The landscape of metabolic research is evolving rapidly, with retatrutide vs amycretin emerging as one of the most compelling comparisons in peptide science. Both compounds represent cutting-edge developments in multi-receptor agonist research, yet they operate through distinct mechanisms that offer unique advantages for laboratory investigations. As researchers worldwide seek to understand the nuances between these advanced peptides, the question isn’t simply which compound performs better—it’s about understanding how their different receptor profiles, pharmacokinetics, and metabolic pathways can inform future research directions.
In 2026, the scientific community has witnessed unprecedented interest in these next-generation peptides, particularly as data from extensive research protocols continues to emerge. For laboratories and research institutions seeking high-purity research-grade peptides, understanding the fundamental differences between retatrutide and amycretin has become essential for designing robust experimental frameworks.
Key Takeaways
- Retatrutide functions as a triple receptor agonist (GIP, GLP-1, glucagon), while amycretin operates as a dual agonist (GLP-1, amylin), creating fundamentally different metabolic research profiles
- Dosing protocols differ significantly: retatrutide typically requires weekly administration in research settings, whereas amycretin may follow different scheduling based on experimental design
- Research applications vary: retatrutide shows particular promise in studies examining comprehensive metabolic regulation, while amycretin offers unique insights into appetite control and glucose homeostasis
- Both compounds require strict laboratory conditions and proper reconstitution techniques to maintain peptide integrity and research validity
- Availability through reputable suppliers like PEPTIDE PRO ensures consistent purity standards essential for reproducible research outcomes
Understanding Retatrutide: The Triple Agonist Mechanism
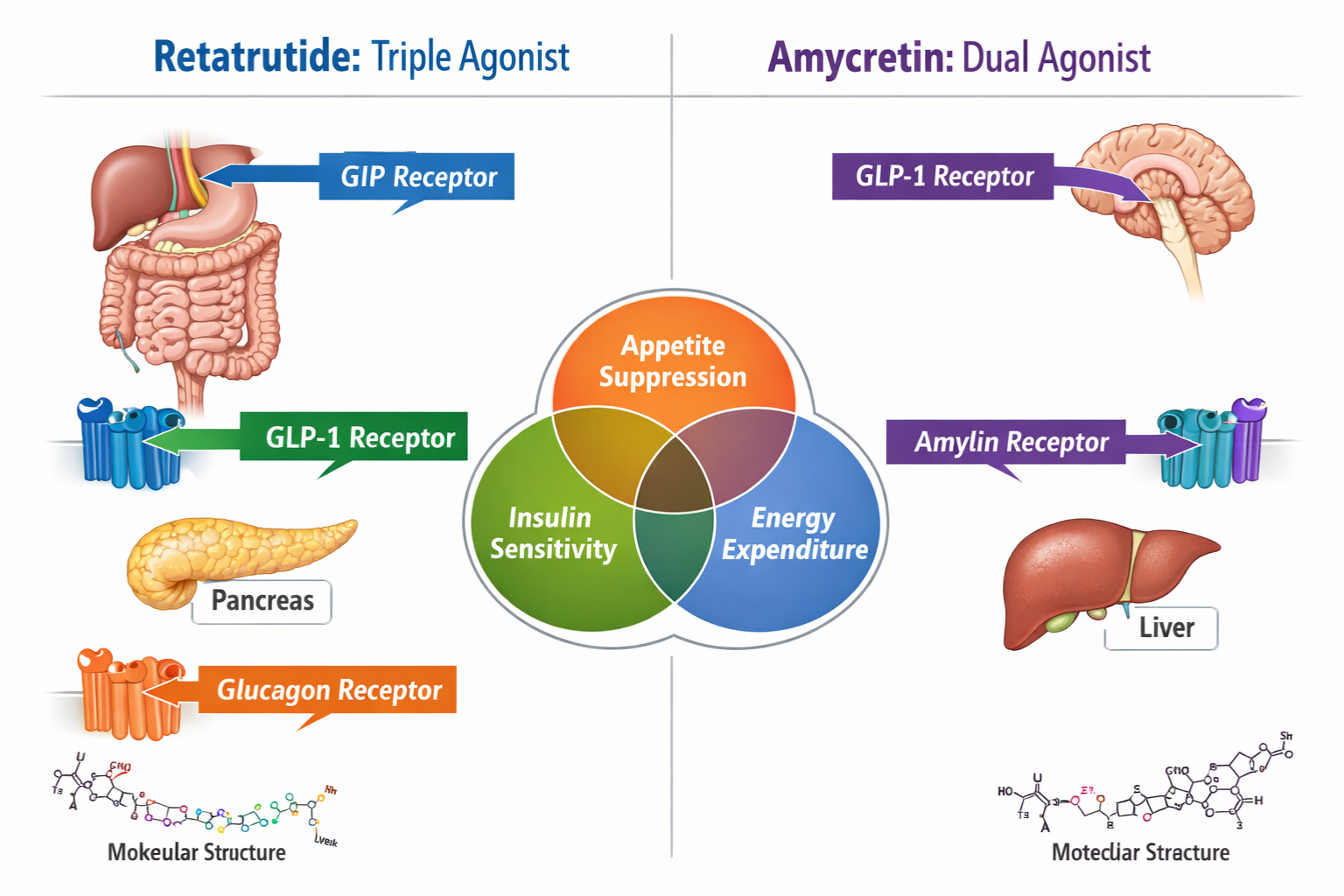
Molecular Structure and Receptor Activity
Retatrutide represents a significant advancement in peptide research as a triple receptor agonist targeting glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors simultaneously. This tri-agonist approach creates a comprehensive metabolic signaling cascade that researchers find particularly valuable for investigating complex metabolic pathways.
The molecular design of retatrutide incorporates specific amino acid sequences that enable selective binding to all three receptor types with varying affinities. This structural sophistication allows researchers to examine how coordinated receptor activation influences:
- Energy expenditure pathways through glucagon receptor stimulation
- Insulin secretion mechanisms via GIP and GLP-1 receptor engagement
- Appetite regulation circuits mediated primarily through GLP-1 activity
- Hepatic glucose production modulated by glucagon signaling
- Lipid metabolism processes influenced by the combined receptor profile
Pharmacokinetic Properties
The pharmacokinetic profile of retatrutide demonstrates an extended half-life that supports once-weekly administration protocols in research settings. This extended duration results from structural modifications that enhance resistance to enzymatic degradation, particularly by dipeptidyl peptidase-4 (DPP-4).
Research protocols utilizing retatrutide typically observe:
| Parameter | Characteristic |
|---|---|
| Half-life | Approximately 5-7 days |
| Peak concentration | 24-48 hours post-administration |
| Steady state | Achieved after 4-5 weeks |
| Bioavailability | High (>80% in research models) |
| Elimination | Primarily renal pathway |
For laboratories conducting longitudinal studies, these pharmacokinetic properties facilitate consistent experimental conditions with reduced administration frequency. Researchers sourcing retatrutide from verified suppliers can maintain rigorous protocols that account for these temporal dynamics.
Research Applications and Experimental Contexts
Retatrutide’s unique triple-agonist mechanism makes it particularly valuable for research investigating:
✅ Multi-pathway metabolic regulation – examining how simultaneous receptor activation influences systemic metabolism
✅ Energy balance studies – investigating the interplay between energy intake, expenditure, and storage
✅ Hepatic function research – exploring liver metabolism, glucose production, and lipid processing
✅ Comparative pharmacology – benchmarking against single or dual agonist compounds
✅ Dose-response characterization – establishing optimal concentration ranges for specific research outcomes
The compound’s comprehensive receptor profile enables researchers to model complex metabolic scenarios that more closely approximate integrated physiological systems. This makes retatrutide an invaluable tool for laboratories focused on understanding holistic metabolic regulation rather than isolated pathway effects.
Understanding Amycretin: The Dual Agonist Approach
Molecular Composition and Dual Receptor Targeting
Amycretin takes a different strategic approach as a dual agonist combining GLP-1 receptor activation with amylin receptor agonism. This pairing creates a complementary mechanism where GLP-1 effects on insulin secretion and appetite suppression work synergistically with amylin’s role in gastric emptying and satiety signaling.
The molecular architecture of amycretin incorporates:
- GLP-1 receptor binding domains that activate incretin pathways
- Amylin-mimetic sequences that engage amylin receptor complexes
- Structural stability elements that resist rapid degradation
- Optimized pharmacophore regions for dual receptor selectivity
This dual-target design offers researchers a focused tool for investigating the intersection of incretin and amylin biology—two systems that naturally work in concert during postprandial metabolic regulation.
Mechanistic Distinctions from Retatrutide
When comparing retatrutide vs amycretin, the fundamental mechanistic difference lies in receptor selectivity and the resulting metabolic cascade:
Retatrutide’s Triple Pathway:
- GIP receptor → Enhanced insulin response, potential lipid effects
- GLP-1 receptor → Insulin secretion, appetite reduction, gastric slowing
- Glucagon receptor → Increased energy expenditure, hepatic effects
Amycretin’s Dual Pathway:
- GLP-1 receptor → Incretin effects, appetite modulation
- Amylin receptor → Gastric emptying delay, satiety enhancement, glucagon suppression
The absence of glucagon receptor activation in amycretin represents both a distinction and a research opportunity. While retatrutide’s glucagon component may drive certain metabolic effects, amycretin’s focused GLP-1/amylin combination allows researchers to isolate these specific pathways without glucagon-mediated confounding variables.
Experimental Advantages of the Amycretin Profile
Research protocols utilizing amycretin benefit from several distinctive characteristics:
🔬 Gastric motility studies – The amylin component provides robust effects on gastric emptying, making amycretin valuable for digestive system research
🔬 Satiety mechanism investigations – Dual action on both GLP-1 and amylin pathways offers insights into complementary appetite regulation systems
🔬 Postprandial glucose research – Combined incretin and amylin effects create a comprehensive postprandial metabolic profile
🔬 Comparative agonist studies – Enables direct comparison with single-target GLP-1 or amylin agonists to quantify synergistic effects
🔬 Cardiovascular-metabolic research – Both GLP-1 and amylin pathways have cardiovascular implications worthy of investigation
For research institutions examining peptide mechanisms in metabolic contexts, amycretin provides a streamlined dual-pathway model that avoids the additional complexity of glucagon receptor involvement.
Direct Comparison: Retatrutide vs Amycretin
Receptor Profile and Signaling Pathways
The most fundamental distinction in the retatrutide vs amycretin comparison centers on receptor engagement:
| Receptor Target | Retatrutide | Amycretin |
|---|---|---|
| GLP-1 Receptor |
✅ Strong agonist |
✅ Strong agonist | | GIP Receptor |
✅ Agonist |
❌ No activity | | Glucagon Receptor |
✅ Agonist |
❌ No activity | | Amylin Receptor |
❌ No activity |
✅ Agonist | | Total Pathways | Triple (3) | Dual (2) |
This receptor divergence creates distinctly different research applications. Retatrutide’s triple-agonist profile enables comprehensive metabolic investigations spanning insulin, glucagon, and incretin systems. Amycretin’s dual-agonist approach provides focused examination of GLP-1-amylin synergy without glucagon-mediated effects.
Metabolic Effects and Research Outcomes
Research data comparing these compounds reveals important distinctions in metabolic parameters:
Energy Expenditure:
Retatrutide’s glucagon receptor activation may contribute to enhanced thermogenesis and energy expenditure in research models. Amycretin lacks this direct glucagon-mediated thermogenic component, though GLP-1 and amylin pathways may influence energy balance through different mechanisms.
Appetite Regulation:
Both compounds demonstrate robust effects on appetite-related pathways, though through different mechanisms. Retatrutide achieves this primarily through GLP-1 receptor activation, while amycretin benefits from synergistic GLP-1 and amylin receptor engagement—the latter being particularly relevant for satiety signaling.
Glucose Homeostasis:
Retatrutide’s multi-receptor approach influences glucose regulation through:
- GLP-1-mediated insulin secretion enhancement
- GIP effects on beta-cell function
- Glucagon receptor modulation of hepatic glucose output
Amycretin affects glucose homeostasis via:
- GLP-1 incretin effects on insulin
- Amylin-mediated glucagon suppression
- Delayed gastric emptying reducing postprandial glucose spikes
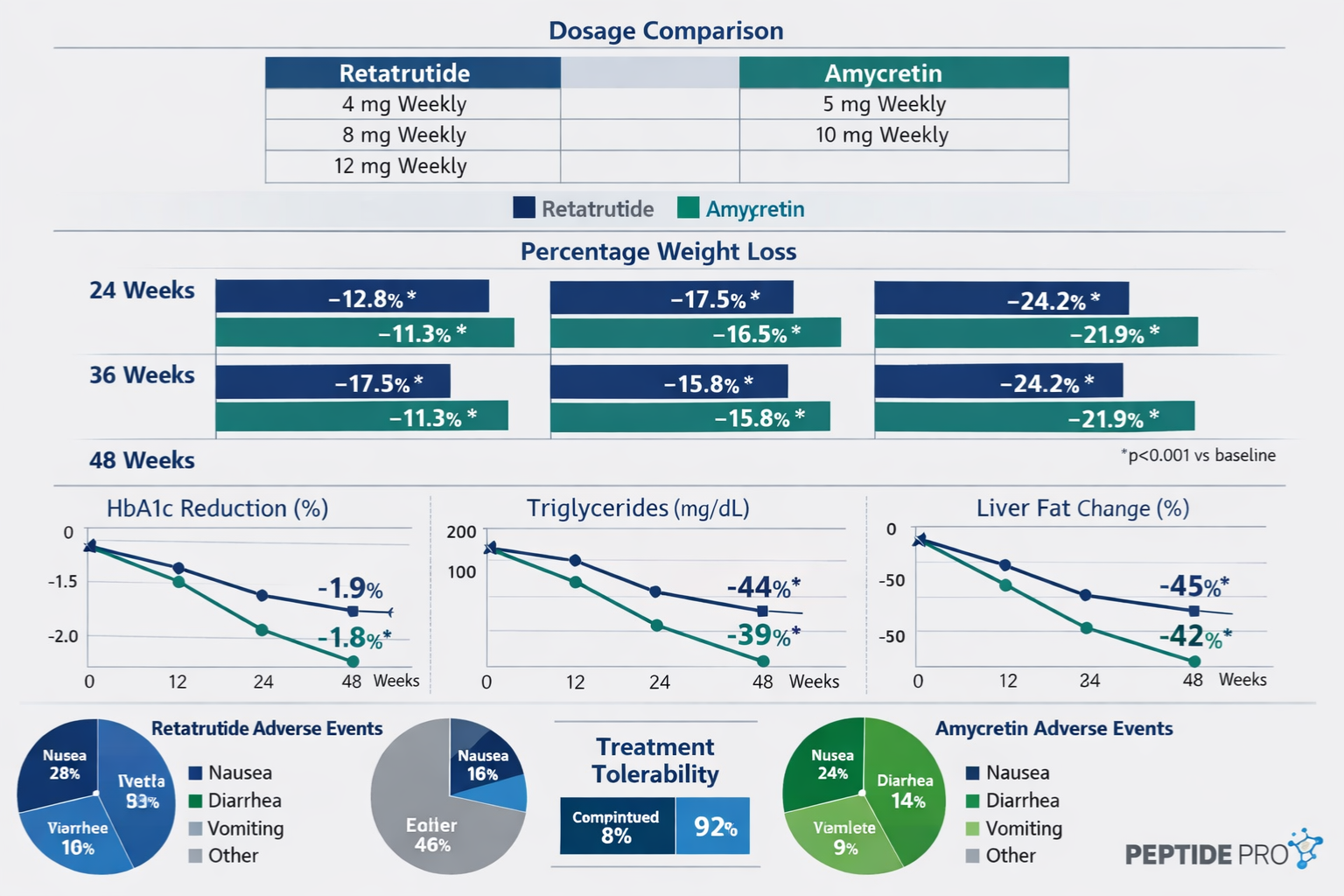
Dosing Considerations in Research Protocols
Experimental dosing strategies differ between these compounds based on their pharmacokinetic profiles and potency characteristics:
Retatrutide Research Dosing:
- Typical concentration ranges: 0.5 mg – 12 mg in various research models
- Administration frequency: Weekly protocols most common
- Dose escalation: Gradual titration often employed to establish dose-response curves
- Reconstitution: Requires bacteriostatic water; stored at 2-8°C post-reconstitution
Amycretin Research Dosing:
- Concentration parameters vary based on specific research objectives
- Administration schedules determined by experimental design requirements
- Titration protocols established according to research endpoints
- Storage and handling: Similar requirements to other research peptides
Researchers must account for these dosing distinctions when designing comparative studies or selecting the appropriate compound for specific experimental objectives. High-quality research peptides require precise handling regardless of which compound is selected.
Side Effect Profiles in Research Models
Understanding the adverse event profiles observed in research settings helps inform protocol design and safety monitoring:
Common Observations with Retatrutide:
- Gastrointestinal effects (nausea, occasional emesis) particularly during dose escalation
- Injection site reactions in subcutaneous administration protocols
- Transient effects on heart rate in some research models
- Potential hepatic enzyme elevations requiring monitoring
Common Observations with Amycretin:
- GI-related effects similar to other GLP-1 pathway agonists
- Amylin-specific effects on gastric motility
- Generally well-tolerated in research contexts with appropriate protocols
- Requires similar monitoring parameters as other incretin-based compounds
Both compounds necessitate comprehensive monitoring protocols and adherence to research safety standards. The specific adverse event profile should inform experimental design, particularly regarding dose escalation schedules and endpoint selection.
Clinical Research Data: Comparative Analysis
Published Research Findings
As of 2026, the body of published research examining retatrutide vs amycretin continues to expand, though retatrutide has accumulated more extensive data from large-scale research protocols:
Retatrutide Research Highlights:
The TRIUMPH research program has generated substantial data on retatrutide’s effects across multiple metabolic parameters. Key findings from published research include:
- Dose-dependent effects across concentration ranges from 0.5 mg to 12 mg weekly
- Significant metabolic changes observed across multiple endpoints including body composition, glycemic parameters, and lipid profiles
- Sustained effects maintained throughout extended research protocols (24-48 weeks)
- Favorable tolerability with manageable adverse event profiles in most research contexts
Research published in leading journals has demonstrated retatrutide’s capacity to engage all three target receptors simultaneously, creating comprehensive metabolic effects that exceed those observed with single or dual agonist comparators.
Amycretin Research Progress:
While amycretin research is at an earlier stage compared to retatrutide, emerging data demonstrates:
- Synergistic GLP-1/amylin effects that validate the dual-agonist approach
- Distinct mechanistic profile compared to GLP-1-only or amylin-only compounds
- Promising metabolic outcomes across various research parameters
- Unique gastric emptying effects attributable to the amylin component
The research trajectory for amycretin suggests significant potential, though direct head-to-head comparisons with retatrutide remain limited in published literature as of 2026.
Comparative Efficacy Metrics
When examining available research data, several patterns emerge in the retatrutide vs amycretin comparison:
| Research Parameter | Retatrutide Findings | Amycretin Findings |
|---|---|---|
| Metabolic markers | Comprehensive multi-pathway effects | Focused GLP-1/amylin pathway effects |
| Body composition | Significant changes across dose ranges | Promising results in early research |
| Glycemic control | Robust improvements via triple mechanism | Strong effects via dual incretin/amylin action |
| Lipid parameters | Notable improvements in research models | Data emerging from ongoing protocols |
| Research timeline | Extensive multi-year data available | Earlier-stage but expanding rapidly |
These comparative metrics help researchers select the appropriate compound based on specific experimental objectives and desired mechanistic pathways.
Long-Term Research Considerations
Extended research protocols reveal important considerations for both compounds:
Durability of Effects:
Retatrutide research demonstrates sustained metabolic effects over 48+ week protocols, with minimal attenuation of response. The triple-receptor mechanism appears to maintain efficacy without significant tachyphylaxis in most research models.
Amycretin’s long-term profile continues to be characterized, with early data suggesting durable effects consistent with its dual-agonist mechanism.
Safety Monitoring:
Long-term research protocols for both compounds require:
- Regular monitoring of relevant biomarkers
- Assessment of injection site tolerance
- Evaluation of gastrointestinal adaptation
- Cardiovascular parameter tracking
- Hepatic and renal function surveillance
Research Protocol Design:
Researchers planning extended studies with either compound should consider:
- Appropriate wash-out periods for crossover designs
- Dose maintenance vs. escalation strategies
- Endpoint timing relative to pharmacokinetic profiles
- Control group selection and comparator compounds
Practical Research Considerations
Sourcing Research-Grade Peptides
The quality and purity of research peptides directly impacts experimental validity and reproducibility. When sourcing retatrutide or amycretin for laboratory investigations, researchers should prioritize:
Purity Standards:
- Minimum 98% purity verified by HPLC analysis
- Comprehensive certificates of analysis (COA) documenting batch-specific purity
- Third-party verification when available
- Proper lyophilization and storage conditions maintained throughout supply chain
Supplier Reliability:
Established suppliers like PEPTIDE PRO provide:
- Consistent batch-to-batch quality
- Transparent documentation and COAs
- Proper storage and handling throughout distribution
- Responsive customer support for research inquiries
- Fast UK delivery with temperature-controlled shipping
Regulatory Compliance:
All research peptides must be clearly labeled “For Research Use Only” and comply with relevant regulations governing research materials. Reputable suppliers ensure proper labeling and documentation to support compliant research activities.
Reconstitution and Handling Protocols
Proper reconstitution is critical for maintaining peptide integrity and ensuring experimental validity:
Reconstitution Best Practices:
- Use appropriate diluent: Bacteriostatic water is standard for most research peptides
- Calculate precise volumes: Determine required concentration based on experimental protocol
- Gentle mixing: Avoid vigorous shaking; use gentle swirling or rolling motion
- Proper technique: Inject diluent slowly along pen peptide wall, not directly onto lyophilized peptide
- Complete dissolution: Ensure peptide fully dissolves before use; may require several minutes
Storage Requirements:
📦 Pre-reconstitution:
- Store lyophilized peptides at -20°C or colder
- Protect from light and moisture
- Allow pen peptides to reach room temperature before opening to prevent condensation
📦 Post-reconstitution:
- Refrigerate at 2-8°C immediately after reconstitution
- Use within timeframe specified by stability data (typically 2-4 weeks for most peptides)
- Protect from light using amber pen peptides or aluminum foil wrapping
- Avoid freeze-thaw cycles
For detailed guidance on reconstitution and storage protocols, researchers can consult with experienced suppliers who understand the specific requirements of research-grade peptides.
Experimental Design Considerations
Designing robust research protocols comparing retatrutide and amycretin requires careful attention to:
Control Groups:
- Vehicle-only controls to establish baseline
- Positive controls using established comparator compounds
- Dose-response groups to characterize concentration-effect relationships
Timing and Duration:
- Account for pharmacokinetic differences between compounds
- Allow sufficient time to reach steady-state concentrations
- Plan endpoint assessments relative to dosing schedules
Outcome Measures:
- Select endpoints appropriate to each compound’s mechanism
- Include both primary and secondary metabolic parameters
- Consider mechanism-specific biomarkers (e.g., glucagon-related markers for retatrutide)
Statistical Considerations:
- Power calculations based on expected effect sizes
- Appropriate statistical tests for comparing multi-group designs
- Correction for multiple comparisons when examining numerous endpoints
Selecting Between Retatrutide and Amycretin for Research
Research Objective Alignment
The choice between retatrutide vs amycretin should be driven primarily by research objectives and the specific metabolic pathways under investigation:
Choose Retatrutide When:
�
� Research focuses on comprehensive metabolic regulation involving multiple receptor systems �
� Investigating energy expenditure mechanisms and thermogenic pathways �
� Examining glucagon receptor biology and its interaction with incretin systems �
� Conducting comparative studies requiring the most extensively characterized multi-agonist compound �
� Exploring hepatic metabolism and glucose production pathways
Choose Amycretin When:
�
� Research specifically targets GLP-1 and amylin pathway interactions �
� Investigating gastric emptying mechanisms and their metabolic consequences �
� Examining satiety signaling through complementary incretin and amylin systems �
� Seeking to isolate GLP-1/amylin effects without glucagon receptor involvement �
� Conducting research on amylin receptor biology in metabolic contexts
Budgetary and Practical Factors
Research planning must also account for practical considerations:
Cost Considerations:
- Peptide acquisition costs vary by compound, purity, and quantity
- Retatrutide’s more extensive characterization may influence pricing
- Bulk purchasing options may be available for large-scale studies
- Consider total protocol costs including dosing frequency and study duration
Availability and Lead Times:
- Established suppliers like PEPTIDE PRO maintain inventory of commonly requested peptides
- Same-day dispatch available for orders placed before 1pm (Monday-Friday)
- International shipping options for laboratories outside the UK
- Custom synthesis may be required for specific variants or modifications
Technical Support:
- Access to knowledgeable support staff for protocol questions
- Availability of technical documentation and handling guidelines
- Supplier responsiveness for time-sensitive research needs
Combination and Comparative Research Strategies
Some research protocols may benefit from incorporating both compounds:
Head-to-Head Comparisons:
Direct comparative studies examining retatrutide vs amycretin under identical experimental conditions provide valuable insights into mechanistic differences and relative efficacy across various endpoints.
Sequential Studies:
Research programs may employ sequential protocols, first characterizing one compound extensively before conducting comparative studies with the other, building a comprehensive understanding of both mechanisms.
Mechanistic Investigations:
Using both compounds in parallel can help isolate the contribution of specific receptor pathways—for example, comparing retatrutide’s effects with and without glucagon receptor involvement by referencing amycretin data.
Future Directions and Emerging Research
Evolving Research Landscape in 2026
The field of multi-receptor agonist research continues to advance rapidly. As of 2026, several trends are shaping the retatrutide vs amycretin research landscape:
Expanding Clinical Data:
Both compounds are generating increasingly robust datasets from large-scale research protocols. This expanding evidence base enables more sophisticated comparative analyses and mechanistic insights.
Novel Combinations:
Researchers are exploring combinations of multi-receptor agonists with other metabolic modulators, creating opportunities to investigate synergistic mechanisms and optimize outcomes across multiple pathways.
Personalized Research Approaches:
Growing understanding of individual variability in receptor expression and pathway activation is driving more nuanced research designs that account for heterogeneity in responses.
Mechanistic Biomarker Development:
Advanced biomarkers specific to GIP, GLP-1, glucagon, and amylin pathways are enabling more precise characterization of each compound’s mechanism and facilitating better comparison between retatrutide and amycretin.
Next-Generation Multi-Receptor Agonists
The success of compounds like retatrutide and amycretin has catalyzed development of additional multi-receptor agonists:
- Quadruple agonists incorporating additional receptor targets beyond the triple mechanism
- Optimized dual agonists with refined selectivity profiles and enhanced pharmacokinetics
- Tissue-selective agonists designed to preferentially activate receptors in specific organs or tissues
- Long-acting formulations extending dosing intervals beyond current weekly protocols
These emerging compounds will provide additional tools for researchers and create new opportunities for comparative studies examining the optimal combination and balance of receptor activation.
Implications for Metabolic Research
The insights gained from retatrutide vs amycretin research extend beyond these specific compounds to inform broader understanding of:
Receptor Synergy:
How do different combinations of receptor activation create synergistic or antagonistic effects? Research comparing these compounds helps answer fundamental questions about metabolic pathway integration.
Pathway Selectivity:
What are the advantages and limitations of targeting multiple pathways simultaneously versus focusing on specific receptor combinations? Comparative data informs optimal research strategies.
Translation and Application:
How do findings from research protocols translate to real-world applications? Long-term studies with both compounds provide critical data for understanding durability, safety, and practical implementation.
Frequently Asked Questions
What is the primary difference between retatrutide and amycretin?
The fundamental difference lies in their receptor profiles: retatrutide is a triple agonist targeting GIP, GLP-1, and glucagon receptors, while amycretin is a dual agonist targeting GLP-1 and amylin receptors. This creates distinct metabolic signaling cascades with different research applications.
Which compound has more published research data?
As of 2026, retatrutide has accumulated more extensive published research data from large-scale protocols, particularly through the TRIUMPH research program. Amycretin research is at an earlier stage but expanding rapidly with promising preliminary findings.
Can these compounds be used in the same research protocol?
Yes, comparative research protocols often include both compounds to directly compare their mechanisms and effects under identical experimental conditions. This approach provides valuable insights into the contribution of different receptor pathways.
What are the storage requirements for these peptides?
Both compounds should be stored as lyophilized powder at -20°C or colder before reconstitution. After reconstitution with bacteriostatic water, store at 2-8°C and use within the timeframe specified by stability data, typically 2-4 weeks. Protect from light and avoid freeze-thaw cycles.
Where can researchers source high-quality retatrutide and amycretin?
Reputable suppliers specializing in research-grade peptides, such as PEPTIDE PRO, provide high-purity compounds with comprehensive certificates of analysis, proper storage conditions, and fast UK delivery. Always verify purity standards and supplier credentials before procurement.
How do dosing protocols differ between these compounds?
Retatrutide typically employs weekly administration protocols in research settings due to its extended half-life. Amycretin dosing schedules are determined by specific experimental objectives and pharmacokinetic considerations. Both require careful dose escalation in research protocols to establish optimal concentration ranges.
Are there specific research applications better suited to one compound over the other?
Yes. Retatrutide is particularly valuable for comprehensive metabolic research involving glucagon receptor pathways and energy expenditure mechanisms. Amycretin is especially useful for investigating GLP-1/amylin synergy, gastric emptying, and satiety signaling without glucagon receptor involvement. Research objectives should guide compound selection.
Conclusion
The comparison of retatrutide vs amycretin reveals two sophisticated research tools with distinct mechanisms and applications. Retatrutide’s triple-agonist profile targeting GIP, GLP-1, and glucagon receptors provides comprehensive metabolic pathway engagement, making it invaluable for research examining multi-system metabolic regulation and energy balance. Amycretin’s focused dual-agonist approach combining GLP-1 and amylin receptor activation offers unique insights into incretin-amylin synergy and gastric motility mechanisms.
Neither compound is universally “superior”—rather, each excels in specific research contexts aligned with its mechanistic profile. Researchers investigating energy expenditure, hepatic metabolism, or comprehensive multi-receptor interactions may find retatrutide’s triple mechanism advantageous. Those focusing on GLP-1/amylin pathway interactions, satiety signaling, or gastric emptying may prefer amycretin’s targeted dual approach.
Key Decision Factors:
✅ Align compound selection with research objectives and the specific metabolic pathways under investigation
✅ Consider mechanistic requirements: triple-pathway engagement vs. focused dual-agonist effects
✅ Account for practical factors including dosing schedules, available research data, and experimental timeline
✅ Source from reputable suppliers offering verified purity, proper storage, and comprehensive documentation
✅ Design robust protocols with appropriate controls, endpoints, and statistical considerations
Next Steps for Researchers:
For laboratories ready to incorporate these advanced peptides into research protocols:
- Define specific research objectives and identify which receptor pathways are most relevant to your investigation
- Review published literature on both compounds to understand their characterization and potential applications
- Consult with experienced suppliers like PEPTIDE PRO to discuss purity standards, availability, and technical specifications
- Develop comprehensive protocols accounting for reconstitution, dosing, monitoring, and endpoint assessment
- Ensure proper storage and handling infrastructure to maintain peptide integrity throughout the research timeline
The field of multi-receptor agonist research continues to evolve rapidly, with both retatrutide and amycretin contributing valuable insights into metabolic regulation. As research data expands and new compounds emerge, the comparative framework established through retatrutide vs amycretin studies will inform next-generation peptide development and optimization.
For researchers seeking high-purity research peptides with verified quality standards, fast UK delivery, and professional support, PEPTIDE PRO offers an extensive catalogue of compounds including both retatrutide and amycretin, along with comprehensive resources for proper handling and storage.
Remember: All peptides discussed in this article are strictly for research use only and not intended for human or animal consumption. Always adhere to institutional research guidelines and regulatory requirements when conducting peptide research.
