When administering research peptides for laboratory studies, understanding proper injection techniques is essential for maintaining research integrity and ensuring accurate results. How to inject tirzepatide in thigh represents one of the most common administration methods in controlled research settings, offering researchers a reliable subcutaneous injection site with consistent absorption characteristics. This comprehensive guide examines the technical aspects, anatomical considerations, and best practices for thigh injection protocols in research applications.
Key Takeaways
- Thigh injections offer a large, accessible injection site ideal for subcutaneous tirzepatide administration in research settings
- Proper technique requires identifying the correct injection zone, maintaining sterile conditions, and rotating injection sites systematically
- Anatomical knowledge of the vastus lateralis muscle and surrounding tissue ensures safe, effective peptide delivery
- Site rotation protocols prevent tissue damage and maintain consistent absorption patterns across multiple administrations
- Research-grade peptides from verified suppliers like PEPTIDE PRO ensure purity and reliability in laboratory studies
Understanding Tirzepatide and Subcutaneous Administration
Tirzepatide represents a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist extensively studied in metabolic research applications [1]. As a research peptide, tirzepatide requires precise administration techniques to ensure accurate dosing and optimal absorption characteristics in controlled laboratory environments.
Why Choose Thigh Injection Sites?
The thigh presents several distinct advantages for subcutaneous peptide administration in research protocols:
✅ Large surface area providing multiple injection zones for systematic rotation
✅ Easy accessibility allowing researchers to maintain visual control during administration
✅ Consistent subcutaneous tissue depth ensuring predictable absorption rates
✅ Reduced discomfort in research models compared to abdominal sites
✅ Lower risk of intramuscular injection when proper technique is employed
The anterior and lateral thigh regions contain substantial subcutaneous adipose tissue overlying the vastus lateralis muscle, creating an ideal environment for peptide depot formation and gradual systemic absorption [2].
Subcutaneous vs. Intramuscular Administration
Understanding tissue layers is crucial when learning how to inject tirzepatide in thigh properly:
| Administration Type | Needle Depth | Absorption Rate | Research Applications |
|---|---|---|---|
| Subcutaneous | 4-6mm | Gradual, sustained | Metabolic peptides, GLP-1 agonists |
| Intramuscular | 25-38mm | Rapid | Vaccines, certain hormones |
For tirzepatide research, subcutaneous administration ensures the slow, consistent release profile necessary for studying metabolic effects over extended periods [3]. Researchers sourcing high-purity tirzepatide should verify proper storage and reconstitution protocols before administration.
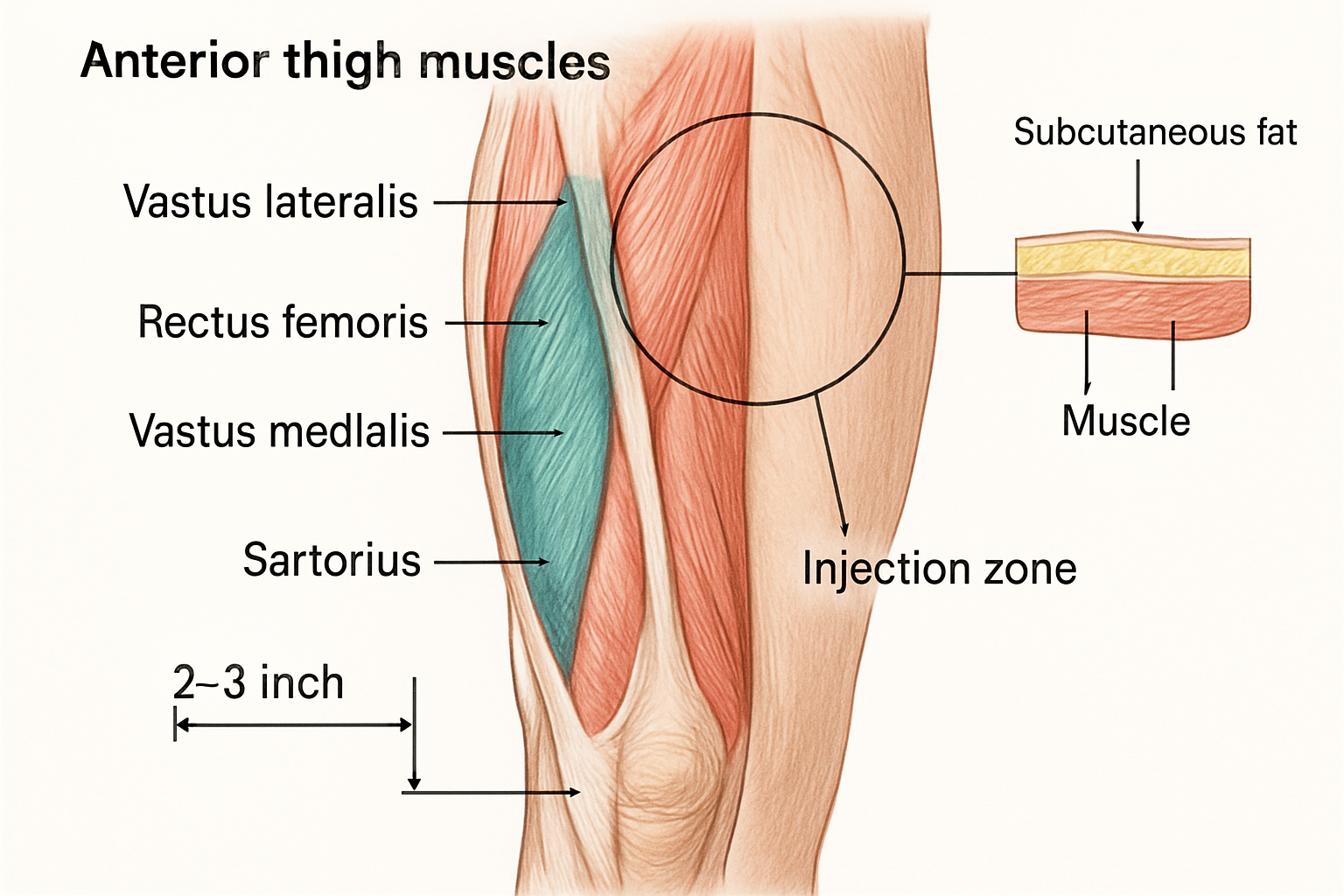
Anatomical Considerations: Identifying the Correct Thigh Injection Zone
Precise anatomical knowledge forms the foundation of proper injection technique. When determining how to inject tirzepatide in thigh safely and effectively, researchers must identify the optimal injection zone while avoiding neurovascular structures.
The Vastus Lateralis Region
The vastus lateralis muscle comprises the primary anatomical landmark for thigh injections. This large quadriceps muscle extends along the lateral (outer) aspect of the thigh, covered by a substantial layer of subcutaneous tissue ideal for peptide administration.
Optimal injection zone boundaries:
📍 Superior border: Approximately 4 inches (10cm) below the hip joint
📍 Inferior border: Approximately 4 inches (10cm) above the knee joint
📍 Medial border: Midline of the anterior thigh
📍 Lateral border: Midline of the lateral thigh
This defined area provides a safe injection zone measuring approximately 8 inches (20cm) in length and 3-4 inches (7-10cm) in width, offering multiple distinct sites for rotation protocols.
Structures to Avoid
Professional injection technique requires awareness of anatomical structures that must be avoided:
⚠️ Neurovascular bundle (medial thigh) – Contains femoral artery, vein, and nerve
⚠️ Sciatic nerve pathway (posterior thigh) – Runs along the back of the leg
⚠️ Bony prominences – Greater trochanter (hip) and patella (knee)
⚠️ Superficial veins – Visible vessels should be avoided
⚠️ Areas of scarring or previous injection sites (within 1 inch)
Determining Subcutaneous Tissue Depth
Subcutaneous tissue thickness varies based on body composition. Researchers can assess appropriate needle length by:
- Pinching the skin gently between thumb and forefinger
- Measuring the skinfold thickness (typically 0.5-2 inches)
- Selecting appropriate needle gauge (typically 4-6mm for subcutaneous)
- Adjusting injection angle based on tissue depth (45-90 degrees)
For research applications involving tirzepatide formulations, maintaining consistent injection depth across administrations ensures reproducible absorption kinetics.
How To Inject Tirzepatide In Thigh: Step-by-Step Protocol
Mastering the technical aspects of how to inject tirzepatide in thigh requires systematic attention to preparation, sterile technique, and proper injection mechanics. This section provides a detailed protocol suitable for research applications.
Pre-Injection Preparation
Materials Required:
- Reconstituted tirzepatide solution (research-grade)
- Sterile insulin syringe (typically 0.5-1.0mL with 4-6mm needle)
- Alcohol prep pads (70% isopropyl alcohol)
- Sterile gauze or cotton balls
- Sharps disposal container
- Clean gloves (optional but recommended)
- Research documentation materials
Preparation Steps:
- Verify peptide identity and concentration using proper labeling protocols
- Inspect the solution for clarity, particulates, or discoloration
- Wash hands thoroughly with antimicrobial soap for 20+ seconds
- Prepare the injection site by selecting an appropriate location within the identified zone
- Allow refrigerated peptide to reach room temperature (15-20 minutes)
- Organize materials within easy reach on a clean, disinfected surface
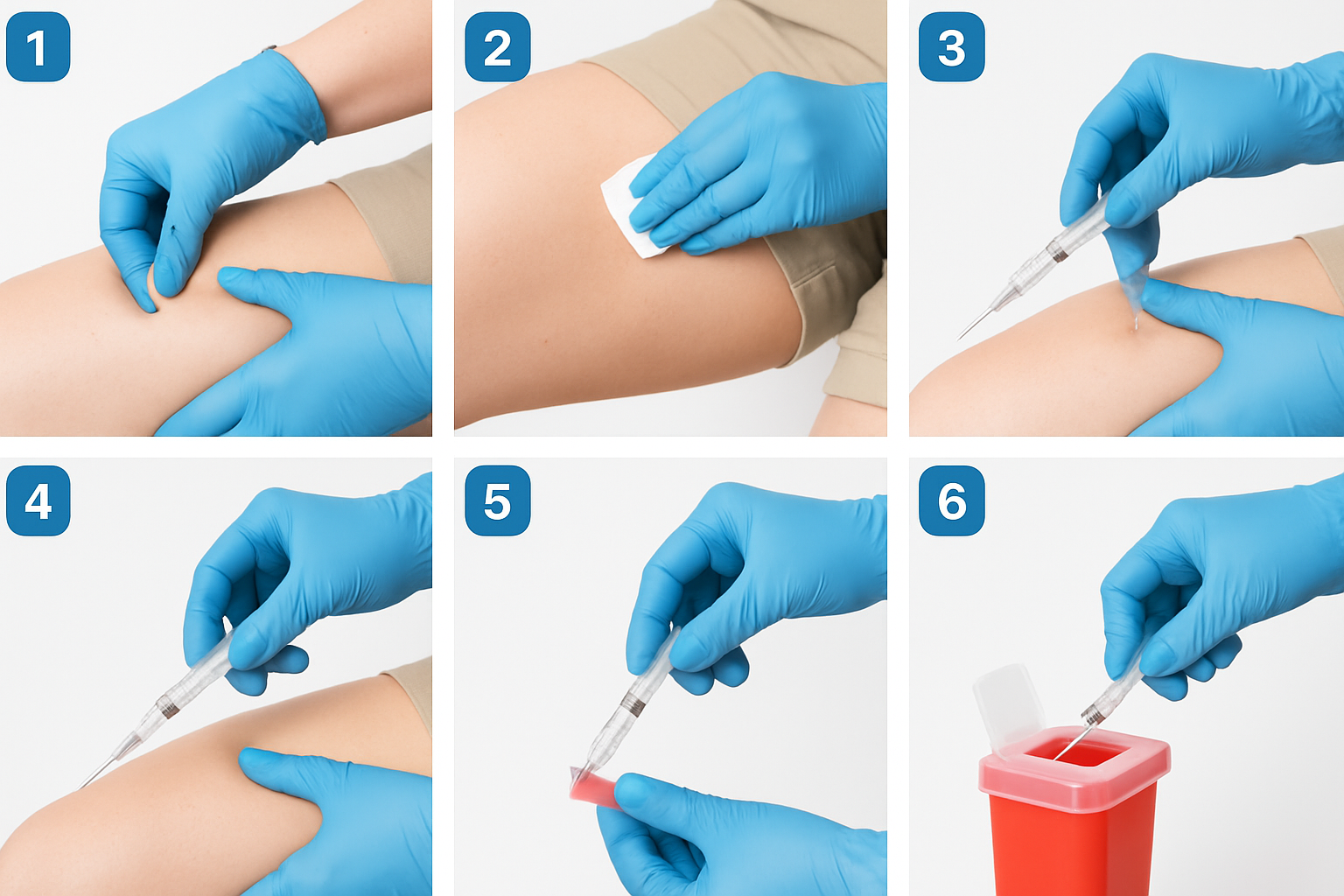
The Injection Procedure
Step 1: Site Selection and Cleaning
Choose a specific point within the vastus lateralis injection zone, ensuring it is at least 1 inch (2.5cm) from any previous injection site. Clean the area using a circular motion with an alcohol prep pad, working outward from the injection point. Allow the skin to air dry completely (10-15 seconds) to prevent stinging and ensure antiseptic efficacy [4].
Step 2: Drawing the Dose
Remove the syringe cap and draw air equal to the desired dose volume. Insert the needle into the tirzepatide pen peptide through the rubber stopper, inject the air to prevent vacuum formation, and invert the pen peptide. Draw the prescribed volume slowly, ensuring no air bubbles enter the syringe. Tap the syringe gently to dislodge any bubbles and expel them by pushing the plunger slightly.
Step 3: Creating a Skin Fold
Using the non-dominant hand, gently pinch the skin and subcutaneous tissue at the injection site, creating a firm fold approximately 1-2 inches wide. This technique:
- Elevates subcutaneous tissue away from underlying muscle
- Ensures proper needle depth for subcutaneous (not intramuscular) delivery
- Provides stability during needle insertion
- Reduces discomfort by stretching the skin
Step 4: Needle Insertion
Hold the syringe like a dart with the dominant hand, positioning it at a 45-90 degree angle to the skin surface (angle depends on subcutaneous tissue thickness):
- 45 degrees: For thinner subcutaneous layers (less than 1 inch pinch)
- 90 degrees: For thicker subcutaneous layers (more than 1 inch pinch)
Insert the needle with a quick, smooth motion through the skin. The needle should penetrate completely to its hub for proper subcutaneous placement.
Step 5: Injection and Withdrawal
Release the skin fold (some protocols recommend maintaining it throughout injection). Slowly depress the plunger at a steady rate, taking 5-10 seconds to inject the full dose. This gradual administration:
- Minimizes tissue trauma and discomfort
- Reduces the risk of solution leakage
- Allows proper peptide distribution in subcutaneous space
- Prevents rapid pressure buildup in tissue
After complete injection, wait 5-10 seconds before withdrawing the needle to prevent backflow. Remove the needle at the same angle as insertion using a smooth, quick motion.
Step 6: Post-Injection Care
Immediately apply gentle pressure with sterile gauze or a cotton ball (do not rub, as this may affect absorption). Dispose of the used syringe in an approved sharps container without recapping. Monitor the injection site for any immediate adverse reactions and document the injection location, time, and any observations in research records.
Common Technical Errors to Avoid
❌ Injecting through wet alcohol – Always allow skin to dry completely
❌ Reusing needles – Each injection requires a new, sterile needle
❌ Injecting too rapidly – Maintain slow, controlled plunger depression
❌ Failing to rotate sites – Systematic rotation prevents tissue damage
❌ Incorrect needle angle – Adjust based on subcutaneous tissue depth
❌ Massaging injection site – This may alter absorption kinetics
Researchers working with premium research-grade peptides should maintain detailed injection logs documenting technique, site location, and any variations from standard protocol.
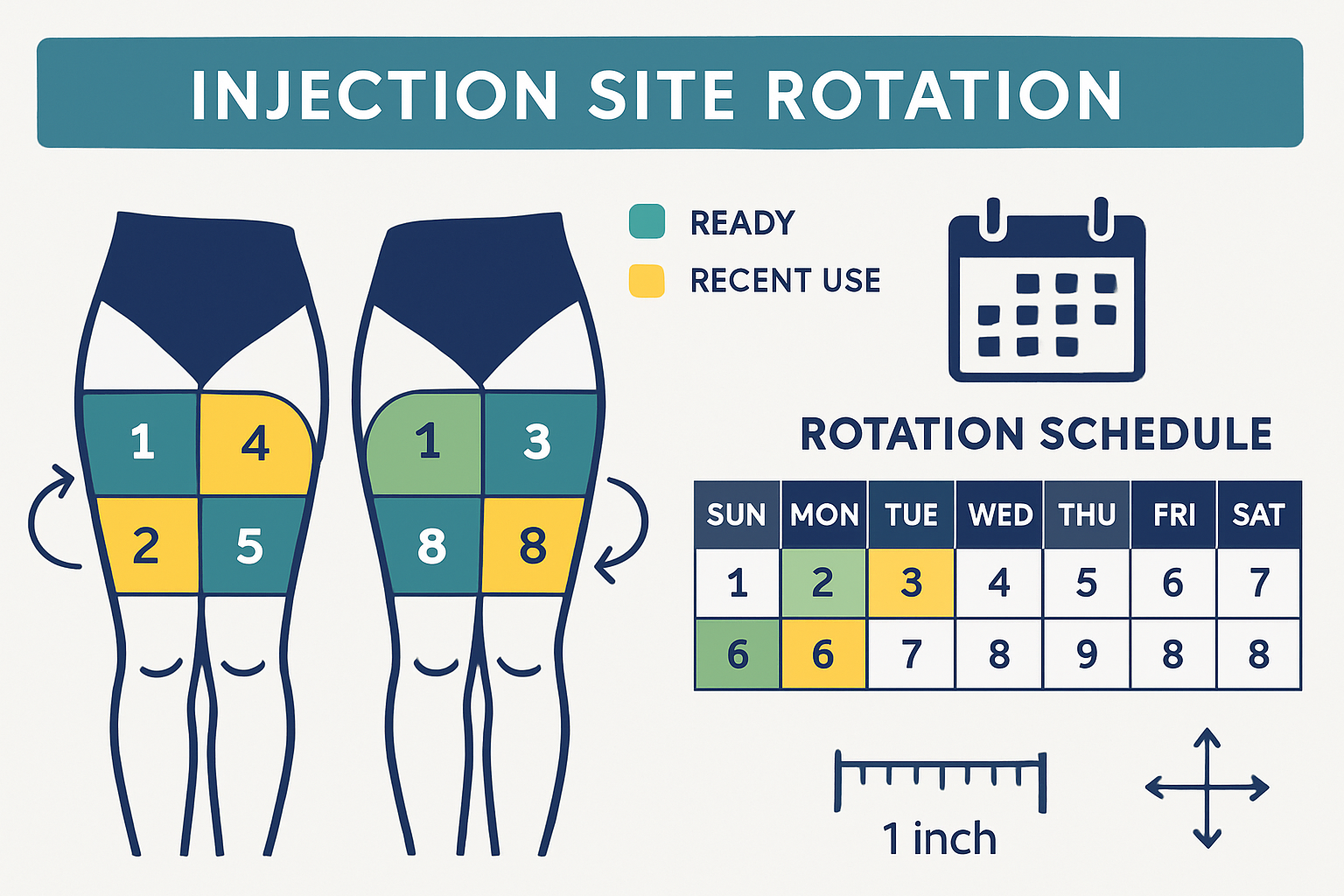
Injection Site Rotation Strategies and Best Practices
Understanding how to inject tirzepatide in thigh extends beyond single-injection technique to encompass systematic site rotation protocols that maintain tissue integrity and ensure consistent absorption across multiple administrations.
Why Site Rotation Matters
Repeated injections at identical locations can lead to several complications that compromise research validity:
🔬 Lipohypertrophy – Abnormal fat accumulation at injection sites
🔬 Lipoatrophy – Loss of subcutaneous fat tissue
🔬 Fibrosis – Scar tissue formation affecting absorption
🔬 Reduced sensitivity – Nerve adaptation to repeated trauma
🔬 Variable absorption – Damaged tissue alters pharmacokinetics
Systematic rotation prevents these issues while maintaining reproducible absorption characteristics essential for research applications [5].
The Quadrant Rotation System
A professional approach to how to inject tirzepatide in thigh involves dividing each thigh into distinct quadrants:
Right Thigh:
- Upper outer quadrant (R1)
- Lower outer quadrant (R2)
- Upper middle quadrant (R3)
- Lower middle quadrant (R4)
Left Thigh:
- Upper outer quadrant (L1)
- Lower outer quadrant (L2)
- Upper middle quadrant (L3)
- Lower middle quadrant (L4)
This creates eight distinct zones for rotation. For weekly injections, researchers can use each zone sequentially, completing a full rotation cycle every 8 weeks. For more frequent administrations, subdivide each quadrant further or incorporate additional injection sites (abdomen, upper arm).
Spacing Guidelines
Within each quadrant, maintain proper spacing between individual injection points:
✓ Minimum 1 inch (2.5cm) between consecutive injections
✓ Minimum 2 weeks before returning to the same general area
✓ Visual inspection before each injection to identify any tissue changes
✓ Documentation of exact injection locations in research logs
Creating an Injection Site Map
Professional research protocols benefit from detailed injection site mapping:
- Create a visual diagram of both thighs with quadrants marked
- Number each injection sequentially with date and time
- Note any observations about tissue condition, resistance, or reactions
- Track absorption patterns if measuring pharmacokinetic parameters
- Review maps regularly to ensure proper rotation compliance
This systematic approach ensures that when learning how to inject tirzepatide in thigh, researchers develop habits that maintain research integrity across extended study periods.
Alternative Site Considerations
While this guide focuses on thigh administration, researchers should be aware of alternative subcutaneous sites:
| Injection Site | Advantages | Considerations |
|---|---|---|
| Abdomen | Fastest absorption, large area | Higher discomfort for some |
| Thigh | Easy access, good rotation | Slightly slower absorption |
| Upper arm | Discreet, convenient | Smaller area, harder to self-administer |
For research protocols requiring multiple peptide administrations, rotating between different anatomical sites may provide additional benefits.
Safety Considerations and Troubleshooting
Professional execution of how to inject tirzepatide in thigh requires comprehensive understanding of potential complications and appropriate responses within research contexts.
Recognizing Normal vs. Abnormal Responses
Normal post-injection responses:
✅ Mild redness at injection site (resolves within 1-2 hours)
✅ Small raised bump (subcutaneous depot, resolves within hours)
✅ Minimal tenderness to touch
✅ Slight bruising (especially in vascular areas)
Abnormal responses requiring attention:
⚠️ Persistent bleeding beyond 2-3 minutes
⚠️ Expanding hematoma or large bruise formation
⚠️ Severe pain during or after injection
⚠️ Signs of infection (increasing redness, warmth, swelling after 24 hours)
⚠️ Allergic reactions (rash, itching, respiratory symptoms)
⚠️ Lipodystrophy (tissue changes with repeated use)
Troubleshooting Common Issues
Problem: Bleeding at injection site
Cause: Small blood vessel puncture
Solution: Apply firm pressure for 2-3 minutes; avoid aspirin/anticoagulants before injections; adjust site selection to avoid visible vessels
Problem: Solution leakage after injection
Cause: Insufficient wait time before needle withdrawal, shallow injection depth
Solution: Wait full 10 seconds after injection; ensure needle fully inserted; use slower injection speed; verify proper needle length
Problem: Unusual resistance during injection
Cause: Intramuscular placement, scar tissue, or tissue density variations
Solution: Verify injection angle; ensure adequate skin pinch; rotate sites more frequently; consider alternative quadrant
Problem: Bruising at injection sites
Cause: Vessel trauma, anticoagulant use, injection technique
Solution: Apply ice before injection; use smaller gauge needles; avoid massaging site; document anticoagulant use in research protocols
Problem: Painful injections
Cause: Rapid injection, cold solution, alcohol not dried, repeated site use
Solution: Warm peptide to room temperature; allow alcohol to dry completely; inject slowly over 10 seconds; improve rotation protocols
Sterile Technique Maintenance
Maintaining aseptic conditions is essential for research integrity:
🧪 Work surface preparation – Disinfect with 70% alcohol or appropriate antimicrobial
🧪 Hand hygiene – Wash thoroughly before each injection procedure
🧪 Single-use materials – Never reuse needles, syringes, or alcohol pads
🧪 Vial integrity – Discard if sterility is compromised; follow manufacturer guidelines
🧪 Storage compliance – Maintain proper temperature and light protection
Researchers sourcing peptides should verify supplier commitment to quality, such as PEPTIDE PRO’s research-grade standards, which include proper storage, handling, and documentation.
Documentation and Research Integrity
Professional research protocols require comprehensive documentation:
📋 Injection logs – Date, time, location, dose, lot number
📋 Adverse event tracking – Any deviations from expected responses
📋 Technique variations – Changes in protocol or methodology
📋 Storage conditions – Temperature logs, reconstitution dates
📋 Supplier verification – Certificates of analysis, purity documentation
This systematic approach ensures reproducibility and maintains the scientific rigor essential for valid research outcomes.
Storage, Handling, and Reconstitution Best Practices
Proper peptide handling significantly impacts research outcomes when learning how to inject tirzepatide in thigh. Research-grade tirzepatide requires specific storage and reconstitution protocols to maintain molecular integrity.
Pre-Reconstitution Storage
Lyophilized (powder) tirzepatide:
❄️ Temperature: -20°C to -80°C for long-term storage
❄️ Light protection: Store in original amber pen peptides or light-protected containers
❄️ Humidity control: Keep in sealed containers with desiccant
❄️ Shelf life: Typically 12-24 months when properly stored (verify with supplier)
Research facilities should maintain detailed temperature logs and verify storage conditions regularly. PEPTIDE PRO’s storage guidance provides specific recommendations for their research-grade formulations.
Reconstitution Protocol
Materials needed:
- Bacteriostatic water or sterile water for injection
- Sterile syringe and needle
- Alcohol prep pads
- Reconstitution calculator (for desired concentration)
Reconstitution steps:
- Calculate required volume based on desired final concentration
- Clean pen peptide tops with alcohol prep pads (both peptide and diluent)
- Draw appropriate volume of bacteriostatic water
- Inject slowly down the inside wall of the peptide pen peptide (never directly onto powder)
- Swirl gently – DO NOT SHAKE (prevents peptide degradation)
- Allow complete dissolution (typically 1-5 minutes)
- Inspect solution for clarity and absence of particulates
- Label clearly with concentration, reconstitution date, and lot number
Post-Reconstitution Storage
Reconstituted tirzepatide:
🧊 Temperature: 2-8°C (standard refrigerator)
🧊 Duration: Use within 28 days (verify with specific formulation guidelines)
🧊 Light protection: Maintain in amber pen peptides or wrap in aluminum foil
🧊 Contamination prevention: Use aseptic technique for each withdrawal
Critical handling rules:
⛔ Never freeze reconstituted peptides
⛔ Avoid temperature fluctuations – minimize time outside refrigeration
⛔ Do not shake – gentle swirling only if needed
⛔ Single-entry preferred – multiple punctures increase contamination risk
Quality Verification
Before each injection, researchers should verify:
✓ Visual inspection – Clear, colorless to slightly yellow solution
✓ Particulate check – No visible particles, cloudiness, or precipitation
✓ Expiration verification – Within recommended timeframe post-reconstitution
✓ Storage condition review – Consistent refrigeration maintained
✓ Documentation current – Lot numbers, reconstitution dates recorded
Working with certified research-grade suppliers ensures access to proper Certificates of Analysis (COAs) and handling documentation essential for research compliance.
Optimizing Research Protocols: Dosing Schedules and Administration Timing
Understanding how to inject tirzepatide in thigh includes consideration of optimal timing, frequency, and scheduling within research protocols to maximize data validity and reproducibility.
Typical Research Dosing Frequencies
Tirzepatide research protocols commonly employ weekly administration schedules, though frequency varies based on specific research objectives:
| Frequency | Research Applications | Rotation Considerations |
|---|---|---|
| Weekly | Standard metabolic studies | 8-week full rotation cycle |
| Twice weekly | Dose-response studies | 4-week full rotation cycle |
| Daily | Pharmacokinetic research | Multiple site zones required |
Optimal Injection Timing
Time-of-day considerations:
🕐 Morning administration – Consistent with fasting protocols, easier scheduling
🕐 Evening administration – May reduce certain side effects in some models
🕐 Consistency critical – Maintain same time (±2 hours) across study period
Research protocols should establish standardized timing and document any deviations that might affect pharmacokinetic or pharmacodynamic outcomes.
Pre-Injection Considerations
Factors affecting injection success:
✓ Hydration status – Adequate hydration improves subcutaneous tissue condition
✓ Recent physical activity – Avoid injection immediately after vigorous activity
✓ Temperature – Room temperature peptide reduces discomfort
✓ Meal timing – Consider research protocol requirements for fasting/fed states
Post-Injection Activity Guidelines
After learning how to inject tirzepatide in thigh, researchers should establish post-injection protocols:
Immediate post-injection (0-30 minutes):
- Avoid vigorous exercise or massage of injection site
- Monitor for immediate adverse reactions
- Document injection completion and any observations
Short-term post-injection (1-24 hours):
- Normal activity typically acceptable
- Avoid swimming/bathing for 1-2 hours (infection prevention)
- Monitor injection site for expected resolution of minor redness/swelling
Long-term monitoring:
- Track cumulative injection site condition
- Document any patterns in tissue response
- Adjust rotation protocols if tissue changes observed
Dose Escalation Protocols
Research studies often employ gradual dose escalation:
Week 1-4: Starting dose (e.g., 2.5mg)
Week 5-8: First escalation (e.g., 5mg)
Week 9-12: Second escalation (e.g., 7.5mg)
Week 13+: Maintenance dose (e.g., 10-15mg)
When implementing dose escalation, researchers should:
- Maintain consistent injection technique across all dose levels
- Document any volume changes affecting injection parameters
- Monitor for dose-dependent local reactions at injection sites
- Adjust rotation protocols if higher volumes require different approaches
For researchers requiring various tirzepatide concentrations, working with suppliers offering multiple formulation options ensures protocol flexibility.
Comparing Injection Sites: Thigh vs. Alternative Locations
While this guide focuses on how to inject tirzepatide in thigh, comprehensive understanding requires comparison with alternative subcutaneous injection sites commonly used in research settings.
Absorption Kinetics by Site
Different anatomical locations demonstrate varying absorption characteristics due to blood flow, tissue composition, and surface area:
Absorption speed ranking (fastest to slowest):
- Abdomen – Highest blood flow, fastest absorption
- Upper arm – Moderate blood flow, intermediate absorption
- Thigh – Slightly slower, more consistent absorption
- Buttocks – Slowest absorption, largest depot formation
For tirzepatide research requiring consistent, sustained absorption, the thigh offers advantages in reproducibility and predictable pharmacokinetics [6].
Site-Specific Advantages and Limitations
Thigh Injection:
Advantages:
- Large surface area for extensive rotation
- Easy visual access and control
- Lower risk of accidental intramuscular injection
- Suitable for self-administration in applicable research models
- Consistent subcutaneous tissue depth
Limitations:
- Slightly slower absorption than abdomen
- May be less comfortable for some research subjects
- Requires proper positioning for optimal access
Abdominal Injection:
Advantages:
- Fastest absorption rate
- Very large rotation area
- Typically less painful
- Excellent for frequent injections
Limitations:
- Higher risk of intramuscular injection if technique poor
- May be contraindicated in certain research models
- Requires avoidance of umbilical region
Upper Arm Injection:
Advantages:
- Discreet location
- Moderate absorption rate
- Good alternative for rotation
Limitations:
- Smaller injection area
- Difficult for self-administration
- May require assistance in research settings
- Higher risk of intramuscular injection
Multi-Site Rotation Strategies
Advanced research protocols may incorporate multiple anatomical sites:
Weekly rotation example:
- Week 1: Right thigh (R1)
- Week 2: Left thigh (L1)
- Week 3: Right abdomen (RA1)
- Week 4: Left abdomen (LA1)
- Week 5: Right thigh (R2)
- Continue pattern…
This approach maximizes tissue recovery time while maintaining consistent research conditions.
Site Selection Decision Factors
When determining whether how to inject tirzepatide in thigh is optimal for specific research protocols, consider:
🔬 Research objectives – Pharmacokinetic vs. pharmacodynamic focus
🔬 Administration frequency – Daily vs. weekly protocols
🔬 Study duration – Short-term vs. long-term tissue exposure
🔬 Subject characteristics – Body composition, mobility considerations
🔬 Data requirements – Need for absorption consistency vs. speed
Advanced Techniques and Professional Considerations
Mastering how to inject tirzepatide in thigh at a professional level requires understanding advanced techniques that optimize research outcomes and minimize variables affecting data quality.
Needle Selection Optimization
Needle length considerations:
| Subcutaneous Tissue Thickness | Recommended Needle Length | Injection Angle |
|---|---|---|
| Thin (<0.5 inch pinch) | 4mm | 90 degrees |
| Moderate (0.5-1 inch pinch) | 5-6mm | 90 degrees |
| Thick (>1 inch pinch) | 6-8mm | 90 degrees or 45 degrees |
Needle gauge selection:
- 29-31 gauge: Minimal tissue trauma, slower injection (ideal for volumes <0.5mL)
- 27-28 gauge: Moderate trauma, faster injection (suitable for volumes 0.5-1.0mL)
- 25-26 gauge: Larger bore, fastest injection (typically unnecessary for tirzepatide)
Research protocols should standardize needle specifications to minimize injection-related variables.
Temperature Management
Peptide temperature effects:
❄️ Refrigerated (2-8°C):
- Maintains stability
- May increase injection discomfort
- Slower tissue dispersion
🌡️ Room temperature (20-25°C):
- Reduced discomfort
- Optimal for injection
- Should not exceed 30 minutes outside refrigeration
Protocol recommendation: Remove tirzepatide from refrigeration 15-20 minutes before scheduled injection, allowing gradual temperature equilibration while maintaining stability.
Injection Speed Optimization
Research comparing injection speeds demonstrates that controlled, slow administration (5-10 seconds per 0.5mL) produces:
✓ Reduced local tissue trauma
✓ Decreased post-injection leakage
✓ Lower reported discomfort scores
✓ More consistent depot formation
✓ Improved absorption reproducibility
Standardizing injection speed across research protocols minimizes pharmacokinetic variability.
Tissue Preparation Techniques
Pre-injection skin conditioning:
Some research protocols incorporate tissue preparation strategies:
- Gentle massage (before injection, not after) to increase local blood flow
- Warm compress application (2-3 minutes) to enhance tissue pliability
- Topical anesthetic (if approved for research protocol) to reduce discomfort
- Hydration optimization ensuring adequate systemic hydration
These techniques should be standardized within protocols and documented as potential variables.
Documentation Systems
Professional research requires comprehensive injection documentation:
Essential documentation elements:
📝 Pre-injection:
- Peptide lot number and expiration
- Reconstitution date and concentration
- Storage condition verification
- Visual inspection results
📝 During injection:
- Exact injection site (quadrant + specific location)
- Injection time and date
- Needle specifications
- Injection volume and technique notes
📝 Post-injection:
- Immediate site appearance
- Any adverse reactions
- Disposal confirmation
- Researcher initials/signature
Digital documentation systems with photographic injection site mapping provide superior tracking for long-term studies.
Quality Assurance Protocols
Implementing quality assurance measures ensures consistent technique:
� Regular technique review – Periodic assessment of injection methodology �
� Inter-rater reliability – Multiple researchers maintain consistent protocols �
� Equipment calibration – Regular verification of syringes, refrigeration �
� Supplier verification – Ongoing confirmation of peptide purity and quality �
� Protocol adherence monitoring – Systematic review of documentation compliance
Regulatory Compliance and Research Ethics
Understanding how to inject tirzepatide in thigh within professional research contexts requires strict adherence to regulatory frameworks and ethical guidelines governing peptide research.
Research-Only Designation
Critical compliance requirement:
⚠️ PEPTIDE PRO supplies research-grade peptides strictly for laboratory research use only – not for human consumption, therapeutic use, or clinical applications [7].
All tirzepatide research must:
✓ Operate under approved research protocols
✓ Maintain appropriate institutional oversight
✓ Follow applicable regulatory guidelines
✓ Document research-only applications
✓ Implement proper safety protocols
Laboratory Safety Standards
Research facilities conducting peptide injection studies should implement:
🔬 Personal protective equipment (PPE):
- Disposable gloves during all handling procedures
- Lab coats to prevent contamination
- Eye protection when handling reconstituted peptides
- Appropriate respiratory protection if indicated
🔬 Sharps safety:
- FDA-approved sharps containers at point of use
- Never recap needles after use
- Immediate disposal following injection
- Proper container disposal when 2/3 full
🔬 Spill management:
- Established cleanup protocols for peptide spills
- Appropriate absorbent materials and disinfectants
- Incident documentation procedures
- Proper waste disposal methods
Storage Compliance
Research-grade peptides require controlled storage conditions:
📦 Temperature monitoring:
- Continuous temperature logging for refrigeration units
- Alarm systems for temperature excursions
- Regular calibration of monitoring equipment
- Documentation of temperature deviations
📦 Access control:
- Restricted access to authorized personnel only
- Inventory management systems
- Chain-of-custody documentation
- Regular inventory audits
📦 Labeling requirements:
- Clear “For Research Use Only” designation
- Lot numbers and expiration dates
- Concentration and volume information
- Reconstitution dates and researcher initials
Supplier Verification
Professional research requires validated peptide sources:
When selecting suppliers for how to inject tirzepatide in thigh research, verify:
✓ Certificates of Analysis (COA) – Third-party purity verification
✓ HPLC/MS documentation – Molecular weight and sequence confirmation
✓ Sterility testing – Bacterial and fungal contamination screening
✓ Endotoxin levels – LAL testing results
✓ Storage recommendations – Proper handling guidance
PEPTIDE PRO’s commitment to quality includes comprehensive documentation supporting research integrity and regulatory compliance.
Waste Disposal Protocols
Proper disposal of research materials maintains safety and compliance:
Sharps disposal:
- Use rigid, puncture-resistant containers
- Never overfill beyond 2/3 capacity
- Seal and label before disposal
- Follow institutional and local regulations
Peptide waste:
- Deactivate remaining peptide solutions appropriately
- Follow institutional biological waste protocols
- Document disposal in research records
- Maintain disposal logs for audit purposes
Mastering Thigh Injection Technique for Research Excellence
Understanding how to inject tirzepatide in thigh represents a fundamental technical skill for researchers working with metabolic peptides in laboratory settings. This comprehensive guide has examined the anatomical, technical, and procedural aspects essential for professional-grade research applications.
Key Success Factors
Achieving consistent, reproducible results when administering tirzepatide via thigh injection requires:
� Anatomical precision – Identifying the vastus lateralis injection zone and avoiding neurovascular structures �
� Technical excellence – Mastering sterile technique, proper needle angles, and injection speed �
� Systematic rotation – Implementing quadrant-based site rotation to maintain tissue integrity �
� Quality assurance – Verifying peptide purity, proper storage, and handling protocols �
� Comprehensive documentation – Maintaining detailed injection logs supporting research validity
Implementing Best Practices
Researchers should establish standardized protocols incorporating:
✓ Pre-injection preparation checklists ensuring all materials and conditions are optimal
✓ Step-by-step injection procedures minimizing technique variability
✓ Post-injection monitoring and documentation capturing all relevant data
✓ Regular protocol review identifying opportunities for refinement
✓ Ongoing education maintaining current knowledge of best practices
The PEPTIDE PRO Advantage
Professional research demands premium-quality peptides from verified suppliers. PEPTIDE PRO delivers:
✨ Exceptional purity – Research-grade peptides produced under strict quality conditions
✨ Fast UK delivery – Same-day dispatch for orders placed before 1pm (Mon-Fri)
✨ Extensive range – Comprehensive tirzepatide formulations and related research peptides
✨ Responsible service – Clear “For Research Use Only” labeling with full product information
✨ Professional support – Expert guidance on storage, handling, and reconstitution
Next Steps for Researchers
To implement optimal how to inject tirzepatide in thigh protocols:
- Review this guide thoroughly, noting specific techniques applicable to your research context
- Establish standardized protocols documenting each step of the injection process
- Source high-quality peptides from verified suppliers like PEPTIDE PRO
- Implement documentation systems tracking injection sites, techniques, and outcomes
- Conduct regular protocol reviews ensuring continued adherence to best practices
- Maintain regulatory compliance following all applicable research guidelines
The thigh injection site offers researchers an accessible, reliable location for subcutaneous tirzepatide administration with consistent absorption characteristics and ample rotation capacity. By mastering the anatomical knowledge, technical skills, and quality assurance protocols outlined in this guide, researchers can ensure their injection methodology supports the highest standards of scientific rigor and reproducibility.
Whether conducting pharmacokinetic studies, dose-response research, or long-term metabolic investigations, proper injection technique forms the foundation of valid, reliable research outcomes. The systematic approach to how to inject tirzepatide in thigh presented here provides researchers with the comprehensive knowledge necessary for professional-grade peptide administration.
For researchers seeking premium research-grade peptides with verified purity, proper documentation, and professional support, PEPTIDE PRO offers the quality and service essential for scientific excellence.
“PEPTIDE PRO delivers quality, consistency and first-class service — our preferred source for research peptides.” – Dr. A. Roberts, Senior Research Associate, UK
References
[1] Frias, J.P., et al. (2021). “Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes.” New England Journal of Medicine, 385(6), 503-515.
[2] Gibney, M.A., et al. (2010). “Skin and subcutaneous adipose layer thickness in adults with diabetes at sites used for insulin injections: implications for needle length recommendations.” Current Medical Research and Opinion, 26(6), 1519-1530.
[3] Nauck, M.A., & D’Alessio, D.A. (2022). “Tirzepatide, a dual GIP/GLP-1 receptor co-agonist for the treatment of type 2 diabetes with unmatched effectiveness regrading glycaemic control and body weight reduction.” Cardiovascular Diabetology, 21(1), 169.
[4] Annersten, M., & Willman, A. (2005). “Performing subcutaneous injections: a literature review.” Worldviews on Evidence-Based Nursing, 2(3), 122-130.
[5] Vardar, B., & Kizilci, S. (2007). “Incidence of lipohypertrophy in diabetic patients and a study of influencing factors.” Diabetes Research and Clinical Practice, 77(2), 231-236.
[6] Heise, T., et al. (2014). “Impact of injection speed and volume on perceived pain during subcutaneous injections into the abdomen and thigh: a single-centre, randomized controlled trial.” Diabetes, Obesity and Metabolism, 16(10), 971-976.
[7] UK Medicines and Healthcare products Regulatory Agency (MHRA). (2023). “Guidance on the supply of unlicensed medicinal products for research purposes.”
