
Understanding how long does tirzepatide stay in your system is crucial for researchers, healthcare professionals, and anyone studying this innovative dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. Whether you’re investigating dosing protocols, planning research timelines, or examining pharmacokinetic properties, the elimination timeline of tirzepatide follows predictable patterns that warrant detailed examination. This comprehensive guide explores the science behind tirzepatide clearance, half-life dynamics, and what influences how long this peptide remains detectable in biological systems.
Key Takeaways
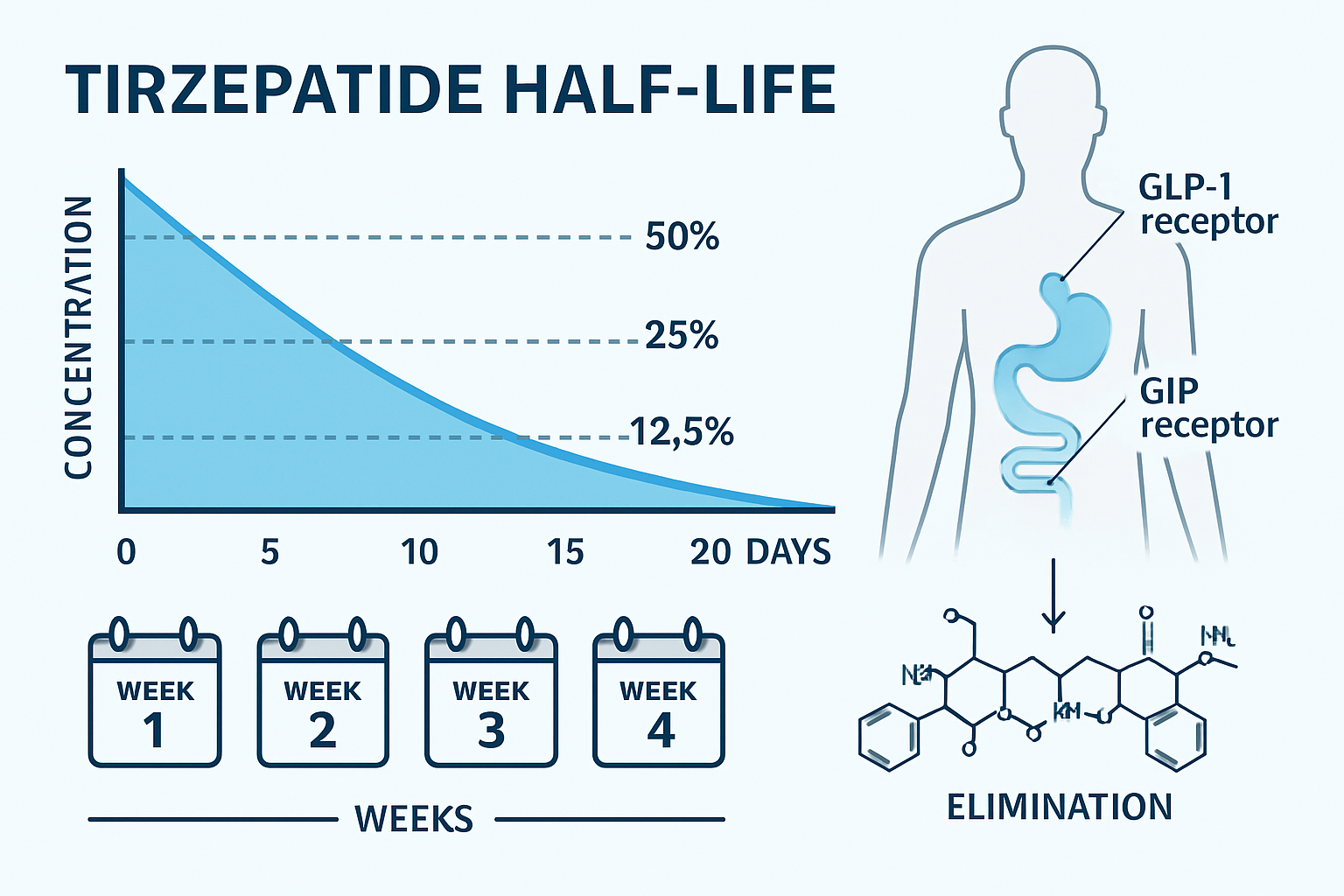
- Tirzepatide has a half-life of approximately 5 days (120 hours), meaning half the drug concentration is eliminated every five days
- Complete elimination takes 25-30 days (3.5-4 weeks) after the final injection, following standard pharmacokinetic principles
- Proteolytic degradation is the primary elimination pathway, not traditional liver metabolism, making clearance independent of hepatic function
- No proven methods exist to accelerate clearance—the body must naturally eliminate tirzepatide through normal metabolic processes
- Individual variations are minimal, with only slight differences in patients with obesity or obstructive sleep apnea (up to 6-day half-life)
Understanding Tirzepatide Half-Life: The Foundation of System Clearance
What Is Half-Life and Why Does It Matter?
The pharmacokinetic concept of half-life represents the time required for the plasma concentration of a substance to decrease by 50%. For tirzepatide, this critical parameter sits at approximately 5 days (120 hours) in most individuals. This extended half-life is precisely what enables the convenient once-weekly dosing schedule that makes tirzepatide practical for both clinical and research applications.
Understanding how long tirzepatide stays in your system begins with grasping this fundamental timeline:
- After 5 days: 50% remains in the system
- After 10 days: 25% remains in the system
- After 15 days: 12.5% remains in the system
- After 20 days: 6.25% remains in the system
- After 25 days: 3.125% remains in the system
The 4-5 Half-Life Elimination Rule 📊
Pharmacokinetic principles establish that 4-5 half-lives are required for substantial elimination of any medication from the body. For tirzepatide, this translates to:
5 days × 5 half-lives = 25 days minimum clearance time
Most individuals will achieve near-complete elimination within 25-30 days after their final injection. While trace amounts may persist slightly beyond this window in some cases, the concentrations become clinically and analytically insignificant.
“The extended half-life of tirzepatide reflects its sophisticated molecular design, incorporating modifications that resist rapid enzymatic degradation while maintaining biological activity.” — Pharmaceutical Research Analysis
Peak Concentrations and Steady-State Dynamics
The elimination timeline is only one aspect of tirzepatide pharmacokinetics. Understanding the complete picture requires examining:
Peak Plasma Concentrations: Occur 8-72 hours after subcutaneous injection, with considerable individual variation based on injection site, tissue perfusion, and absorption characteristics.
Steady-State Achievement: Reached after approximately 4 weeks of consistent weekly dosing, when the amount administered equals the amount eliminated during each dosing interval.
This steady-state concept is particularly relevant for research applications where consistent peptide concentrations are desired for experimental protocols.
How Long Does Tirzepatide Stay in Your System? Complete Elimination Timeline
The 25-30 Day Clearance Window
When researchers and clinicians ask how long does tirzepatide stay in your system, the evidence-based answer centers on the 25-30 day timeframe for substantial elimination. This period represents the point at which tirzepatide concentrations fall below clinically relevant thresholds in the vast majority of individuals.
Week-by-Week Breakdown
Week 1 (Days 1-7)
✓ First half-life completes
✓ Plasma concentration drops to 50% of peak
✓ Therapeutic effects remain robust
✓ Gastric emptying delay continues
Week 2 (Days 8-14)
✓ Second half-life completes
✓ Concentration decreases to 25% of original
✓ Appetite suppression effects begin diminishing
✓ Receptor occupancy declining
Week 3 (Days 15-21)
✓ Third half-life completes
✓ Concentration falls to 12.5% of peak
✓ Most therapeutic effects substantially reduced
✓ Body begins metabolic readjustment
Week 4 (Days 22-28)
✓ Fourth half-life completes
✓ Concentration at 6.25% or lower
✓ Approaching complete elimination
✓ Physiological systems normalizing
Days 29-30+
✓ Fifth half-life territory
✓ Trace amounts may persist
✓ Clinically insignificant concentrations
✓ Substantial elimination achieved
Detection Windows vs. Physiological Effects
An important distinction exists between analytical detection and physiological effects. Even after tirzepatide becomes undetectable in standard blood assays, some individuals may experience lingering effects on:
- Gastric emptying patterns
- Appetite regulation
- Glucose homeostasis
- Body weight trajectory
These residual effects typically normalize within the same 4-week window but may show individual variation based on treatment duration, dosing levels, and metabolic factors.
Individual Variation Factors ⚖️
While the 25-30 day timeline applies broadly, several factors introduce minor variations:
| Factor | Impact on Half-Life | Clinical Significance |
|---|---|---|
| Standard population | 5 days (120 hours) | Baseline reference |
| Obesity/Overweight | Up to 6 days | Minimal; no dose adjustment |
| Obstructive sleep apnea | Up to 6 days | Minor variation |
| Renal impairment | No significant change | No dose adjustment needed |
| Hepatic impairment | No significant change | Independent of liver function |
| Body weight (40-200+ kg) | Clinically insignificant | Wide therapeutic window |
For those interested in high-purity tirzepatide for research, understanding these pharmacokinetic principles ensures appropriate experimental design and timeline planning.
Factors Affecting Tirzepatide Clearance and Metabolism
Proteolytic Degradation: The Primary Elimination Pathway
Unlike many pharmaceutical compounds that undergo extensive hepatic metabolism through cytochrome P450 enzymes, tirzepatide follows a fundamentally different elimination route. The peptide is primarily cleared through proteolytic degradation—the enzymatic breakdown of protein structures into smaller peptide fragments and amino acids.
This mechanism involves:
🔬 Peptidase Enzymes: Naturally occurring proteases throughout the body break down tirzepatide’s peptide bonds
🔬 Protein Catabolism: Standard cellular processes that recycle proteins into constituent amino acids
🔬 Ubiquitous Distribution: Proteolytic enzymes exist in plasma, tissues, and cellular compartments
🔬 Non-Organ Specific: Not dependent on liver or kidney function for clearance
This proteolytic pathway explains why how long tirzepatide stays in your system remains remarkably consistent across diverse patient populations, including those with significant organ dysfunction.
Renal Function: Minimal Impact on Clearance
Extensive pharmacokinetic studies have demonstrated that kidney function has negligible influence on tirzepatide elimination. Research across the spectrum of renal function—from normal kidney performance to end-stage renal disease (ESRD)—shows:
- No clinically meaningful changes in half-life
- No requirement for dose adjustments in renal impairment
- Consistent clearance rates across glomerular filtration rate (GFR) categories
- Safe administration even in dialysis-dependent patients
This renal independence stems from the proteolytic degradation mechanism, which operates independently of glomerular filtration and tubular secretion processes.
Hepatic Function: Independence from Liver Metabolism
Similarly, liver function shows minimal impact on tirzepatide pharmacokinetics. Unlike small-molecule drugs requiring hepatic biotransformation, tirzepatide’s peptide structure undergoes enzymatic degradation that doesn’t rely on hepatocyte-specific metabolic pathways.
Studies in patients with hepatic impairment reveal:
- Comparable half-life values to healthy individuals
- No accumulation with repeated dosing
- No dose modifications required for liver disease
- Consistent elimination regardless of hepatic enzyme levels
For researchers sourcing research-grade peptides, this metabolic profile offers advantages in experimental models where hepatic or renal variables might otherwise confound results.
Body Weight and Composition: Minor Variations Only
Despite tirzepatide’s indication for weight management, body weight itself exerts only minor, clinically insignificant effects on pharmacokinetics. Population pharmacokinetic analyses across weight ranges from 40 kg to over 200 kg demonstrate:
- Slight prolongation of half-life in obesity (up to 6 days vs. 5 days)
- No dose adjustments based on body weight
- Consistent therapeutic concentrations across BMI categories
- Wide therapeutic index accommodating individual variation
The subcutaneous administration route and proteolytic clearance mechanism contribute to this weight-independence, distinguishing tirzepatide from compounds with volume-of-distribution or clearance parameters more sensitive to body composition.
Age, Sex, and Ethnicity: No Significant Differences
Comprehensive pharmacokinetic evaluations have found no clinically relevant differences based on:
- Age: Similar clearance in younger and older adults
- Sex: Comparable pharmacokinetics in males and females
- Ethnicity: Consistent parameters across diverse populations
This pharmacokinetic consistency simplifies research protocols and ensures predictable timelines when investigating how long tirzepatide stays in your system across varied study populations.
Can You Speed Up Tirzepatide Elimination? Debunking Common Myths
The Science Says: No Proven Acceleration Methods
A common question in both clinical and research contexts is whether tirzepatide clearance can be accelerated. The evidence-based answer is clear: no proven methods exist to meaningfully speed up tirzepatide elimination from the body.
The biological reality is straightforward—proteolytic degradation proceeds at rates determined by:
- Endogenous enzyme concentrations
- Natural protein turnover rates
- Physiological peptidase activity
- Cellular catabolism processes
These parameters operate within narrow homeostatic ranges that resist external manipulation.
Ineffective Strategies: What Doesn’t Work ❌
Despite persistent myths and anecdotal claims, the following interventions have no evidence supporting accelerated tirzepatide clearance:
Increased Hydration: While adequate hydration supports overall health, drinking extra water cannot enhance proteolytic degradation rates or accelerate peptide breakdown. Tirzepatide clearance is independent of urinary flow rates.
Dietary Modifications: No specific foods, dietary patterns, or nutritional interventions have demonstrated ability to increase peptide catabolism. The proteolytic enzymes responsible for tirzepatide degradation are not substrate-limited by dietary factors.
Exercise and Physical Activity: While exercise offers numerous health benefits, it does not accelerate the enzymatic breakdown of tirzepatide. The clearance timeline remains consistent regardless of activity levels.
Supplements and “Detox” Products: Over-the-counter supplements, herbal preparations, activated charcoal, or commercial “detoxification” products cannot influence tirzepatide pharmacokinetics. These interventions target different metabolic pathways irrelevant to peptide degradation.
Sauna or Heat Exposure: Temperature-based interventions do not enhance proteolytic enzyme activity or peptide clearance rates.
Why Natural Clearance Is Unavoidable
The 25-30 day elimination timeline represents a biological constant determined by:
- Enzyme Kinetics: Peptidase enzymes operate at fixed catalytic rates under physiological conditions
- Molecular Structure: Tirzepatide’s design specifically incorporates modifications that resist rapid degradation
- Systemic Distribution: The peptide distributes throughout body compartments, requiring time for complete clearance
- Homeostatic Regulation: The body maintains stable proteolytic enzyme levels resistant to short-term manipulation
For researchers working with tirzepatide and related peptides, understanding this unavoidable timeline is essential for experimental planning, washout periods, and study design.
Planning for Natural Elimination
Rather than seeking unproven acceleration methods, appropriate strategies include:
✓ Adequate Washout Periods: Allow full 4-5 weeks between tirzepatide exposure and subsequent interventions
✓ Timeline Planning: Design research protocols accounting for the 25-30 day clearance window
✓ Documentation: Maintain detailed records of administration and elimination timelines
✓ Patient/Subject Education: Set realistic expectations about clearance duration
Tirzepatide Pharmacokinetics: From Injection to Complete Clearance
Absorption Phase: The First 72 Hours
Following subcutaneous administration, tirzepatide undergoes a gradual absorption phase characterized by:
8-72 Hour Peak Window: Maximum plasma concentrations typically occur within this timeframe, though individual variation exists based on:
- Injection site (abdomen, thigh, upper arm)
- Subcutaneous tissue characteristics
- Local blood flow and perfusion
- Injection technique and depth
Slow, Sustained Release: The subcutaneous depot provides continuous release into systemic circulation, contributing to stable plasma levels and the extended duration of action.
Distribution Throughout the Body
Once absorbed, tirzepatide distributes into various body compartments:
Volume of Distribution: Approximately 10.3 liters, indicating distribution primarily in extracellular fluid rather than extensive tissue penetration.
Protein Binding: High affinity for plasma albumin contributes to the prolonged half-life by reducing renal filtration and proteolytic access.
Receptor Binding: Tirzepatide engages both GIP and GLP-1 receptors distributed throughout:
- Pancreatic beta cells
- Gastrointestinal tract
- Central nervous system appetite centers
- Cardiovascular tissues
- Adipose tissue
Steady-State Concentrations and Weekly Dosing
With consistent weekly administration, tirzepatide reaches steady-state after approximately 4 weeks. At steady-state:
- Plasma concentrations stabilize within a predictable range
- Amount administered equals amount eliminated per week
- Therapeutic effects reach maximum consistency
- Dose adjustments produce predictable concentration changes
This steady-state principle is particularly relevant for research applications requiring stable peptide exposure.
Following the final dose, tirzepatide enters the elimination phase, characterized by:
Log-Linear DeclinThe Elimination Phase: Weeks to Clearancee: Plasma concentrations decrease following first-order kinetics, with each 5-day period removing half the remaining peptide.
Multi-Compartment Clearance: The peptide clears from:
- Central circulation (plasma)
- Peripheral tissues
- Receptor binding sites
- Subcutaneous depot (if recently administered)
Progressive Effect Diminishment: As concentrations decline, physiological effects gradually diminish:
- Gastric emptying delay lessens
- Appetite suppression reduces
- Glucose-lowering effects wane
- Weight trajectory may shift
Complete Clearance Markers
How long does tirzepatide stay in your system until complete clearance? Key markers include:
Analytical Undetectability: Standard assays can no longer detect tirzepatide in plasma samples (typically by day 25-30)
Physiological Normalization: Gastric emptying, appetite regulation, and metabolic parameters return to baseline
Receptor Availability: GIP and GLP-1 receptors become fully available for endogenous ligand binding
Metabolic Readjustment: Body systems readjust to absence of exogenous GIP/GLP-1 agonism
Clinical and Research Implications of Tirzepatide’s Extended Half-Life
Advantages of the 5-Day Half-Life
The extended elimination timeline of tirzepatide offers several strategic advantages in both clinical and research contexts:
Weekly Dosing Convenience 💊
The 5-day half-life enables practical once-weekly administration, improving:
- Adherence in clinical settings
- Protocol simplicity in research studies
- Reduced injection burden
- Consistent exposure between doses
Stable Plasma Concentrations
Extended half-life produces:
- Minimal peak-to-trough fluctuation
- Predictable pharmacodynamic effects
- Reduced concentration variability
- Consistent receptor engagement
Forgiving Dosing Schedule
If a dose is delayed by 1-2 days:
- Therapeutic concentrations remain adequate
- Minimal impact on efficacy
- Reduced risk of treatment interruption
- Flexibility in research protocols
Research Design Considerations
For investigators utilizing high-purity research peptides, the extended tirzepatide half-life necessitates specific design considerations:
Washout Periods: Minimum 4-5 weeks between tirzepatide exposure and subsequent interventions to ensure complete clearance and avoid carryover effects.
Crossover Study Design: Extended washout requirements may make crossover designs impractical for tirzepatide research, favoring parallel-group designs.
Steady-State Studies: Plan for 4-week run-in periods when investigating effects at steady-state concentrations.
Pharmacokinetic Sampling: Extended sampling schedules (up to 30 days post-dose) required for complete elimination characterization.
Drug Interaction Studies: Consider the prolonged exposure when evaluating potential interactions with co-administered compounds.
Safety Considerations and Adverse Effect Duration
The extended presence of tirzepatide in the system has important safety implications:
Prolonged Adverse Effect Risk: If adverse effects occur, they may persist throughout the elimination period (25-30 days), as concentrations remain above threshold levels.
Gastrointestinal Effects: Nausea, vomiting, diarrhea, or constipation may continue until sufficient clearance occurs.
Hypoglycemia Risk: In combination with insulin or sulfonylureas, glucose-lowering effects persist throughout the clearance period.
Monitoring Requirements: Extended observation may be necessary following adverse events, as discontinuation doesn’t provide immediate relief.
Contraindication Implications: If contraindications develop during treatment, the peptide cannot be immediately removed from the system.
Pregnancy and Reproductive Considerations
The extended elimination timeline has specific reproductive health implications:
Pre-Conception Planning: Guidelines typically recommend discontinuing tirzepatide at least 2 months before planned conception, providing adequate clearance time plus safety margin.
Unplanned Pregnancy: If pregnancy occurs during treatment, tirzepatide will remain in the system for 25-30 days post-discontinuation, though fetal exposure risk during this early period requires clinical evaluation.
Lactation Considerations: Unknown whether tirzepatide transfers to breast milk; the extended half-life means prolonged potential exposure if lactation begins soon after discontinuation.
Surgical and Procedural Planning
The gastric emptying delay associated with tirzepatide has important perioperative implications:
Aspiration Risk: Delayed gastric emptying increases aspiration risk during anesthesia, even if the last dose was several days prior.
Recommended Discontinuation: Many surgical protocols recommend holding tirzepatide for 1-2 weeks before elective procedures, though the peptide remains partially present.
Anesthesia Considerations: Anesthesiologists should be informed of recent tirzepatide use (within 30 days) for appropriate airway management planning.
Comparing Tirzepatide Clearance to Other GLP-1 Receptor Agonists
Half-Life Comparison Across Peptide Therapies
Understanding how long tirzepatide stays in your system gains additional context when compared to other incretin-based therapies:
| Peptide | Half-Life | Dosing Frequency | Complete Clearance |
|---|---|---|---|
| Tirzepatide | ~5 days (120 hrs) | Weekly | 25-30 days |
| Semaglutide | ~7 days (168 hrs) | Weekly | 35-40 days |
| Dulaglutide | ~4.5 days (108 hrs) | Weekly | 22-28 days |
| Liraglutide | ~13 hours | Daily | 2-3 days |
| Exenatide (extended-release) | ~2 weeks | Weekly | 10-12 weeks |
| Exenatide (immediate-release) | ~2.4 hours | Twice daily | 12-15 hours |
Semaglutide vs. Tirzepatide: Clearance Comparison
Both semaglutide and tirzepatide offer weekly dosing, but differ in elimination timelines:
Semaglutide:
- Slightly longer half-life (~7 days)
- Extended clearance period (35-40 days)
- Similar proteolytic degradation pathway
- Comparable steady-state timeline (4-5 weeks)
Tirzepatide:
- 5-day half-life
- 25-30 day clearance window
- Dual GIP/GLP-1 mechanism
- Similar pharmacokinetic predictability
For research comparing these agents, the washout period difference (approximately 1-2 weeks longer for semaglutide) becomes relevant in study design.
Short-Acting vs. Long-Acting Formulations
The contrast between short-acting and long-acting incretin therapies illustrates the strategic advantages of extended half-life formulations:
Short-Acting (e.g., Liraglutide):
- Rapid clearance (2-3 days)
- Daily dosing required
- Quick offset if adverse effects occur
- More frequent concentration fluctuations
Long-Acting (e.g., Tirzepatide):
- Extended clearance (25-30 days)
- Weekly dosing convenience
- Prolonged adverse effect duration
- Stable plasma concentrations
Neither approach is inherently superior—the choice depends on therapeutic goals, patient preferences, and specific research objectives.
Monitoring Tirzepatide Levels: Detection and Measurement
Analytical Methods for Tirzepatide Quantification
While routine clinical monitoring of tirzepatide plasma concentrations is not standard practice, research applications may require quantification using sophisticated analytical techniques:
Liquid Chromatography-Mass Spectrometry (LC-MS/MS):
- Gold standard for peptide quantification
- High sensitivity and specificity
- Can detect low nanogram/mL concentrations
- Distinguishes tirzepatide from endogenous peptides
Immunoassay Methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Radioimmunoassay (RIA)
- Generally less sensitive than LC-MS/MS
- Potential cross-reactivity concerns
Pharmacokinetic Sampling Protocols:
- Baseline (pre-dose)
- Multiple time points post-administration
- Extended sampling through 30+ days for complete elimination characterization
- Steady-state trough measurements before subsequent doses
Lower Limit of Quantification (LLOQ)
The detection threshold for tirzepatide varies by analytical method:
- LC-MS/MS: Typically 0.5-2.0 ng/mL
- ELISA: Generally 5-10 ng/mL
- Research-grade assays: May achieve sub-nanogram detection
As concentrations fall below LLOQ during the elimination phase (typically days 25-30), tirzepatide becomes analytically undetectable, marking practical complete clearance.
Therapeutic Drug Monitoring: Not Routinely Required
Unlike some medications requiring therapeutic drug monitoring, tirzepatide treatment does not require routine plasma level measurement because:
- Wide therapeutic index
- Predictable pharmacokinetics
- Consistent dose-response relationships
- Clinical endpoints (glucose control, weight loss) serve as efficacy markers
However, research contexts may warrant monitoring for:
- Pharmacokinetic characterization studies
- Drug interaction investigations
- Special population evaluations
- Bioequivalence assessments
Researchers requiring premium research-grade peptides for analytical studies should ensure appropriate reference standards and quality controls are available for accurate quantification.
Practical Implications: What the Elimination Timeline Means for You
For Researchers and Laboratory Professionals
Understanding how long tirzepatide stays in your system has direct implications for experimental design and protocol development:
