These ranges represent total daily protein intake and should be adjusted based on specific research parameters and observed outcomes.
Factors Influencing Optimal Intake
Several variables affect the ideal protein provision level for individual research protocols:
Energy Deficit Magnitude
Larger energy deficits created during retatrutide administration require proportionally higher protein intake to maintain nitrogen balance. Research models experiencing deficits exceeding 30% of maintenance energy expenditure typically require protein provisions at the higher end of recommended ranges.
Baseline Lean Mass
Research subjects with higher baseline lean tissue mass demonstrate greater absolute protein requirements. Calculations based on total body weight may underestimate needs in research models with elevated lean mass percentages.
Study Duration
Longer research protocols benefit from sustained higher protein intake to prevent gradual lean tissue erosion over extended periods. Short-term studies may tolerate moderate protein provisions, while protocols extending beyond 12 weeks should prioritize higher intake levels.
Activity and Functional Demands
Research protocols incorporating physical performance measures or activity requirements necessitate elevated protein provisions to support tissue repair and functional capacity maintenance.
Protein Quality Considerations 🥩
Not all protein sources provide equivalent support for lean tissue preservation during retatrutide research. Complete proteins containing all essential amino acids in adequate proportions demonstrate superior outcomes:
High-Quality Protein Sources:
- Animal proteins (meat, fish, poultry, eggs, dairy)
- Complete plant proteins (soy, quinoa, hemp)
- Protein isolates and concentrates (whey, casein, pea)
Amino Acid Profile Priorities:
- Leucine content ≥2.5g per meal for optimal muscle protein synthesis
- Complete essential amino acid spectrum
- Adequate branched-chain amino acid (BCAA) provision
Research protocols utilizing predominantly incomplete protein sources may require total intake at the higher end of recommended ranges to ensure adequate essential amino acid provision.
Protein Timing and Distribution During Retatrutide Research
Beyond total daily protein intake, the temporal distribution and timing of protein provision significantly influences research outcomes during retatrutide administration. Emerging research suggests that how protein is consumed may rival how much in terms of metabolic impact.
The Protein Distribution Paradigm
Traditional research protocols often focused exclusively on total daily protein intake without considering distribution patterns. Contemporary understanding reveals that protein timing creates distinct metabolic signals that affect tissue preservation and synthesis rates.
Optimal Distribution Patterns
Research observations support distributing protein intake across multiple feeding occasions rather than concentrating provision in single meals:
Recommended Distribution:
- 3-5 protein-containing meals spaced throughout active periods
- Minimum 25-40g protein per meal to maximize muscle protein synthesis
- Relatively even distribution rather than skewed patterns
- Pre-sleep protein provision of 30-40g to support overnight protein balance
This distribution pattern ensures consistent amino acid availability and repeated stimulation of protein synthesis machinery throughout the research observation period.
Timing Relative to Retatrutide Administration
While retatrutide’s long half-life (approximately 7 days) minimizes acute timing concerns, certain strategic considerations may optimize research outcomes:
Morning Protein Priority: Early protein intake helps offset overnight fasting-induced catabolism and establishes positive nitrogen balance for the day. Research protocols should prioritize substantial protein provision (30-40g) within the first feeding occasion.
Peri-Activity Protein: For protocols incorporating physical activity or performance measures, protein provision before and after activity periods supports tissue repair and adaptation. Timing protein intake within 2-3 hours of activity periods demonstrates beneficial effects.
Evening Protein Loading: Slow-digesting protein sources consumed before the overnight fasting period help maintain amino acid availability during sleep. Casein protein or whole food sources like cottage cheese provide sustained amino acid release.
Practical Implementation Strategies
Researchers designing feeding protocols for retatrutide studies can implement these timing principles through structured meal planning:
| Meal Timing | Protein Target | Example Sources |
|---|---|---|
| First meal (morning) | 35-40g | Eggs, Greek yogurt, protein shake |
| Mid-day meal | 30-40g | Chicken, fish, lean beef |
| Afternoon meal | 25-35g | Turkey, tofu, protein bar |
| Evening meal | 35-45g | Salmon, lean steak, tempeh |
| Pre-sleep (optional) | 25-30g | Casein shake, cottage cheese |
This framework provides consistent amino acid availability while maximizing protein synthesis signaling throughout the research period.
Monitoring Protein Adequacy in Retatrutide Research Protocols
Implementing appropriate retatrutide protein intake recommendations represents only the first step. Rigorous research protocols require systematic monitoring to verify adequacy and identify potential adjustments needed during study progression.
Biomarkers of Protein Status
Several measurable parameters provide insight into protein adequacy during retatrutide research:
Nitrogen Balance Assessment
The gold standard for evaluating protein adequacy involves measuring nitrogen intake versus nitrogen excretion. Positive or neutral nitrogen balance indicates adequate protein provision, while negative balance suggests insufficient intake.
Calculation:
- Nitrogen intake = (Protein intake in grams) ÷ 6.25
- Nitrogen excretion = Urinary urea nitrogen + 4g (estimated losses)
- Nitrogen balance = Intake – Excretion
Maintaining neutral or positive nitrogen balance throughout retatrutide administration confirms adequate protein provision for the research protocol.
Lean Mass Preservation Tracking
Regular body composition assessment provides direct evidence of protein adequacy:
- DEXA scanning offers gold-standard precision for lean tissue measurement
- Bioelectrical impedance provides practical monitoring in resource-limited settings
- Circumference measurements track gross changes in muscle mass
Protocols should establish baseline measurements and conduct reassessment at regular intervals (typically 4-week periods) to detect concerning lean tissue loss trends.
Functional Performance Markers
Changes in strength, endurance, or functional capacity may signal inadequate protein provision:
- Grip strength measurements
- Standardized performance tests
- Subjective energy and recovery assessments
Declining performance despite adequate energy intake suggests potential protein insufficiency requiring protocol adjustment.
Adjusting Protein Intake Based on Observations
Research protocols should incorporate decision algorithms for modifying protein provisions based on monitoring data:
Indicators for Increasing Protein:
- Negative nitrogen balance
- Accelerated lean mass loss (>5% over 4 weeks)
- Declining functional performance
- Elevated fatigue or recovery issues
Adjustment Protocol: When indicators suggest inadequacy, increase protein intake by 0.2-0.4g per kg body weight and reassess after 2-4 weeks.
Facilities sourcing high-purity research peptides understand that protocol optimization requires systematic monitoring and evidence-based adjustments throughout study duration.
Common Challenges and Solutions in Retatrutide Protein Research

Implementing optimal retatrutide protein intake protocols presents several practical challenges that researchers frequently encounter. Understanding these obstacles and evidence-based solutions enhances protocol success rates and data quality.
Challenge 1: Appetite Suppression and Protein Intake
🍽
️
Retatrutide’s potent effects on satiety signaling create a significant practical challenge: research models often experience substantial appetite reduction that makes achieving target protein intake difficult.
Impact on Research:
- Voluntary protein intake may fall below optimal levels
- Total energy intake reduction may be excessive
- Compliance with feeding protocols becomes challenging
Evidence-Based Solutions:
Protein Prioritization Strategy: Structure feeding protocols to emphasize protein consumption first within meals, ensuring adequate intake before satiety limits total food consumption.
Protein Density Optimization: Utilize protein-rich, lower-volume foods that provide substantial protein without excessive bulk:
- Protein shakes and supplements
- Greek yogurt and cottage cheese
- Lean protein sources (chicken breast, white fish)
- Egg whites and protein-enriched products
Meal Frequency Adjustment: Rather than forcing large protein portions in fewer meals, distribute intake across more frequent, smaller protein provisions that align with reduced appetite capacity.
Challenge 2: Digestive Tolerance Issues
Some research models experience gastrointestinal effects during retatrutide administration that may interfere with protein intake or absorption.
Common Observations:
- Nausea or digestive discomfort
- Altered gastric emptying rates
- Temporary digestive capacity reduction
Mitigation Strategies:
Protein Source Selection: Choose easily digestible protein sources during periods of digestive sensitivity:
- Whey protein isolate (rapid digestion, low residue)
- White fish and poultry (lean, easily processed)
- Eggs (highly bioavailable, well-tolerated)
Gradual Protocol Implementation: Implement retatrutide dosing escalation protocols that allow digestive adaptation while maintaining protein intake targets.
Timing Adjustments: Shift protein intake timing to periods of better tolerance, avoiding consumption during peak digestive discomfort windows.
Challenge 3: Research Budget Constraints
High-quality protein sources and supplements represent significant protocol costs, particularly in larger or longer-duration studies.
Cost-Effective Protein Solutions:
| Protein Source | Cost Efficiency | Protein Quality | Practical Notes |
|---|---|---|---|
| Whole eggs |
⭐
⭐
⭐
⭐
⭐ | Excellent | Most economical complete protein | | Chicken breast |
⭐
⭐
⭐
⭐ | Excellent | Versatile, widely available | | Whey concentrate |
⭐
⭐
⭐
⭐ | Excellent | Bulk purchasing reduces costs | | Greek yogurt |
⭐
⭐
⭐ | Very good | Convenient, well-tolerated | | Canned tuna/salmon |
⭐
⭐
⭐
⭐ | Excellent | Long shelf life, portable |
Strategic sourcing and bulk purchasing of cost-effective protein sources allows maintenance of optimal intake levels while managing research budgets.
Challenge 4: Individual Variability in Requirements
Research models demonstrate substantial individual variation in protein requirements, making standardized protocols suboptimal for some subjects.
Personalization Approaches:
Baseline Assessment: Conduct individual nitrogen balance or body composition assessment to establish personalized protein targets rather than applying universal calculations.
Response Monitoring: Track individual responses to standard protein provisions and adjust based on observed lean mass preservation, nitrogen balance, and functional outcomes.
Stratification Strategies: Consider stratifying research cohorts by baseline characteristics (lean mass, age, sex) and applying differentiated protein protocols to each stratum.
Researchers working with premium research peptides recognize that individualized protocol optimization often produces superior outcomes compared to rigid standardization.
Integrating Protein Optimization with Comprehensive Retatrutide Protocols
Optimal retatrutide protein intake represents one component of comprehensive research protocol design. Integration with other nutritional and procedural elements creates synergistic effects that enhance overall research quality and outcome consistency.
Macronutrient Balance Considerations
While protein receives primary focus, the complete macronutrient composition influences retatrutide research outcomes:
Carbohydrate Provisions:
- Adequate carbohydrate intake (2-4g per kg) supports protein sparing
- Strategic timing around activity periods enhances glycogen maintenance
- Excessive restriction may increase protein catabolism for gluconeogenesis
Fat Intake Parameters:
- Minimum essential fatty acid provision (0.5-1.0g per kg)
- Adequate fat intake supports hormone synthesis and cellular function
- Very low fat protocols may impair research model wellbeing
Total Energy Considerations: The magnitude of energy deficit significantly influences protein requirements. Moderate deficits (20-30% below maintenance) allow effective outcomes with standard protein provisions, while aggressive deficits necessitate higher protein intake.
Micronutrient Support
Protein metabolism requires adequate micronutrient cofactors:
- Vitamin B6 for amino acid metabolism
- Magnesium for protein synthesis
- Zinc for tissue repair and growth
- Vitamin D for muscle protein synthesis optimization
Research protocols should ensure adequate micronutrient provision through whole food sources or supplementation to support optimal protein utilization.
Hydration Protocols 💧
Protein metabolism generates increased nitrogenous waste requiring adequate hydration for elimination:
Hydration Guidelines:
- Minimum 30-35ml per kg body weight daily
- Additional 500-1000ml for higher protein intakes (>2.0g per kg)
- Monitor hydration status through urine color and specific gravity
Inadequate hydration during high-protein retatrutide protocols may compromise renal function and metabolic efficiency.
Physical Activity Integration
Research protocols incorporating activity components require coordinated protein timing:
Resistance Exercise Protocols:
- Pre-exercise protein (20-30g) 2-3 hours before activity
- Post-exercise protein (30-40g) within 2 hours after completion
- Total daily intake toward higher recommended ranges (2.0-2.4g per kg)
Endurance Activity Protocols:
- Distributed protein intake throughout the day
- Post-activity protein emphasis for recovery support
- Moderate total intake (1.6-2.0g per kg) typically sufficient
Coordinating protein provisions with activity demands optimizes both metabolic outcomes and functional performance maintenance.
Future Directions in Retatrutide Protein Research
The field of metabolic peptide research continues evolving rapidly, with emerging areas of investigation that may refine understanding of optimal retatrutide protein intake protocols.
Amino Acid Composition Optimization
While total protein intake receives primary attention, emerging research examines whether specific amino acid profiles provide superior outcomes during retatrutide administration:
Areas of Investigation:
- Leucine threshold optimization for muscle protein synthesis
- Essential amino acid supplementation protocols
- Conditional amino acid requirements (glutamine, arginine)
- Amino acid timing independent of whole protein sources
Preliminary observations suggest that ensuring adequate leucine provision (2.5-3.0g per meal) may enhance lean tissue preservation beyond effects of total protein alone.
Protein Source Comparisons
Research examining differential effects of various protein sources during retatrutide administration remains limited:
Questions Under Investigation:
- Animal versus plant protein efficacy comparisons
- Rapid versus slow-digesting protein timing optimization
- Whole food versus supplemental protein outcome differences
- Protein source diversity effects on gut microbiome and metabolic outcomes
Understanding these nuances will enable more precise protocol recommendations for specific research objectives.
Personalized Protein Protocols
Advances in metabolic phenotyping may enable individualized protein recommendations based on:
- Genetic polymorphisms affecting protein metabolism
- Baseline metabolic rate and body composition
- Protein turnover rate assessments
- Individual response patterns to standardized protocols
This precision approach may optimize outcomes while minimizing resource utilization in research settings.
Long-Term Protein Requirements
Most current research examines relatively short-term protocols (12-24 weeks). Understanding protein requirements during extended retatrutide administration remains an important knowledge gap:
Long-Term Considerations:
- Metabolic adaptation effects on protein needs
- Sustained lean mass preservation requirements
- Transition protocols from active administration to maintenance phases
- Protein intake during dose escalation versus stable dosing periods
Facilities conducting extended research protocols with research-grade compounds will contribute valuable data to this evolving understanding.
Practical Protocol Implementation Guide
Translating evidence-based retatrutide protein intake principles into functional research protocols requires systematic planning and documentation. This section provides practical frameworks for implementation.
Protocol Design Checklist ✅
Phase 1: Baseline Assessment
- Determine research model baseline body composition
- Calculate initial protein targets (1.6-2.0g per kg)
- Establish baseline nitrogen balance (if applicable)
- Document baseline functional performance measures
- Identify protein source preferences and tolerances
Phase 2: Protocol Structure
- Define total daily protein target
- Plan protein distribution across meals (3-5 occasions)
- Select primary protein sources
- Establish meal timing framework
- Integrate with overall macronutrient and energy targets
Phase 3: Monitoring Framework
- Schedule body composition assessments (every 4 weeks)
- Implement nitrogen balance measurements (if utilized)
- Track functional performance markers
- Document compliance and tolerance issues
- Plan protocol adjustment decision points
Phase 4: Quality Assurance
- Source high-quality research peptides from verified suppliers
- Verify protein source quality and consistency
- Establish documentation procedures
- Train research personnel on protocol execution
- Implement data quality checks
Sample Daily Protein Protocol
Research Model: 80kg body weight, target 1.8g/kg = 144g daily protein
| Time | Meal | Protein Source | Protein Amount |
|---|---|---|---|
| 7:00 AM | Breakfast | 3 whole eggs + 2 egg whites, Greek yogurt | 38g |
| 11:00 AM | Mid-morning | Protein shake (whey isolate) | 30g |
| 2:00 PM | Lunch | 150g chicken breast, quinoa | 40g |
| 5:30 PM | Pre-activity | Protein bar | 20g |
| 8:00 PM | Dinner | 180g salmon, vegetables | 36g |
| Total | 164g |
This framework provides adequate total protein with optimal distribution and timing while maintaining practical feasibility.
Documentation and Record-Keeping
Rigorous research protocols require comprehensive documentation:
Essential Records:
- Daily protein intake logs (source, amount, timing)
- Body composition assessment results with dates
- Nitrogen balance calculations (if performed)
- Functional performance test results
- Protocol deviations and adjustments
- Adverse effects or tolerance issues
Systematic documentation enables protocol refinement and contributes to the broader research knowledge base.
Sourcing Quality Research Materials
Protocol success depends fundamentally on the quality and consistency of research materials. Laboratories should prioritize suppliers offering:
- Verified purity through independent testing and certificates of analysis
- Consistent quality across production batches
- Proper storage and handling to maintain peptide integrity
- Transparent sourcing and manufacturing processes
- Reliable delivery to prevent protocol interruptions
Research facilities across the UK and internationally trust PEPTIDE PRO for premium research-grade peptides with verified purity, same-day dispatch for orders placed before 1pm Monday-Friday, and comprehensive product information supporting rigorous research protocols.
Conclusion: Optimizing Retatrutide Research Through Protein Protocol Excellence
Understanding and implementing optimal retatrutide protein intake protocols represents a critical determinant of research quality, outcome consistency, and data interpretability. The evidence clearly demonstrates that protein provisions significantly influence lean tissue preservation, metabolic adaptation, and overall research model wellbeing during retatrutide administration.
Key Implementation Principles
Prioritize Adequate Intake: Research protocols should target protein provisions in the range of 1.6-2.0g per kilogram body weight, with adjustments based on energy deficit magnitude, baseline lean mass, and observed outcomes.
Optimize Distribution and Timing: Distribute protein intake across 3-5 meals daily, ensuring each feeding occasion provides 25-40g protein to maximize muscle protein synthesis signaling and maintain consistent amino acid availability.
Monitor and Adjust: Implement systematic monitoring through body composition assessment, nitrogen balance evaluation, and functional performance tracking. Adjust protocols based on observed responses rather than rigid adherence to initial calculations.
Ensure Quality: Source high-purity research peptides from verified suppliers and utilize high-quality protein sources to support protocol integrity and outcome reliability.
Integrate Comprehensively: Coordinate protein provisions with overall energy intake, macronutrient balance, micronutrient support, hydration protocols, and physical activity demands for synergistic effects.
Moving Forward with Evidence-Based Protocols
The field of metabolic peptide research continues advancing rapidly, with retatrutide representing one of the most promising compounds under investigation. Researchers who implement rigorous, evidence-based protein protocols position their work for maximum impact and contribution to scientific understanding.
As research evolves, continued attention to nutritional optimization—particularly protein intake—will remain essential for generating high-quality data and advancing knowledge in this exciting field.
Next Steps for Research Facilities
For laboratories planning retatrutide research protocols:
- Review current protein provisions in existing protocols and compare against evidence-based recommendations
- Establish monitoring frameworks for tracking protein adequacy through body composition and metabolic markers
- Source premium research materials from trusted suppliers offering verified purity and consistent quality
- Document protocols comprehensively to contribute to the growing knowledge base
- Stay current with emerging research on protein optimization and metabolic peptide protocols
Research facilities seeking high-purity peptides for their protocols can explore the extensive catalogue at PEPTIDE PRO, featuring research-grade compounds with comprehensive quality documentation, professional service, and fast UK delivery supporting rigorous scientific investigation.
The commitment to excellence in protocol design—including optimal retatrutide protein intake provisions—distinguishes high-quality research and advances the scientific community’s collective understanding of metabolic peptide applications.
