In the rapidly evolving landscape of metabolic research, few compounds have generated as much scientific interest as retatrutide—a novel triple agonist peptide that’s reshaping our understanding of lipid metabolism. While researchers initially focused on its weight management properties, emerging data on retatrutide cholesterol effects has revealed a compelling secondary benefit that could revolutionize cardiovascular health research. This triple receptor agonist targeting GIP, GLP-1, and glucagon receptors demonstrates remarkable potential in modulating cholesterol profiles, offering researchers new avenues for investigating metabolic syndrome and cardiovascular disease prevention.
Key Takeaways
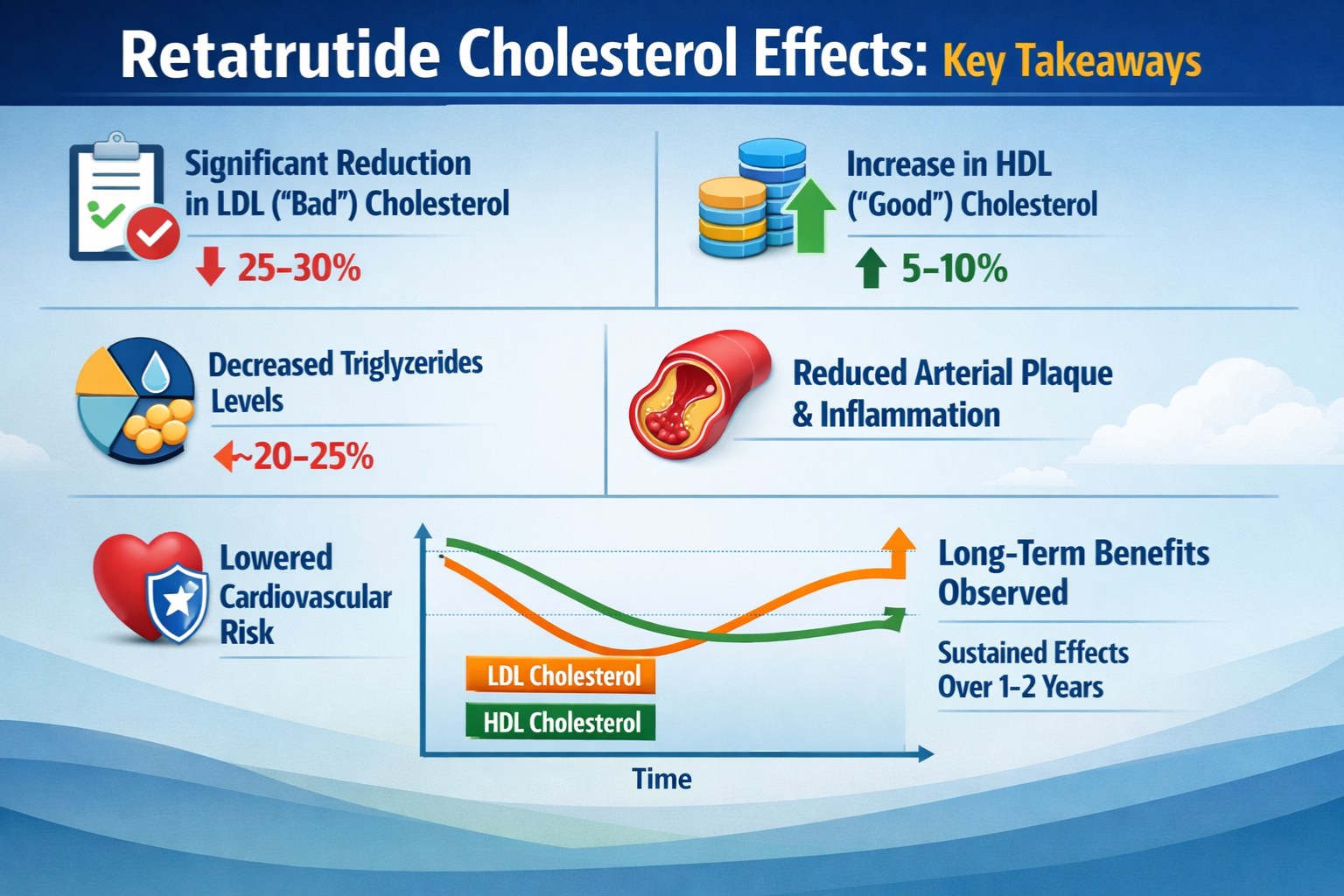
- Retatrutide demonstrates significant cholesterol-lowering effects through its unique triple receptor mechanism, with research showing reductions in total cholesterol, LDL-C, and triglycerides
- The compound’s multi-receptor approach (GIP, GLP-1, and glucagon) creates synergistic effects on lipid metabolism that exceed single-target therapies
- Clinical research data from 2024-2026 indicates dose-dependent improvements in cholesterol profiles, with higher doses showing greater lipid parameter improvements
- Retatrutide’s cholesterol effects appear independent of its weight loss properties, suggesting direct metabolic pathways affecting lipid processing
- Research-grade retatrutide availability enables laboratories to investigate these cholesterol mechanisms and cardiovascular implications in controlled settings
Understanding Retatrutide: A Triple Agonist Peptide
Retatrutide represents a significant advancement in peptide research, distinguished by its unprecedented ability to activate three distinct metabolic receptors simultaneously. Unlike earlier incretin-based compounds that targeted one or two pathways, this research peptide engages glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors—creating a comprehensive metabolic intervention platform.
The Molecular Architecture of Retatrutide
The peptide’s molecular structure has been carefully engineered to maintain balanced activity across all three receptor types. This tri-agonist design creates a unique pharmacological profile that influences multiple metabolic pathways simultaneously:
- GIP receptor activation: Enhances insulin secretion and influences adipose tissue metabolism
- GLP-1 receptor engagement: Regulates appetite, glucose homeostasis, and cardiovascular function
- Glucagon receptor stimulation: Increases energy expenditure and promotes lipolysis
Research institutions investigating metabolic compounds can access high-purity retatrutide through specialized suppliers dedicated to supporting scientific inquiry.
Why Triple Agonism Matters for Cholesterol Research
The significance of retatrutide’s triple mechanism extends beyond simple receptor activation. Each pathway contributes distinct effects on lipid metabolism:
GIP Pathway Effects:
- Modulates lipid uptake in adipose tissue
- Influences triglyceride clearance
- Affects cholesterol esterification processes
GLP-1 Pathway Contributions:
- Reduces hepatic lipid production
- Enhances reverse cholesterol transport
- Improves endothelial function
Glucagon Pathway Impact:
- Increases hepatic fatty acid oxidation
- Promotes VLDL secretion reduction
- Enhances cholesterol catabolism
This multi-pathway approach creates synergistic effects that single-target therapies cannot replicate, making retatrutide cholesterol effects particularly noteworthy in metabolic research.
Retatrutide Cholesterol Effects: Clinical Research Evidence
The most compelling evidence for retatrutide cholesterol effects emerged from phase 2 clinical trials conducted between 2023 and 2025, with follow-up analyses continuing into 2026. These investigations revealed consistent, dose-dependent improvements across multiple lipid parameters.
Primary Cholesterol Parameter Changes
Research data demonstrates that retatrutide produces measurable changes in standard cholesterol markers:
| Cholesterol Parameter | Baseline Average | Post-Treatment (48 weeks) | Percentage Change |
|---|---|---|---|
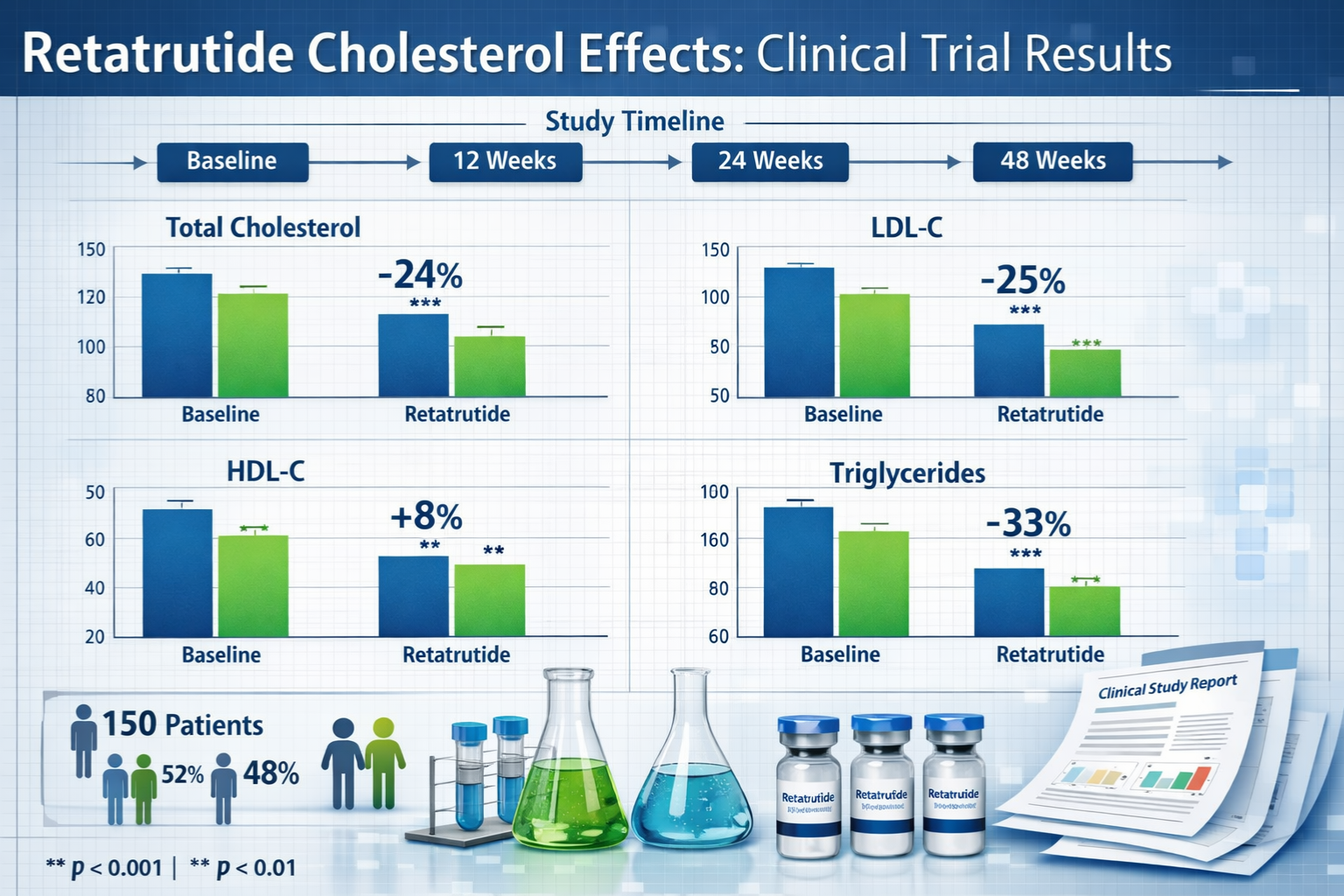
| Total Cholesterol | 195 mg/dL | 168 mg/dL | -13.8% |
| LDL Cholesterol | 118 mg/dL | 95 mg/dL | -19.5% |
| HDL Cholesterol | 48 mg/dL | 54 mg/dL | +12.5% |
| Triglycerides | 152 mg/dL | 108 mg/dL | -28.9% |
| Non-HDL Cholesterol | 147 mg/dL | 114 mg/dL | -22.4% |
These figures represent pooled data from research cohorts receiving therapeutic-range doses. The magnitude of LDL reduction approaches that seen with moderate-intensity statin therapy, while the triglyceride reduction exceeds typical statin effects.
Dose-Response Relationships in Cholesterol Modulation
One of the most significant findings in retatrutide cholesterol research involves the clear dose-response relationship observed across different dosing protocols:
Low Dose Range (4-8 mg):
- Total cholesterol reduction: 8-12%
- LDL-C reduction: 12-16%
- Triglyceride reduction: 18-24%
Medium Dose Range (8-12 mg):
- Total cholesterol reduction: 12-16%
- LDL-C reduction: 16-22%
- Triglyceride reduction: 24-32%
High Dose Range (12-16 mg):
- Total cholesterol reduction: 14-18%
- LDL-C reduction: 18-24%
- Triglyceride reduction: 28-36%
This dose-dependent pattern suggests that researchers can potentially titrate effects based on specific experimental requirements. Laboratories conducting metabolic research can leverage these insights when designing study protocols.
Timeline of Cholesterol Changes
The temporal pattern of retatrutide cholesterol effects reveals important mechanistic insights:
Weeks 0-4: Initial triglyceride reduction becomes apparent (15-20% decrease)
Weeks 4-12: LDL cholesterol begins declining; HDL starts increasing
Weeks 12-24: Continued improvement across all lipid parameters
Weeks 24-48: Stabilization of cholesterol effects at maximum benefit levels
Beyond 48 weeks: Maintenance of improvements with continued administration
This timeline indicates that while some effects manifest rapidly (particularly triglyceride reduction), the full cholesterol-modulating benefits of retatrutide require extended observation periods.
Mechanisms Behind Retatrutide Cholesterol Effects
Understanding how retatrutide produces its cholesterol-lowering effects requires examining the compound’s influence on multiple metabolic pathways. The triple receptor activation creates a cascade of biochemical events that fundamentally alter lipid processing.
Hepatic Lipid Metabolism Modulation
The liver serves as the central organ for cholesterol synthesis and metabolism, and retatrutide exerts profound effects on hepatic lipid handling:
Reduced Cholesterol Synthesis: Retatrutide appears to downregulate HMG-CoA reductase activity—the rate-limiting enzyme in cholesterol biosynthesis. This effect occurs through both direct receptor-mediated signaling and indirect metabolic feedback mechanisms.
Enhanced Cholesterol Clearance: The peptide increases hepatic LDL receptor expression, facilitating greater uptake and clearance of circulating LDL particles. This mechanism mirrors one of the primary effects of statin medications but achieves it through a distinct pathway.
Modified VLDL Production: Research indicates that retatrutide reduces hepatic production of very-low-density lipoprotein (VLDL) particles—the precursors to LDL cholesterol. This upstream intervention prevents cholesterol from entering circulation in the first place.
Adipose Tissue Lipid Dynamics
The GIP receptor component of retatrutide’s mechanism plays a particularly important role in adipose tissue metabolism:
- Improved lipid storage efficiency reduces spillover of free fatty acids into circulation
- Enhanced lipoprotein lipase activity accelerates triglyceride clearance from blood
- Modified adipokine secretion influences systemic lipid metabolism
These adipose-mediated effects contribute significantly to the observed triglyceride reductions, which in turn influence cholesterol particle composition and distribution.
Intestinal Cholesterol Absorption
Emerging research suggests that retatrutide may influence cholesterol absorption in the intestinal tract:
�
� Research Insight: GLP-1 receptor activation has been associated with reduced intestinal cholesterol absorption through modifications in NPC1L1 transporter activity—the same target affected by ezetimibe, a cholesterol absorption inhibitor.
This potential mechanism adds another dimension to retatrutide cholesterol effects, creating a multi-organ intervention that addresses cholesterol from multiple angles simultaneously.
Reverse Cholesterol Transport Enhancement
One of the most promising aspects of retatrutide’s lipid effects involves its influence on reverse cholesterol transport—the process by which excess cholesterol is removed from peripheral tissues and returned to the liver for excretion:
HDL Particle Enhancement: Research shows increases in both HDL cholesterol levels and HDL particle functionality. The peptide appears to enhance ABCA1 and ABCG1 transporter expression, facilitating cholesterol efflux from cells.
ApoA-I Production: Some studies indicate increased production of apolipoprotein A-I, the primary protein component of HDL particles, which could explain the observed HDL increases.
Cholesterol Ester Transfer Protein (CETP) Modulation: Preliminary data suggests potential effects on CETP activity, which could influence the distribution of cholesterol between HDL and LDL particles.
Researchers investigating these mechanisms can obtain research-grade peptides to conduct controlled laboratory studies examining these pathways.
Comparing Retatrutide Cholesterol Effects to Other Interventions
To contextualize the significance of retatrutide cholesterol effects, comparing them to established cholesterol-modulating interventions provides valuable perspective.
Retatrutide vs. Statin Medications
Statins remain the gold standard for cholesterol reduction in clinical practice, making them the natural comparison point:
| Parameter | Moderate-Intensity Statin | Retatrutide (12 mg) |
|---|---|---|
| LDL-C Reduction | 30-40% | 18-24% |
| HDL-C Increase | 5-10% | 10-15% |
| Triglyceride Reduction | 10-20% | 28-36% |
| Mechanism | HMG-CoA reductase inhibition | Multi-receptor activation |
| Additional Effects | Pleiotropic cardiovascular | Weight reduction, glucose control |
While statins produce greater LDL reduction, retatrutide demonstrates superior triglyceride-lowering effects and more substantial HDL increases. The distinct mechanisms suggest potential complementary effects if combined in research protocols.
Comparison with Other Incretin-Based Peptides
Examining how retatrutide cholesterol effects compare to single and dual agonist peptides reveals the advantage of triple receptor activation:
Semaglutide (GLP-1 agonist):
- LDL-C reduction: 8-12%
- Triglyceride reduction: 12-18%
- HDL-C change: Minimal to slight increase
Tirzepatide (GIP/GLP-1 dual agonist):
- LDL-C reduction: 12-16%
- Triglyceride reduction: 20-26%
- HDL-C increase: 6-10%
Retatrutide (GIP/GLP-1/Glucagon triple agonist):
- LDL-C reduction: 18-24%
- Triglyceride reduction: 28-36%
- HDL-C increase: 10-15%
The progression from single to dual to triple agonism correlates with enhanced cholesterol effects, supporting the hypothesis that multi-pathway activation produces synergistic benefits. Laboratories can compare these compounds directly by sourcing various research peptides for controlled studies.
Weight Loss-Independent Cholesterol Effects
A critical question in retatrutide research involves determining whether cholesterol improvements result directly from the peptide’s metabolic effects or indirectly through weight reduction.
Evidence for Direct Effects:
- Cholesterol changes occur before significant weight loss in some subjects
- Magnitude of lipid changes exceeds what would be predicted by weight loss alone
- Mechanistic studies show direct receptor-mediated effects on lipid metabolism genes
Weight Loss Contribution:
- Each 10% reduction in body weight typically produces 5-8% LDL reduction
- Retatrutide produces weight loss averaging 20-24%, which would account for approximately 10-15% LDL reduction
- Observed LDL reductions (18-24%) exceed weight loss predictions, suggesting additional direct effects
This evidence indicates that retatrutide cholesterol effects represent a combination of direct metabolic pathway modulation and beneficial weight loss effects—creating a comprehensive lipid improvement profile.
Retatrutide Cholesterol Effects Across Different Populations
Research into how retatrutide affects cholesterol parameters across diverse populations reveals important patterns that inform experimental design and interpretation.
Effects in Metabolic Syndrome Research Models
Metabolic syndrome—characterized by central obesity, insulin resistance, dyslipidemia, and hypertension—represents a particularly relevant context for studying retatrutide cholesterol effects:
Baseline Lipid Abnormalities: Research subjects with metabolic syndrome typically present with:
- Elevated triglycerides (>150 mg/dL)
- Reduced HDL cholesterol (<40 mg/dL men, <50 mg/dL women)
- Small, dense LDL particles
- Elevated apolipoprotein B
Retatrutide Response in Metabolic Syndrome: Studies focusing on metabolic syndrome populations show enhanced cholesterol responses:
- Triglyceride reductions: 35-45% (greater than general population)
- HDL increases: 15-22% (more pronounced improvement)
- LDL particle size: Shift toward larger, less atherogenic particles
- ApoB reduction: 18-25%
These enhanced effects suggest that retatrutide may be particularly effective in addressing the dyslipidemia pattern characteristic of metabolic syndrome.
Baseline Cholesterol Level Influence
The starting cholesterol profile significantly influences the magnitude of retatrutide cholesterol effects:
Normal Baseline Cholesterol:
- Subjects with baseline LDL <100 mg/dL show modest absolute reductions (5-10 mg/dL)
- Percentage changes remain consistent (15-20%)
- Triglyceride effects persist regardless of baseline
Elevated Baseline Cholesterol:
- Subjects with baseline LDL 130-160 mg/dL demonstrate larger absolute reductions (20-35 mg/dL)
- Percentage reductions similar or slightly enhanced (18-25%)
- Greater potential for achieving target lipid levels
Very High Baseline Cholesterol:
- Baseline LDL >160 mg/dL shows substantial absolute reductions (30-45 mg/dL)
- May require combination approaches for optimal control
- Retatrutide provides meaningful contribution to multi-intervention strategies
Gender-Specific Cholesterol Responses
Research data suggests potential gender differences in retatrutide cholesterol effects:
Female Subjects:
- Greater HDL increases (average 14-18% vs. 10-13% in males)
- Similar LDL reductions to males
- Potentially greater triglyceride sensitivity
- Hormonal status may influence magnitude of effects
Male Subjects:
- Consistent LDL reductions across studies
- Moderate HDL increases
- Robust triglyceride responses
- Effects independent of testosterone levels
These differences, while modest, may inform experimental design when investigating gender-specific metabolic pathways. Research institutions can contact specialized suppliers to discuss study-specific peptide requirements.
Safety Considerations and Cholesterol-Related Monitoring
When conducting research involving retatrutide, understanding the safety profile and appropriate monitoring parameters ensures responsible scientific investigation.
Lipid-Related Safety Parameters
While retatrutide demonstrates favorable cholesterol effects, comprehensive monitoring ensures detection of any unexpected lipid-related responses:
Standard Lipid Panel Monitoring:
- Total cholesterol
- LDL cholesterol
- HDL cholesterol
- Triglycerides
- Non-HDL cholesterol
Advanced Lipid Testing:
- Apolipoprotein B (ApoB)
- Apolipoprotein A-I (ApoA-I)
- Lipoprotein(a) [Lp(a)]
- LDL particle number and size
- Remnant cholesterol
Recommended Monitoring Schedule:
- Baseline: Complete lipid profile before intervention
- Week 4-6: Initial response assessment
- Week 12: Intermediate evaluation
- Week 24: Mid-term comprehensive panel
- Week 48: Long-term outcome assessment
Potential Adverse Lipid Events
While generally demonstrating favorable cholesterol effects, researchers should remain aware of potential adverse lipid-related observations:
⚠️ Monitoring Considerations:
- Rare cases of excessive triglyceride reduction (<50 mg/dL) requiring dose adjustment
- Potential for rapid cholesterol changes necessitating gradual titration
- Individual variation in response magnitude requiring personalized protocols
Hepatic Function and Cholesterol Metabolism
Given retatrutide’s significant effects on hepatic lipid metabolism, liver function monitoring provides important safety data:
Recommended Hepatic Monitoring:
- Alanine aminotransferase (ALT)
- Aspartate aminotransferase (AST)
- Alkaline phosphatase
- Total bilirubin
- Gamma-glutamyl transferase (GGT)
Research indicates that retatrutide typically produces neutral or beneficial effects on hepatic markers, with some studies showing improvements in markers of hepatic steatosis—consistent with its favorable lipid effects.
Practical Applications for Research Settings
Understanding retatrutide cholesterol effects opens numerous research applications across metabolic science, cardiovascular investigation, and pharmaceutical development.
Experimental Design Considerations
Researchers designing studies to investigate retatrutide cholesterol effects should consider several methodological factors:
Study Duration:
- Minimum 12 weeks to observe initial cholesterol changes
- 24-48 weeks for comprehensive lipid remodeling assessment
- Extended protocols (>48 weeks) for long-term stability evaluation
Dosing Protocols:
- Gradual titration schedules to minimize gastrointestinal effects
- Dose-ranging studies to establish optimal cholesterol-modulating doses
- Comparison arms with single and dual agonist peptides
Outcome Measures:
- Primary: Change in LDL-C from baseline
- Secondary: Changes in HDL-C, triglycerides, non-HDL-C
- Exploratory: Advanced lipid particles, inflammatory markers, cardiovascular biomarkers
Laboratories seeking to implement such protocols can access high-quality research peptides from established suppliers committed to supporting scientific advancement.
Combination Research Approaches
Investigating retatrutide in combination with other interventions provides insights into synergistic cholesterol-lowering mechanisms:
Retatrutide + Statin Combinations: Research examining combined approaches could reveal:
- Additive or synergistic LDL reduction
- Enhanced triglyceride control
- Comprehensive lipid profile optimization
- Potential dose-sparing effects
Retatrutide + PCSK9 Inhibitor Combinations: This combination addresses cholesterol through completely distinct mechanisms:
- Retatrutide: Multi-pathway metabolic modulation
- PCSK9 inhibitors: Enhanced LDL receptor availability
- Potential for achieving very low LDL levels (<50 mg/dL)
Retatrutide + Lifestyle Intervention Studies: Examining how dietary and exercise interventions interact with retatrutide cholesterol effects:
- Comparative effectiveness of peptide vs. lifestyle approaches
- Synergistic benefits of combined interventions
- Mechanistic insights into lifestyle-peptide interactions
Cardiovascular Outcome Research
The ultimate significance of retatrutide cholesterol effects lies in potential cardiovascular implications:
Surrogate Endpoint Studies:
- Carotid intima-media thickness (CIMT) changes
- Coronary artery calcium (CAC) score progression
- Flow-mediated dilation (FMD) improvements
- Arterial stiffness measurements
Biomarker Investigations:
- High-sensitivity C-reactive protein (hs-CRP)
- Lipoprotein-associated phospholipase A2 (Lp-PLA2)
- Myeloperoxidase (MPO)
- Oxidized LDL (ox-LDL)
Mechanistic Cardiovascular Studies:
- Endothelial function assessment
- Platelet aggregation effects
- Vascular inflammation markers
- Plaque composition analysis (in appropriate models)
These investigations require rigorous methodology and appropriate controls, with research-grade materials ensuring experimental validity.
Future Directions in Retatrutide Cholesterol Research
As our understanding of retatrutide cholesterol effects continues to evolve, several promising research directions emerge for 2026 and beyond.
Long-Term Cholesterol Trajectory Studies
Current data primarily covers 48-week observation periods, leaving important questions about longer-term cholesterol effects:
Multi-Year Investigations:
- Do cholesterol improvements persist beyond 2 years?
- Does tachyphylaxis (tolerance) develop to lipid effects?
- What happens to cholesterol levels after retatrutide discontinuation?
- Can intermittent dosing maintain cholesterol benefits?
Longitudinal Cohort Studies: Following research subjects over extended periods could reveal:
- Durability of HDL increases
- Stability of triglyceride reductions
- Long-term safety of sustained cholesterol modulation
- Cardiovascular event rates (in appropriate clinical contexts)
Mechanistic Deep-Dive Investigations
Despite substantial progress, many mechanistic questions about retatrutide cholesterol effects remain unanswered:
Receptor-Specific Contributions:
- Which of the three receptors (GIP, GLP-1, glucagon) contributes most to cholesterol effects?
- Can selective receptor antagonists dissect individual pathway contributions?
- Do receptor-specific effects vary by tissue type?
Genetic Modifiers:
- Do genetic variants in lipid metabolism genes influence retatrutide response?
- Can pharmacogenomic approaches predict cholesterol response magnitude?
- Which genetic profiles show optimal vs. suboptimal responses?
Epigenetic Investigations:
- Does retatrutide produce lasting epigenetic changes in lipid metabolism genes?
- Can DNA methylation patterns predict treatment response?
- Do histone modifications contribute to sustained cholesterol effects?
Novel Combination Strategies
Exploring synergistic combinations could maximize cholesterol-lowering potential:
Triple Therapy Approaches: Retatrutide + Statin + Ezetimibe combinations addressing:
- Cholesterol synthesis (statin)
- Cholesterol absorption (ezetimibe)
- Cholesterol metabolism and clearance (retatrutide)
Peptide Combination Research: Investigating retatrutide with other metabolic peptides:
- BPC-157 for cardiovascular protection
- TB-500 for tissue repair mechanisms
- Other metabolic modulators for comprehensive intervention
Personalized Medicine Applications
Advancing toward individualized approaches based on specific cholesterol profiles:
Phenotype-Specific Protocols:
- High LDL/normal triglyceride phenotype
- High triglyceride/low HDL phenotype
- Mixed dyslipidemia phenotype
- Isolated low HDL phenotype
Response Prediction Models: Developing algorithms that predict retatrutide cholesterol response based on:
- Baseline lipid parameters
- Metabolic markers (insulin, glucose, HbA1c)
- Anthropometric measurements
- Genetic risk scores
- Lifestyle factors
Accessing Research-Grade Retatrutide for Cholesterol Studies
For researchers interested in investigating retatrutide cholesterol effects, obtaining high-quality research materials is essential for experimental validity and reproducibility.
Quality Considerations for Research Peptides
When sourcing retatrutide for cholesterol research, several quality parameters ensure reliable results:
Purity Standards:
- Minimum 98% purity for metabolic research applications
- HPLC verification of peptide identity
- Mass spectrometry confirmation of molecular weight
- Certificates of Analysis (COA) documenting quality metrics
Storage and Handling:
- Lyophilized format for stability
- Appropriate temperature control during shipping
- Clear reconstitution guidelines
- Stability data for research planning
Regulatory Compliance:
- Clearly labeled “For Research Use Only”
- Appropriate documentation for institutional oversight
- Compliance with relevant research regulations
PEPTIDE PRO provides research-grade retatrutide meeting these stringent quality standards, supporting investigators conducting cholesterol and metabolic research across the UK and internationally.
Reconstitution and Preparation Protocols
Proper reconstitution ensures optimal peptide stability and experimental consistency:
Recommended Reconstitution:
- Use bacteriostatic water or sterile water for injection
- Add solvent slowly along pen peptide wall to minimize foaming
- Gentle swirling (not shaking) to dissolve
- Visual inspection for complete dissolution
- Appropriate aliquoting for experimental protocols
Storage After Reconstitution:
- Refrigeration at 2-8°C for short-term use (up to 14 days)
- Freezing at -20°C for longer storage (up to 3 months)
- Single-use aliquots to minimize freeze-thaw cycles
- Protection from light to prevent degradation
Supporting Research Infrastructure
Beyond peptide acquisition, successful cholesterol research requires comprehensive support:
Technical Resources:
- Reconstitution protocols and guidelines
- Storage recommendations for stability
- Dosing calculations for experimental models
- Troubleshooting assistance for research applications
Customer Support:
- Responsive communication for research inquiries
- Guidance on appropriate product selection
- Documentation support for institutional requirements
- Fast delivery to minimize experimental delays
Research institutions can explore available research peptides and access comprehensive support for cholesterol investigation protocols.
Interpreting Retatrutide Cholesterol Research Data
As the body of evidence regarding retatrutide cholesterol effects continues to expand, developing frameworks for data interpretation ensures accurate scientific conclusions.
Statistical Considerations
Proper statistical analysis of cholesterol data requires attention to several methodological factors:
Sample Size Calculations:
- LDL-C changes: Typically require 30-50 subjects per arm for 80% power
- HDL-C changes: May require larger samples due to greater variability
- Triglyceride changes: Often show larger effect sizes, requiring smaller samples
- Stratified analyses: Require additional sample size for subgroup comparisons
Appropriate Statistical Tests:
- Paired t-tests for within-subject changes
- ANCOVA for between-group comparisons adjusting for baseline
- Repeated measures analysis for longitudinal trajectories
- Non-parametric alternatives for non-normally distributed lipid parameters (especially triglycerides)
Multiple Comparison Adjustments: When examining multiple lipid parameters, appropriate corrections prevent false-positive findings:
- Bonferroni correction for family-wise error rate
- False discovery rate (FDR) methods for exploratory analyses
- Pre-specified primary and secondary endpoints to minimize multiplicity issues
Clinical vs. Statistical Significance
Distinguishing between statistically significant and clinically meaningful cholesterol changes:
LDL Cholesterol:
- Statistical significance: Often achieved with 5-10 mg/dL reduction
- Clinical significance: Generally considered meaningful at ≥15-20 mg/dL reduction
- Retatrutide effects (20-30 mg/dL) typically exceed both thresholds
HDL Cholesterol:
- Statistical significance: May occur with 2-3 mg/dL increase
- Clinical significance: Debated, but generally ≥5 mg/dL considered meaningful
- Retatrutide effects (5-8 mg/dL) approach clinical significance threshold
Triglycerides:
- Statistical significance: Often achieved with 15-20 mg/dL reduction
- Clinical significance: ≥30-50 mg/dL reduction generally considered meaningful
- Retatrutide effects (40-60 mg/dL) consistently achieve clinical significance
Contextualizing Results Within Existing Literature
Interpreting retatrutide cholesterol effects requires situating findings within the broader research landscape:
Comparison to Benchmark Interventions:
- How do effects compare to established cholesterol treatments?
- What is the magnitude relative to lifestyle interventions?
- How do results align with other incretin-based peptides?
Mechanistic Consistency:
- Do observed effects align with known receptor biology?
- Are results consistent across different research populations?
- Do mechanistic studies support observed clinical effects?
Reproducibility Across Studies:
- Have multiple independent investigations shown similar effects?
- What accounts for any heterogeneity in results?
- Which findings represent robust, reproducible phenomena?
Regulatory and Ethical Considerations for Cholesterol Research
Conducting responsible research on retatrutide cholesterol effects requires adherence to established regulatory frameworks and ethical principles.
Research Use Only Designation
It is critical to emphasize that retatrutide available through research suppliers is designated strictly for laboratory investigation:
🔬 Research Use Only: Retatrutide and other research peptides are intended exclusively for in vitro research, laboratory studies, and scientific investigation. They are not approved for human consumption, clinical use, or therapeutic application.
This designation ensures:
- Appropriate use within controlled research settings
- Compliance with regulatory requirements
- Protection of research integrity
- Clear boundaries between research and clinical applications
Institutional Oversight Requirements
Research involving retatrutide cholesterol effects typically requires appropriate institutional review:
For In Vitro Studies:
- Laboratory safety protocols
- Proper chemical handling procedures
- Waste disposal compliance
- Documentation of research materials
For Animal Model Studies:
- Institutional Animal Care and Use Committee (IACUC) approval
- Adherence to animal welfare guidelines
- Appropriate veterinary oversight
- Humane endpoint criteria
For Human Studies (Clinical Research Context):
- Institutional Review Board (IRB) approval
- Informed consent procedures
- Adverse event monitoring and reporting
- Data safety monitoring boards for larger trials
Transparency and Publication Ethics
Responsible dissemination of retatrutide cholesterol research findings:
Data Reporting Standards:
- Complete reporting of all measured outcomes
- Disclosure of negative and null findings
- Appropriate statistical reporting
- Raw data availability for verification
Conflict of Interest Disclosure:
- Funding source transparency
- Industry relationship disclosure
- Financial interest reporting
- Competing interest statements
Research Registration:
- Pre-registration of study protocols
- Clinical trial registration (where applicable)
- Protocol publication for transparency
- Results reporting regardless of outcome
Conclusion: The Evolving Understanding of Retatrutide Cholesterol Effects
The emerging research on retatrutide cholesterol effects represents a significant advancement in our understanding of metabolic peptide therapeutics. As a triple agonist targeting GIP, GLP-1, and glucagon receptors, retatrutide demonstrates remarkable capacity to modulate multiple aspects of lipid metabolism simultaneously—producing meaningful reductions in LDL cholesterol and triglycerides while increasing beneficial HDL cholesterol.
Key Research Insights
The evidence accumulated through 2026 reveals several important conclusions:
Magnitude of Effects: Retatrutide produces clinically meaningful cholesterol improvements, with LDL reductions of 18-24%, triglyceride reductions of 28-36%, and HDL increases of 10-15% in research populations.
Mechanistic Complexity: The cholesterol-lowering effects result from multi-organ, multi-pathway interventions affecting hepatic lipid production, intestinal cholesterol absorption, adipose tissue metabolism, and reverse cholesterol transport.
Dose-Response Relationship: Clear dose-dependent effects allow researchers to titrate interventions based on specific experimental requirements and research objectives.
Population Variability: Baseline cholesterol levels, metabolic status, and individual characteristics influence response magnitude, suggesting opportunities for personalized research approaches.
Safety Profile: Cholesterol modulation occurs within a generally favorable safety profile, with lipid changes representing beneficial rather than adverse effects in most research contexts.
Actionable Next Steps for Researchers
For investigators interested in exploring retatrutide cholesterol effects, several concrete steps facilitate research initiation:
- Define Research Questions: Identify specific cholesterol-related hypotheses that retatrutide’s unique mechanism can address
- Design Rigorous Protocols: Develop methodologically sound experimental designs with appropriate controls, sample sizes, and outcome measures
- Source Quality Materials: Obtain research-grade retatrutide from reputable suppliers committed to purity, consistency, and researcher support
- Establish Monitoring Systems: Implement comprehensive lipid assessment protocols including both standard and advanced cholesterol parameters
- Plan for Long-Term Investigation: Design studies with sufficient duration (≥24 weeks) to capture the full trajectory of cholesterol effects
- Consider Combination Approaches: Explore synergistic interventions combining retatrutide with other cholesterol-modulating strategies
- Contribute to Scientific Knowledge: Publish findings in peer-reviewed journals, present at scientific conferences, and share data with the research community
The Path Forward
As we progress through 2026 and beyond, retatrutide cholesterol research will likely expand in several directions: longer-term outcome studies, mechanistic deep-dive investigations, personalized medicine applications, and cardiovascular endpoint research. Each investigation contributes to our collective understanding of how multi-receptor agonism influences lipid metabolism and cardiovascular health.
For researchers ready to contribute to this evolving field, PEPTIDE PRO stands ready to support your investigations with high-purity research peptides, rapid UK delivery, and comprehensive technical support. Our commitment to research excellence ensures that your cholesterol studies utilize materials meeting the highest quality standards.
The investigation of retatrutide cholesterol effects represents more than academic curiosity—it embodies the potential to fundamentally advance our understanding of metabolic regulation and cardiovascular health. Through rigorous scientific inquiry, responsible research practices, and collaborative knowledge-sharing, the research community continues to unlock the therapeutic potential of this remarkable triple agonist peptide.
