
When exploring novel peptide therapeutics, understanding potential safety signals becomes paramount—and the retatrutide thyroid cancer warning represents a critical consideration for research professionals working with this triple-agonist compound. As laboratories across the UK and internationally investigate retatrutide’s metabolic effects, the thyroid-related precautionary information derived from preclinical studies demands careful attention and proper contextualisation within research protocols.
Retatrutide, a triple receptor agonist targeting GLP-1, GIP, and glucagon receptors, has emerged as a promising research peptide in metabolic studies. However, like other compounds in the incretin-based therapeutic class, it carries specific warnings related to thyroid C-cell tumours observed in rodent models. This comprehensive guide examines the retatrutide thyroid cancer warning, its scientific basis, regulatory context, and implications for responsible research practices in 2026.
Key Takeaways
- Rodent-specific findings: Thyroid C-cell tumours (medullary thyroid carcinoma) have been observed in rodent studies with GLP-1 receptor agonists, forming the basis for the retatrutide thyroid cancer warning
- Human relevance uncertain: The direct applicability of rodent thyroid findings to human physiology remains scientifically debated due to significant species differences in C-cell density and GLP-1 receptor expression
- Contraindications established: Research protocols should exclude subjects with personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Monitoring protocols essential: Baseline and periodic calcitonin measurements represent standard precautionary measures in research settings involving incretin-based peptides
- Regulatory compliance required: All retatrutide research must adhere to strict “Research Use Only” guidelines with appropriate institutional oversight and safety monitoring
Understanding Retatrutide: Mechanism and Research Applications
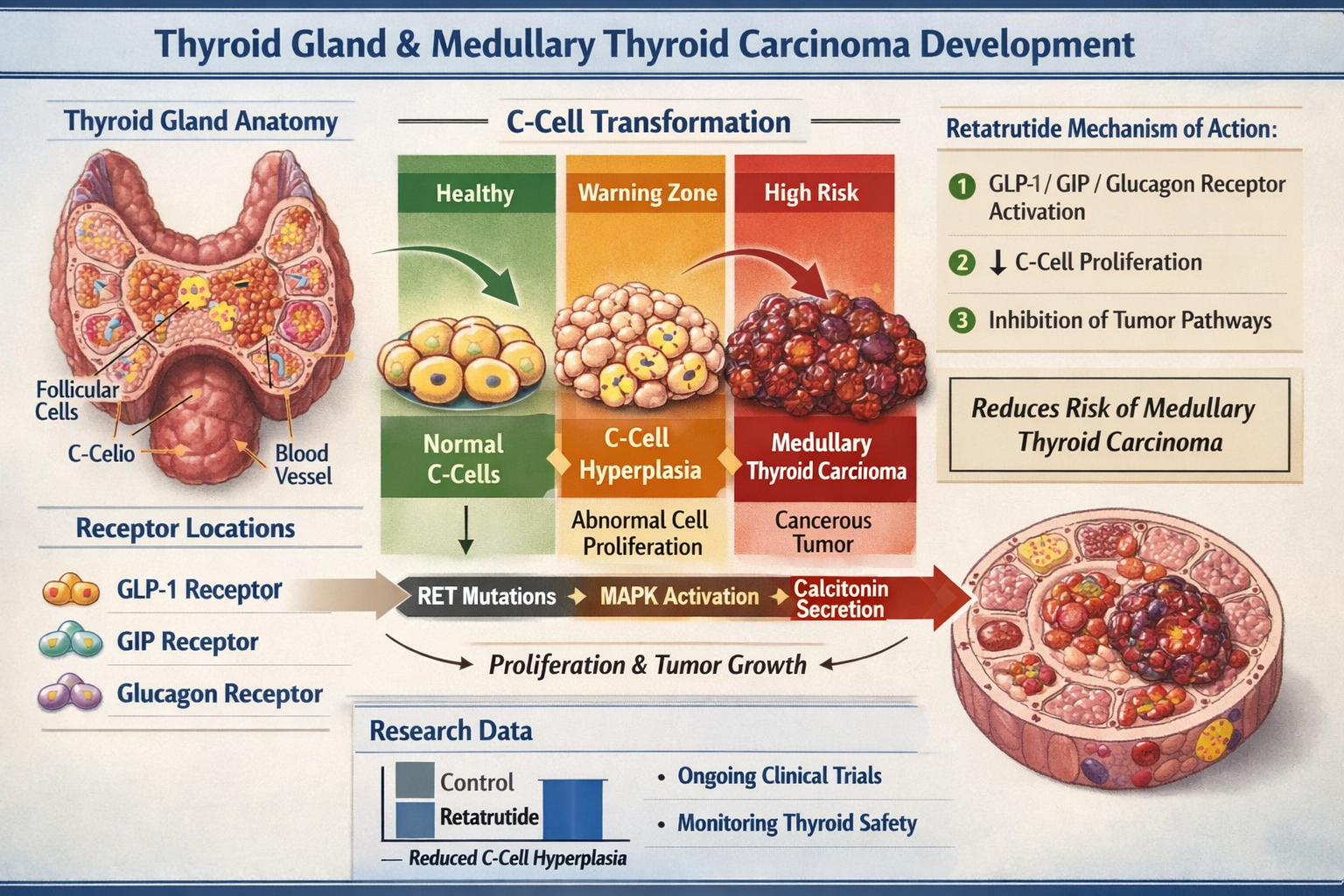
Retatrutide represents an innovative class of research peptides known as triple agonists, simultaneously activating three distinct metabolic pathways through GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors. This multi-targeted approach distinguishes retatrutide from earlier single or dual agonist compounds, offering researchers unique opportunities to investigate complex metabolic regulation.
Molecular Mechanism of Action
The compound’s triple-agonist activity creates a comprehensive metabolic effect profile:
GLP-1 Receptor Activation
- Enhances glucose-dependent insulin secretion
- Reduces glucagon release
- Slows gastric emptying
- Influences satiety signalling pathways
GIP Receptor Activation
- Potentiates insulin secretion
- May influence lipid metabolism
- Contributes to bone metabolism regulation
- Modulates adipose tissue function
Glucagon Receptor Activation
- Increases energy expenditure
- Promotes lipolysis
- Enhances hepatic fat oxidation
- Influences thermogenic pathways
This multi-receptor engagement makes retatrutide particularly valuable for research into metabolic syndrome, obesity mechanisms, and energy homeostasis. Laboratories sourcing high-purity research peptides require comprehensive safety information to design appropriate protocols.
Research Applications in 2026
Current research applications for retatrutide span multiple investigational areas:
| Research Domain | Investigation Focus | Methodological Considerations |
|---|---|---|
| Metabolic Studies | Energy balance mechanisms, substrate utilisation | Requires comprehensive metabolic monitoring |
| Obesity Research | Weight regulation pathways, adipose tissue dynamics | Long-term observation protocols needed |
| Glucose Homeostasis | Insulin sensitivity, beta-cell function | Glucose tolerance assessments essential |
| Cardiovascular Metabolism | Lipid profiles, vascular function | Cardiovascular parameter tracking |
| Hepatic Function | Fatty liver mechanisms, hepatic metabolism | Liver enzyme and imaging protocols |
Researchers working with retatrutide must implement rigorous safety monitoring that includes awareness of the thyroid cancer warning derived from preclinical toxicology studies.
The Scientific Basis of the Retatrutide Thyroid Cancer Warning
The retatrutide thyroid cancer warning originates from observations in long-term rodent carcinogenicity studies, a standard component of pharmaceutical development for incretin-based compounds. Understanding the scientific foundation of this warning requires examination of both the preclinical findings and the mechanistic hypotheses underlying thyroid C-cell responses to GLP-1 receptor agonists.
Preclinical Toxicology Findings
Rodent carcinogenicity studies with GLP-1 receptor agonists, including compounds structurally and mechanistically related to retatrutide, have consistently demonstrated:
C-Cell Hyperplasia and Tumour Formation
- Dose-dependent increases in thyroid C-cell hyperplasia
- Development of medullary thyroid carcinoma (MTC) in rats and mice
- Statistically significant increases at exposures relevant to therapeutic investigations
- Time-dependent progression from hyperplasia to adenoma to carcinoma
Calcitonin Elevation
- Sustained increases in serum calcitonin levels
- Correlation between calcitonin elevation and C-cell proliferation
- Dose-response relationship in rodent models
- Persistence throughout exposure duration
These findings led regulatory agencies to require specific warnings and contraindications for GLP-1 receptor agonists, which extend to multi-agonist compounds like retatrutide that incorporate GLP-1 receptor activity.
Mechanistic Understanding: GLP-1 Receptors and Thyroid C-Cells
The proposed mechanism linking GLP-1 receptor activation to rodent thyroid tumours involves:
🔬 GLP-1 Receptor Expression: Rodent thyroid C-cells express significant levels of GLP-1 receptors, making them responsive to agonist stimulation.
🔬 Proliferative Signalling: Chronic GLP-1 receptor activation may trigger proliferative pathways in C-cells, leading to hyperplasia.
🔬 Calcitonin Production: Stimulated C-cells increase calcitonin synthesis and secretion, serving as a biomarker of C-cell activation.
🔬 Neoplastic Transformation: Prolonged hyperplastic stimulation may progress to neoplastic changes in susceptible rodent models.
This mechanistic framework, while well-established in rodents, faces significant questions regarding human relevance—a critical consideration for research protocol design.
Species Differences: Rodent vs. Human Thyroid Physiology
A fundamental question surrounding the retatrutide thyroid cancer warning centres on species-specific differences in thyroid C-cell biology:
Critical Distinctions Include:
| Characteristic | Rodents | Humans |
|---|---|---|
| C-Cell Density | High concentration | Low concentration (scattered distribution) |
| GLP-1R Expression | Abundant on C-cells | Minimal to absent on C-cells |
| Calcitonin Response | Marked elevation with GLP-1 agonists | Minimal or no elevation |
| Spontaneous MTC | Relatively common in aging rats | Rare (< 0.001% population) |
| C-Cell Proliferative Response | Robust | Minimal observed response |
These substantial physiological differences have led many researchers to question the direct translatability of rodent thyroid findings to human risk assessment. However, regulatory frameworks adopt precautionary principles, maintaining warnings until definitive human safety data accumulates.
Epidemiological Evidence in Human Populations
As of 2026, extensive post-marketing surveillance and clinical trial data for earlier GLP-1 receptor agonists (liraglutide, semaglutide, dulaglutide) spanning millions of patient-years have not demonstrated increased medullary thyroid carcinoma incidence in human populations. This real-world evidence supports the hypothesis that rodent thyroid findings may not translate to human risk.
However, this reassuring epidemiological pattern does not eliminate the need for appropriate precautions in research settings, particularly given:
- The relatively recent introduction of triple agonists like retatrutide
- The long latency period for thyroid cancer development
- Individual genetic susceptibility factors (MEN 2 syndrome)
- Regulatory requirements for comprehensive safety monitoring
Research institutions must therefore maintain vigilant safety protocols that acknowledge both the precautionary warnings and the evolving understanding of species-specific thyroid physiology.
Regulatory Framework and Contraindications for Retatrutide Research
The retatrutide thyroid cancer warning exists within a comprehensive regulatory framework governing research peptide use. Understanding these requirements ensures compliant, ethical, and safe research practices in laboratory settings throughout 2026.
Regulatory Classification and Requirements
Retatrutide falls under strict regulatory oversight as an investigational compound:
Research-Only Status ⚠️
- Classified exclusively for research applications
- Not approved for human therapeutic use
- Requires institutional review and oversight
- Must be clearly labelled “For Research Use Only”
Quality Standards
- Manufacturing under controlled conditions
- Purity verification through independent testing
- Certificate of Analysis (COA) documentation
- Proper storage and handling protocols
Reputable suppliers like PEPTIDE PRO provide research-grade peptides with comprehensive documentation, supporting compliant research practices.
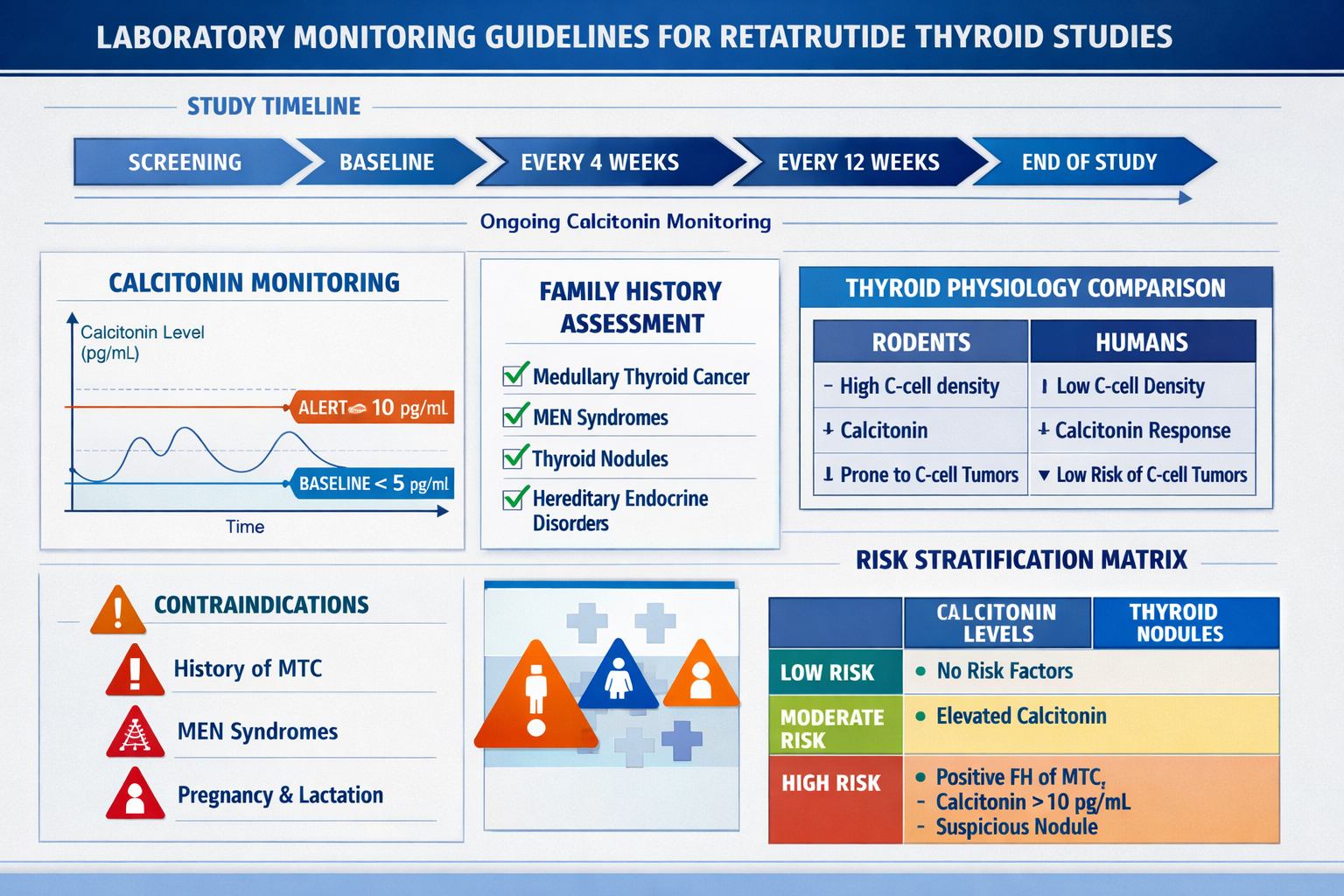
Established Contraindications Based on Thyroid Cancer Warning
Research protocols involving retatrutide should implement specific exclusion criteria derived from the thyroid cancer warning:
Absolute Contraindications:
✖️ Personal History of Medullary Thyroid Carcinoma (MTC)
- Any documented MTC diagnosis
- Previous thyroidectomy for MTC
- Elevated baseline calcitonin levels (>50 pg/mL typically considered significant)
✖️ Family History of MTC
- First-degree relatives with MTC diagnosis
- Known familial MTC patterns
- Uncertain family thyroid cancer history requiring investigation
✖️ Multiple Endocrine Neoplasia Syndrome Type 2 (MEN 2)
- Personal diagnosis of MEN 2A or MEN 2B
- Family history of MEN 2 syndrome
- Known RET proto-oncogene mutations associated with MEN 2
- Pheochromocytoma or hyperparathyroidism suggesting MEN 2
✖️ Thyroid Nodules with Uncertain Pathology
- Undiagnosed thyroid nodules
- Suspicious ultrasound findings pending evaluation
- Elevated calcitonin without established benign cause
Relative Considerations:
⚠️ Pre-existing Thyroid Conditions
- Goiter or thyroid enlargement
- History of thyroid surgery (non-MTC)
- Thyroid hormone replacement therapy
- Autoimmune thyroid disease
⚠️ Pregnancy and Reproductive Considerations
- Research protocols typically exclude pregnancy
- Reproductive toxicology data limited
- Contraceptive requirements in research settings
These contraindications should be incorporated into research subject screening protocols, with appropriate documentation and institutional review board (IRB) approval.
Informed Consent and Disclosure Requirements
Ethical research practice demands comprehensive disclosure of the retatrutide thyroid cancer warning:
Essential Disclosure Elements:
- Rodent Study Findings: Clear explanation of thyroid tumours in animal models
- Human Relevance Uncertainty: Honest discussion of species differences and unknown human risk
- Contraindication Rationale: Explanation of exclusion criteria based on MTC/MEN 2 risk
- Monitoring Procedures: Description of calcitonin testing and thyroid surveillance
- Warning Signs: Education about symptoms requiring immediate medical evaluation
- Research-Only Status: Explicit statement that retatrutide is not approved for therapeutic use
Research institutions must ensure participants or research subjects (where applicable to research models) receive this information in accessible language, with opportunities for questions and clarification.
Institutional Oversight and Safety Monitoring
Responsible retatrutide research requires multi-layered institutional oversight:
Institutional Review Board (IRB) Requirements:
- Protocol review including thyroid safety considerations
- Risk-benefit assessment specific to research objectives
- Monitoring plan approval
- Adverse event reporting procedures
- Annual continuing review with safety data
Safety Monitoring Committees:
- Independent data safety monitoring for longer-term studies
- Periodic review of thyroid-related parameters
- Authority to halt studies based on safety signals
- Expertise in endocrinology and thyroid pathology
Laboratory Safety Protocols:
- Proper peptide handling and storage procedures
- Waste disposal compliance
- Exposure prevention for laboratory personnel
- Emergency response procedures
These oversight mechanisms ensure that the retatrutide thyroid cancer warning translates into practical protective measures within research environments.
Implementing Thyroid Monitoring Protocols in Retatrutide Research
Translating the retatrutide thyroid cancer warning into actionable research protocols requires systematic thyroid monitoring strategies. These evidence-based approaches balance precautionary principles with practical research implementation.
Baseline Screening Recommendations
Comprehensive baseline assessment forms the foundation of thyroid safety monitoring:
Pre-Research Thyroid Evaluation:
📋 Medical History Assessment
- Personal thyroid disease history
- Family history of thyroid cancer (particularly MTC)
- MEN 2 syndrome screening questions
- Previous thyroid surgery or radiation exposure
- Current thyroid medications
📋 Physical Examination
- Thyroid palpation for nodules or enlargement
- Cervical lymph node assessment
- Documentation of baseline thyroid size
- Notation of any asymmetry or irregularities
📋 Laboratory Testing
- Serum Calcitonin: Most critical baseline marker
- Normal reference range: < 10 pg/mL (varies by assay)
- Elevated levels (>50-100 pg/mL) warrant investigation
- Gender differences: slightly higher in males
- Stimulation testing (pentagastrin or calcium) if borderline elevated
- Thyroid Function Tests:
- TSH (thyroid-stimulating hormone)
- Free T4 (thyroxine)
- Free T3 (triiodothyronine) if indicated
- Thyroid Antibodies (if autoimmune disease suspected):
- Anti-TPO (thyroid peroxidase antibodies)
- Anti-thyroglobulin antibodies
📋 Imaging Studies (When Indicated)
- Thyroid Ultrasound:
- Recommended if palpable nodules detected
- Baseline documentation for high-risk subjects
- Characterization of any suspicious findings
- Measurement of nodule size for monitoring
- Fine Needle Aspiration (FNA):
- Required for suspicious nodules
- Cytological evaluation before research participation
- Calcitonin immunostaining if MTC suspected
Periodic Monitoring During Research
Ongoing surveillance throughout research protocols enables early detection of thyroid changes:
Recommended Monitoring Schedule:
| Time Point | Calcitonin Testing | Thyroid Function | Physical Exam | Imaging |
|---|---|---|---|---|
| Baseline |
✓ | Month 3 |
✓ | Month 6 |
✓ | Month 12 |
✓ | Annually thereafter |
✓ Required | As clinically indicated |
Calcitonin Monitoring Interpretation:
🔍 Stable Normal Levels (< 10 pg/mL)
- Continue scheduled monitoring
- No additional intervention required
- Document in research records
🔍 Mild Elevation (10-50 pg/mL)
- Repeat measurement for confirmation
- Consider stimulation testing
- Increase monitoring frequency
- Thyroid ultrasound evaluation
- Review for secondary causes (renal insufficiency, proton pump inhibitors, thyroiditis)
🔍 Moderate Elevation (50-100 pg/mL)
- Immediate repeat measurement
- Stimulation testing strongly recommended
- Thyroid ultrasound required
- Endocrinology consultation
- Consider research protocol modification or discontinuation
🔍 Significant Elevation (> 100 pg/mL)
- Immediate research discontinuation
- Urgent endocrinology referral
- Comprehensive thyroid imaging
- Consideration of surgical evaluation
- Detailed documentation and reporting
Calcitonin Testing: Technical Considerations
Accurate calcitonin measurement requires attention to methodological details:
Assay Selection and Standardization:
- Multiple commercial assays available with varying reference ranges
- Two-site immunometric assays most commonly used
- Lack of international standardization complicates interpretation
- Consistency in assay methodology essential for serial monitoring
- Laboratory-specific reference ranges must be applied
Pre-analytical Factors Affecting Results:
- Sample timing: fasting vs. non-fasting (minimal effect)
- Sample handling: serum separation within 2 hours
- Storage: frozen if not analyzed immediately
- Interfering substances: heterophile antibodies, biotin supplementation
- Physiological variations: gender, age, renal function
Stimulation Testing Protocols:
When baseline calcitonin is borderline elevated (10-50 pg/mL), stimulation testing may improve diagnostic accuracy:
- Calcium Stimulation Test: IV calcium gluconate administration with serial calcitonin measurements
- Pentagastrin Stimulation: (Limited availability in some regions) Synthetic gastrin administration
- Interpretation: Exaggerated calcitonin response suggests C-cell pathology
- Sensitivity/Specificity: Higher diagnostic accuracy than basal calcitonin alone
Managing Abnormal Findings
Research protocols must include clear pathways for managing thyroid abnormalities:
Decision Algorithm for Elevated Calcitonin:
Elevated Calcitonin Detected
↓
Confirm with Repeat Measurement
↓
Persistent Elevation?
↓
┌────┴────┐
Yes No
↓ ↓
Thyroid Continue
Ultrasound Monitoring
↓
Nodules Present?
↓
┌───┴───┐
Yes No
↓ ↓
FNA Consider
Biopsy Stimulation
Testing
Research Protocol Modifications:
- Temporary hold pending evaluation
- Dose reduction consideration
- Permanent discontinuation if MTC suspected
- Enhanced monitoring for continued participation (if appropriate)
- Documentation of decision-making rationale
Specialist Referral Criteria:
- Calcitonin > 50 pg/mL on repeated testing
- Any thyroid nodule with elevated calcitonin
- Suspicious ultrasound findings
- Positive family history with any calcitonin elevation
- Participant anxiety or request for specialist evaluation
These comprehensive monitoring protocols translate the retatrutide thyroid cancer warning into practical safety measures, protecting research integrity while maintaining participant welfare.
Comparative Context: Thyroid Warnings Across Incretin-Based Peptides
Understanding the retatrutide thyroid cancer warning benefits from comparative analysis across the broader class of incretin-based research peptides. This contextualisation helps researchers appreciate both class-wide considerations and compound-specific nuances.
GLP-1 Receptor Agonist Class Warnings
The thyroid cancer warning associated with retatrutide reflects a class effect observed across GLP-1 receptor agonists:
Established GLP-1 Agonists with Thyroid Warnings:
| Compound | Receptor Activity | Rodent MTC Findings | Human Epidemiology (2026) |
|---|---|---|---|
| Liraglutide | GLP-1 agonist | Dose-dependent C-cell tumours in rats/mice | No increased MTC incidence in >10 years post-marketing surveillance |
| Semaglutide | GLP-1 agonist | C-cell hyperplasia and tumours in rodents | Extensive trial data showing no human MTC signal |
| Dulaglutide | GLP-1 agonist | Thyroid C-cell tumours in rodent studies | Post-marketing data reassuring for human risk |
| Exenatide | GLP-1 agonist | C-cell proliferation in preclinical models | Long-term human use without MTC association |
| Tirzepatide | GLP-1/GIP dual agonist | Rodent thyroid findings consistent with class | Clinical trial data showing no human MTC cases |
| Retatrutide | GLP-1/GIP/Glucagon triple agonist | Preclinical findings consistent with GLP-1 class | Limited human data; research-only status in 2026 |
This pattern demonstrates that the thyroid warning represents a class characteristic rather than a retatrutide-specific concern, stemming from the GLP-1 receptor agonism component of these compounds.
Dual and Triple Agonist Considerations
Retatrutide’s multi-receptor activity raises questions about whether additional receptor targets modify thyroid risk:
GIP Receptor Considerations:
- GIP receptors not significantly expressed on thyroid C-cells
- No independent thyroid tumour signal from GIP agonism
- Dual GLP-1/GIP agonists (like tirzepatide) show thyroid findings similar to GLP-1-only agonists
- GIP component unlikely to contribute to thyroid risk
Glucagon Receptor Considerations:
- Glucagon receptors not prominently expressed on thyroid tissue
- No established thyroid proliferative effects from glucagon receptor activation
- Limited preclinical data specifically examining glucagon agonism and thyroid outcomes
- Glucagon component not expected to modify thyroid cancer risk
Conclusion: The retatrutide thyroid cancer warning likely derives primarily from its GLP-1 receptor agonist activity, with GIP and glucagon components not substantially modifying this risk profile.
Lessons from Long-Term Human Data with Related Compounds
The extensive human experience with earlier GLP-1 receptor agonists provides valuable context:
Reassuring Epidemiological Patterns:
📊 Large-Scale Safety Databases
- Millions of patient-years of exposure to liraglutide, semaglutide, dulaglutide
- Comprehensive post-marketing surveillance systems
- No statistically significant increase in MTC incidence
- Background MTC rate (0.001% annually) unchanged in exposed populations
📊 Clinical Trial Safety Data
- Systematic calcitonin monitoring in major trials
- No consistent pattern of calcitonin elevation in humans
- Rare MTC cases occur at background rates
- No dose-response relationship observed in human studies
📊 Mechanistic Studies in Human Tissue
- Human thyroid C-cells show minimal GLP-1 receptor expression
- In vitro studies demonstrate lack of proliferative response
- Calcitonin secretion not stimulated by GLP-1 agonists in human C-cells
- Fundamental species differences confirmed at molecular level
Implications for Retatrutide Research:
While this accumulated evidence is reassuring, several important caveats apply to retatrutide specifically:
⚠️ Limited Human Exposure: As a newer investigational compound, retatrutide lacks the extensive human safety database of older GLP-1 agonists
⚠️ Unique Pharmacology: Triple agonism represents a distinct pharmacological profile requiring independent safety assessment
⚠️ Long Latency Periods: Thyroid cancer development occurs over years to decades, requiring prolonged surveillance
⚠️ Regulatory Precaution: Despite reassuring data for related compounds, regulatory frameworks maintain warnings until compound-specific long-term data accumulates
Practical Implications for Research Design
This comparative context informs evidence-based research protocol development:
Risk Stratification Approach:
🔬 Lower Risk Research
- Short-term mechanistic studies (days to weeks)
- In vitro or ex vivo investigations
- Animal models with appropriate species selection
- Minimal thyroid monitoring requirements
🔬 Moderate Risk Research
- Medium-term investigations (months)
- Repeated dosing protocols
- Baseline and periodic calcitonin monitoring
- Standard exclusion criteria (MTC/MEN 2 history)
🔬 Higher Scrutiny Research
- Long-term exposure studies (> 6 months)
- High-dose investigations
- Enhanced thyroid monitoring protocols
- Comprehensive baseline thyroid evaluation
- More frequent surveillance intervals
Monitoring Intensity Calibration:
Research teams can calibrate monitoring intensity based on:
- Study duration and cumulative exposure
- Dose levels relative to proposed therapeutic ranges
- Participant risk factors and baseline thyroid status
- Institutional risk tolerance and oversight requirements
- Availability of resources for comprehensive monitoring
This nuanced approach acknowledges both the precautionary thyroid warning and the reassuring human experience with related compounds, enabling scientifically sound and ethically responsible research practices.
For researchers seeking high-quality retatrutide for laboratory investigations, understanding these comparative considerations supports informed protocol design and appropriate safety monitoring implementation.
Clinical Presentation and Warning Signs: What Researchers Should Monitor
Beyond structured monitoring protocols, researchers working with retatrutide must maintain vigilance for clinical presentations that might indicate thyroid pathology. Understanding the warning signs associated with medullary thyroid carcinoma ensures prompt recognition and appropriate response.
Medullary Thyroid Carcinoma: Clinical Features
Medullary thyroid carcinoma (MTC), the specific thyroid malignancy observed in rodent studies, presents with characteristic clinical features:
Early-Stage MTC:
In early stages, MTC is often asymptomatic and detected only through:
- Incidental findings on imaging performed for other reasons
- Screening in individuals with MEN 2 syndrome or family history
- Elevated calcitonin discovered during routine monitoring
- Palpable thyroid nodule discovered on physical examination
Advanced MTC Presentations:
When symptomatic, MTC may present with:
• Local Symptoms
- Palpable neck mass or thyroid nodule
- Neck pain or discomfort
- Dysphagia (difficulty swallowing)
- Hoarseness or voice changes (recurrent laryngeal nerve involvement)
- Cervical lymphadenopathy (enlarged neck lymph nodes)
• Systemic Symptoms (Advanced Disease)
- Diarrhea (from calcitonin or other peptide secretion)
- Flushing episodes
- Constitutional symptoms (fatigue, weight loss)
- Symptoms from distant metastases (bone pain, respiratory symptoms)
MEN 2-Associated Symptoms
- Pheochromocytoma symptoms: episodic hypertension, headaches, palpitations, sweating
- Hyperparathyroidism symptoms: kidney stones, bone pain, cognitive changes
- Mucosal neuromas (in MEN 2B): visible oral lesions, distinctive facial appearance
Warning Signs Requiring Immediate Evaluation
Research participants or subjects should be educated about symptoms warranting immediate medical assessment:
Critical Warning Signs:
⚠️ Rapidly Enlarging Neck Mass
- Any new thyroid nodule or neck swelling
- Progressive enlargement over weeks to months
- Firm, fixed, or irregular masses on palpation
⚠️ Voice or Swallowing Changes
- New hoarseness persisting beyond 2 weeks
- Progressive difficulty swallowing
- Sensation of pressure or fullness in neck
⚠️ Persistent Neck Pain
- Localized thyroid region pain
- Pain radiating to ear or jaw
- Pain unresponsive to simple analgesics
⚠️ Unexplained Diarrhea
- New-onset chronic diarrhea
- Secretory diarrhea (large volume, watery)
- Diarrhea unresponsive to dietary modifications
⚠️ Concerning Systemic Symptoms
- Unexplained weight loss
- Persistent fatigue
- New bone pain (potential metastases)
Distinguishing MTC from Benign Thyroid Conditions
Research protocols must account for the high prevalence of benign thyroid conditions in the general population:
Common Benign Thyroid Presentations:
| Condition | Prevalence | Key Features | Calcitonin Level |
|---|---|---|---|
| Benign Thyroid Nodules | 50-60% of adults by age 60 | Usually asymptomatic, discovered incidentally | Normal |
| Hashimoto’s Thyroiditis | 5-10% of population | Hypothyroid symptoms, positive antibodies | Normal |
| Graves’ Disease | 1-2% of population | Hyperthyroid symptoms, ophthalmopathy | Normal |
| Multinodular Goiter | Common with aging | Multiple nodules, possible compression symptoms | Normal |
| Thyroid Cysts | Very common | Fluid-filled, benign on ultrasound | Normal |
Key Distinguishing Feature: Normal calcitonin levels effectively exclude MTC in the vast majority of cases, making calcitonin measurement the critical discriminating test.
False Positive Considerations:
Calcitonin elevation can occur in non-MTC conditions:
- Chronic kidney disease (impaired clearance)
- Proton pump inhibitor use (gastric hypochlorhydria)
- Chronic thyroiditis
- Other neuroendocrine tumours (rare)
- C-cell hyperplasia (non-neoplastic)
Proper clinical correlation and specialist evaluation help distinguish these scenarios from true MTC.
Documentation and Reporting Requirements
Research protocols must establish clear documentation and reporting pathways:
Adverse Event Reporting:
📝 Immediate Reporting (Within 24 Hours)
- Any suspected thyroid malignancy
- Calcitonin elevation > 100 pg/mL
- New thyroid nodule with concerning features
- Participant-reported warning symptoms
📝 Expedited Reporting (Within 7 Days)
- Confirmed thyroid pathology requiring intervention
- Moderate calcitonin elevation (50-100 pg/mL)
- Abnormal thyroid imaging findings
- Specialist referral for thyroid concerns
📝 Routine Reporting (Periodic Safety Updates)
- Mild calcitonin fluctuations within monitoring range
- Benign thyroid findings on evaluation
- Scheduled monitoring results
- Participant withdrawals for thyroid concerns
Documentation Standards:
Complete documentation should include:
- Detailed symptom description with onset timing
- Physical examination findings
- All laboratory results with reference ranges
- Imaging reports and interpretations
- Specialist consultation notes
- Clinical decision-making rationale
- Participant communication records
- Protocol modification decisions
This comprehensive approach to clinical monitoring ensures that the retatrutide thyroid cancer warning translates into practical vigilance, enabling early detection and appropriate management of any thyroid-related concerns.
Risk Communication and Ethical Considerations in Retatrutide Research
The retatrutide thyroid cancer warning raises important ethical considerations regarding risk communication, informed consent, and the balance between scientific advancement and participant protection. Navigating these ethical dimensions requires thoughtful approaches grounded in research ethics principles.
Principles of Effective Risk Communication
Communicating the thyroid cancer warning effectively requires balancing scientific accuracy with accessibility:
Key Communication Principles:
✅ Transparency Without Alarmism
- Present rodent findings honestly and completely
- Contextualize with species differences and human data from related compounds
- Avoid minimizing concerns while preventing undue alarm
- Acknowledge scientific uncertainties openly
✅ Accessible Language
- Translate technical terminology into plain language
- Use visual aids (diagrams, charts) to explain complex concepts
- Provide written materials at appropriate literacy levels
- Offer multiple communication formats (verbal, written, video)
✅ Contextualized Risk Information
- Compare thyroid cancer baseline risk (very rare) with potential research-related risk (unknown but likely minimal based on class data)
- Explain monitoring procedures that mitigate risk
- Describe early detection capabilities and treatment options
- Frame risk within broader research risk-benefit context
✅ Participant-Centered Approach
- Assess individual understanding and concerns
- Allow adequate time for questions and reflection
- Respect cultural and personal values affecting risk perception
- Support informed decision-making without coercion
Informed Consent Challenges and Best Practices
The retatrutide thyroid cancer warning presents specific informed consent challenges:
Challenge 1: Communicating Uncertain Risk
The disconnect between rodent findings and reassuring human data for related compounds creates communication complexity:
Best Practice Approach:
- Explicitly state: “Studies in rats and mice showed thyroid tumours, but extensive human experience with similar medications has not shown this problem in people”
- Explain species differences in thyroid biology
- Acknowledge: “We cannot be certain this will never occur in humans, which is why we monitor carefully”
- Emphasize monitoring as risk mitigation strategy
Challenge 2: Addressing Individual Risk Factors
Personal and family history create variable individual risk profiles:
Best Practice Approach:
- Conduct thorough family history assessment
- Explain why certain histories (MTC, MEN 2) lead to exclusion
- For participants without risk factors, contextualize their lower baseline risk
- Discuss how monitoring is tailored to individual risk profile
Challenge 3: Long-Term Uncertainty
Thyroid cancer’s long latency period means definitive safety data requires decades:
Best Practice Approach:
- Acknowledge: “Long-term effects beyond the research period are not fully known”
- Discuss plans for follow-up monitoring after research participation ends
- Provide contact information for reporting future concerns
- Commit to sharing new safety information as it emerges
Challenge 4: Research-Only Status
Retatrutide’s investigational status requires clear communication:
Best Practice Approach:
- Explicitly state: “This compound is not approved for any medical use and is strictly for research purposes”
- Clarify that participation offers no therapeutic benefit
- Explain the distinction between research and clinical treatment
- Ensure understanding that findings may not translate to personal health applications
Vulnerable Populations and Special Protections
Certain populations require additional safeguards in retatrutide research:
Populations Requiring Enhanced Protection:
🛡️ Individuals with Limited Health Literacy
- Additional educational sessions with teach-back methods
- Simplified consent documents with visual aids
- Family member or advocate involvement (with permission)
- Extended time for decision-making
🛡️ Economically Disadvantaged Participants
- Careful assessment of undue financial inducement
- Provision of monitoring and follow-up at no cost
- Transportation and time compensation that facilitates participation without coercion
- Clear communication that declining participation has no negative consequences
🛡️ Individuals with Previous Thyroid Conditions
- Enhanced baseline evaluation and specialist consultation
- Individualized risk-benefit assessment
- Potentially more intensive monitoring protocols
- Lower threshold for research discontinuation if concerns arise
🛡️ Research Staff and Laboratory Personnel
- Occupational health considerations for peptide handling
- Appropriate personal protective equipment
- Exposure monitoring if applicable
- Clear protocols for accidental exposure
Institutional Responsibilities and Oversight
Research institutions bear responsibility for ethical retatrutide research conduct:
Institutional Safeguards:
🏛️ Robust IRB Review
- Thyroid safety expertise on review panels
- Detailed protocol evaluation of monitoring adequacy
- Consent document review for clarity and completeness
- Ongoing oversight through continuing review process
🏛️ Data Safety Monitoring
- Independent review of accumulating safety data
- Authority to modify or halt research based on safety signals
- Periodic assessment of risk-benefit balance
- Communication channels with regulatory authorities
️ Investigator Training and Competence
- Required training in thyroid pathology recognition
- Familiarity with MTC and MEN 2 syndrome
- Competence in informed consent processes
- Understanding of ethical research principles
️ Participant Support Systems
- Access to medical consultation for concerns
- Clear pathways for reporting adverse events
- Psychological support if concerning findings emerge
- Financial protection for research-related injuries
Balancing Scientific Progress with Precaution
The retatrutide thyroid cancer warning exemplifies the tension between scientific advancement and precautionary principles:
Justification for Proceeding with Research Despite Warning:
✓ Substantial scientific interest in triple-agonist mechanisms ✓ Potential for significant metabolic research insights ✓ Reassuring human data from related GLP-1 receptor agonists ✓ Availability of effective monitoring strategies ✓ Ability to exclude highest-risk individuals ✓ Informed, voluntary participation by research subjects
Ongoing Ethical Obligations:
• Continuous reassessment of risk-benefit balance as data accumulates
• Prompt modification of protocols if safety signals emerge
• Transparent reporting of all thyroid-related findings
• Sharing of safety information across research community
• Commitment to long-term follow-up when feasible
• Contribution to scientific understanding of human thyroid safety
This ethical framework ensures that retatrutide research proceeds responsibly, respecting both the pursuit of scientific knowledge and the fundamental principle of participant protection.
Researchers sourcing peptides from reputable suppliers like PEPTIDE PRO should ensure their institutional frameworks support these ethical standards, with appropriate oversight and monitoring capabilities in place.
Future Directions: Evolving Understanding of Retatrutide Thyroid Safety
As research with retatrutide expands in 2026 and beyond, the scientific understanding of thyroid safety continues to evolve. Examining emerging research directions and knowledge gaps helps contextualize current precautions and anticipate future developments.
Ongoing Research Addressing Thyroid Safety Questions
Multiple research streams are actively investigating thyroid safety aspects of incretin-based peptides:
Mechanistic Studies in Human Tissue:
🔬 C-Cell Biology Investigations
- Advanced molecular characterization of human thyroid C-cells
- Single-cell RNA sequencing to definitively map GLP-1 receptor expression
- Functional studies of human C-cell responses to GLP-1 agonists
- Comparison of proliferative signaling pathways across species
Expected Impact: These studies will provide definitive molecular evidence regarding human C-cell responsiveness to GLP-1 receptor activation, potentially resolving the species extrapolation question.
🔬 Calcitonin Regulation Studies
- Detailed investigation of calcitonin secretion mechanisms in humans
- Identification of factors causing calcitonin elevation in non-MTC conditions
- Development of more specific MTC biomarkers beyond calcitonin
- Improved diagnostic algorithms for calcitonin interpretation
Expected Impact: Enhanced ability to distinguish clinically significant calcitonin elevations from benign fluctuations, reducing false-positive concerns.
Long-Term Epidemiological Surveillance
As triple-agonist compounds transition from research to potential therapeutic applications, large-scale surveillance becomes critical:
Post-Marketing Surveillance Frameworks:
📊 Registry-Based Studies
- Establishment of international registries tracking long-term outcomes
- Systematic collection of thyroid-related events
- Linkage with cancer registries for comprehensive MTC surveillance
- Comparison of exposed vs. unexposed populations
📊 Electronic Health Record (EHR) Studies
- Big data approaches analyzing millions of patient records
- Real-world evidence of thyroid outcomes with incretin-based therapies
- Detection of rare events through population-scale analysis
- Rapid signal detection systems for emerging safety concerns
📊 Comparative Effectiveness Research
- Head-to-head comparisons of thyroid safety across different incretin-based compounds
- Assessment of dose-response relationships in human populations
- Identification of genetic or demographic risk modifiers
- Evaluation of monitoring strategy effectiveness
Timeline Considerations: Definitive long-term thyroid safety data requires decades of follow-up, given MTC’s long latency period. However, intermediate analyses at 5-10 year intervals will provide progressively stronger evidence.
Genetic and Biomarker Research
Precision medicine approaches may enable more individualized thyroid risk assessment:
Genetic Risk Stratification:
🧬 RET Proto-Oncogene Screening
- Broader implementation of RET mutation testing
- Identification of novel genetic variants affecting MTC risk
- Integration of genetic risk scores into research eligibility criteria
- Personalized monitoring intensity based on genetic profile
🧬 Pharmacogenomic Studies
- Genetic variants affecting GLP-1 receptor signaling
- Individual differences in peptide metabolism and exposure
- Genetic modifiers of thyroid C-cell biology
- Polygenic risk scores for thyroid cancer susceptibility
Novel Biomarker Development:
🔍 Beyond Calcitonin
- Procalcitonin and calcitonin precursors as supplementary markers
- Chromogranin A and other neuroendocrine markers
- MicroRNA signatures specific to MTC
- Circulating tumour DNA (ctDNA) for ultra-sensitive detection
- Imaging biomarkers (molecular imaging, advanced ultrasound techniques)
Expected Impact: More precise risk stratification enabling tailored monitoring approaches, reducing unnecessary interventions for low-risk individuals while intensifying surveillance for higher-risk populations.
Regulatory Evolution and Guideline Development
Regulatory frameworks governing retatrutide research continue to evolve based on accumulating evidence:
Anticipated Regulatory Developments:
📋 Refined Monitoring Guidelines
- Evidence-based recommendations for calcitonin testing frequency
- Clarified thresholds for intervention based on calcitonin levels
- Standardized protocols for stimulation testing
- Harmonized international approaches to thyroid monitoring
📋 Risk Communication Standards
- Model informed consent language balancing accuracy and accessibility
- Required educational materials for research participants
- Standardized risk communication training for investigators
- Patient decision aids incorporating latest evidence
📋 Adaptive Regulatory Approaches
- Real-world evidence integration into safety assessments
- Accelerated reassessment of warnings as human data accumulates
- Risk-based regulatory frameworks tailored to individual compounds
- International regulatory harmonization for consistent standards
Integration of Artificial Intelligence and Advanced Analytics
Emerging technologies offer new approaches to thyroid safety monitoring:
AI Applications in Thyroid Safety:
🤖 Predictive Modeling
- Machine learning algorithms predicting individual thyroid cancer risk
- Integration of genetic, demographic, and exposure data
- Early warning systems for concerning patterns in monitoring data
- Optimization of monitoring schedules based on risk prediction
🤖 Image Analysis
- AI-enhanced thyroid ultrasound interpretation
- Automated nodule characterization and risk stratification
- Longitudinal tracking of thyroid changes
- Integration with pathology analysis for comprehensive assessment
🤖 Natural Language Processing
- Automated extraction of thyroid-related symptoms from research records
- Systematic analysis of adverse event narratives
- Early signal detection from unstructured clinical documentation
- Enhanced pharmacovigilance through comprehensive data mining
Knowledge Gaps and Research Priorities
Despite substantial progress, important questions remain:
Critical Unanswered Questions:
❓ Duration-Dependent Risk: Does thyroid cancer risk (if any in humans) increase with longer exposure duration, or is there a threshold beyond which risk plateaus?
❓ Dose-Response Relationships: Is there a dose below which thyroid effects are absent in humans, enabling safer research protocols?
❓ Reversibility: If C-cell changes occur in humans, are they reversible upon discontinuation?
❓ Combination Effects: Do multi-receptor agonists like retatrutide have different thyroid safety profiles than single-receptor agonists?
❓ Population Variability: Are certain ethnic, age, or gender groups at different risk levels?
Research Priorities for 2026-2030:
Definitive characterization of GLP-1 receptor expression on human thyroid C-cells using multiple methodologies
Long-term prospective cohort studies with systematic thyroid monitoring
Mechanistic studies explaining rodent-human species differences
Development and validation of improved MTC biomarkers
Genetic studies identifying individual susceptibility factors
Cost-effectiveness analyses of various monitoring strategies
Patient-reported outcome studies assessing impact of monitoring on quality of life
Implications for Current Research Practice
While awaiting these future developments, current research must proceed based on existing evidence:
Practical Guidance for 2026:
✅ Maintain current precautionary monitoring protocols ✅ Implement established contraindications (MTC/MEN 2 history) ✅ Stay informed about emerging evidence and guideline updates ✅ Participate in registries and surveillance systems when available ✅ Contribute to knowledge base through transparent safety reporting ✅ Balance precaution with scientific opportunity ✅ Engage in ongoing ethical reflection as evidence evolves
The retatrutide thyroid cancer warning, while based on preclinical findings, serves an important precautionary function while the scientific community works toward definitive answers about human thyroid safety. Responsible research practices honor both this precautionary principle and the pursuit of scientific knowledge that may ultimately benefit human health.
For research teams seeking premium research-grade peptides with comprehensive documentation and quality assurance, partnering with suppliers committed to transparency and safety supports these evolving best practices.
Responsible Research with Retatrutide in 2026
The retatrutide thyroid cancer warning represents a critical safety consideration for research professionals investigating this innovative triple-agonist peptide. While based primarily on rodent carcinogenicity studies that may not directly translate to human risk, this warning serves essential precautionary functions that responsible researchers must integrate into protocol design, monitoring strategies, and ethical frameworks.
Key Principles for Responsible Retatrutide Research
Synthesizing the comprehensive information presented throughout this guide, several core principles emerge:
🔬 Evidence-Based Risk Assessment
- Acknowledge rodent findings while contextualizing with species differences
- Incorporate reassuring human data from related GLP-1 receptor agonists
- Recognize remaining uncertainties requiring ongoing vigilance
- Maintain proportionate precautions aligned with current evidence
🔬 Rigorous Safety Monitoring
- Implement comprehensive baseline thyroid evaluation
- Conduct systematic calcitonin monitoring at appropriate intervals
- Establish clear protocols for abnormal finding management
- Document all thyroid-related observations thoroughly
🔬 Strict Exclusion Criteria
- Exclude individuals with personal or family MTC history
- Screen for MEN 2 syndrome and related genetic conditions
- Evaluate and address uncertain thyroid findings before research participation
- Apply evidence-based contraindications consistently
🔬 Transparent Risk Communication
- Provide complete, accurate information about thyroid cancer warning
- Use accessible language while maintaining scientific accuracy
- Support informed decision-making through comprehensive education
- Acknowledge uncertainties honestly and openly
🔬 Ethical Research Conduct
- Ensure robust institutional oversight and IRB review
- Protect vulnerable populations with enhanced safeguards
- Balance scientific advancement with participant protection
- Contribute to collective knowledge through transparent reporting
Practical Action Steps for Research Teams
Research teams planning or conducting retatrutide studies should take these concrete actions:
✅ Protocol Development Phase
- Incorporate thyroid safety considerations into research design
- Develop comprehensive monitoring schedules appropriate to study duration
- Establish clear exclusion criteria and screening procedures
- Create detailed adverse event management algorithms
- Secure institutional approvals with thyroid safety documentation
✅ Participant Recruitment Phase
- Develop clear, accessible informed consent materials
- Implement thorough baseline thyroid evaluation procedures
- Conduct comprehensive family history assessments
- Provide adequate time for questions and decision-making
- Document informed consent process meticulously
✅ Active Research Phase
- Execute monitoring protocols consistently and completely
- Maintain vigilance for clinical warning signs
- Respond promptly to abnormal findings with appropriate evaluation
- Document all thyroid-related observations systematically
- Maintain open communication channels with participants
✅ Data Analysis and Reporting Phase
- Analyze and report all thyroid-related safety data
- Contribute findings to broader safety databases and registries
- Publish results transparently, including negative findings
- Share lessons learned with research community
- Update protocols based on emerging evidence
The Broader Context: Advancing Metabolic Research Safely
Retatrutide represents an exciting frontier in metabolic research, offering unique opportunities to investigate triple-agonist mechanisms and their physiological effects. The thyroid cancer warning, while requiring serious attention, should not prevent scientifically valuable and ethically conducted research when appropriate safeguards are implemented.
The extensive human experience with related GLP-1 receptor agonists provides substantial reassurance that the rodent thyroid findings likely do not translate to significant human risk. However, the relatively limited human exposure to retatrutide specifically, combined with its novel triple-agonist pharmacology, justifies maintaining precautionary monitoring until more definitive long-term data accumulates.
Looking Forward: Contributing to Scientific Knowledge
Every research study involving retatrutide contributes to the collective understanding of its safety profile. By implementing rigorous thyroid monitoring, documenting findings comprehensively, and sharing results transparently, the research community progressively builds the evidence base that will ultimately determine whether the current thyroid cancer warning requires modification, intensification, or—potentially—relaxation based on reassuring human data.
Researchers have both the privilege and responsibility of advancing scientific knowledge while protecting those who participate in or are affected by their work. The retatrutide thyroid cancer warning exemplifies how precautionary principles, grounded in animal toxicology but informed by evolving human evidence, guide this balance.
Resources and Support for Researchers
Research teams seeking to conduct responsible retatrutide studies should leverage available resources:
Quality Peptide Sources: Partner with reputable suppliers like PEPTIDE PRO that provide research-grade peptides with comprehensive quality documentation, proper labeling, and expert support.
Institutional Support: Engage institutional review boards, data safety monitoring committees, and endocrinology consultants early in protocol development.
Professional Networks: Participate in research networks and professional societies focused on peptide research and metabolic investigations.
Regulatory Guidance: Consult current regulatory guidelines and maintain awareness of evolving recommendations.
Scientific Literature: Stay current with emerging research on incretin-based peptide safety and thyroid cancer epidemiology.
The retatrutide thyroid cancer warning reflects the scientific community’s commitment to precautionary principles in research involving novel compounds. While based on animal findings that may not directly predict human risk, this warning serves critical functions: protecting potentially vulnerable individuals, ensuring systematic safety monitoring, and maintaining ethical standards in research conduct.
As the research community continues investigating retatrutide’s metabolic effects and safety profile, each study conducted with appropriate thyroid monitoring contributes valuable data. The coming years will progressively clarify whether retatrutide follows the reassuring human safety pattern of related GLP-1 receptor agonists or presents unique considerations requiring modified approaches.
Until definitive long-term human data becomes available, responsible research practice demands implementing the precautions outlined in this guide: rigorous screening, systematic monitoring, transparent communication, and ethical oversight. These measures enable scientifically valuable research to proceed while honoring the fundamental principle of protecting research participants and advancing knowledge responsibly.
For research professionals committed to the highest standards of scientific excellence and ethical conduct, understanding and appropriately addressing the retatrutide thyroid cancer warning represents not a barrier to research, but rather an essential component of responsible investigation that serves both scientific progress and human welfare.
