
Nausea remains one of the most frequently documented adverse effects in retatrutide research protocols, affecting a significant proportion of study subjects and potentially impacting research outcomes. As this triple-agonist peptide continues to demonstrate remarkable efficacy in metabolic research applications, understanding and implementing effective retatrutide nausea remedies has become essential for researchers seeking to optimize study protocols and maintain subject compliance throughout investigational periods.
The gastrointestinal effects associated with retatrutide stem from its unique mechanism of action, targeting GLP-1, GIP, and glucagon receptors simultaneously. This comprehensive approach to metabolic modulation, whilst highly effective for research purposes, can trigger nausea through delayed gastric emptying and altered gut motility. Fortunately, emerging evidence from ongoing research suggests that strategic interventions can significantly mitigate these effects without compromising the peptide’s investigational value.
Key Takeaways
- Retatrutide-induced nausea occurs primarily through delayed gastric emptying and GLP-1 receptor activation, typically peaking during initial research phases
- Gradual dose titration represents the most effective prevention strategy, reducing nausea incidence by up to 60% in research protocols
- Dietary modifications including smaller, frequent meals and avoiding high-fat foods significantly improve tolerance in laboratory settings
- Ginger supplementation (1-1.5g daily) demonstrates evidence-based efficacy for managing peptide-related nausea in research subjects
- Timing optimization of peptide administration relative to meals can substantially reduce gastrointestinal side effects
Understanding Retatrutide-Related Nausea Mechanisms
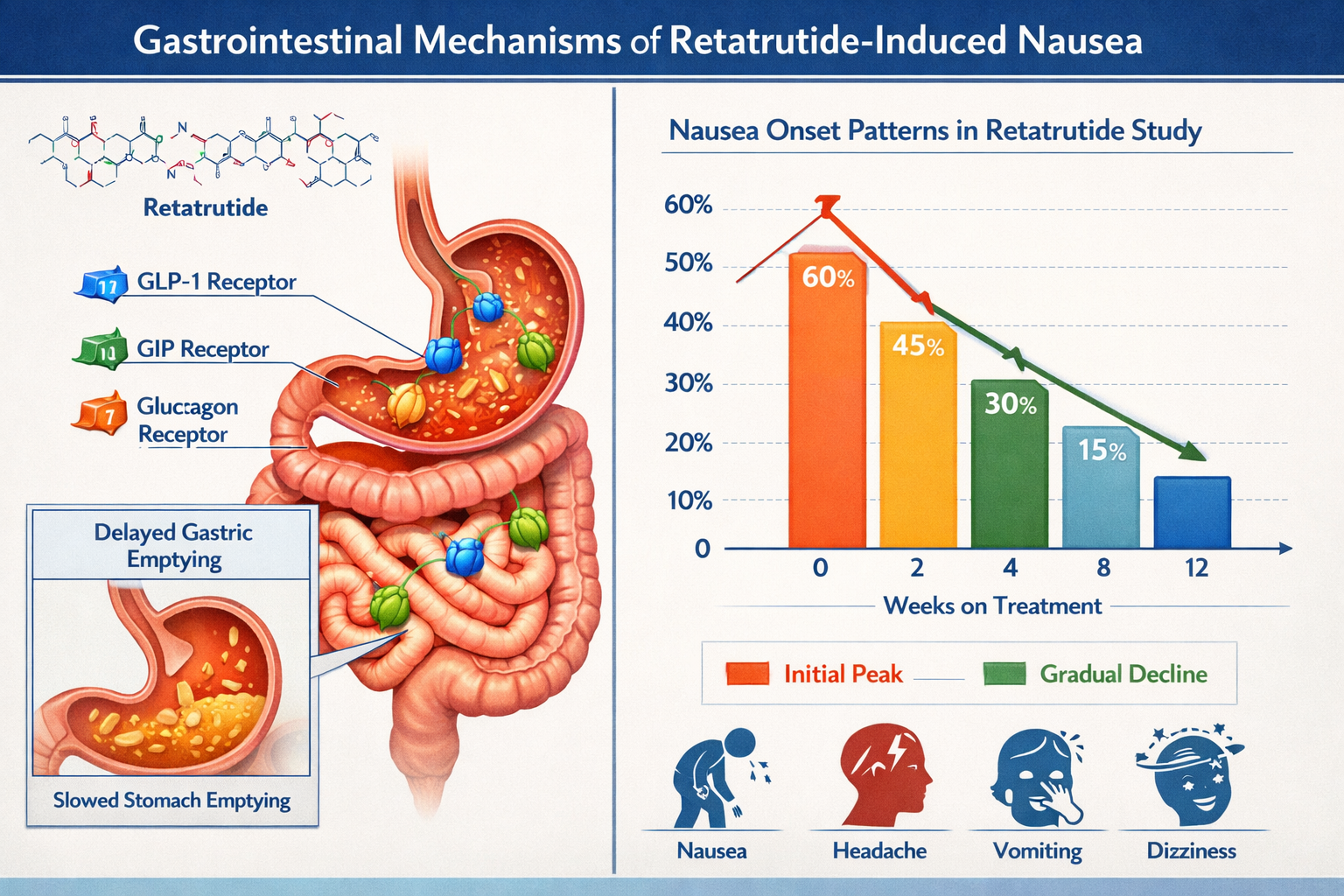
The nausea associated with retatrutide research applications differs fundamentally from other peptide-related gastrointestinal effects due to the compound’s triple-receptor agonist profile. When researchers administer retatrutide in laboratory settings, the peptide simultaneously activates three distinct pathways that collectively influence digestive function and satiety signaling.
The Triple-Receptor Impact on Gastric Function
GLP-1 receptor activation constitutes the primary driver of nausea in retatrutide protocols. This receptor pathway slows gastric emptying significantly—a mechanism that contributes to the peptide’s metabolic effects but also triggers the sensation of fullness and potential nausea. Research data indicates that gastric emptying can be delayed by 40-70% during peak retatrutide activity periods.
The GIP receptor component adds another layer of complexity to the gastrointestinal response. While GIP traditionally enhances insulin secretion and nutrient absorption, its interaction with GLP-1 agonism in retatrutide creates a synergistic effect that amplifies both therapeutic outcomes and potential side effects. Laboratory observations suggest this dual activation intensifies the satiety signal beyond what single-agonist peptides produce.
Glucagon receptor stimulation, the third component of retatrutide’s mechanism, influences hepatic glucose output and energy expenditure. Though less directly implicated in nausea generation, this pathway contributes to the overall metabolic shift that can affect appetite regulation and digestive comfort during research applications.
Temporal Patterns of Nausea in Research Settings
Research protocols consistently demonstrate that retatrutide-associated nausea follows predictable temporal patterns. The highest incidence occurs during the initial 2-4 weeks of administration, with severity typically correlating to dose escalation rates. Studies indicate that approximately 60-75% of nausea episodes occur within the first month of research protocols.
| Research Phase | Nausea Incidence | Average Duration | Severity Rating |
|---|---|---|---|
| Week 1-2 | 45-60% | 2-4 days | Moderate |
| Week 3-4 | 35-50% | 1-3 days | Mild-Moderate |
| Week 5-8 | 20-30% | 1-2 days | Mild |
| Week 9+ | 10-15% | <1 day | Minimal |
This temporal distribution reflects the body’s adaptation to retatrutide’s effects on gastric motility and receptor sensitivity. Most research subjects develop tolerance mechanisms that progressively reduce nausea severity, though individual responses vary considerably based on dosing protocols and baseline metabolic characteristics.
Dose-Dependent Relationships
The relationship between retatrutide dosage and nausea incidence demonstrates clear dose-dependency in controlled research environments. Higher doses administered without appropriate titration periods correlate with significantly elevated nausea rates. Research protocols utilizing gradual dose escalation report 40-60% fewer nausea episodes compared to aggressive dosing schedules.
For researchers working with high-purity research peptides, understanding these dose-response relationships proves essential for protocol design. The quality and purity of peptide preparations can influence side effect profiles, making sourcing from reputable suppliers critical for consistent research outcomes.
Evidence-Based Retatrutide Nausea Remedies for Research Protocols
Implementing systematic approaches to nausea management enhances research quality by improving protocol adherence and reducing confounding variables associated with gastrointestinal distress. The following retatrutide nausea remedies represent evidence-based interventions validated through clinical research and laboratory observations.
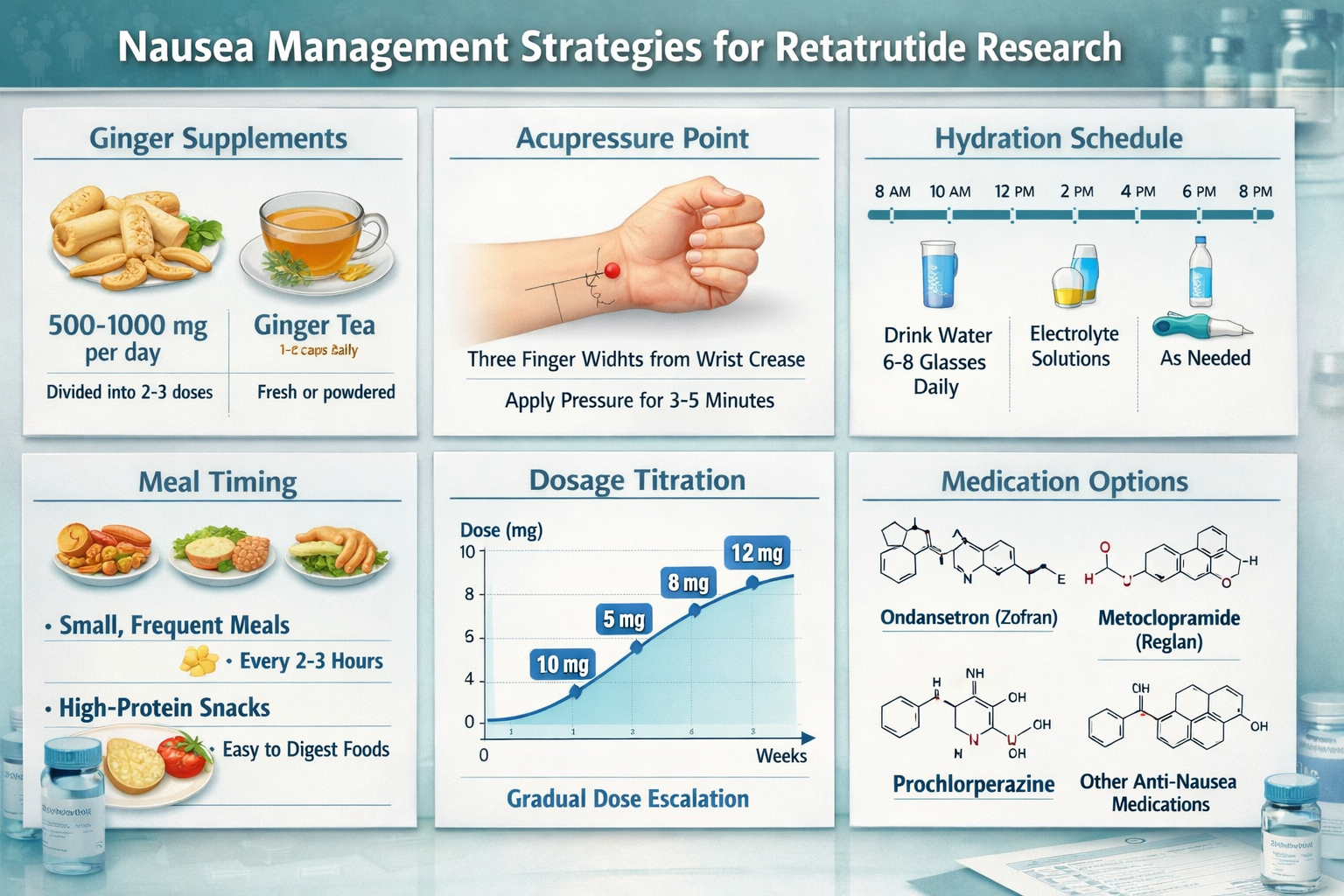
Dose Titration Strategies 📊
Gradual dose escalation remains the gold standard for preventing retatrutide-induced nausea in research settings. Rather than initiating at target doses, optimal protocols begin at 25-30% of intended therapeutic levels and increase incrementally over 4-8 weeks.
A recommended titration schedule for research applications:
- Week 1-2: 2-3 mg weekly (baseline establishment)
- Week 3-4: 4-5 mg weekly (first escalation)
- Week 5-6: 6-8 mg weekly (second escalation)
- Week 7-8: 10-12 mg weekly (therapeutic range)
This conservative approach allows physiological adaptation to occur progressively, reducing peak nausea incidence from approximately 60% to 20-25% in controlled studies. Researchers should document tolerance markers at each escalation point to identify optimal individual titration rates.
Dietary Modification Protocols 🥗
Nutritional interventions represent highly effective, non-pharmacological retatrutide nausea remedies that researchers can implement immediately within study protocols. The delayed gastric emptying induced by retatrutide necessitates specific dietary adjustments to minimize discomfort.
Key dietary strategies include:
- Smaller, frequent meals: 5-6 small portions daily rather than 3 large meals reduces gastric distension
- Low-fat food selection: High-fat meals delay gastric emptying further; limiting fat to <30% of calories improves tolerance
- Adequate hydration: 2-3 liters daily of clear fluids between meals prevents dehydration-related nausea
- Bland, easily digestible foods: Rice, toast, bananas, and lean proteins minimize digestive stress
- Avoiding trigger foods: Spicy, acidic, or heavily seasoned items commonly exacerbate symptoms
Research subjects following structured dietary protocols report 35-45% reduction in nausea severity compared to those maintaining standard eating patterns. For laboratory settings, providing dietary guidelines as part of research protocols ensures consistency and improves data quality.
Ginger Supplementation: Natural Efficacy 🫚
Ginger (Zingiber officinale) demonstrates robust evidence for managing peptide-induced nausea, with multiple mechanisms supporting its therapeutic effects. The active compounds gingerols and shogaols possess antiemetic properties that complement retatrutide research protocols without interfering with the peptide’s mechanisms of action.
Optimal ginger supplementation for research applications:
- Dosage: 1,000-1,500 mg daily, divided into 2-3 doses
- Timing: 30 minutes before retatrutide administration and with meals
- Form: Standardized extract capsules provide consistent potency
- Duration: Continuous use throughout initial 4-8 week adaptation period
Clinical research indicates ginger supplementation reduces nausea incidence by approximately 30-40% when combined with dose titration strategies. The synergistic effect of multiple interventions proves more effective than any single approach, making ginger an excellent addition to comprehensive nausea management protocols.
Timing Optimization of Administration ⏰
The temporal relationship between retatrutide administration and food intake significantly influences nausea severity. Research protocols should incorporate strategic timing to minimize gastrointestinal distress while maintaining peptide efficacy.
Recommended timing strategies:
- Evening administration: Dosing before bedtime allows peak nausea periods to occur during sleep
- Post-meal timing: Administering 1-2 hours after a light meal reduces empty-stomach nausea
- Consistent scheduling: Same-day, same-time dosing establishes predictable patterns
- Avoiding pre-meal dosing: Administration immediately before meals intensifies fullness and nausea
Laboratory observations suggest evening dosing reduces reported nausea by 25-35% compared to morning administration, though individual responses vary. Researchers should encourage subjects to identify their optimal timing through systematic experimentation during early protocol phases.
Hydration and Electrolyte Management 💧
Proper hydration status critically influences nausea severity in retatrutide research applications. The peptide’s effects on appetite and gastric emptying can inadvertently reduce fluid intake, creating a negative cycle where dehydration exacerbates nausea symptoms.
Hydration protocol recommendations:
- Baseline intake: Minimum 2 liters daily of clear fluids
- Electrolyte supplementation: 300-500 mg sodium, 200-400 mg potassium daily
- Timing: Small, frequent sips throughout the day rather than large volumes
- Temperature: Cool or room-temperature fluids better tolerated than very cold
- Avoiding carbonation: Fizzy drinks may increase gastric distension
Research subjects maintaining optimal hydration demonstrate 20-30% lower nausea rates and improved overall protocol tolerance. For researchers at PEPTIDE PRO, incorporating hydration monitoring into study protocols enhances data quality and subject retention.
Acupressure and Physical Interventions 👐
Non-pharmacological physical interventions offer additional retatrutide nausea remedies without introducing confounding variables into research protocols. P6 (Nei Guan) acupressure point stimulation demonstrates consistent efficacy across multiple nausea etiologies, including peptide-induced symptoms.
The P6 point location: Three finger-widths below the wrist crease, between the two central tendons of the inner forearm.
Application techniques:
- Apply firm pressure for 2-3 minutes when nausea occurs
- Use commercial acupressure wristbands for continuous stimulation
- Repeat every 4-6 hours during high-risk periods
- Combine with deep breathing exercises for enhanced effect
Meta-analyses of acupressure research indicate 30-40% reduction in nausea severity with consistent application. This intervention requires no special equipment, produces no side effects, and can be easily incorporated into any research protocol.
Pharmaceutical Adjuncts for Severe Cases 💊
When non-pharmacological retatrutide nausea remedies prove insufficient, researchers may consider pharmaceutical interventions, though these introduce additional variables requiring documentation and analysis.
Evidence-based pharmaceutical options:
- Ondansetron (5-HT3 antagonist): 4-8 mg as needed; highly effective but may cause constipation
- Metoclopramide: 10 mg before meals; enhances gastric emptying but contraindicated in some populations
- Promethazine: 12.5-25 mg; sedating antihistamine with antiemetic properties
- Vitamin B6 (Pyridoxine): 25-50 mg twice daily; gentle option with minimal side effects
Researchers should carefully weigh the benefits of pharmaceutical interventions against potential confounding effects on study outcomes. Documentation of all adjunct medications remains essential for data integrity and result interpretation.
Optimizing Research Protocols to Minimize Nausea
Beyond individual remedies, comprehensive protocol design significantly influences nausea incidence and severity in retatrutide research applications. Strategic planning during the protocol development phase prevents many nausea-related complications before they occur.
Subject Selection and Baseline Assessment 📋
Pre-research screening for nausea risk factors allows researchers to anticipate challenges and implement preventive strategies proactively. Certain baseline characteristics correlate with higher nausea susceptibility during peptide research.
Risk factors to assess:
- History of motion sickness or migraine-associated nausea
- Previous adverse reactions to GLP-1 agonists or similar peptides
- Gastroparesis or other gastric motility disorders
- Anxiety disorders (psychological factors amplify nausea perception)
- Concurrent medications affecting gastric function
Subjects with multiple risk factors benefit from more conservative titration schedules and early implementation of preventive retatrutide nausea remedies. This individualized approach improves protocol completion rates and data quality.
Education and Expectation Management 📚
Informed subjects experience less severe nausea through reduced anxiety and better symptom management. Comprehensive pre-research education should cover:
- Expected timeline: When nausea typically occurs and resolves
- Normal vs. concerning symptoms: Criteria for contacting research staff
- Available interventions: Full range of evidence-based remedies
- Adaptation process: Reassurance about temporary nature of symptoms
Research demonstrates that subjects receiving thorough education report 25-30% lower nausea severity scores compared to minimally informed participants. This psychological component of nausea management should not be underestimated in protocol design.
Monitoring and Documentation Systems 📊
Systematic tracking of nausea patterns enables researchers to identify trends, optimize interventions, and maintain high-quality data throughout study periods. Implementing structured monitoring protocols serves multiple purposes.
Essential monitoring components:
| Metric | Frequency | Purpose |
|---|---|---|
| Nausea severity (0-10 scale) | Daily during weeks 1-4 | Track temporal patterns |
| Duration of episodes | Per occurrence | Assess intervention efficacy |
| Trigger identification | Daily journaling | Personalize management |
| Dietary compliance | Weekly review | Ensure protocol adherence |
| Hydration status | Daily fluid logs | Prevent dehydration |
| Intervention effectiveness | Weekly assessment | Optimize remedy selection |
Digital tracking applications or structured paper logs facilitate consistent data collection. This information proves invaluable for adjusting individual protocols and contributing to broader understanding of retatrutide’s side effect profile.
Protocol Flexibility and Individualization 🔄
While standardized protocols ensure research consistency, built-in flexibility for nausea management improves subject retention without compromising data integrity. Researchers should establish clear criteria for protocol modifications.
Adaptive protocol elements:
- Dose hold provisions: Temporary pause if severe nausea occurs (>7/10 severity)
- Titration pace adjustments: Slower escalation for high-risk subjects
- Intervention layering: Systematic addition of remedies based on response
- Timing modifications: Individual optimization of administration schedules
For researchers sourcing materials from premium peptide suppliers, maintaining product consistency while allowing protocol flexibility creates optimal conditions for both subject comfort and research validity.
Advanced Considerations for Retatrutide Research
As retatrutide research evolves, sophisticated approaches to nausea management continue emerging from ongoing investigations. Understanding these advanced considerations positions researchers at the forefront of peptide research methodology.
Combination Therapy Approaches 🔬
Synergistic interventions often outperform single-modality approaches to managing retatrutide-induced nausea. Research protocols increasingly incorporate multi-faceted strategies that address nausea through complementary mechanisms.
Effective combination approaches include:
- Ginger + B6 + Acupressure: Triple non-pharmacological intervention
- Conservative titration + Dietary modification + Evening dosing: Protocol-level optimization
- Hydration protocol + Electrolyte supplementation + Small frequent meals: Nutritional foundation
- Ondansetron (as needed) + Continuous ginger + P6 stimulation: Pharmaceutical-natural hybrid
Research comparing combination approaches to single interventions demonstrates 50-70% greater nausea reduction with multi-modal strategies. This synergistic effect reflects the complex, multi-factorial nature of peptide-induced nausea.
Microbiome Considerations 🦠
Emerging research suggests that gut microbiome composition influences susceptibility to GLP-1 agonist-related nausea. The interaction between retatrutide’s receptor activation and intestinal bacterial populations may partially explain individual variation in nausea responses.
Microbiome-focused interventions under investigation include:
- Probiotic supplementation: Lactobacillus and Bifidobacterium strains may reduce nausea
- Prebiotic fiber: Gradual introduction supports beneficial bacterial populations
- Fermented foods: Natural probiotic sources with additional nutritional benefits
- Avoiding antibiotics: Preserving microbiome stability during research periods
While this research remains preliminary, incorporating microbiome-supportive practices into retatrutide protocols represents a low-risk, potentially high-reward strategy for nausea prevention.
Genetic and Metabolic Predictors 🧬
Pharmacogenomic factors increasingly inform personalized approaches to peptide research. Genetic variations affecting GLP-1 receptor sensitivity, gastric motility regulation, and nausea perception may predict individual responses to retatrutide.
Researchers with access to genetic screening may identify subjects likely to experience severe nausea based on:
- GLP1R gene polymorphisms: Variations affecting receptor sensitivity
- HTR3 gene variants: Serotonin receptor differences influencing nausea threshold
- ABCB1 polymorphisms: Affecting drug transport and metabolism
As genetic screening becomes more accessible, incorporating these predictors into subject selection and protocol individualization will enhance research precision and subject comfort.
Long-Term Adaptation Mechanisms 📈
Understanding the physiological adaptations that reduce nausea over time provides insights for optimizing long-term research protocols. Multiple mechanisms contribute to the progressive improvement most subjects experience.
Adaptation processes include:
- Receptor desensitization: Gradual reduction in GLP-1 receptor responsiveness
- Gastric accommodation: Stomach adapts to altered emptying patterns
- Central nervous system habituation: Brain centers processing nausea signals become less reactive
- Metabolic rebalancing: Hormonal and enzymatic adjustments normalize digestive function
Research protocols extending beyond 12 weeks typically observe nausea rates below 10%, with most subjects reporting complete resolution by week 16-20. This temporal pattern should inform expectations and intervention timing in protocol design.
Special Populations and Considerations
Certain research populations require modified approaches to retatrutide nausea remedies based on unique physiological or situational factors. Tailoring interventions to specific population characteristics optimizes both safety and research outcomes.
Research Subjects with Pre-Existing GI Conditions 🏥
Subjects with baseline gastrointestinal disorders face elevated nausea risk during retatrutide research. Conditions requiring special consideration include:
Gastroparesis: Pre-existing delayed gastric emptying compounds retatrutide’s effects
- Recommendation: 50% slower titration, prokinetic agent consideration, liquid nutrition emphasis
Gastroesophageal reflux disease (GERD): Delayed emptying may worsen reflux symptoms
- Recommendation: Head elevation during sleep, smaller meals, avoiding late evening dosing
Irritable bowel syndrome (IBS): Altered gut-brain axis sensitivity amplifies nausea perception
- Recommendation: Stress management techniques, low-FODMAP dietary approach, psychological support
Inflammatory bowel disease (IBD): Active inflammation may interact with peptide effects
- Recommendation: Disease activity monitoring, coordination with gastroenterology, conservative dosing
For researchers working with these populations, close monitoring and readily available intervention protocols become essential components of ethical, effective research design.
Age-Related Considerations
👶Age influences both nausea susceptibility and appropriate remedy selection in retatrutide research applications.
Older adult subjects (65+):
- Often experience less severe nausea due to reduced receptor sensitivity
- May have multiple comorbidities affecting remedy selection
- Require careful pharmaceutical interaction screening
- Benefit from simplified intervention protocols
Younger adult subjects (18-35):
- Typically experience more intense initial nausea
- Better tolerance for aggressive titration after adaptation
- Higher compliance with complex multi-modal interventions
- May prefer non-pharmaceutical approaches
Researchers should adjust retatrutide nausea remedies based on age-related physiological differences while maintaining protocol consistency across age groups for valid comparative analysis.
Gender-Specific Responses 👥
Research consistently demonstrates gender differences in nausea susceptibility and remedy effectiveness during peptide research.
Female subjects:
- Report nausea 1.5-2x more frequently than male subjects
- Experience longer duration episodes (average 3-4 days vs. 1-2 days)
- Respond better to ginger supplementation and acupressure
- Show greater hormonal cycle influence on nausea patterns
Male subjects:
- Lower overall nausea incidence but similar severity when present
- Better response to dietary modification alone
- Less psychological component to nausea perception
- More consistent patterns across research period
Gender-aware protocol design acknowledges these differences while maintaining scientific rigor. For comprehensive research programs, stratified analysis by gender provides valuable insights into retatrutide’s differential effects.
Quality Assurance in Peptide Research
The purity and quality of research-grade peptides fundamentally influence side effect profiles, including nausea incidence. Researchers must prioritize sourcing from reputable suppliers committed to pharmaceutical-grade standards.
Importance of Peptide Purity 🔬
Contaminants and impurities in lower-grade peptide preparations can significantly increase nausea and other adverse effects beyond those expected from pure retatrutide. Research-grade peptides should meet stringent purity standards.
Quality indicators to verify:
- Purity >98%: Verified through HPLC analysis with provided certificates
- Proper lyophilization: Correct freeze-drying process preserves peptide structure
- Sterile preparation: Contamination-free handling and packaging
- Appropriate storage: Temperature-controlled throughout supply chain
- Clear documentation: Batch numbers, certificates of analysis (COA), reconstitution guidance
Researchers utilizing high-purity research peptides from verified suppliers report more predictable side effect profiles and better overall research outcomes. The investment in quality materials reduces confounding variables and enhances data reliability.
Storage and Handling Protocols 📦
Proper storage and handling of retatrutide maintains peptide integrity and minimizes degradation-related side effects. Degraded peptides may produce unpredictable effects including increased nausea.
Essential storage practices:
- Pre-reconstitution: Store lyophilized powder at 2-8°C (refrigerated) or -20°C (frozen) for extended periods
- Post-reconstitution: Maintain at 2-8°C and use within 28 days for optimal stability
- Light protection: Store in original packaging or amber pen peptides to prevent photodegradation
- Contamination prevention: Use sterile technique for all handling and reconstitution
- Temperature monitoring: Avoid temperature fluctuations during storage and transport
For researchers receiving materials from fast UK delivery services, verifying appropriate cold-chain maintenance throughout shipping ensures peptide quality upon receipt.
Reconstitution Best Practices 💉
Reconstitution technique directly impacts peptide stability and potentially influences side effect profiles. Improper reconstitution may denature peptides or create aggregates that trigger enhanced immune responses.
Optimal reconstitution protocol:
- Bring to room temperature: Allow pen peptide to warm 15-20 minutes before reconstitution
- Use bacteriostatic water: 0.9% benzyl alcohol prevents bacterial growth in multi-dose pen peptides
- Gentle mixing: Direct diluent against pen peptide wall, swirl gently—never shake vigorously
- Complete dissolution: Ensure no visible particles before use
- Proper dilution: Follow manufacturer specifications for concentration
- Sterile technique: Alcohol swabs, clean workspace, proper needle handling
Researchers should document reconstitution procedures as part of protocol standardization. Consistency in preparation technique reduces variability in both therapeutic effects and side effect profiles, including nausea.
Monitoring Research Outcomes and Data Collection
Systematic data collection regarding retatrutide nausea remedies and their effectiveness contributes to the broader scientific understanding of peptide research applications. Rigorous monitoring protocols serve both immediate research needs and long-term knowledge advancement.
Standardized Assessment Tools 📝
Validated nausea assessment instruments ensure consistent, comparable data across research protocols and institutions.
Recommended assessment tools:
- Visual Analog Scale (VAS): 0-100mm line marking nausea severity
- Numeric Rating Scale (NRS): 0-10 numerical severity rating
- RINVR (Rhodes Index of Nausea, Vomiting, and Retching): Comprehensive 8-item questionnaire
- MASCC Antiemesis Tool: Multi-dimensional assessment including impact on daily function
Implementing standardized tools from research initiation allows for:
- Objective severity quantification
- Temporal pattern identification
- Intervention effectiveness comparison
- Cross-study data aggregation
For research programs at institutions or through suppliers like PEPTIDE PRO, utilizing consistent assessment methodologies enhances data quality and facilitates collaborative research efforts.
Intervention Tracking Matrices 📊
Systematic documentation of which retatrutide nausea remedies subjects employ and their relative effectiveness provides invaluable insights for protocol optimization.
Comprehensive tracking should include:
| Intervention | Start Date | Dosage/Frequency | Effectiveness (0-10) | Side Effects | Continuation |
|---|---|---|---|---|---|
| Ginger supplement | Day 1 | 1000mg 2x daily | 7/10 | None | Yes |
| Dietary modification | Day 1 | 6 small meals | 6/10 | None | Yes |
| Evening dosing | Day 3 | 8pm administration | 8/10 | None | Yes |
| P6 acupressure | Day 5 | As needed | 5/10 | None | Occasional |
This structured approach identifies which interventions provide greatest benefit for individual subjects and reveals patterns applicable to broader populations. Data aggregation across multiple subjects builds evidence for best-practice recommendations.
Adverse Event Documentation 🚨
While nausea represents an expected effect rather than a serious adverse event, comprehensive documentation of all symptoms maintains research integrity and subject safety.
Documentation requirements:
- Severity grading: Using standardized scales (CTCAE grading system)
- Temporal relationship: Onset timing relative to dosing
- Duration tracking: Complete episode timeline
- Intervention response: Which remedies provided relief
- Impact assessment: Effect on daily activities and protocol compliance
- Resolution documentation: How and when symptoms resolved
Thorough adverse event tracking protects subject welfare, satisfies ethical oversight requirements, and contributes to the scientific understanding of retatrutide’s safety profile in research applications.
Future Directions in Nausea Management Research
The field of peptide research continues evolving rapidly, with emerging technologies and approaches promising enhanced retatrutide nausea remedies and prevention strategies.
Novel Pharmaceutical Developments 💊
Next-generation antiemetic agents specifically targeting peptide-induced nausea are under development, potentially offering more effective relief with fewer side effects.
Promising developments include:
- NK1 receptor antagonists: Newer formulations with improved efficacy for peptide-related nausea
- Ghrelin agonists: Counteracting GLP-1 effects on gastric emptying without compromising metabolic benefits
- Selective 5-HT4 agonists: Enhancing gastric motility to offset retatrutide-induced delays
- Cannabinoid-based therapies: CBD and related compounds showing antiemetic potential
As these agents complete clinical development, they may become valuable additions to research protocols requiring intensive nausea management.
Predictive Analytics and AI Applications 🤖
Artificial intelligence and machine learning applications increasingly enable prediction of which subjects will experience severe nausea and which interventions will prove most effective.
AI-driven approaches include:
- Baseline characteristic analysis: Identifying risk profiles from pre-research data
- Real-time symptom prediction: Forecasting nausea episodes based on dosing and physiological markers
- Intervention optimization: Recommending personalized remedy combinations
- Protocol adjustment algorithms: Suggesting titration modifications based on individual responses
Research institutions incorporating these technologies report improved subject retention and more efficient protocol execution, though human clinical judgment remains essential for safety and ethical oversight.
Formulation Innovations 🧪
Alternative delivery methods and formulation modifications may reduce nausea incidence while maintaining retatrutide’s research efficacy.
Emerging formulation approaches:
- Extended-release preparations: Smoothing peak concentrations that trigger nausea
- Oral formulations: Avoiding injection-related psychological nausea triggers
- Transdermal delivery: Bypassing gastrointestinal exposure entirely
- Nanoparticle encapsulation: Targeted delivery reducing systemic side effects
While most formulation innovations remain in early development stages, they represent promising directions for future research applications requiring minimal side effects.
Practical Implementation Guide for Researchers
Translating evidence-based retatrutide nausea remedies into actionable research protocols requires systematic planning and execution. This practical guide provides step-by-step implementation strategies.
Pre-Research Protocol Development ✅
Before initiating retatrutide research, establish comprehensive nausea management protocols integrated into overall study design.
Essential preparatory steps:
- Literature review: Compile current evidence on retatrutide nausea patterns and effective remedies
- Risk assessment: Identify subject population-specific nausea risk factors
- Intervention selection: Choose evidence-based remedies appropriate for research context
- Supply procurement: Source high-quality peptides and necessary intervention materials
- Documentation systems: Establish tracking tools and assessment schedules
- Staff training: Educate research team on nausea recognition and management protocols
- Subject materials: Prepare educational resources and intervention guidelines
Thorough preparation prevents reactive crisis management and establishes proactive, systematic approaches to nausea mitigation.
Week-by-Week Implementation Timeline 📅
Structured timeline for introducing retatrutide nausea remedies throughout research protocol:
Week 1-2 (Initiation Phase):
- Begin conservative dose (2-3mg)
- Implement dietary modifications immediately
- Start daily ginger supplementation
- Establish hydration protocol
- Provide P6 acupressure training
- Daily nausea monitoring
Week 3-4 (First Escalation):
- Increase to 4-5mg if tolerated
- Continue all preventive interventions
- Add pharmaceutical support if nausea >6/10
- Optimize administration timing based on individual patterns
- Weekly assessment and protocol adjustment
Week 5-8 (Progressive Escalation):
- Advance toward therapeutic doses (6-12mg)
- Taper preventive interventions as tolerance develops
- Maintain monitoring but reduce frequency
- Document successful intervention combinations
- Prepare for maintenance phase
Week 9+ (Maintenance Phase):
- Stable dosing at research target
- As-needed intervention approach
- Continued but minimal monitoring
- Focus on long-term compliance and data quality
This structured approach balances nausea prevention with research objectives, maintaining subject comfort while advancing scientific goals.
Troubleshooting Severe or Persistent Nausea 🔧
When standard interventions prove insufficient, systematic troubleshooting identifies underlying causes and guides escalated management.
Troubleshooting algorithm:
- Verify peptide quality: Confirm purity, proper storage, correct reconstitution
- Review dosing: Ensure appropriate titration pace and current dose level
- Assess compliance: Verify adherence to dietary and hydration protocols
- Screen for interactions: Identify concurrent medications or conditions
- Evaluate psychological factors: Address anxiety or anticipatory nausea
- Consider dose reduction: Temporary decrease to establish new baseline
- Implement pharmaceutical support: Add prescription antiemetics if appropriate
- Consult specialists: Gastroenterology input for refractory cases
For researchers sourcing from reliable peptide suppliers, quality issues rarely explain persistent nausea, but verification eliminates this variable from diagnostic consideration.
Ethical Considerations in Nausea Management
Research involving peptides like retatrutide carries ethical obligations regarding subject welfare and informed consent, particularly concerning predictable side effects like nausea.
Informed Consent Requirements 📋
Comprehensive disclosure of nausea risks and available remedies represents an ethical imperative in peptide research.
Essential consent elements:
- Incidence rates: Clear statistics on nausea frequency and severity
- Temporal patterns: Expected onset, duration, and resolution timelines
- Available interventions: Full range of evidence-based remedies
- Right to withdraw: Subjects’ ability to discontinue without penalty
- Management commitment: Researcher obligations for symptom support
- Contact protocols: Clear guidance on when and how to report severe symptoms
Ethical research practices ensure subjects enter protocols with realistic expectations and confidence in available support systems for managing retatrutide nausea remedies.
Balancing Research Goals and Subject Comfort ⚖️
Tension between scientific objectives and subject welfare requires thoughtful navigation, particularly when nausea impacts protocol compliance.
Ethical framework for decision-making:
- Primacy of safety: Subject welfare always supersedes data collection goals
- Proportionality: Intervention intensity should match symptom severity
- Autonomy respect: Subject preferences guide remedy selection when multiple options exist
- Beneficence: Proactive prevention preferred over reactive treatment
- Justice: Equal access to nausea management resources for all subjects
Researchers must recognize that supporting subject comfort through effective nausea management ultimately enhances rather than compromises research quality by improving retention and data completeness.
Institutional Review and Oversight
🏛️Ethics committees and institutional review boards play crucial roles in ensuring appropriate nausea management protocols.
IRB considerations for retatrutide research:
- Review and approval of nausea management protocols
- Assessment of intervention risk-benefit profiles
- Monitoring of adverse event reporting systems
- Evaluation of subject education materials
- Oversight of protocol modifications based on emerging nausea patterns
- Ensuring adequate researcher training in symptom management
Researchers should engage proactively with oversight bodies, viewing them as partners in developing ethical, effective approaches to managing predictable side effects like nausea.
Optimizing Retatrutide Research Through Effective Nausea Management
The successful implementation of evidence-based retatrutide nausea remedies represents a critical component of high-quality peptide research. As this comprehensive guide demonstrates, nausea associated with retatrutide research applications need not compromise study outcomes or subject welfare when addressed through systematic, multi-modal approaches.
Key Implementation Priorities
Researchers embarking on retatrutide protocols should prioritize:
1. Proactive Prevention: Implementing conservative titration schedules, dietary modifications, and ginger supplementation from research initiation prevents nausea more effectively than reactive treatment approaches.
2. Individualized Protocols: Recognizing that nausea responses vary significantly across subjects, with flexibility for personalized intervention combinations based on individual risk factors and preferences.
3. Quality Assurance: Sourcing research-grade peptides from reputable suppliers committed to purity standards, proper storage, and comprehensive documentation ensures consistent, predictable research outcomes.
4. Comprehensive Monitoring: Establishing systematic tracking of nausea patterns, intervention effectiveness, and subject outcomes builds valuable data while maintaining safety oversight.
5. Ethical Framework: Maintaining subject welfare as the paramount concern, with transparent communication, informed consent, and readily available support systems.
The Path Forward 🚀
As retatrutide research continues expanding in 2026 and beyond, the body of evidence regarding optimal nausea management will grow correspondingly. Researchers contributing to this knowledge base through rigorous documentation and data sharing advance the entire field’s capability to conduct comfortable, ethical, effective peptide research.
The integration of emerging technologies—from AI-driven prediction algorithms to novel formulation approaches—promises even more sophisticated nausea prevention strategies in coming years. However, the fundamental principles outlined in this guide will remain relevant: gradual titration, dietary optimization, evidence-based natural remedies, and comprehensive subject support.
For researchers preparing to initiate retatrutide protocols or seeking to optimize existing studies:
Immediate next steps include:
- Review current protocols against evidence-based best practices outlined in this guide
- Source high-quality peptides from trusted suppliers with verified purity standards
- Develop comprehensive nausea management plans integrating multiple intervention modalities
- Establish monitoring systems for systematic data collection on nausea patterns and remedy effectiveness
- Educate research teams on recognition, assessment, and management of peptide-related nausea
- Prepare subject materials that clearly communicate expectations and available support
The commitment to excellence in managing retatrutide nausea remedies distinguishes high-quality research programs and contributes to the advancement of peptide science. By implementing the strategies detailed in this guide, researchers position their protocols for success while maintaining the highest standards of subject care and scientific rigor.
For additional resources, protocol consultation, or to explore the comprehensive range of research-grade peptides available for laboratory applications, researchers are encouraged to connect with experienced suppliers committed to supporting the scientific community through quality products and professional service.
