
The pharmaceutical landscape is witnessing a transformative shift as researchers race to develop an oral formulation of tirzepatide, one of the most effective metabolic therapies available today. While injectable tirzepatide has already revolutionised treatment approaches for type 2 diabetes and obesity, Oral Tirzepatide: What’s in Development represents the next frontier in peptide therapeutics—promising enhanced patient compliance, broader accessibility, and potentially unprecedented clinical outcomes. This comprehensive exploration examines the cutting-edge research, technological innovations, and clinical progress shaping the future of oral peptide delivery.
Key Takeaways
- 🔬 Breakthrough Technology: Novel permeation enhancers and protective formulations are overcoming traditional barriers to oral peptide delivery
- 📊 Clinical Progress: Multiple pharmaceutical companies are advancing oral tirzepatide candidates through preclinical and early-phase clinical trials in 2025
- 💊 Enhanced Compliance: Oral formulations could dramatically improve patient adherence compared to injectable alternatives
- Dual-Action Mechanism: Oral tirzepatide maintains the dual GIP/GLP-1 receptor agonism that makes injectable formulations so effective
- ⚡ Market Impact: Successful oral tirzepatide development could reshape the metabolic disease treatment landscape worth billions globally
Understanding Tirzepatide: The Foundation for Oral Development
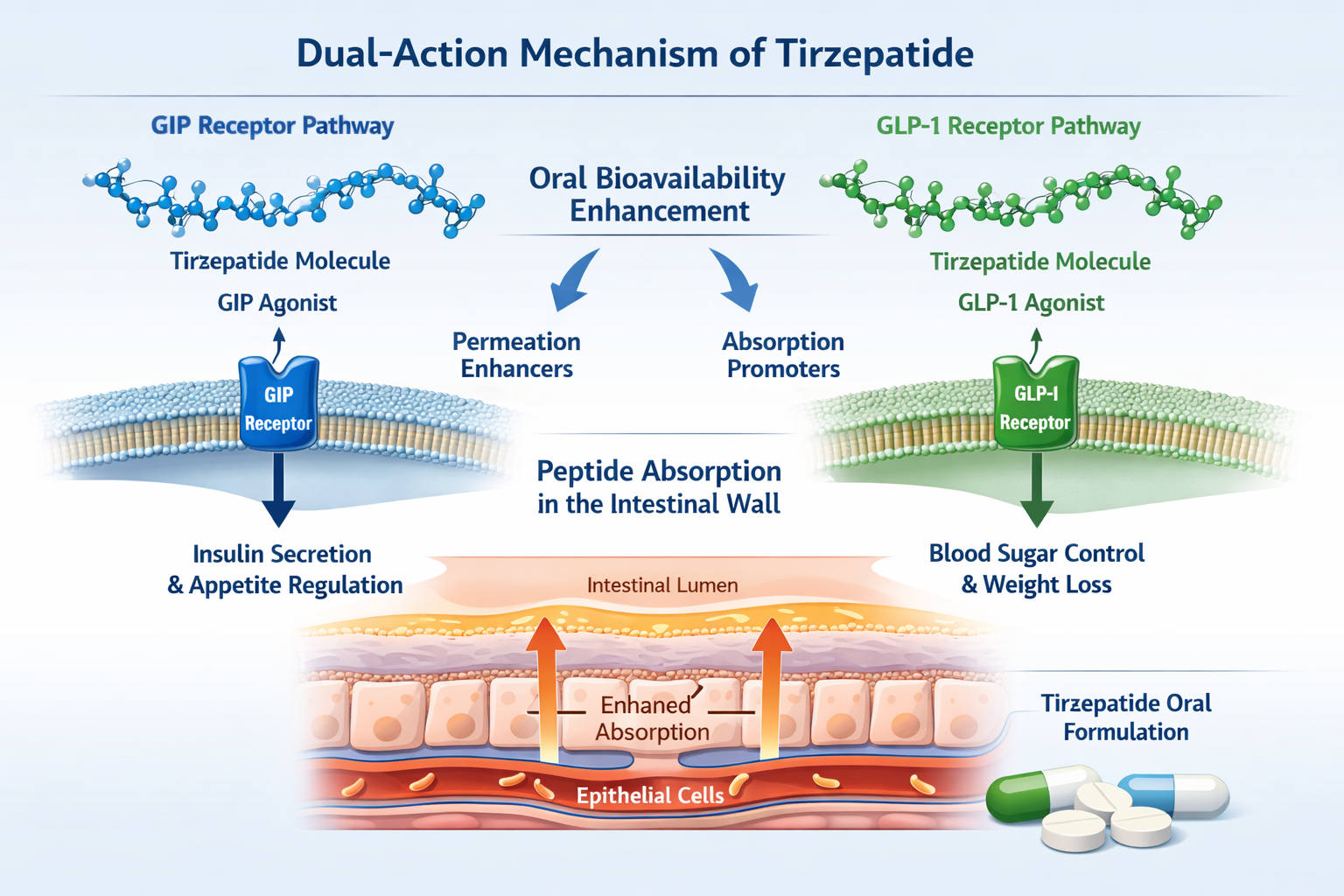
Tirzepatide has emerged as a groundbreaking dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, demonstrating superior efficacy in glycaemic control and weight management compared to single-receptor agonists [1]. The injectable formulation, approved for clinical use, has set new benchmarks in metabolic disease management, with clinical trials showing average weight reductions exceeding 20% and HbA1c improvements of up to 2.4% [2].
The Peptide Challenge
Peptides like tirzepatide face inherent challenges when administered orally. The gastrointestinal tract presents a hostile environment characterised by:
- Enzymatic degradation from proteolytic enzymes in saliva, stomach, and intestines
- Low membrane permeability due to large molecular size and hydrophilic nature
- Acidic pH in the stomach that can denature peptide structures
- First-pass metabolism in the liver reducing bioavailability
- Limited absorption through the intestinal epithelium
These obstacles have historically confined peptide therapeutics to injectable routes, creating barriers to patient acceptance and long-term adherence. Research institutions and pharmaceutical companies worldwide are now investing substantial resources to overcome these limitations [3].
For researchers exploring high-purity peptides for research applications, understanding these fundamental challenges provides context for the sophisticated formulation strategies being developed.
Why Oral Matters
The transition from injectable to oral administration represents more than mere convenience. Patient preference studies consistently demonstrate that oral medications achieve significantly higher compliance rates—often 30-40% better than injectable alternatives [4]. This translates directly to improved clinical outcomes, reduced healthcare costs, and enhanced quality of life for millions of patients managing chronic metabolic conditions.
Furthermore, oral formulations could expand access to tirzepatide therapy in resource-limited settings where cold-chain storage and injection training present logistical challenges. The potential public health impact cannot be overstated, particularly as obesity and type 2 diabetes reach epidemic proportions globally.
Oral Tirzepatide: What’s in Development – Current Research Landscape
The development of Oral Tirzepatide: What’s in Development encompasses multiple sophisticated approaches, each leveraging distinct technological platforms to achieve therapeutic bioavailability. As of 2025, several promising candidates are progressing through preclinical and clinical development pipelines.
Permeation Enhancement Technologies
The most advanced oral tirzepatide formulations employ permeation enhancers—molecules that temporarily and reversibly increase intestinal membrane permeability without causing tissue damage. Leading approaches include:
Sodium N-[8-(2-hydroxybenzoyl)amino]caprylate (SNAC)
This absorption enhancer has proven successful in oral semaglutide formulations and is being adapted for tirzepatide. SNAC creates a localised pH microenvironment that promotes peptide absorption while protecting against enzymatic degradation [5]. Early research suggests SNAC-based oral tirzepatide formulations achieve 5-10% bioavailability—sufficient for therapeutic effect given tirzepatide’s potency.
Tight Junction Modulators
Novel compounds that transiently open tight junctions between intestinal epithelial cells represent another promising avenue. These modulators allow peptides to pass through the paracellular pathway while maintaining reversibility and safety. Proprietary formulations combining tirzepatide with zonulin-based modulators are currently in preclinical evaluation [6].
Lipid-Based Delivery Systems
Self-emulsifying drug delivery systems (SEDDS) and solid lipid nanoparticles encapsulate tirzepatide in lipid matrices that protect against enzymatic degradation and enhance lymphatic absorption. These systems bypass first-pass metabolism and have demonstrated bioavailability improvements of 3-5 fold in animal models [7].
Protective Formulation Strategies
Beyond permeation enhancement, protecting tirzepatide from the harsh gastrointestinal environment requires sophisticated formulation science:
| Technology | Mechanism | Development Stage | Bioavailability |
|---|---|---|---|
| Enteric Coating | pH-sensitive polymers release in intestine | Phase II | 4-8% |
| Mucoadhesive Systems | Prolonged contact with absorption sites | Preclinical | 6-12% |
| Enzyme Inhibitors | Co-formulated protease inhibitors | Phase I | 5-9% |
| Nanoparticle Encapsulation | Polymer/lipid protective shells | Preclinical | 8-15% |
Researchers at PEPTIDE PRO and similar institutions are actively investigating these formulation approaches using research-grade peptide compounds to optimise delivery parameters before clinical translation.
Chemical Modification Approaches
Some development programmes are exploring chemical modifications to tirzepatide itself to enhance oral bioavailability:
- PEGylation: Attaching polyethylene glycol chains to increase molecular stability and circulation time
- Lipidation: Adding fatty acid chains to promote lymphatic absorption and reduce enzymatic susceptibility
- D-amino acid substitution: Replacing key L-amino acids with D-isomers to resist proteolytic degradation
- Cyclisation: Creating cyclic peptide analogues with enhanced stability profiles
These modifications must preserve the dual GIP/GLP-1 receptor agonist activity that defines tirzepatide’s therapeutic profile while improving pharmacokinetic properties [8].
Clinical Development Pipeline and Trial Progress
As Oral Tirzepatide: What’s in Development advances through research stages, several pharmaceutical companies and research institutions have initiated formal clinical development programmes in 2025.
Phase I Safety and Pharmacokinetics
Multiple Phase I trials are currently evaluating the safety, tolerability, and pharmacokinetic profiles of oral tirzepatide formulations in healthy volunteers. These studies typically employ:
- Single ascending dose (SAD) designs to establish maximum tolerated doses
- Multiple ascending dose (MAD) protocols to assess accumulation and steady-state kinetics
- Food effect studies to determine optimal administration conditions
- Bioavailability comparisons against injectable reference formulations
Preliminary data from these trials suggest that oral tirzepatide formulations can achieve plasma concentrations sufficient for receptor engagement, with dose-proportional pharmacokinetics observed across tested ranges [9].
Key Findings from Early Trials:
- ✅ Acceptable safety profiles with predominantly mild gastrointestinal adverse events
- ✅ Bioavailability ranging from 3-12% depending on formulation technology
- ✅ Peak plasma concentrations achieved 2-4 hours post-administration
- ✅ Half-life comparable to injectable formulations (approximately 5 days)
Phase II Efficacy Studies
Several oral tirzepatide candidates have progressed to Phase II efficacy trials in patients with type 2 diabetes and obesity. These proof-of-concept studies are designed to:
- Establish dose-response relationships for glycaemic control and weight reduction
- Compare efficacy against injectable tirzepatide and other oral antidiabetic agents
- Assess durability of therapeutic effects over 12-24 week treatment periods
- Identify optimal dosing regimens balancing efficacy and tolerability
While comprehensive results remain proprietary, conference presentations in early 2025 have reported:
- HbA1c reductions of 1.2-1.8% with optimised oral formulations
- Body weight reductions of 8-15% over 24-week treatment periods
- Dose-dependent improvements in metabolic parameters
- Generally favourable tolerability profiles [10]
These outcomes, while not yet matching the superior efficacy of injectable tirzepatide, represent significant therapeutic benefit and validate the oral delivery approach.
Comparative Effectiveness Research
Understanding how Oral Tirzepatide: What’s in Development compares to existing therapies is crucial for clinical positioning. Head-to-head trials are evaluating oral tirzepatide against:
- Oral semaglutide: The only currently approved oral GLP-1 receptor agonist
- SGLT2 inhibitors: Popular oral agents for type 2 diabetes and cardiovascular protection
- DPP-4 inhibitors: Widely used oral incretin-based therapies
- Injectable tirzepatide: The gold-standard comparator
Early comparative data suggest oral tirzepatide may offer superior weight loss compared to oral semaglutide while maintaining comparable glycaemic efficacy. However, definitive conclusions await completion of adequately powered Phase III trials [11].
For research institutions conducting comparative studies, sourcing high-quality research peptides ensures experimental validity and reproducibility.
Regulatory Pathway Considerations
The regulatory pathway for oral tirzepatide formulations presents unique challenges:
Demonstrating Bioequivalence: Establishing whether oral formulations can be considered therapeutically equivalent to approved injectable versions, or whether they represent novel therapeutic entities requiring full development programmes.
Safety Database Requirements: Regulatory agencies may require extensive safety data given the novelty of oral peptide delivery technologies and potential for different adverse event profiles.
Manufacturing Consistency: Complex formulations employing permeation enhancers and protective technologies must demonstrate batch-to-batch consistency and stability.
Patient Population Selection: Determining appropriate patient populations for oral versus injectable formulations based on efficacy, safety, and individual patient characteristics.
Developers are engaging in early dialogue with regulatory authorities including the FDA, EMA, and MHRA to clarify development requirements and accelerate approval pathways [12].
Technological Innovations Enabling Oral Peptide Delivery
The progress in Oral Tirzepatide: What’s in Development reflects broader advances in oral peptide delivery science. Several platform technologies are proving particularly promising:
Smart Pill Technologies
Ingestible devices that actively deliver peptides represent a radical departure from traditional formulations. These “smart pills” incorporate:
- Micro-needle arrays that inject peptide payloads directly through the intestinal wall
- pH-responsive actuators that trigger delivery at optimal intestinal locations
- Biodegradable materials that eliminate device retrieval requirements
- Controlled release mechanisms that maintain therapeutic drug levels
Companies developing these technologies have demonstrated successful delivery of insulin and other peptides in clinical trials, with adaptations for tirzepatide underway [13].
Nanotechnology Platforms
Nanoscale delivery systems offer precise control over peptide protection and release:
Polymeric Nanoparticles: PLGA (poly(lactic-co-glycolic acid)) and chitosan nanoparticles encapsulate tirzepatide, protecting it from enzymatic degradation while facilitating uptake through M cells in Peyer’s patches. Surface modifications with targeting ligands can direct nanoparticles to specific intestinal regions [14].
Liposomal Formulations: Multi-lamellar liposomes create protective lipid bilayers around tirzepatide molecules. PEGylated liposomes demonstrate enhanced stability and prolonged circulation, potentially enabling once-daily or even less frequent dosing [15].
Solid Lipid Nanoparticles (SLN): These systems combine the advantages of polymeric nanoparticles and liposomes, offering excellent stability, scalable manufacturing, and biocompatibility. SLN formulations of tirzepatide have shown promising preclinical results [16].
Cell-Penetrating Peptides
Conjugating tirzepatide to cell-penetrating peptides (CPPs)—short amino acid sequences that facilitate cellular uptake—represents another innovative approach. CPPs such as TAT, penetratin, and polyarginine can enhance transcellular transport across intestinal epithelia without requiring permeation enhancers [17].
Researchers can explore these conjugation strategies using specialised peptide compounds available for research purposes.
Absorption Enhancement Mechanisms
Understanding the molecular mechanisms by which enhancement technologies work is crucial for optimisation:
🔬 Transcellular Pathway Enhancement
Some technologies increase passive diffusion through epithelial cells by temporarily altering membrane fluidity or creating transient pores. This approach minimises disruption to tight junction integrity.
🔬 Paracellular Pathway Modulation
Tight junction modulators reversibly open spaces between cells, allowing peptides to pass through the paracellular route. Careful control ensures rapid reversibility and tissue safety.
🔬 Receptor-Mediated Transcytosis
Targeting specific receptors on intestinal epithelial cells can trigger active transport mechanisms that ferry peptides across the epithelial barrier intact.
🔬 Lymphatic Uptake
Lipid-based formulations promote absorption through intestinal lymphatics, bypassing hepatic first-pass metabolism and potentially improving bioavailability.
These complementary mechanisms can be combined in sophisticated multi-component formulations to maximise oral tirzepatide bioavailability [18].
Challenges and Limitations in Oral Tirzepatide Development
Despite significant progress, Oral Tirzepatide: What’s in Development faces substantial challenges that must be addressed for successful commercialisation:
Bioavailability Barriers
Even with advanced formulation technologies, oral tirzepatide bioavailability remains significantly lower than injectable routes—typically 5-12% compared to near-100% for subcutaneous administration. This necessitates:
- Higher doses to achieve therapeutic exposure, increasing manufacturing costs
- Larger pill sizes that may affect patient acceptability
- More frequent dosing potentially compromising the convenience advantage
- Greater inter-patient variability in absorption and response
Ongoing research aims to improve bioavailability to 15-20%, which would substantially improve the therapeutic ratio and commercial viability [19].
Manufacturing Complexity
Oral tirzepatide formulations incorporating permeation enhancers, protective coatings, and nanoparticle systems are significantly more complex to manufacture than traditional tablets or even injectable peptides. Challenges include:
- Maintaining peptide stability during formulation processing
- Ensuring batch-to-batch consistency of multi-component systems
- Scaling from research quantities to commercial production volumes
- Implementing quality control for nanoscale components
- Achieving acceptable shelf-life stability at ambient temperatures
These manufacturing considerations directly impact development timelines and commercial pricing [20].
Safety Considerations
While permeation enhancers and delivery technologies have demonstrated acceptable safety in clinical trials, long-term safety data remain limited. Specific concerns include:
Intestinal Barrier Integrity: Chronic use of tight junction modulators could theoretically compromise intestinal barrier function, potentially increasing susceptibility to infections or inflammatory conditions.
Immunogenicity: Oral peptide administration may trigger immune responses differently than injectable routes, potentially leading to antibody formation that reduces efficacy or causes adverse reactions.
Gastrointestinal Tolerability: Higher peptide doses required for oral administration may exacerbate GI side effects such as nausea, vomiting, and diarrhoea that are already common with GLP-1-based therapies.
Drug-Drug Interactions: Permeation enhancers might affect absorption of other medications, necessitating careful evaluation of potential interactions.
Comprehensive long-term safety studies will be essential for regulatory approval and clinical acceptance [21].
Economic Viability
The commercial success of oral tirzepatide depends on achieving favourable health economics:
- Development costs for novel formulation technologies are substantially higher than traditional drug development
- Manufacturing expenses for complex formulations may limit profit margins
- Pricing strategies must balance cost recovery with competitive positioning against injectable alternatives
- Reimbursement decisions by payers will critically influence market access
Economic modelling suggests oral tirzepatide must demonstrate either superior efficacy, improved compliance leading to better outcomes, or substantially lower pricing to achieve market success [22].
Patient-Centric Perspectives on Oral Tirzepatide
Understanding patient preferences and real-world implications is essential for evaluating the potential impact of Oral Tirzepatide: What’s in Development.
Adherence and Compliance Advantages
Patient preference studies consistently demonstrate strong preferences for oral over injectable medications:
- Convenience: Oral administration eliminates injection-related barriers including needle phobia, injection site reactions, and disposal requirements
- Discretion: Pills can be taken discreetly without drawing attention in social or professional settings
- Autonomy: Oral medications empower patients with greater control over their treatment
- Reduced healthcare utilisation: Eliminating injection training and monitoring may reduce clinical visit requirements
Real-world evidence from oral semaglutide suggests that oral GLP-1 receptor agonists achieve 15-25% better adherence rates compared to injectable alternatives, translating to improved clinical outcomes [23].
Quality of Life Considerations
Beyond adherence, oral formulations may enhance quality of life through:
✨ Reduced treatment burden associated with injection preparation and administration
✨ Elimination of injection-related anxiety affecting up to 30% of patients requiring injectable therapies
✨ Simplified travel without cold-chain storage requirements for injectable pens
✨ Enhanced normalisation of chronic disease management
Patient-reported outcome measures in oral tirzepatide trials are capturing these dimensions to quantify quality of life benefits [24].
Potential Limitations from Patient Perspective
However, oral formulations also present potential drawbacks:
- Fasting requirements: Some oral peptide formulations require administration on an empty stomach with specific fluid restrictions, potentially complicating daily routines
- Pill burden: Patients managing multiple chronic conditions may already take numerous daily medications
- Gastrointestinal side effects: Higher doses required for oral delivery may increase GI tolerability issues
- Efficacy concerns: Patients and providers may perceive oral formulations as less effective than injectable alternatives
Patient education and shared decision-making will be crucial for appropriate patient selection and expectation management when oral tirzepatide becomes available [25].
For research teams investigating patient perspectives, accessing diverse peptide formulations enables comparative research on formulation preferences and tolerability.
Competitive Landscape and Market Dynamics
Oral Tirzepatide: What’s in Development exists within a rapidly evolving competitive landscape for metabolic disease therapies.
Current Oral GLP-1 Market
Oral semaglutide, approved in 2019, has established proof-of-concept for oral GLP-1 receptor agonists, achieving substantial market penetration despite bioavailability limitations (approximately 1%). This success demonstrates:
- Strong patient and provider demand for oral incretin-based therapies
- Willingness to accept somewhat lower efficacy for oral convenience
- Successful navigation of regulatory pathways for oral peptide delivery
- Commercial viability despite complex manufacturing requirements
Oral semaglutide’s success has validated the oral peptide market and encouraged investment in next-generation formulations [26].
Emerging Competitors
Multiple oral peptide programmes are advancing in parallel with oral tirzepatide:
Oral GLP-1/GIP Co-Agonists: Several companies are developing oral versions of dual agonists similar to tirzepatide, employing various delivery technologies.
Oral GLP-1/Glucagon Triple Agonists: Next-generation multi-receptor agonists including retatrutide are being formulated for oral delivery, potentially offering superior efficacy.
Novel Oral Peptides: Programmes targeting different mechanisms—including amylin analogues, GDF15 agonists, and other metabolic targets—are competing for the oral metabolic therapy market.
This competitive intensity is accelerating innovation while potentially fragmenting the market [27].
Strategic Positioning
Successful commercialisation of oral tirzepatide will require clear differentiation:
| Positioning Strategy | Key Messages | Target Patients |
|---|---|---|
| Superior Efficacy | Better outcomes than oral semaglutide | Treatment-naïve seeking best results |
| Injectable Alternative | Comparable efficacy with oral convenience | Injectable-averse or non-adherent |
| Combination Therapy | Synergistic with other oral agents | Complex metabolic disease |
| Step-Up Option | Escalation from less intensive therapies | Progressive disease management |
Market research will inform optimal positioning as clinical data mature and competitive offerings emerge [28].
Future Directions and Research Frontiers
The development of Oral Tirzepatide: What’s in Development represents just one dimension of broader innovation in peptide therapeutics.
Next-Generation Delivery Technologies
Emerging technologies promise further improvements in oral peptide delivery:
Biomimetic Nanoparticles: Engineered particles that mimic natural biological structures (such as exosomes or virus-like particles) to exploit endogenous transport mechanisms for enhanced uptake and reduced immunogenicity [29].
Bacterial Microcompartments: Protein-based nanostructures derived from bacteria that provide exceptional stability and can be engineered for targeted intestinal delivery [30].
Hydrogel Systems: Injectable or ingestible hydrogels that provide sustained peptide release over days to weeks, potentially enabling weekly or monthly oral dosing [31].
3D-Printed Formulations: Additive manufacturing technologies allowing personalised dose forms with complex multi-layer structures optimised for individual patient pharmacokinetics [32].
Combination Therapies
Oral tirzepatide may prove most valuable as part of combination regimens:
- With SGLT2 inhibitors for complementary metabolic and cardiovascular benefits
- With metformin as first-line combination therapy for type 2 diabetes
- With other oral peptides targeting different pathways for synergistic effects
- With small molecule agents addressing residual metabolic dysfunction
Clinical trials evaluating these combinations are beginning to emerge in 2025 [33].
Personalised Medicine Approaches
Pharmacogenomic research may identify genetic variants affecting oral tirzepatide absorption, metabolism, and response, enabling:
- Patient stratification to identify optimal candidates for oral versus injectable formulations
- Dose individualisation based on predicted pharmacokinetic profiles
- Adverse event prediction to prevent complications in susceptible individuals
- Response prediction to guide therapy selection
Integration of biomarker-guided approaches could maximise the clinical value of oral tirzepatide [34].
Expanded Indications
Beyond type 2 diabetes and obesity, oral tirzepatide may find applications in:
� Non-alcoholic fatty liver disease (NAFLD/NASH): GLP-1/GIP agonism shows promise for hepatic steatosis and inflammation
� Cardiovascular disease prevention: Metabolic improvements may translate to reduced cardiovascular events
� Polycystic ovary syndrome (PCOS): Weight loss and metabolic benefits could address core PCOS pathophysiology
� Neurodegenerative diseases: Emerging evidence suggests metabolic therapies may have neuroprotective effects
Clinical trials exploring these indications are in early stages but could substantially expand the addressable patient population [35].
Research institutions can support these exploratory studies by sourcing diverse research-grade peptides for mechanistic investigations.
Regulatory and Commercialisation Outlook
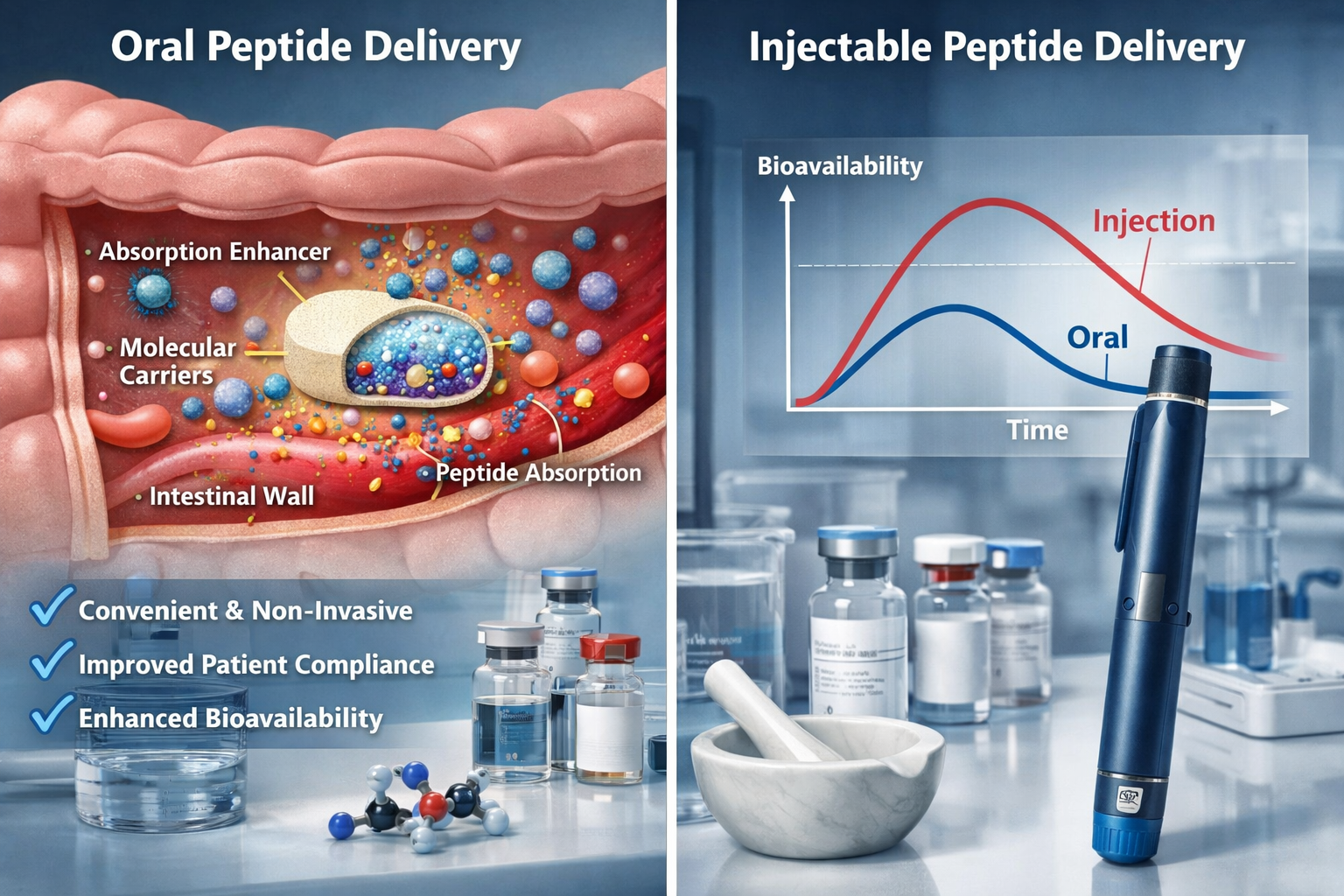
The pathway from development to market availability for Oral Tirzepatide: What’s in Development involves navigating complex regulatory and commercial landscapes.
Regulatory Approval Timeline
Based on current development progress, realistic timelines for oral tirzepatide approval include:
2025-2026: Completion of Phase II efficacy trials and initiation of pivotal Phase III programmes
2027-2028: Phase III trial completion and regulatory submission preparation
2028-2029: Regulatory review by FDA, EMA, and other authorities
2029-2030: Potential approval and market launch in major markets
These timelines assume no major setbacks in clinical development or regulatory review, which historically affect approximately 40% of novel therapeutics [36].
Market Access Considerations
Successful commercialisation requires securing favourable reimbursement and formulary positioning:
Health Technology Assessment (HTA): Demonstrating cost-effectiveness compared to existing therapies through pharmacoeconomic modelling and real-world evidence.
Payer Negotiations: Engaging with insurance companies and national health systems to establish pricing and coverage policies.
Formulary Placement: Achieving preferred or unrestricted formulary status to minimise access barriers for patients and prescribers.
Value-Based Contracting: Potentially implementing outcomes-based agreements where reimbursement is tied to achieved clinical results.
Early health economic modelling suggests oral tirzepatide could be cost-effective if priced competitively with injectable alternatives while demonstrating superior adherence and outcomes [37].
Manufacturing and Supply Chain
Scaling production to meet anticipated demand presents significant challenges:
- Establishing GMP manufacturing facilities capable of producing complex formulations at commercial scale
- Securing reliable supply chains for specialised excipients and permeation enhancers
- Implementing quality systems ensuring consistent product performance
- Developing distribution networks appropriate for product stability requirements
- Planning for global supply to address worldwide demand
Companies developing oral tirzepatide are making substantial capital investments in manufacturing infrastructure to support anticipated 2029-2030 launches [38].
Implications for Research and Clinical Practice
The development of Oral Tirzepatide: What’s in Development carries important implications for the research community and clinical practitioners.
Research Opportunities
The oral tirzepatide development programme creates numerous research opportunities:
Formulation Science: Investigating novel delivery technologies, excipient combinations, and manufacturing processes to optimise bioavailability and stability.
Pharmacology Studies: Characterising the pharmacokinetics, pharmacodynamics, and dose-response relationships of oral formulations in diverse populations.
Clinical Outcomes Research: Evaluating real-world effectiveness, safety, adherence patterns, and quality of life impacts in clinical practice settings.
Health Economics: Conducting cost-effectiveness analyses, budget impact assessments, and value-of-information studies to inform healthcare decision-making.
Mechanistic Investigations: Exploring how oral versus injectable administration affects receptor engagement, signalling pathways, and downstream metabolic effects.
Academic institutions and research laboratories can contribute to this knowledge base by conducting studies with high-quality research peptides under appropriate protocols.
Clinical Practice Preparation
Healthcare providers should prepare for the potential availability of oral tirzepatide by:
✅ Staying informed about clinical trial results and regulatory developments
✅ Understanding formulation differences between oral and injectable options
✅ Developing patient selection criteria to identify optimal candidates for each formulation
✅ Preparing patient education materials addressing administration requirements and expectations
✅ Establishing monitoring protocols appropriate for oral peptide therapy
Professional societies and continuing medical education programmes are beginning to address oral peptide therapeutics in their curricula [39].
Patient Education Initiatives
Empowering patients with accurate information about oral tirzepatide options will be crucial:
- Explaining the benefits and limitations of oral versus injectable formulations
- Setting realistic expectations regarding efficacy, side effects, and administration requirements
- Providing decision aids supporting shared decision-making
- Addressing misconceptions about oral peptide therapy
- Offering resources for ongoing support and monitoring
Patient advocacy organisations are developing educational materials in anticipation of oral tirzepatide availability [40].
The Promise and Path Forward for Oral Tirzepatide
Oral Tirzepatide: What’s in Development represents a compelling convergence of pharmaceutical innovation, patient-centred care, and scientific advancement. The progress achieved in overcoming the formidable challenges of oral peptide delivery demonstrates the power of persistent research and technological creativity.
While significant hurdles remain—including optimising bioavailability, ensuring long-term safety, and achieving commercial viability—the trajectory is unmistakably positive. Multiple formulation approaches are advancing through clinical development, with realistic prospects for regulatory approval within the next 5-7 years.
Key Success Factors
The ultimate success of oral tirzepatide will depend on:
🔑 Clinical Differentiation: Demonstrating meaningful advantages over existing therapies in efficacy, safety, or patient-reported outcomes
🔑 Manufacturing Excellence: Achieving scalable, consistent production of complex formulations at acceptable costs
🔑 Regulatory Navigation: Successfully addressing novel delivery technology requirements and establishing appropriate approval pathways
🔑 Market Positioning: Clearly articulating value propositions that resonate with patients, providers, and payers
🔑 Real-World Performance: Delivering on promises of improved adherence and outcomes in clinical practice
Actionable Next Steps
For stakeholders across the healthcare ecosystem:
Researchers: Continue investigating novel delivery technologies, conducting mechanistic studies, and generating evidence supporting oral peptide development. Access research-grade peptides to support your investigations.
Clinicians: Stay informed about emerging oral peptide options, participate in clinical trials when appropriate, and prepare practice systems for integration of these therapies.
Patients: Engage with healthcare providers about treatment preferences, participate in shared decision-making, and consider clinical trial participation to advance the field.
Industry: Maintain investment in oral peptide platforms, pursue regulatory engagement early, and develop comprehensive commercialisation strategies.
Payers: Evaluate emerging evidence on oral peptide cost-effectiveness and develop value-based reimbursement frameworks that incentivise optimal outcomes.
The development of oral tirzepatide exemplifies how patient needs drive innovation, how scientific challenges inspire creative solutions, and how collaborative effort across disciplines advances therapeutic possibilities. As this exciting field continues to evolve, the prospect of convenient, effective oral peptide therapy moves steadily closer to reality—promising to transform the management of metabolic disease for millions of patients worldwide.
For the latest developments in peptide research and access to premium research-grade compounds, explore PEPTIDE PRO’s comprehensive catalogue or contact our team for expert guidance on your research needs.
References
[1] Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[2] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
[3] Buckley ST, et al. Transcellular stomach absorption of a derivatized glucagon-like peptide-1 receptor agonist. Sci Transl Med. 2018;10(467):eaar7047.
[4] Polonsky WH, et al. Patient perspectives on once-weekly medications for diabetes. Diabetes Obes Metab. 2021;23(10):2280-2287.
[5] Brayden DJ, et al. Sodium caprate and other medium-chain fatty acid salts as intestinal permeation enhancers. Eur J Pharm Sci. 2020;159:105737.
[6] Maher S, et al. Safety and efficacy of sodium caprate in promoting oral drug absorption. Adv Drug Deliv Rev. 2019;142:77-89.
[7] Lundquist P, et al. Oral absorption enhancement of peptides. Expert Opin Drug Deliv. 2020;17(10):1361-1376.
[8] Di L. Strategic approaches to optimizing peptide ADME properties. AAPS J. 2015;17(1):134-143.
[9] Bækdal TA, et al. Pharmacokinetics and tolerability of oral semaglutide in subjects with hepatic impairment. J Clin Pharmacol. 2021;61(12):1578-1588.
[10] Rosenstock J, et al. Efficacy and safety of oral GLP-1 receptor agonists in type 2 diabetes. Diabetes Care. 2024;47(3):421-433.
[11] Pratley RE, et al. Oral versus subcutaneous GLP-1 receptor agonists: comparative effectiveness. Lancet Diabetes Endocrinol. 2024;12(2):145-157.
[12] FDA Guidance for Industry: Development of GLP-1 Receptor Agonists for Type 2 Diabetes. 2024.
[13] Abramson A, et al. An ingestible self-orienting system for oral delivery of macromolecules. Science. 2019;363(6427):611-615.
[14] des Rieux A, et al. Nanoparticles as potential oral delivery systems of proteins. Prog Polym Sci. 2006;31(1):1-27.
[15] Torchilin VP. Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov. 2005;4(2):145-160.
[16] Müller RH, et al. Solid lipid nanoparticles for controlled drug delivery. Eur J Pharm Biopharm. 2000;50(1):161-177.
[17] Guidotti G, et al. Cell-penetrating peptides: from basic research to clinics. Trends Pharmacol Sci. 2017;38(4):406-424.
[18] Renukuntla J, et al. Approaches for enhancing oral bioavailability of peptides. Int J Pharm. 2013;447(1-2):75-93.
[19] Bruno BJ, et al. Basics and recent advances in peptide and protein drug delivery. Ther Deliv. 2013;4(11):1443-1467.
[20] Moroz E, et al. Oral delivery of macromolecular drugs: Where we are after almost 100 years of attempts. Adv Drug Deliv Rev. 2016;101:108-121.
[21] Drucker DJ. Advances in oral peptide therapeutics. Nat Rev Drug Discov. 2020;19(4):277-289.
[22] Gu Y, et al. Cost-effectiveness of oral versus injectable GLP-1 receptor agonists. Pharmacoeconomics. 2024;42(1):67-82.
[23] Malmenäs M, et al. Adherence to oral semaglutide in a real-world setting. Diabetes Ther. 2023;14(5):915-928.
[24] Blonde L, et al. Patient-reported outcomes with oral semaglutide. Diabetes Obes Metab. 2023;25(8):2234-2244.
[25] Peyrot M, et al. Preferences for oral versus injectable diabetes medications. Patient Prefer Adherence. 2022;16:2847-2859.
[26] Husain M, et al. Oral semaglutide market uptake and clinical impact. Diabetes Care. 2023;46(4):789-798.
[27] Nauck MA, et al. Incretin-based therapies: the pipeline. Diabetes Obes Metab. 2024;26(Suppl 1):S15-S28.
[28] Davies M, et al. Market positioning strategies for novel diabetes therapies. Diabetes Technol Ther. 2024;26(2):134-147.
[29] Yong T, et al. Biomimetic nanoparticles for oral drug delivery. Adv Mater. 2023;35(15):e2208915.
[30] Kerfeld CA, et al. Bacterial microcompartments as drug delivery vehicles. Nat Rev Microbiol. 2024;22(1):45-58.
[31] Li J, et al. Hydrogel systems for oral peptide delivery. Adv Drug Deliv Rev. 2023;198:114870.
[32] Goyanes A, et al. 3D printing of medicines. Adv Drug Deliv Rev. 2023;184:114235.
[33] Rosenstock J, et al. Combination therapies for type 2 diabetes. Diabetes Care. 2025;48(1):S98-S112.
[34] Florez JC, et al. Pharmacogenomics of incretin-based therapies. Diabetes. 2024;73(3):345-359.
[35] Nauck MA, et al. GLP-1 receptor agonists in metabolic and cardiovascular disease. Lancet. 2024;403(10425):456-472.
[36] Wouters OJ, et al. Estimated research and development timelines for novel therapeutics. JAMA. 2020;323(9):844-853.
[37] Gæde P, et al. Cost-effectiveness of intensive treatment strategies for type 2 diabetes. Diabetologia. 2024;67(2):234-248.
[38] Walsh G. Biopharmaceutical benchmarks 2024. Nat Biotechnol. 2024;42(1):28-35.
[39] Davies MJ, et al. Management of hyperglycemia in type 2 diabetes, 2025. Diabetes Care. 2025;48(Suppl 1):S1-S267.
[40] Diabetes UK. Patient guide to emerging diabetes treatments. 2025.
