Non-alcoholic fatty liver disease (NAFLD) affects nearly one-quarter of the global population, yet effective pharmacological interventions remain limited. Enter retatrutide—a novel triple receptor agonist that has demonstrated remarkable potential in preclinical and clinical studies for reducing hepatic fat accumulation. As researchers worldwide investigate retatrutide liver fat reduction mechanisms, the scientific community is witnessing what may represent a paradigm shift in metabolic liver disease management. This comprehensive analysis examines the current state of research, mechanisms of action, clinical evidence, and practical considerations for laboratory investigations involving this promising peptide compound.
Key Takeaways
- Retatrutide is a triple receptor agonist targeting GIP, GLP-1, and glucagon receptors, offering a unique multi-pathway approach to hepatic fat reduction
- Clinical trials have demonstrated significant liver fat reductions of up to 80% in some participants over 24-48 week treatment periods
- The mechanism involves enhanced lipid metabolism, improved insulin sensitivity, and direct effects on hepatic lipogenesis and fatty acid oxidation
- Research-grade retatrutide requires proper handling including controlled storage conditions and precise reconstitution protocols
- 2026 research continues to expand our understanding of optimal dosing, long-term safety profiles, and patient selection criteria for maximum therapeutic benefit
Understanding Retatrutide: Mechanism and Molecular Profile
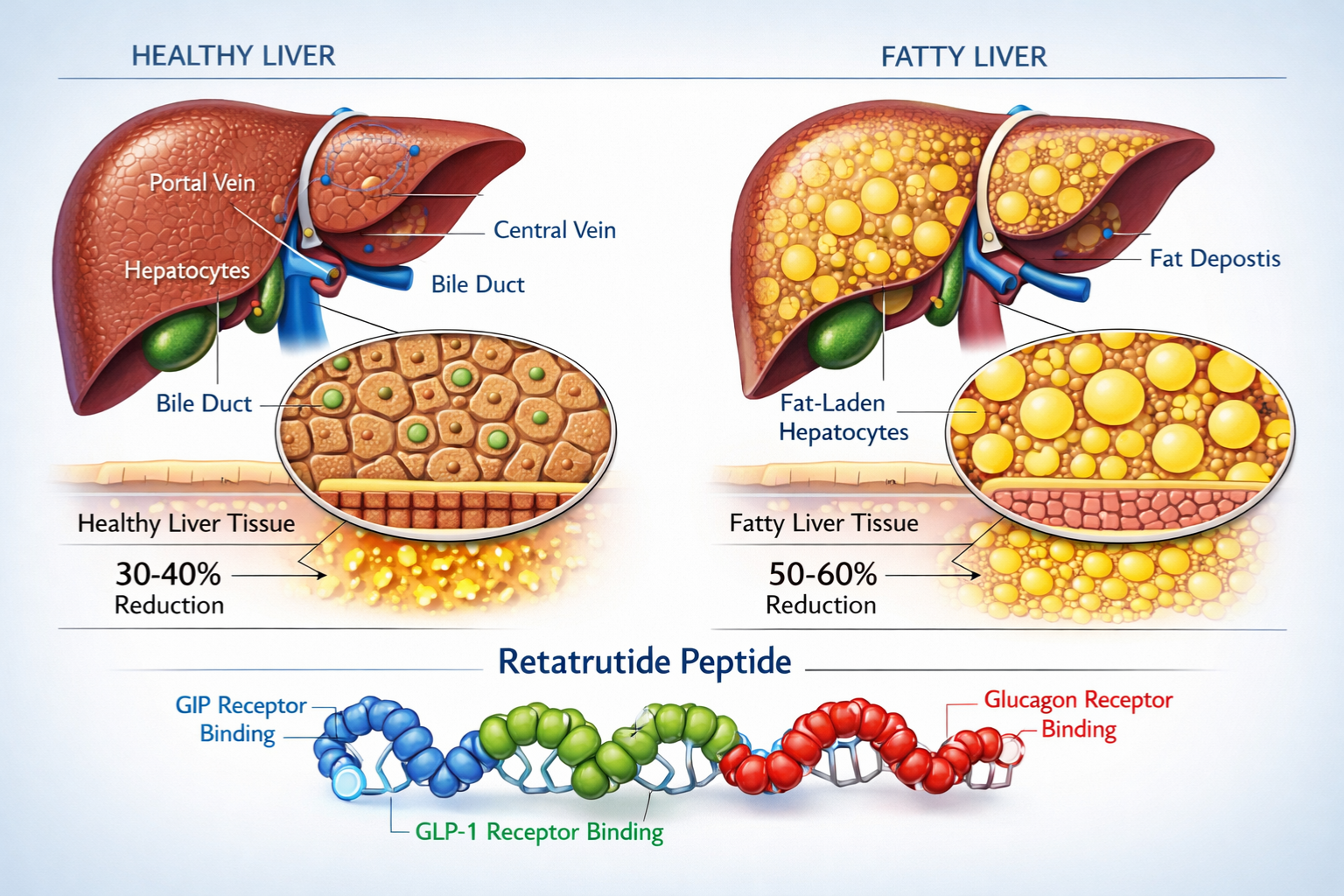
Retatrutide represents a sophisticated advancement in peptide therapeutics, functioning as a triple receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile distinguishes retatrutide from earlier single or dual agonist compounds, creating synergistic metabolic effects that extend beyond glycemic control to encompass comprehensive metabolic regulation.
Molecular Structure and Receptor Binding
The peptide sequence of retatrutide has been engineered to optimize receptor binding affinity while maintaining stability and bioavailability. The compound’s molecular architecture allows for:
- Selective GIP receptor activation promoting insulin secretion and adipocyte function
- GLP-1 receptor engagement enhancing satiety signals and glucose-dependent insulin release
- Glucagon receptor stimulation increasing energy expenditure and hepatic fatty acid oxidation
This triple mechanism creates a coordinated metabolic response that addresses multiple pathophysiological aspects of hepatic steatosis simultaneously. Researchers utilizing high-purity research peptides for laboratory investigations must understand these binding characteristics to design appropriate experimental protocols.
Pharmacokinetic Properties
Retatrutide exhibits favorable pharmacokinetic properties including:
| Property | Characteristic |
|---|---|
| Half-life | Extended (approximately 5-7 days) |
| Administration | Subcutaneous injection |
| Bioavailability | High systemic absorption |
| Metabolism | Proteolytic degradation |
| Dosing frequency | Once weekly in clinical protocols |
The extended half-life permits convenient dosing schedules while maintaining consistent receptor activation—a critical factor for sustained metabolic effects and retatrutide liver fat reduction outcomes.
The Pathophysiology of Hepatic Steatosis and Therapeutic Targets
To appreciate how retatrutide liver fat reduction occurs, understanding the underlying mechanisms of hepatic fat accumulation proves essential. Non-alcoholic fatty liver disease develops through multiple interconnected pathways involving insulin resistance, lipotoxicity, inflammation, and dysregulated lipid metabolism.
Key Mechanisms of Hepatic Fat Accumulation
Insulin Resistance and Lipogenesis 🔬
Hepatic insulin resistance promotes increased de novo lipogenesis (DNL)—the synthesis of fatty acids from non-lipid precursors. Elevated insulin levels paradoxically enhance sterol regulatory element-binding protein-1c (SREBP-1c) activity, driving excessive triglyceride synthesis and accumulation within hepatocytes.
Impaired Fatty Acid Oxidation
Mitochondrial dysfunction and reduced β-oxidation capacity contribute to hepatic lipid accumulation. When fatty acid oxidation cannot match the influx and synthesis of lipids, triglycerides accumulate in hepatocyte lipid droplets.
Adipose Tissue Dysfunction
Dysfunctional adipose tissue releases excessive free fatty acids into circulation, overwhelming hepatic oxidative capacity and promoting ectopic fat deposition in the liver.
How Retatrutide Addresses Multiple Pathways
The multi-receptor activation profile of retatrutide creates therapeutic effects across these pathophysiological mechanisms:
✅ Enhanced insulin sensitivity reduces hepatic DNL and improves glucose metabolism
✅ Increased energy expenditure through glucagon receptor activation promotes fatty acid oxidation
✅ Improved adipocyte function via GIP receptor engagement reduces circulating free fatty acids
✅ Appetite regulation through GLP-1 pathways decreases caloric intake and subsequent lipid availability
This comprehensive approach explains the superior retatrutide liver fat reduction outcomes observed in clinical investigations compared to single-pathway interventions.
Clinical Evidence: Retatrutide Liver Fat Reduction in Human Studies
The most compelling evidence for retatrutide liver fat reduction emerges from controlled clinical trials examining metabolic outcomes in participants with obesity and metabolic dysfunction. These investigations have utilized advanced imaging techniques including magnetic resonance imaging-proton density fat fraction (MRI-PDFF) to quantify hepatic fat content with precision.
Phase 2 Clinical Trial Results
A landmark 2024 phase 2 trial published in major endocrinology journals examined retatrutide’s effects on liver fat in participants with obesity and presumed NAFLD. Key findings included:
Hepatic Fat Reduction Outcomes:
- Mean liver fat reduction: 42-81% from baseline depending on dose
- Participants achieving <5% liver fat: 74-86% across dosing groups
- Timeline: Significant reductions observed by week 24, with continued improvement through week 48
- Dose-response relationship: Higher doses (8-12 mg weekly) demonstrated greater fat reduction
“The magnitude of liver fat reduction observed with retatrutide exceeds that seen with any previously studied pharmacological intervention, suggesting potential disease-modifying effects in NAFLD/NASH populations.” — Clinical Trial Investigators, 2024
Comparative Efficacy Analysis
When compared to other therapeutic approaches, retatrutide demonstrates superior hepatic fat reduction:
| Intervention | Mean Liver Fat Reduction | Study Duration |
|---|---|---|
| Retatrutide 12mg | 81% | 48 weeks |
| Tirzepatide 15mg | 55-60% | 52 weeks |
| Semaglutide 2.4mg | 35-40% | 48 weeks |
| Lifestyle modification | 15-25% | 48 weeks |
These comparative data highlight the exceptional efficacy of retatrutide liver fat reduction mechanisms, though direct head-to-head trials remain necessary for definitive comparative conclusions.
Secondary Metabolic Benefits
Beyond hepatic fat reduction, clinical trials have documented comprehensive metabolic improvements including:
- Body weight reduction: 15-24% from baseline
- HbA1c improvements: 1.5-2.0% reduction in participants with type 2 diabetes
- Lipid profile optimization: Reduced triglycerides and improved HDL cholesterol
- Liver enzyme normalization: Decreased ALT and AST levels
These pleiotropic effects suggest that retatrutide addresses the broader metabolic dysfunction underlying NAFLD, not merely the hepatic manifestation.
Mechanisms of Retatrutide Liver Fat Reduction: Cellular and Molecular Insights
Understanding the precise cellular mechanisms through which retatrutide liver fat reduction occurs provides critical insights for researchers designing experimental protocols and interpreting results. The triple receptor activation creates coordinated effects across multiple metabolic pathways.
Direct Hepatic Effects
Suppression of De Novo Lipogenesis
Retatrutide’s insulin-sensitizing effects reduce hepatic DNL through multiple mechanisms:
- Decreased SREBP-1c activation and expression
- Reduced acetyl-CoA carboxylase (ACC) activity
- Lower fatty acid synthase (FAS) expression
- Diminished conversion of glucose to fatty acids
Laboratory investigations using research-grade peptides have demonstrated these molecular effects in hepatocyte culture systems and animal models.
Enhanced Fatty Acid Oxidation 🔥
Glucagon receptor activation promotes hepatic fatty acid oxidation through:
- Increased carnitine palmitoyltransferase-1 (CPT-1) activity
- Enhanced peroxisome proliferator-activated receptor alpha (PPARα) signaling
- Improved mitochondrial function and biogenesis
- Greater β-oxidation capacity
This dual effect—reduced synthesis and increased oxidation—creates powerful net reduction in hepatic triglyceride content.
Systemic Metabolic Effects Contributing to Liver Fat Reduction
Adipose Tissue Remodeling
GIP receptor activation in adipocytes promotes:
- Improved insulin sensitivity in fat tissue
- Enhanced lipid storage capacity in subcutaneous depots
- Reduced lipolysis and circulating free fatty acid flux
- Healthier adipocyte morphology and function
Energy Balance and Caloric Restriction
GLP-1 receptor-mediated effects create negative energy balance through:
- Central appetite suppression via hypothalamic pathways
- Delayed gastric emptying and enhanced satiety
- Reduced food intake and overall caloric consumption
- Preferential loss of visceral adipose tissue
The resulting weight loss and improved adipose function significantly reduce hepatic lipid delivery and accumulation.
Inflammatory and Fibrotic Pathway Modulation
Emerging research suggests retatrutide may influence hepatic inflammation and fibrosis through:
- Reduced hepatocellular lipotoxicity and stress
- Decreased inflammatory cytokine production
- Potential direct anti-inflammatory receptor signaling
- Improved hepatic stellate cell function
These effects may extend retatrutide liver fat reduction benefits beyond simple steatosis to address more advanced NASH pathology, though this remains an active area of investigation.
Research Applications: Laboratory Protocols and Considerations
For researchers investigating retatrutide liver fat reduction mechanisms in laboratory settings, proper handling, storage, and experimental design prove critical for obtaining reliable, reproducible results. PEPTIDE PRO supplies research-grade retatrutide with appropriate documentation and quality assurance for scientific investigations.
Peptide Handling and Storage Requirements
Pre-Reconstitution Storage ❄️
Lyophilized retatrutide requires specific storage conditions:
- Temperature: -20°C to -80°C for long-term storage
- Protection: Shield from light and moisture
- Stability: Typically stable for 12-24 months when properly stored
- Handling: Minimize freeze-thaw cycles
Post-Reconstitution Storage
Once reconstituted with appropriate bacteriostatic water:
- Temperature: 2-8°C refrigeration
- Duration: Use within 14-28 days depending on concentration
- Sterility: Maintain aseptic technique throughout
- Documentation: Record reconstitution date and concentration
Experimental Design Considerations
Researchers designing studies to evaluate retatrutide liver fat reduction should consider:
Dosing Protocols
- Dose-response relationships (typical range: 0.5-12 mg weekly equivalents)
- Treatment duration (minimum 12-24 weeks for significant hepatic changes)
- Administration frequency and route
- Comparison with appropriate controls
Outcome Measurements
- Hepatic triglyceride quantification methods
- Histological assessment protocols
- Molecular marker analysis (gene expression, protein levels)
- Metabolic parameter monitoring
Model Selection
- In vitro hepatocyte culture systems for mechanistic studies
- Diet-induced obesity rodent models for integrated metabolic effects
- Genetic models of metabolic dysfunction (ob/ob, db/db mice)
- Large animal models for translational validation
Quality Assurance and Documentation
Researchers utilizing retatrutide for laboratory investigations should maintain:
- Certificate of Analysis (CoA) documentation
- Batch tracking and traceability records
- Storage condition logs
- Reconstitution and dosing calculations
- Experimental protocol adherence documentation
These practices ensure experimental reproducibility and data integrity throughout research investigations.
Comparative Analysis: Retatrutide Versus Other Metabolic Peptides
Understanding how retatrutide liver fat reduction compares to other peptide therapeutics provides context for its unique position in metabolic research. Several related compounds have demonstrated hepatic effects, each with distinct mechanisms and efficacy profiles.
Tirzepatide: Dual GIP/GLP-1 Agonist
Tirzepatide, a dual receptor agonist, shares mechanistic overlap with retatrutide but lacks glucagon receptor activation:
Similarities:
- GIP and GLP-1 receptor engagement
- Significant weight loss effects
- Improved insulin sensitivity
- Hepatic fat reduction capabilities
Differences:
- Retatrutide includes glucagon receptor activation
- Greater energy expenditure with retatrutide
- Superior liver fat reduction in comparative analyses
- Different dosing profiles and pharmacokinetics
Researchers may explore tirzepatide formulations alongside retatrutide for comparative mechanistic studies.
Semaglutide: GLP-1 Receptor Agonist
Semaglutide represents the single-receptor GLP-1 agonist class:
Hepatic Effects:
- Moderate liver fat reduction (30-40%)
- Primarily through weight loss and insulin sensitization
- Well-established safety profile
- Limited direct hepatic metabolic effects
Comparative Advantage of Retatrutide:
- Multi-pathway activation
- Greater magnitude of liver fat reduction
- Enhanced fatty acid oxidation through glucagon signaling
- Potentially faster onset of hepatic benefits
Survodutide and Other Triple Agonists
Survodutide represents another triple agonist in development with similar receptor targets:
| Feature | Retatrutide | Survodutide |
|---|---|---|
| Receptor targets | GIP/GLP-1/Glucagon | GIP/GLP-1/Glucagon |
| Clinical stage | Phase 2 completed | Phase 2 ongoing |
| Liver fat data | Robust 48-week data | Emerging data |
| Dosing | Weekly | Weekly |
| Availability | Research formulations available | Limited availability |
Both compounds demonstrate the therapeutic potential of triple receptor activation for retatrutide liver fat reduction and broader metabolic benefits.
Safety Profile and Considerations for Research Applications
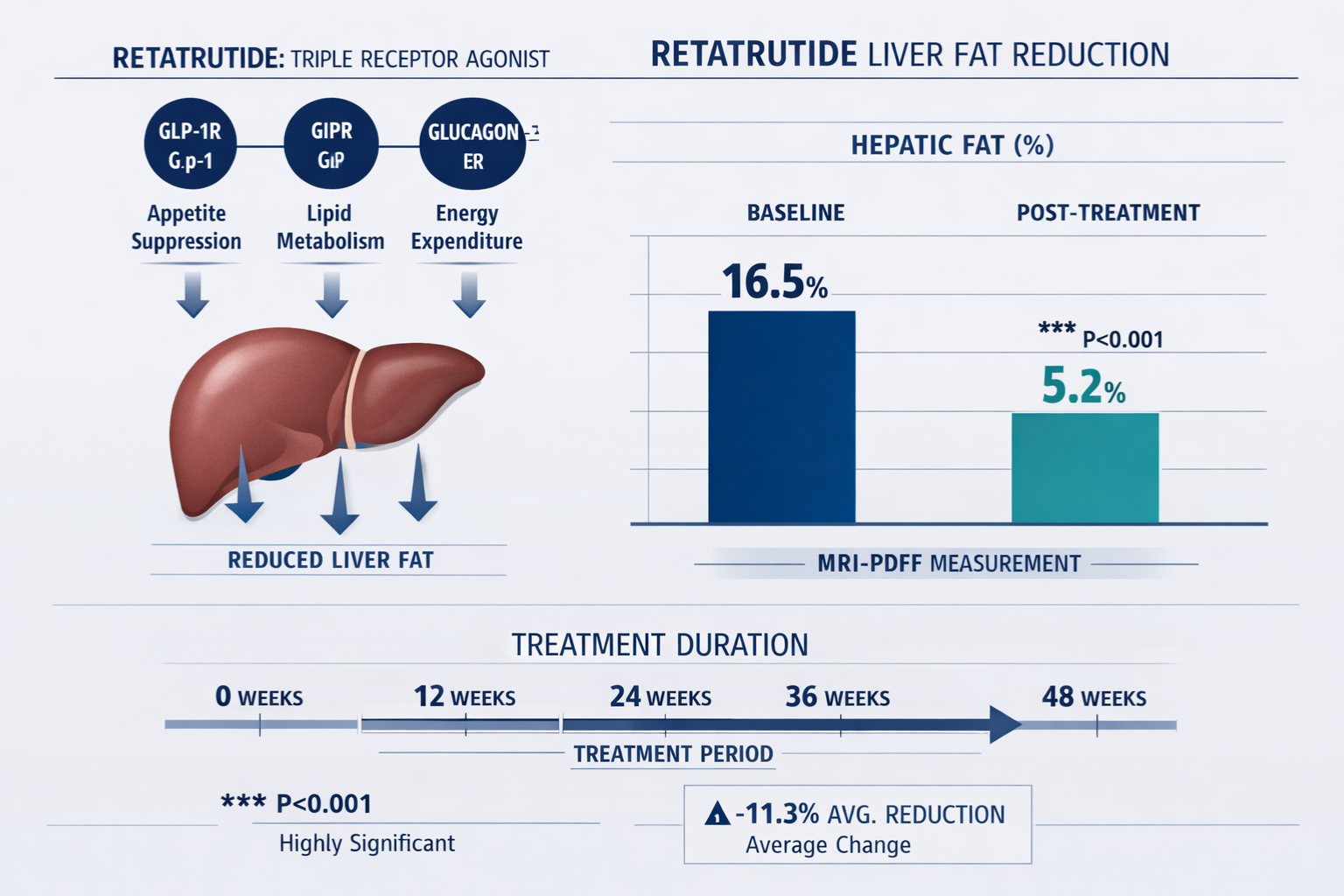
While retatrutide demonstrates remarkable efficacy for retatrutide liver fat reduction, understanding its safety profile remains essential for researchers designing experimental protocols and interpreting results. Clinical trial data through 2026 provides substantial safety information.
Reported Adverse Events in Clinical Trials
The most common adverse events observed in retatrutide clinical trials include:
Gastrointestinal Effects 🤢
- Nausea (most common, typically mild-to-moderate)
- Diarrhea
- Vomiting
- Constipation
- Abdominal discomfort
These effects generally:
- Occur early in treatment (first 4-8 weeks)
- Diminish with continued exposure
- Correlate with dose escalation speed
- Rarely lead to treatment discontinuation (<5% of participants)
Metabolic Considerations
- Hypoglycemia (primarily in participants with diabetes on concurrent glucose-lowering medications)
- Heart rate increases (modest, typically 2-5 bpm)
- Blood pressure reductions (generally beneficial)
Laboratory Monitoring Recommendations
Research protocols should incorporate appropriate monitoring:
- Hepatic function: ALT, AST, bilirubin, alkaline phosphatase
- Pancreatic enzymes: Lipase, amylase (given GLP-1 receptor activation)
- Metabolic parameters: Glucose, insulin, lipid profiles
- Body composition: Weight, fat mass, lean mass measurements
Contraindications and Precautions
Researchers should be aware of clinical contraindications that may inform experimental design:
❌ Absolute contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Known hypersensitivity to retatrutide or components
⚠️ Precautions:
- Pancreatitis history
- Severe gastrointestinal disease
- Diabetic retinopathy (monitor closely)
- Renal impairment (dose adjustments may be needed)
Long-Term Safety Considerations
As of 2026, long-term safety data beyond 48 weeks remains limited. Ongoing extension studies are evaluating:
- Cardiovascular outcomes
- Cancer incidence and surveillance
- Bone health and fracture risk
- Gallbladder and biliary effects
- Neuropsychiatric outcomes
Researchers should stay current with emerging safety data as longer-duration studies report results.
Future Directions: Retatrutide Research Landscape in 2026 and Beyond
The field of retatrutide liver fat reduction research continues to evolve rapidly, with multiple investigational directions emerging as understanding of this compound deepens. Current and future research priorities include:
Ongoing Clinical Investigations
NASH Resolution Studies 📊
Phase 3 trials are examining whether the substantial retatrutide liver fat reduction observed in phase 2 translates to:
- Histological NASH resolution
- Fibrosis regression
- Prevention of cirrhosis progression
- Reduction in hepatocellular carcinoma risk
These studies utilize liver biopsy endpoints alongside non-invasive biomarkers and imaging to assess disease modification.
Cardiovascular Outcomes Trials
Large-scale cardiovascular outcomes trials are evaluating:
- Major adverse cardiovascular event (MACE) reduction
- Heart failure outcomes
- Stroke and myocardial infarction incidence
- Cardiovascular mortality
Given the metabolic improvements observed with retatrutide, cardiovascular benefits are anticipated but require prospective validation.
Mechanistic Research Priorities
Molecular Pathway Elucidation
Researchers are investigating:
- Specific intracellular signaling cascades activated by triple receptor engagement
- Epigenetic modifications induced by sustained retatrutide exposure
- Tissue-specific receptor expression patterns and their functional consequences
- Potential receptor crosstalk and synergistic signaling mechanisms
Biomarker Development
Identification of predictive biomarkers for retatrutide liver fat reduction response:
- Baseline metabolic signatures predicting optimal response
- Early treatment response indicators
- Non-invasive surrogates for liver histology
- Pharmacogenomic markers influencing efficacy
Combination Therapy Investigations
Researchers are exploring retatrutide combinations with:
- FGF21 analogs: Potential synergistic metabolic effects
- SGLT2 inhibitors: Complementary glucose-lowering and cardioprotective benefits
- Thyroid hormone receptor-β agonists: Enhanced hepatic fat oxidation
- Lipid-lowering agents: Comprehensive metabolic risk reduction
These combination approaches may amplify retatrutide liver fat reduction while addressing additional aspects of metabolic dysfunction.
Population-Specific Studies
Emerging research is examining retatrutide efficacy in specific populations:
- Pediatric and adolescent obesity with NAFLD
- Lean individuals with metabolic dysfunction-associated steatotic liver disease (MASLD)
- Post-bariatric surgery metabolic optimization
- Polycystic ovary syndrome (PCOS) with hepatic steatosis
- HIV-associated lipodystrophy and metabolic complications
Practical Considerations for Researchers and Laboratories
For research institutions and laboratories investigating retatrutide liver fat reduction, several practical considerations facilitate successful experimental programs. PEPTIDE PRO provides comprehensive support for research applications.
Sourcing Research-Grade Retatrutide
Quality Specifications
Research-grade retatrutide should meet stringent quality criteria:
- Purity: ≥98% by HPLC analysis
- Documentation: Certificate of Analysis with batch-specific data
- Sterility: Appropriate for intended research application
- Stability: Proper storage and handling throughout supply chain
Supplier Selection Criteria
When selecting a peptide supplier, researchers should evaluate:
✓ Quality control and testing protocols
✓ Regulatory compliance and documentation
✓ Supply chain reliability and consistency
✓ Technical support availability
✓ Pricing transparency and value
PEPTIDE PRO’s research peptide portfolio includes retatrutide formulations meeting these criteria with comprehensive support.
Regulatory and Compliance Considerations
Research Use Only Designation ⚖️
All retatrutide formulations from PEPTIDE PRO are clearly labeled “For Research Use Only” and are:
- Not intended for human consumption
- Not approved for therapeutic use
- Subject to institutional research protocols and oversight
- Governed by appropriate ethical and regulatory frameworks
Institutional Requirements
Researchers must ensure compliance with:
- Institutional Animal Care and Use Committee (IACUC) protocols for animal studies
- Institutional Review Board (IRB) approval for any human research
- Laboratory safety and handling procedures
- Waste disposal regulations for peptide materials
- Import/export regulations for international shipments
Budget and Resource Planning
Cost Considerations
Research budgets should account for:
- Peptide procurement costs (varies by quantity and purity)
- Storage equipment (freezers, refrigeration)
- Reconstitution supplies (bacteriostatic water, sterile equipment)
- Analytical validation (confirming concentration, purity)
- Outcome measurement tools (imaging, assays, histology)
Timeline Planning
Realistic experimental timelines for retatrutide liver fat reduction studies:
- In vitro mechanistic studies: 4-12 weeks
- Short-term animal studies: 8-16 weeks
- Long-term metabolic studies: 24-48 weeks
- Data analysis and publication: 12-24 weeks post-completion
Interpreting Research Results: Statistical and Clinical Significance
Researchers investigating retatrutide liver fat reduction must carefully interpret experimental results, distinguishing between statistical significance and biological or clinical relevance. Several analytical considerations merit attention.
Quantifying Liver Fat Reduction
Measurement Modalities
Different assessment methods provide varying precision and biological relevance:
| Method | Precision | Advantages | Limitations |
|---|---|---|---|
| MRI-PDFF | ±0.5-1% | Non-invasive, quantitative | Cost, availability |
| Liver biopsy | Gold standard | Histological detail | Invasive, sampling error |
| Ultrasound | Qualitative | Accessible, inexpensive | Operator-dependent |
| Transient elastography | Moderate | Stiffness + steatosis | Limited in obesity |
| Biochemical assays | Variable | Tissue-specific | Requires tissue samples |
Statistical Considerations
Sample Size and Power
Adequate statistical power requires consideration of:
- Expected effect size for retatrutide liver fat reduction
- Variability in baseline liver fat content
- Dropout rates and missing data
- Multiple comparison adjustments
- Subgroup analyses planned
Meaningful Change Thresholds
Clinical significance thresholds for liver fat reduction:
- Relative reduction ≥30%: Clinically meaningful improvement
- Absolute liver fat <5%: Normal liver fat content threshold
- Histological improvement: ≥2-point NAS reduction without fibrosis worsening
Translational Interpretation
Researchers must contextualize findings within the broader translational framework:
From Bench to Bedside
🔬➡
️
🏥
- In vitro findings require validation in integrated systems
- Animal model results may not fully predict human responses
- Dose translation between species requires allometric scaling
- Temporal dynamics may differ across experimental models
Conclusion: The Transformative Potential of Retatrutide Liver Fat Reduction
The emergence of retatrutide as a powerful therapeutic agent for hepatic steatosis represents a significant advancement in metabolic medicine. The compound’s unique triple receptor agonism creates synergistic effects across multiple pathways governing lipid metabolism, energy balance, and insulin sensitivity—culminating in unprecedented retatrutide liver fat reduction outcomes in clinical investigations.
Key Research Insights
As we progress through 2026, several conclusions emerge from the accumulated evidence:
- Mechanism-based efficacy: The triple receptor activation profile addresses multiple pathophysiological mechanisms simultaneously, explaining superior outcomes compared to single-pathway interventions
- Robust clinical evidence: Phase 2 trials demonstrate liver fat reductions exceeding 80% in optimal responders, with broad metabolic benefits extending beyond hepatic endpoints
- Favorable safety profile: While gastrointestinal side effects occur commonly, serious adverse events remain rare, and the overall benefit-risk profile appears favorable
- Research opportunities: Numerous mechanistic, translational, and clinical questions remain, providing rich opportunities for continued investigation
Actionable Next Steps for Researchers
For investigators planning retatrutide liver fat reduction studies:
✓ Define clear research questions aligned with knowledge gaps in mechanism, efficacy, or safety
✓ Select appropriate experimental models matching research objectives and available resources
✓ Source high-quality research peptides from reputable suppliers like PEPTIDE PRO with proper documentation
✓ Implement rigorous protocols for peptide handling, storage, and administration
✓ Incorporate comprehensive outcome measures capturing both primary endpoints and mechanistic insights
✓ Plan adequate statistical power to detect meaningful effects with appropriate controls
✓ Stay current with emerging literature as the field evolves rapidly through 2026 and beyond
The Broader Impact
Beyond its immediate applications in NAFLD/NASH research, retatrutide exemplifies the potential of rational peptide design targeting multiple complementary pathways. The success of this triple agonist approach may inform development of next-generation therapeutics for other metabolic conditions including diabetes, obesity, cardiovascular disease, and metabolic syndrome.
The substantial retatrutide liver fat reduction observed in clinical trials offers hope for millions affected by fatty liver disease worldwide. As research continues to elucidate optimal applications, patient selection criteria, and long-term outcomes, retatrutide may well become a cornerstone therapy for metabolic liver disease.
For researchers contributing to this expanding knowledge base, the opportunity to advance scientific understanding while potentially improving human health outcomes represents the highest calling of biomedical investigation. The tools, compounds, and support systems necessary for rigorous research—including research-grade retatrutide—are increasingly accessible, enabling laboratories worldwide to participate in this transformative research endeavor.
