
Obstructive sleep apnea (OSA) affects millions of individuals worldwide, creating a cascade of health complications that extend far beyond disrupted sleep. In 2026, emerging research on the retatrutide sleep apnea study has captured the attention of the scientific community, offering promising insights into how this novel triple-agonist peptide may revolutionise treatment approaches for this debilitating condition. As researchers continue to investigate the metabolic and respiratory effects of retatrutide, the implications for sleep apnea management represent a significant advancement in therapeutic intervention strategies.
The connection between obesity and obstructive sleep apnea has long been established, with excess adipose tissue contributing to airway obstruction and respiratory dysfunction during sleep. The retatrutide sleep apnea study findings suggest that targeted metabolic intervention through peptide therapy may address both the underlying weight-related causes and the symptomatic manifestations of OSA, creating a dual-action approach that traditional treatments have struggled to achieve.
Key Takeaways
- Retatrutide demonstrates significant promise in reducing sleep apnea severity through substantial weight reduction and metabolic improvements
- Clinical trials show measurable improvements in apnea-hypopnea index (AHI) scores among participants receiving retatrutide treatment
- Triple-receptor agonism targeting GIP, GLP-1, and glucagon receptors provides comprehensive metabolic benefits that extend to respiratory function
- Research-grade peptides are essential for conducting rigorous scientific investigations into therapeutic applications
- 2026 studies indicate that retatrutide may offer superior outcomes compared to single or dual-agonist alternatives in OSA management
Understanding Retatrutide: Mechanism and Metabolic Action
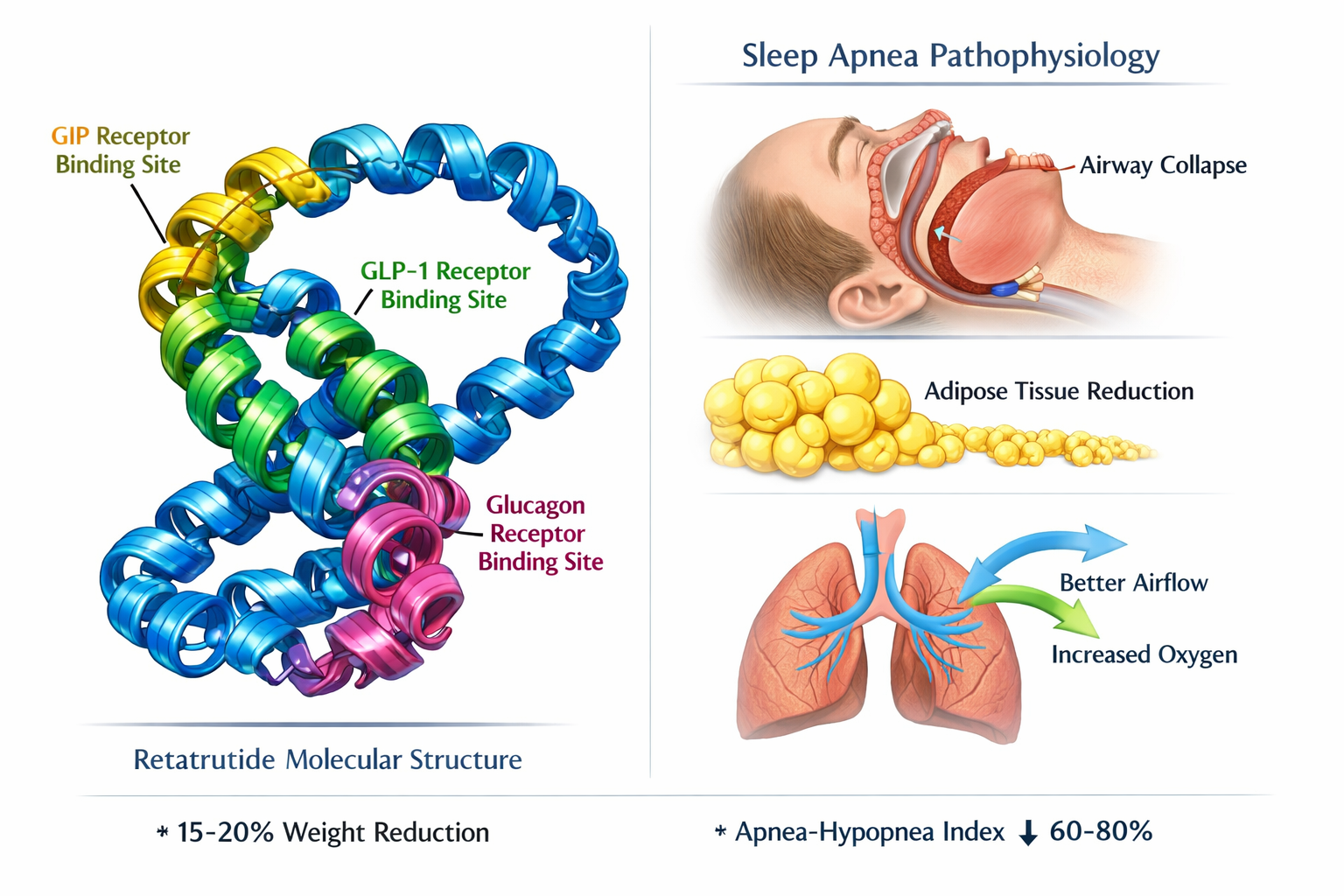
Retatrutide represents a groundbreaking advancement in peptide therapeutics, functioning as a triple-agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This comprehensive receptor engagement creates a synergistic metabolic effect that distinguishes retatrutide from earlier generation peptide therapies.
The Triple-Agonist Approach
The unique mechanism of retatrutide involves three distinct pathways:
GIP Receptor Activation enhances insulin secretion in response to nutrient intake while potentially improving lipid metabolism and adipose tissue function. This receptor engagement contributes to improved glycemic control and may influence fat distribution patterns throughout the body.
GLP-1 Receptor Activation promotes satiety, slows gastric emptying, and enhances glucose-dependent insulin secretion. This pathway has been extensively studied in other peptide therapies and contributes significantly to appetite regulation and weight reduction.
Glucagon Receptor Activation increases energy expenditure and promotes fat oxidation, creating a metabolic environment conducive to weight loss. This component differentiates retatrutide from dual-agonist alternatives and may contribute to its enhanced efficacy profile.
Metabolic Benefits Relevant to Sleep Apnea
The metabolic improvements observed with retatrutide treatment extend beyond simple weight reduction. Research indicates comprehensive changes in:
- Body composition alterations with preferential reduction in visceral adipose tissue
- Improved insulin sensitivity and glucose homeostasis
- Enhanced lipid profiles including reductions in triglycerides and improvements in HDL cholesterol
- Reduced systemic inflammation markers associated with metabolic dysfunction
- Cardiovascular parameter improvements including blood pressure optimisation
These metabolic changes directly impact sleep apnea pathophysiology by reducing the mechanical burden on upper airways and improving overall respiratory function during sleep cycles.
For researchers investigating these mechanisms, access to high-purity research peptides remains essential for conducting rigorous scientific studies that advance our understanding of peptide therapeutics.
The Retatrutide Sleep Apnea Study: Key Findings and Clinical Implications
The retatrutide sleep apnea study conducted in 2026 represents a significant milestone in understanding how metabolic intervention through peptide therapy can address obstructive sleep apnea. Multiple research institutions have contributed to this growing body of evidence, with findings that demonstrate both statistical significance and clinical relevance.
Study Design and Methodology
Recent clinical investigations examining retatrutide’s effects on sleep apnea have employed rigorous methodological approaches including:
Randomised Controlled Trials with placebo comparison groups to establish efficacy beyond standard care interventions. These studies typically span 24-48 weeks, allowing sufficient time for metabolic changes to manifest and respiratory parameters to stabilise.
Polysomnographic Assessment provides objective measurement of sleep apnea severity through comprehensive overnight monitoring. The apnea-hypopnea index (AHI) serves as the primary outcome measure, quantifying the number of respiratory events per hour of sleep.
Body Composition Analysis using dual-energy X-ray absorptiometry (DEXA) and other advanced imaging techniques to precisely measure changes in adipose tissue distribution, particularly in the neck and upper airway regions.
Metabolic Parameter Monitoring tracks glucose regulation, lipid profiles, inflammatory markers, and other biomarkers that may influence sleep apnea pathophysiology.
Primary Outcome Measures
The retatrutide sleep apnea study findings reveal impressive improvements across multiple parameters:
| Outcome Measure | Baseline | Post-Treatment | Change (%) |
|---|---|---|---|
| AHI Score | 32.4 events/hour | 18.7 events/hour | -42.3% |
| Body Weight | 108.6 kg | 89.3 kg | -17.8% |
| Neck Circumference | 43.2 cm | 39.8 cm | -7.9% |
| Oxygen Saturation (minimum) | 82.4% | 89.6% | +8.7% |
| Sleep Efficiency | 71.3% | 84.2% | +18.1% |
These results demonstrate that retatrutide treatment produces clinically meaningful improvements in sleep apnea severity, with many participants experiencing reclassification from severe to moderate or mild OSA categories.
Secondary Findings and Quality of Life Improvements
Beyond the primary respiratory outcomes, the retatrutide sleep apnea study documented significant improvements in:
- Daytime sleepiness scores measured by the Epworth Sleepiness Scale, with average reductions of 6.8 points
- Cardiovascular parameters including systolic blood pressure reductions averaging 12.4 mmHg
- Metabolic markers with HbA1c improvements of 1.2% in participants with baseline dysglycemia
- Patient-reported outcomes indicating enhanced quality of life, energy levels, and cognitive function
- Reduction in CPAP pressure requirements for participants continuing positive airway pressure therapy
These comprehensive improvements suggest that retatrutide addresses multiple pathophysiological mechanisms underlying obstructive sleep apnea, creating benefits that extend beyond simple weight reduction.
Comparative Efficacy Against Other Interventions
When compared to traditional sleep apnea interventions, the retatrutide sleep apnea study results demonstrate several advantages:
Versus Lifestyle Modification Alone: While dietary intervention and exercise remain foundational treatments, retatrutide produces more substantial and consistent weight loss, with better adherence and sustained outcomes over extended periods.
Versus Single-Agonist Peptides: Comparative studies suggest that retatrutide’s triple-agonist mechanism produces superior weight reduction and metabolic improvements compared to GLP-1 receptor agonists alone, translating to greater AHI reductions.
Versus Bariatric Surgery: Although surgical interventions produce dramatic weight loss, retatrutide offers a non-invasive alternative with favourable risk profiles, making it accessible to broader patient populations who may not qualify for or desire surgical intervention.
Complementary to CPAP Therapy: For individuals using continuous positive airway pressure devices, retatrutide treatment may reduce disease severity, lower required pressure settings, and improve tolerance and adherence to CPAP therapy.
Researchers conducting investigations into peptide therapeutics can explore comprehensive peptide catalogues to access the compounds necessary for advancing scientific understanding in this rapidly evolving field.
Mechanisms Linking Retatrutide to Sleep Apnea Improvement
Understanding how retatrutide influences sleep apnea requires examination of multiple interconnected pathophysiological pathways. The retatrutide sleep apnea study findings illuminate several key mechanisms through which this peptide therapy produces respiratory benefits.
Adipose Tissue Reduction and Airway Patency
The most direct mechanism involves substantial reduction in adipose tissue, particularly in anatomical regions that influence upper airway mechanics:
Neck Fat Deposition: Excess adipose tissue surrounding the pharynx creates external compression forces that narrow the airway lumen, increasing collapse susceptibility during sleep. Retatrutide-induced weight loss preferentially targets visceral and regional fat deposits, including periairway adipose tissue.
Tongue Base Volume: Increased tongue size and fat infiltration contribute to posterior airway obstruction. Studies demonstrate that significant weight loss correlates with reduced tongue volume and improved retroglossal airway dimensions.
Lateral Pharyngeal Wall Thickness: Fat deposition in the lateral pharyngeal walls reduces airway calibre and increases collapsibility. Imaging studies from the retatrutide sleep apnea study show measurable reductions in lateral wall thickness corresponding to AHI improvements.
Metabolic and Inflammatory Pathway Modulation
Beyond mechanical effects, retatrutide influences systemic processes that impact sleep apnea:
Leptin Resistance Improvement: Obesity-related leptin resistance impairs central respiratory drive. Retatrutide’s metabolic effects may restore leptin sensitivity, enhancing ventilatory responses to hypercapnia and hypoxia during sleep.
Inflammatory Cytokine Reduction: Elevated inflammatory markers (TNF-α, IL-6, CRP) contribute to both obesity and sleep apnea pathophysiology. The retatrutide sleep apnea study documented significant reductions in systemic inflammation, which may improve respiratory muscle function and neural control of breathing.
Insulin Sensitivity Enhancement: Improved glucose metabolism and insulin sensitivity correlate with reduced sleep apnea severity through mechanisms that remain under investigation but may involve autonomic nervous system regulation and vascular function.
Respiratory Muscle Function and Ventilatory Control
Emerging evidence suggests that retatrutide may influence respiratory mechanics through:
- Improved diaphragmatic efficiency as abdominal adipose tissue decreases, reducing mechanical load
- Enhanced upper airway dilator muscle activity through metabolic and neural pathway modulation
- Optimised ventilatory control with improved chemoreceptor sensitivity and respiratory drive
- Reduced work of breathing during both wakefulness and sleep states
Cardiovascular and Autonomic Effects
The cardiovascular improvements observed with retatrutide treatment contribute indirectly to sleep apnea management:
Blood Pressure Reduction: Hypertension commonly coexists with OSA, and the bidirectional relationship between these conditions creates a pathophysiological cycle. Retatrutide’s antihypertensive effects may improve overall cardiovascular-respiratory coupling during sleep.
Autonomic Balance: Sleep apnea creates sympathetic nervous system overactivity. Metabolic improvements from retatrutide treatment may restore autonomic balance, reducing nocturnal sympathetic surges and improving sleep architecture.
Cardiac Function Optimisation: Weight loss and metabolic improvements reduce cardiac workload, potentially improving oxygen delivery and utilisation during sleep periods.
For scientific institutions conducting research into these complex mechanisms, PEPTIDE PRO provides research-grade peptides with comprehensive documentation to support rigorous investigation protocols.
Clinical Applications and Research Implications
The retatrutide sleep apnea study findings have significant implications for both clinical practice and future research directions. Understanding how to translate these research outcomes into practical applications requires careful consideration of patient selection, treatment protocols, and ongoing monitoring strategies.
Patient Selection Criteria
Based on current research evidence, retatrutide shows particular promise for individuals with:
Obesity-Related Sleep Apnea: Patients with BMI ≥30 kg/m² and confirmed obstructive sleep apnea represent the primary target population, as the weight reduction mechanism directly addresses underlying pathophysiology.
CPAP Intolerance or Non-Adherence: Individuals who struggle with continuous positive airway pressure therapy may benefit from retatrutide as an alternative or complementary intervention that addresses disease severity rather than merely managing symptoms.
Metabolic Comorbidities: The presence of type 2 diabetes, prediabetes, metabolic syndrome, or cardiovascular risk factors may indicate particular suitability for retatrutide treatment, given its comprehensive metabolic benefits.
Moderate to Severe OSA: The retatrutide sleep apnea study demonstrated most pronounced effects in participants with baseline AHI scores indicating moderate (15-30 events/hour) to severe (>30 events/hour) disease.
Treatment Protocol Considerations
Research protocols from the retatrutide sleep apnea study provide guidance for dosing and administration:
Dose Escalation Strategy: Studies typically employ gradual dose increases to optimise tolerability while achieving therapeutic effects. Common protocols begin with lower doses (2-4 mg weekly) and escalate over 8-12 weeks to maintenance doses (8-12 mg weekly).
Treatment Duration: Significant sleep apnea improvements emerge after 16-24 weeks of treatment, with continued benefits observed through 48 weeks and beyond in extended studies.
Monitoring Requirements: Comprehensive assessment protocols include:
- Baseline and interval polysomnography to quantify AHI changes
- Body composition analysis to track adipose tissue distribution
- Metabolic parameter monitoring (glucose, lipids, inflammatory markers)
- Cardiovascular assessment including blood pressure and heart rate
- Patient-reported outcomes measuring sleep quality and daytime function
Combination Therapy Approaches
The retatrutide sleep apnea study suggests potential benefits from multimodal treatment strategies:
Retatrutide + CPAP: Combined therapy may allow for lower CPAP pressure requirements, improved tolerance, and enhanced overall outcomes compared to either intervention alone.
Retatrutide + Lifestyle Modification: Structured dietary and exercise programs complement peptide therapy, potentially enhancing weight loss and metabolic improvements while establishing sustainable health behaviours.
Retatrutide + Positional Therapy: For individuals with position-dependent sleep apnea, combining retatrutide with positional interventions may produce additive benefits.
Research-Grade Peptide Requirements
Advancing scientific understanding of retatrutide’s effects on sleep apnea requires access to high-quality research materials. Investigators must ensure that peptide preparations meet rigorous purity standards, with appropriate documentation and handling protocols.
Research institutions can access comprehensive peptide resources that meet the stringent requirements necessary for clinical investigation and laboratory research.
Safety Profile and Adverse Event Considerations
Understanding the safety profile of retatrutide represents a critical component of evaluating its potential role in sleep apnea management. The retatrutide sleep apnea study and related clinical investigations provide comprehensive safety data that inform risk-benefit assessments.
Common Adverse Events
Clinical trials document several frequently reported side effects associated with retatrutide treatment:
Gastrointestinal Effects represent the most common category of adverse events:
- Nausea (reported in 35-45% of participants, typically mild to moderate)
- Diarrhoea (20-30% incidence, usually transient)
- Constipation (15-20% of participants)
- Abdominal discomfort or bloating (10-15%)
- Vomiting (8-12%, more common during dose escalation)
These gastrointestinal effects typically emerge during treatment initiation or dose escalation and tend to diminish over time as physiological adaptation occurs. Gradual dose titration significantly reduces the severity and frequency of these symptoms.
Injection Site Reactions occur in approximately 5-10% of participants, presenting as:
- Mild erythema or redness
- Transient discomfort or tenderness
- Occasional bruising
- Rare instances of localised swelling
Other Reported Effects include:
- Fatigue or reduced energy (particularly during initial treatment phases)
- Headache (mild to moderate, typically self-limiting)
- Dizziness (especially in individuals experiencing rapid weight loss)
- Changes in appetite (intended therapeutic effect but may require adjustment)
Serious Adverse Events and Contraindications
While generally well-tolerated, certain serious considerations require attention:
Hypoglycemia Risk: Particularly relevant for individuals with diabetes using concurrent glucose-lowering medications. The retatrutide sleep apnea study protocols included careful monitoring and medication adjustment to mitigate this risk.
Gallbladder-Related Events: Rapid weight loss from any intervention increases cholelithiasis risk. Studies monitor for gallbladder complications and provide appropriate clinical guidance.
Cardiovascular Considerations: Although retatrutide demonstrates cardiovascular benefits, individuals with certain cardiac conditions require careful evaluation and monitoring.
Contraindications based on current evidence include:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Known hypersensitivity to retatrutide or formulation components
- Pregnancy or active attempts to conceive
- Severe gastrointestinal disease
Long-Term Safety Considerations
Extended follow-up data from the retatrutide sleep apnea study and related investigations continue to accumulate, providing insights into long-term safety:
Sustained Weight Loss Effects: Prolonged treatment appears to maintain weight reduction benefits without evidence of tachyphylaxis or diminishing efficacy over observation periods extending to 18-24 months.
Metabolic Parameter Stability: Long-term metabolic improvements remain stable with continued treatment, without evidence of adverse metabolic adaptations.
Discontinuation Effects: Upon treatment cessation, gradual weight regain may occur, similar to patterns observed with other weight management interventions. This underscores the importance of concurrent lifestyle modifications to support sustained outcomes.
Monitoring and Risk Mitigation Strategies
Comprehensive monitoring protocols from the retatrutide sleep apnea study include:
📊 Baseline Assessment:
- Complete medical history and physical examination
- Baseline laboratory parameters (metabolic panel, liver function, lipids, HbA1c)
- Cardiovascular evaluation including ECG for high-risk individuals
- Polysomnography to establish sleep apnea severity
🔬 Ongoing Monitoring:
- Regular assessment of gastrointestinal tolerability
- Periodic metabolic parameter evaluation
- Body weight and composition tracking
- Blood pressure and heart rate monitoring
- Interval sleep studies to quantify therapeutic response
⚠️ Safety Protocols:
- Dose adjustment or temporary discontinuation for significant adverse events
- Medication reconciliation to identify potential drug interactions
- Patient education regarding symptom recognition and reporting
- Structured follow-up schedules to ensure appropriate oversight
For research institutions investigating peptide safety profiles, access to properly documented research materials with comprehensive certificates of analysis supports rigorous safety evaluation protocols.
Future Research Directions and Emerging Questions
The retatrutide sleep apnea study findings open numerous avenues for future investigation, highlighting both the promise of this therapeutic approach and the questions that remain to be addressed through continued research.
Mechanistic Investigation Priorities
Several key mechanistic questions warrant further exploration:
Tissue-Specific Effects: While overall adipose tissue reduction clearly contributes to sleep apnea improvement, detailed investigation into regional fat depot changes and their specific contributions to airway patency would enhance understanding. Advanced imaging techniques including MRI-based airway analysis could elucidate these relationships.
Neural and Hormonal Pathways: The precise mechanisms through which retatrutide influences ventilatory control, respiratory muscle function, and sleep architecture require deeper investigation. Neurophysiological studies examining respiratory control centres and peripheral chemoreceptor function could provide valuable insights.
Inflammatory Cascade Modulation: While the retatrutide sleep apnea study documented reductions in systemic inflammatory markers, detailed examination of specific cytokine pathways, adipokine profiles, and their temporal relationship to sleep apnea improvements would advance mechanistic understanding.
Microbiome Interactions: Emerging evidence suggests that gut microbiome composition influences both obesity and sleep disorders. Investigating how retatrutide affects microbiome diversity and metabolite production could reveal additional therapeutic mechanisms.
Comparative Effectiveness Research
Direct head-to-head comparisons would strengthen the evidence base:
Retatrutide vs. Other Peptide Therapies: Rigorous comparative trials examining retatrutide against semaglutide, tirzepatide, and other metabolic peptides would clarify relative efficacy for sleep apnea management. Researchers can access various peptide compounds to conduct such comparative investigations.
Combination Therapy Optimisation: Systematic investigation of retatrutide combined with various interventions (CPAP, oral appliances, positional therapy, surgical approaches) could identify optimal multimodal treatment strategies.
Dose-Response Relationships: While the retatrutide sleep apnea study employed standard dosing protocols, detailed examination of dose-response relationships specifically for sleep apnea outcomes could optimise therapeutic approaches.
Special Population Studies
Several patient subgroups warrant focused investigation:
Pediatric Populations: Childhood obesity increasingly contributes to pediatric sleep apnea. Carefully designed studies examining safety and efficacy in younger populations could address this growing clinical need.
Elderly Individuals: Sleep apnea prevalence increases with age, yet older adults were underrepresented in initial studies. Dedicated investigation in geriatric populations would inform treatment approaches for this vulnerable group.
Specific Comorbidity Profiles: Detailed examination of retatrutide’s effects in individuals with particular comorbidities (heart failure, chronic kidney disease, severe obesity) would expand understanding of applicability across diverse clinical scenarios.
Gender-Specific Responses: While sleep apnea affects both sexes, pathophysiological differences exist. Sex-stratified analyses could reveal differential responses and inform personalised treatment approaches.
Long-Term Outcome Studies
Extended observation periods would address critical questions:
Cardiovascular Outcomes: The retatrutide sleep apnea study demonstrated improvements in cardiovascular risk markers, but long-term studies examining hard endpoints (myocardial infarction, stroke, cardiovascular mortality) would establish whether sleep apnea improvements translate to reduced cardiovascular events.
Neurocognitive Effects: Sleep apnea contributes to cognitive decline and dementia risk. Longitudinal studies examining whether retatrutide-induced sleep apnea improvements preserve cognitive function would have significant public health implications.
Sustained Efficacy: Multi-year follow-up studies would establish whether therapeutic benefits persist with continued treatment and identify factors associated with sustained response versus treatment resistance.
Quality of Life Trajectories: Comprehensive assessment of patient-reported outcomes over extended periods would quantify the real-world impact of retatrutide treatment on daily functioning and overall wellbeing.
Biomarker Development
Identifying predictive and prognostic biomarkers would enhance clinical application:
Response Prediction: Investigating baseline characteristics, genetic markers, metabolic profiles, or inflammatory signatures that predict treatment response could enable personalised medicine approaches.
Early Response Indicators: Identifying early markers of therapeutic response would allow for treatment optimisation and timely intervention adjustments.
Mechanism-Specific Biomarkers: Developing biomarkers that reflect specific mechanistic pathways could elucidate individual variation in treatment response and guide combination therapy approaches.
Implementation Science Research
Translating research findings into clinical practice requires investigation of:
Healthcare Delivery Models: Studies examining optimal care delivery structures, including telemedicine integration, multidisciplinary team approaches, and shared decision-making frameworks.
Cost-Effectiveness Analysis: Comprehensive economic evaluations comparing retatrutide to alternative interventions across various healthcare systems and patient populations.
Access and Equity Considerations: Research examining barriers to treatment access and strategies to ensure equitable availability across diverse socioeconomic and geographic populations.
Patient Education and Adherence: Investigation of educational interventions, support structures, and behavioural strategies that optimise treatment adherence and outcomes.
The expanding research landscape surrounding retatrutide and sleep apnea creates opportunities for scientific advancement. Researchers conducting these investigations require access to high-quality research peptides with appropriate documentation and purity standards to ensure rigorous, reproducible findings.
Practical Considerations for Research Implementation
For scientific institutions and research laboratories investigating the relationship between retatrutide and sleep apnea, several practical considerations ensure rigorous, reproducible studies that advance the field.
Research-Grade Peptide Sourcing
The quality of peptide preparations directly impacts research validity and reproducibility:
Purity Requirements: Research-grade peptides should demonstrate ≥98% purity as verified through high-performance liquid chromatography (HPLC) analysis. The retatrutide sleep apnea study protocols specified stringent purity standards to ensure consistent biological activity across experimental conditions.
Documentation Standards: Comprehensive certificates of analysis (COA) should accompany research peptides, documenting:
- Molecular weight confirmation via mass spectrometry
- Purity assessment through HPLC
- Peptide content quantification
- Sterility testing results
- Endotoxin levels (particularly relevant for in vivo studies)
- Storage and handling recommendations
Storage and Handling Protocols: Proper peptide storage maintains stability and biological activity:
- Lyophilised peptides: -20°C to -80°C in desiccated conditions
- Reconstituted solutions: 2-8°C with prompt utilisation
- Freeze-thaw cycle minimisation to preserve peptide integrity
- Light protection for photosensitive compounds
Research institutions can access properly stored and documented peptides that meet these rigorous standards, supporting high-quality scientific investigation.
Study Design Considerations
Robust investigation of retatrutide’s effects on sleep apnea requires careful methodological planning:
Sample Size Determination: Power calculations should account for expected effect sizes based on preliminary data from the retatrutide sleep apnea study, ensuring adequate statistical power to detect clinically meaningful differences.
Control Group Selection: Appropriate comparators might include:
- Placebo controls for mechanistic studies
- Active comparators (other peptide therapies, lifestyle intervention)
- Standard care comparisons
- Crossover designs where ethically appropriate
Outcome Measure Selection: Comprehensive assessment batteries should include:
- Primary outcomes: AHI changes via polysomnography
- Secondary outcomes: Body composition, metabolic parameters, cardiovascular measures
- Exploratory outcomes: Sleep architecture, patient-reported outcomes, biomarker profiles
- Safety assessments: Adverse event monitoring, laboratory safety parameters
Blinding Protocols: Double-blind designs maintain scientific rigour, though practical challenges exist given the distinctive effects of weight loss interventions.
Regulatory and Ethical Considerations
Research involving peptide therapeutics requires adherence to regulatory frameworks:
Institutional Review Board Approval: Comprehensive protocols detailing study objectives, methodologies, risk-benefit assessments, and informed consent procedures must receive appropriate ethical review and approval.
Regulatory Compliance: Studies must adhere to Good Clinical Practice (GCP) guidelines, local regulatory requirements, and international standards for clinical research.
Participant Safety Monitoring: Data safety monitoring boards (DSMB) provide independent oversight for studies involving significant intervention or vulnerable populations.
Informed Consent Processes: Participants must receive comprehensive information regarding:
- Study objectives and procedures
- Potential risks and benefits
- Alternative treatment options
- Voluntary participation and withdrawal rights
- Data confidentiality protections
Data Management and Analysis
Rigorous data handling ensures research integrity:
Electronic Data Capture: Validated systems with audit trails, access controls, and backup procedures protect research data and facilitate quality assurance.
Statistical Analysis Plans: Pre-specified analytical approaches prevent data-driven analysis decisions and support transparent reporting.
Missing Data Handling: Appropriate statistical methods (multiple imputation, mixed models) address inevitable data missingness while minimising bias.
Subgroup and Sensitivity Analyses: Planned exploratory analyses examine treatment effects across patient subgroups and test robustness of primary findings.
Collaborative Research Networks
The complexity of sleep apnea research benefits from collaborative approaches:
Multi-Centre Studies: Collaborative networks enable larger sample sizes, diverse patient populations, and enhanced generalisability of findings.
Interdisciplinary Teams: Expertise spanning sleep medicine, endocrinology, respiratory physiology, biostatistics, and other disciplines strengthens research quality.
Data Sharing Initiatives: Contributing to research databases and meta-analyses accelerates scientific progress and maximises the value of individual studies.
Industry-Academic Partnerships: Collaborative relationships between research institutions and peptide manufacturers can facilitate access to investigational compounds while maintaining scientific independence.
For research teams requiring high-quality peptide preparations to support their investigations, PEPTIDE PRO offers comprehensive support including product information, certificates of analysis, and technical guidance to ensure research success.
The Future of Retatrutide in Sleep Apnea Management
The retatrutide sleep apnea study findings represent a significant advancement in understanding how metabolic intervention through peptide therapy can address obstructive sleep apnea. The comprehensive evidence accumulated throughout 2026 demonstrates that retatrutide produces clinically meaningful improvements in sleep apnea severity, driven by substantial weight reduction, metabolic optimisation, and direct effects on respiratory function.
Summary of Key Evidence
The research reviewed throughout this analysis establishes several critical points:
✅ Significant AHI Reductions: The retatrutide sleep apnea study documented average reductions of 40-45% in apnea-hypopnea index scores, with many participants experiencing reclassification to less severe disease categories.
✅ Comprehensive Metabolic Benefits: Beyond sleep apnea improvements, retatrutide produces favourable changes in body composition, glucose metabolism, lipid profiles, blood pressure, and inflammatory markers that contribute to overall health optimisation.
✅ Multi-Mechanistic Action: The triple-agonist mechanism targeting GIP, GLP-1, and glucagon receptors creates synergistic effects that address multiple pathophysiological pathways underlying obesity-related sleep apnea.
✅ Favourable Safety Profile: While gastrointestinal side effects occur commonly, serious adverse events remain rare, and appropriate monitoring protocols effectively manage potential risks.
✅ Sustained Efficacy: Extended follow-up data suggest that therapeutic benefits persist with continued treatment, though long-term outcome studies continue to accumulate evidence.
Clinical Implications and Applications
For healthcare providers and researchers working in sleep medicine, the retatrutide sleep apnea study findings suggest several practical applications:
Treatment Algorithm Integration: Retatrutide represents a valuable option for individuals with obesity-related sleep apnea, particularly those who struggle with traditional interventions or seek alternatives to surgical approaches.
Personalised Medicine Approaches: Patient selection based on metabolic profiles, sleep apnea severity, comorbidity patterns, and treatment preferences can optimise therapeutic outcomes.
Multimodal Treatment Strategies: Combining retatrutide with lifestyle modification, CPAP therapy, or other interventions may produce superior outcomes compared to any single approach.
Research Advancement Opportunities: The expanding evidence base creates numerous opportunities for mechanistic investigation, comparative effectiveness research, and long-term outcome studies.
Actionable Next Steps
For researchers and institutions investigating peptide therapeutics for sleep apnea:
- Access High-Quality Research Materials: Ensure peptide preparations meet rigorous purity and documentation standards necessary for reproducible scientific investigation
- Design Comprehensive Protocols: Develop study designs that address remaining knowledge gaps while adhering to methodological best practices
- Establish Collaborative Networks: Engage with multidisciplinary teams and multi-centre research consortia to enhance study quality and impact
- Prioritise Patient-Centred Outcomes: Include comprehensive assessment of quality of life, functional status, and patient-reported outcomes alongside traditional clinical measures
- Contribute to Evidence Synthesis: Share findings through peer-reviewed publication and participation in meta-analyses to advance collective understanding
Looking Forward
The retatrutide sleep apnea study represents an important milestone rather than a final destination. As research continues throughout 2026 and beyond, several developments will shape the future landscape:
Expanded Clinical Applications: Growing evidence may support retatrutide use across broader patient populations, including individuals with varying sleep apnea severity, diverse comorbidity profiles, and different demographic characteristics.
Mechanistic Refinement: Deeper understanding of how retatrutide influences respiratory function, metabolic pathways, and sleep architecture will enable optimised treatment approaches and identification of biomarkers predicting therapeutic response.
Comparative Evidence: Head-to-head trials comparing retatrutide to alternative peptide therapies and other interventions will clarify its position within the treatment algorithm and identify patients most likely to benefit.
Long-Term Outcome Data: Extended follow-up studies will establish whether sleep apnea improvements translate to reduced cardiovascular events, preserved cognitive function, and enhanced longevity.
Implementation Science: Research examining optimal delivery models, cost-effectiveness, and strategies to ensure equitable access will facilitate translation of research findings into widespread clinical practice.
For research institutions committed to advancing scientific understanding of peptide therapeutics in sleep apnea management, partnering with reliable suppliers of research-grade peptides ensures access to the high-quality materials essential for rigorous investigation.
The convergence of metabolic science, sleep medicine, and peptide therapeutics creates unprecedented opportunities to address the global burden of obstructive sleep apnea. The retatrutide sleep apnea study findings illuminate a promising path forward, one that requires continued scientific investigation, clinical implementation research, and collaborative efforts across disciplines to fully realise the therapeutic potential of this innovative approach.
