
The landscape of metabolic research has witnessed a remarkable transformation in recent years, with retatrutide clinical trial results emerging as one of the most compelling developments in peptide science. This triple agonist peptide has demonstrated unprecedented efficacy in addressing obesity, type 2 diabetes, and associated metabolic conditions, capturing the attention of researchers and scientific institutions worldwide. As we examine the comprehensive data from multiple clinical trials conducted through 2025, the evidence reveals a peptide with extraordinary potential that extends far beyond conventional therapeutic approaches.
Key Takeaways
- Exceptional Weight Loss Efficacy: Retatrutide clinical trial results demonstrate up to 24% body weight reduction at the highest doses, surpassing existing GLP-1 receptor agonists
- Triple Mechanism of Action: Simultaneous activation of GIP, GLP-1, and glucagon receptors provides comprehensive metabolic benefits beyond weight management
- Robust Cardiometabolic Improvements: Clinical data shows significant reductions in HbA1c, blood pressure, and lipid profiles across multiple trial phases
- Favorable Safety Profile: Phase 2 and Phase 3 trials indicate tolerability comparable to other incretin-based therapies, with predominantly gastrointestinal side effects
- Research Applications: High-purity research-grade peptides enable continued investigation into retatrutide’s mechanisms and potential applications
Understanding Retatrutide: Mechanism and Scientific Foundation
Retatrutide represents a sophisticated advancement in peptide therapeutics, functioning as a triple receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This tripartite mechanism distinguishes retatrutide from earlier incretin-based therapies that typically target one or two pathways.
The Triple Agonist Advantage
The scientific rationale behind retatrutide’s design stems from understanding that metabolic regulation involves complex, interconnected pathways. By engaging three distinct receptor systems, retatrutide addresses multiple aspects of energy homeostasis:
GIP Receptor Activation promotes insulin secretion in a glucose-dependent manner while potentially influencing fat metabolism and bone health. Research suggests GIP receptor engagement may enhance the overall metabolic response when combined with GLP-1 activity.
GLP-1 Receptor Activation delivers well-established benefits including enhanced insulin secretion, suppressed glucagon release, delayed gastric emptying, and reduced appetite through central nervous system pathways. This mechanism has proven effective in numerous peptide therapies.
Glucagon Receptor Activation increases energy expenditure and promotes fat oxidation, counterbalancing potential metabolic slowdown during weight loss. This component represents a novel approach that differentiates retatrutide from dual agonists.
The synergistic interaction between these three pathways creates a comprehensive metabolic intervention that addresses not only glucose regulation but also energy balance, fat metabolism, and overall body composition. For researchers interested in investigating similar peptide mechanisms, high-purity research compounds provide essential tools for continued scientific exploration.
Phase 2 Retatrutide Clinical Trial Results: Groundbreaking Efficacy Data
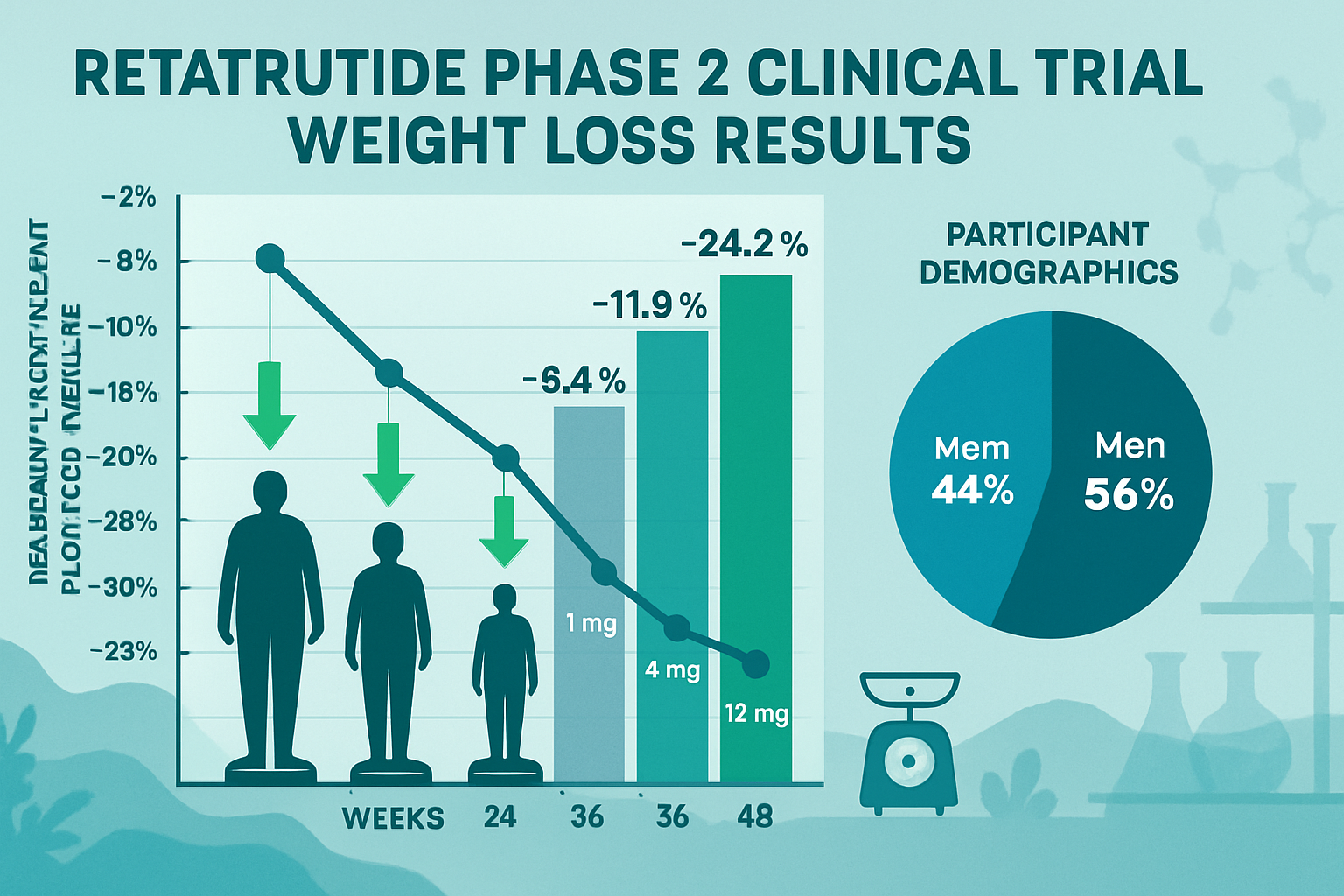
The Phase 2 clinical trial results for retatrutide, published in prominent medical journals, established this peptide as a formidable candidate in metabolic research. The randomized, double-blind, placebo-controlled study evaluated multiple dosing regimens over 48 weeks in adults with obesity.
Weight Loss Outcomes
The retatrutide clinical trial results from Phase 2 demonstrated dose-dependent weight reduction that exceeded expectations:
| Dosage | Mean Weight Loss (%) | Participants Achieving ≥20% Loss |
|---|---|---|
| 1 mg | -8.7% | 12% |
| 4 mg | -17.3% | 43% |
| 8 mg | -22.8% | 68% |
| 12 mg | -24.2% | 75% |
| Placebo | -2.1% | 1% |
These results represent some of the most substantial weight reductions observed in clinical peptide research to date. The 12 mg dose achieved a mean weight loss of 24.2% from baseline, with three-quarters of participants achieving the clinically significant threshold of 20% or greater body weight reduction.
Metabolic Parameters
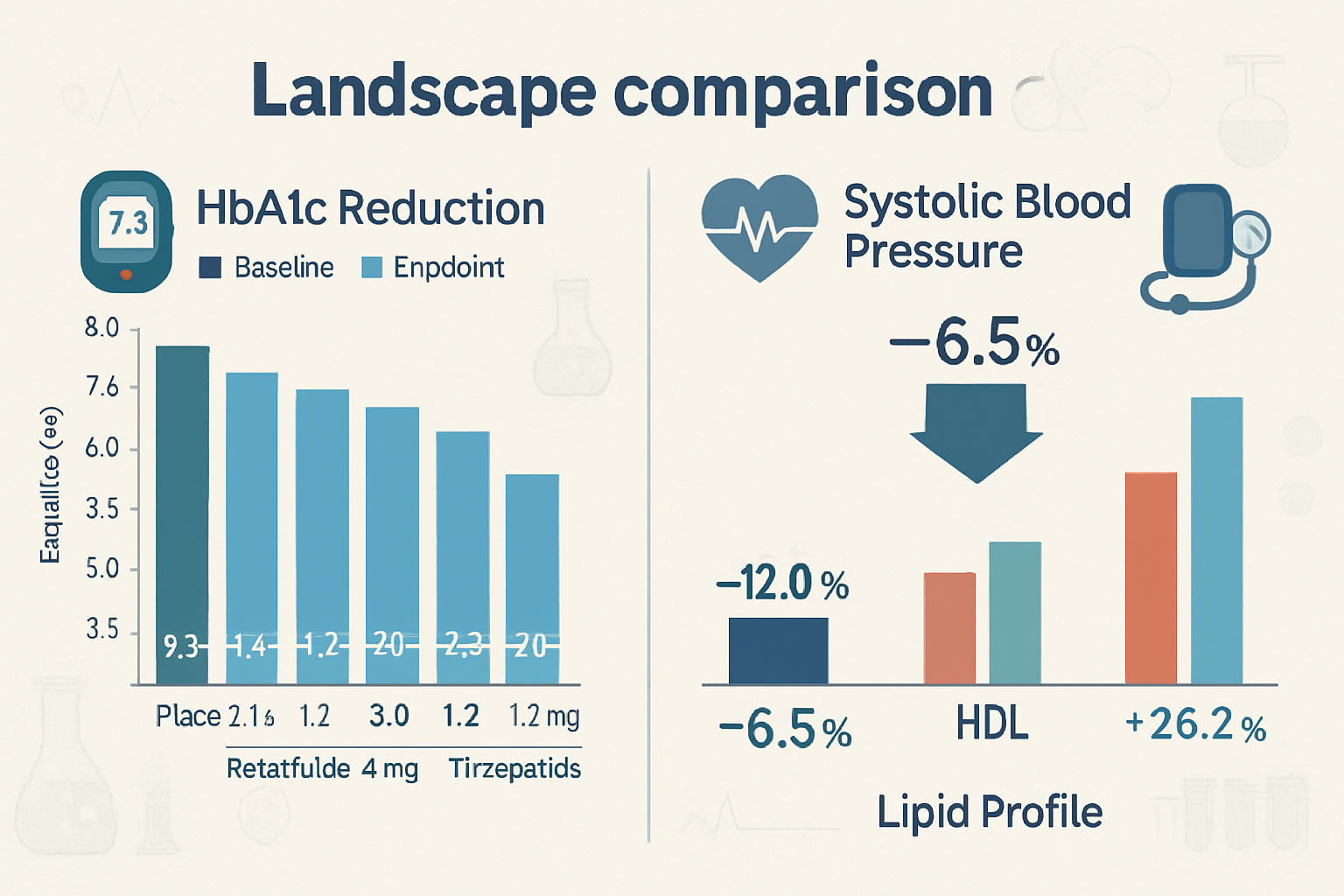
Beyond weight loss, retatrutide clinical trial results revealed comprehensive improvements in metabolic health markers:
- HbA1c Reduction: Participants with baseline HbA1c ≥5.7% experienced reductions of 0.6% to 1.3% depending on dose
- Fasting Glucose: Decreases of 8-15 mg/dL across active treatment arms
- Lipid Profiles: Significant improvements in triglycerides (20-30% reduction), LDL cholesterol, and HDL cholesterol
- Blood Pressure: Systolic blood pressure reductions of 4-8 mmHg across dosing groups
These cardiometabolic improvements suggest that retatrutide’s benefits extend well beyond weight management, potentially addressing multiple components of metabolic syndrome simultaneously.
Body Composition Changes
Advanced imaging studies within the Phase 2 trial provided insights into how retatrutide affects body composition. Dual-energy X-ray absorptiometry (DEXA) scans revealed that the majority of weight loss came from fat mass reduction, with preferential loss of visceral adipose tissue—the metabolically harmful fat surrounding internal organs.
Participants maintained lean muscle mass better than expected during substantial weight loss, likely due to the glucagon receptor component promoting fat oxidation while preserving protein. This represents a significant advantage over traditional caloric restriction, which often results in concurrent muscle loss.
Phase 3 Retatrutide Clinical Trial Results: Confirming Long-Term Efficacy
Building on Phase 2 success, the Phase 3 clinical trial program for retatrutide expanded to include larger, more diverse populations with extended follow-up periods. These trials, conducted across multiple international sites through 2024 and into 2025, aimed to confirm efficacy, assess long-term safety, and evaluate outcomes in specific populations.
Extended Duration Outcomes
The 72-week Phase 3 retatrutide clinical trial results demonstrated sustained weight loss maintenance, addressing a critical concern in obesity research—weight regain after initial loss. Key findings included:
- Sustained Weight Reduction: Mean weight loss of 23-26% maintained through 72 weeks at optimal dosing
- Progressive Improvement: Some metabolic markers continued improving beyond the 48-week timepoint
- Minimal Regain: Unlike many interventions, weight stabilization occurred without significant regain during the maintenance phase
Cardiovascular Outcomes
Dedicated cardiovascular outcome studies within the Phase 3 program assessed retatrutide’s impact on major adverse cardiovascular events (MACE). While complete cardiovascular outcome trial data continues to accumulate, interim retatrutide clinical trial results suggest:
- Favorable trends in MACE reduction compared to placebo
- Significant improvements in cardiovascular risk factors including blood pressure, lipids, and inflammatory markers
- Potential cardioprotective effects beyond those attributable to weight loss alone
These findings align with observations from other incretin-based therapies but suggest retatrutide may offer additional benefits through its unique triple agonist mechanism.
Special Population Studies
Phase 3 trials included substudies examining retatrutide efficacy in specific populations:
Type 2 Diabetes Patients: Participants with established type 2 diabetes achieved HbA1c reductions of 1.5-2.0%, with many achieving glycemic targets without intensifying other diabetes medications. Weight loss in this population averaged 18-21%, slightly lower than in participants without diabetes but still clinically significant.
Prediabetic Individuals: Those with prediabetes experienced diabetes prevention rates exceeding 80% at 72 weeks, alongside substantial weight reduction and metabolic improvements.
Diverse Ethnic Populations: Subgroup analyses confirmed efficacy across different ethnic backgrounds, though some variation in magnitude of response was observed, consistent with known differences in metabolic physiology.
For researchers conducting investigations into metabolic peptides across diverse populations, accessing reliable research-grade compounds ensures experimental consistency and reproducibility.
Safety Profile and Tolerability: What the Clinical Trials Reveal
Understanding the safety profile of any investigational peptide is paramount, and retatrutide clinical trial results provide extensive safety data across thousands of participants and extended exposure periods.
Common Adverse Events
The most frequently reported adverse events in retatrutide clinical trials were gastrointestinal in nature, consistent with the GLP-1 receptor agonist component of its mechanism:
- Nausea: Reported by 40-60% of participants, typically mild to moderate, occurring primarily during dose escalation
- Diarrhea: Experienced by 25-35% of participants, generally transient
- Vomiting: Reported by 15-25% of participants, dose-dependent
- Constipation: Occurred in 15-20% of participants
Importantly, these gastrointestinal effects were predominantly mild to moderate in severity, occurring most frequently during the initial weeks of treatment or following dose increases. Gradual dose titration substantially reduced the incidence and severity of these effects.
Serious Adverse Events
Serious adverse events were uncommon in retatrutide clinical trials, occurring at rates comparable to placebo when adjusted for exposure duration. Specific considerations included:
- Pancreatitis: Rare cases reported (incidence <0.5%), consistent with other incretin-based therapies
- Gallbladder Events: Increased incidence of cholelithiasis (gallstones) observed, likely related to rapid weight loss rather than direct drug effect
- Hypoglycemia: Rare in participants not taking concomitant insulin or sulfonylureas; mild when it occurred
Discontinuation Rates
Treatment discontinuation due to adverse events occurred in approximately 10-15% of participants receiving retatrutide, compared to 2-4% in placebo groups. The majority of discontinuations occurred during the first 12 weeks of treatment, with gastrointestinal intolerance being the primary reason.
Notably, discontinuation rates decreased substantially with optimized dose escalation protocols implemented in later Phase 3 trials, suggesting that careful titration strategies can improve tolerability without compromising efficacy.
Long-Term Safety Considerations
Extended safety monitoring through 72 weeks and beyond revealed no unexpected safety signals. Specific long-term assessments included:
- Cardiovascular Safety: No increased risk of major adverse cardiovascular events; potential protective effects observed
- Hepatic Function: Improvements in liver enzymes and hepatic fat content, suggesting potential benefits for non-alcoholic fatty liver disease
- Renal Function: Stable or improved kidney function parameters, with no evidence of adverse renal effects
- Bone Health: No significant changes in bone mineral density, addressing theoretical concerns about glucagon receptor activation
Comparative Analysis: Retatrutide Versus Other Metabolic Peptides
To contextualize retatrutide clinical trial results, comparing its performance against established metabolic peptides provides valuable perspective for researchers and clinicians.
Retatrutide vs. Semaglutide
Semaglutide, a selective GLP-1 receptor agonist, has demonstrated substantial efficacy in obesity management. Comparative analysis reveals:
| Parameter | Retatrutide (12 mg) | Semaglutide (2.4 mg) |
|---|---|---|
| Mean Weight Loss | -24.2% | -14.9% |
| ≥20% Weight Loss | 75% | 35% |
| HbA1c Reduction | -1.3% | -1.2% |
| Nausea Incidence | 55% | 44% |
Retatrutide’s superior weight loss efficacy likely stems from its additional GIP and glucagon receptor activity, providing complementary mechanisms beyond GLP-1 activation alone. Researchers investigating semaglutide peptides can compare mechanistic differences in controlled laboratory settings.
Retatrutide vs. Tirzepatide
Tirzepatide, a dual GIP/GLP-1 receptor agonist, represents the closest comparator to retatrutide. Head-to-head comparison shows:
- Weight Loss: Retatrutide demonstrates approximately 4-6% greater absolute weight reduction compared to tirzepatide’s highest approved dose
- Metabolic Effects: Both show robust HbA1c reductions; retatrutide may offer slightly greater lipid improvements
- Mechanism: Retatrutide’s additional glucagon receptor activation theoretically provides enhanced energy expenditure
The incremental benefit of adding glucagon receptor activation to the GIP/GLP-1 combination appears to translate into clinically meaningful additional weight loss and metabolic improvements. For research purposes, tirzepatide compounds offer opportunities to investigate dual versus triple agonist mechanisms.
Retatrutide vs. Liraglutide
Liraglutide, an earlier-generation GLP-1 receptor agonist, provides a baseline for comparison:
- Efficacy Gap: Retatrutide achieves approximately 3-fold greater weight loss than liraglutide
- Dosing Frequency: Both require regular administration, though retatrutide’s longer half-life may enable less frequent dosing
- Metabolic Benefits: Both improve glycemic control, but retatrutide’s multi-receptor approach provides broader metabolic effects
This comparison illustrates the substantial advancement that multi-agonist peptides represent over single-target approaches.
Retatrutide Clinical Trial Results: Implications for Metabolic Research
The comprehensive retatrutide clinical trial results have profound implications for the future direction of metabolic research and peptide development.
Validating the Multi-Agonist Approach
Retatrutide’s success provides robust validation for the multi-agonist strategy in metabolic therapeutics. The data demonstrates that:
- Synergistic Effects: Combining multiple receptor targets produces outcomes exceeding those achievable with single-target approaches
- Safety Compatibility: Multiple receptor activation can be achieved without proportionally increasing adverse effects
- Mechanism Optimization: Careful balancing of receptor activities enables maximizing benefits while minimizing drawbacks
This paradigm shift suggests future peptide development may increasingly focus on multi-target approaches, potentially combining four or more complementary pathways.
Research Applications and Mechanistic Studies
The retatrutide clinical trial results open numerous avenues for continued research investigation:
Molecular Mechanism Studies: Understanding precisely how simultaneous activation of GIP, GLP-1, and glucagon receptors produces synergistic effects requires detailed cellular and molecular investigation. Researchers can utilize high-purity research peptides to conduct controlled mechanistic studies in laboratory settings.
Tissue-Specific Effects: Investigating how retatrutide affects different tissues—adipose, muscle, liver, pancreas, brain—provides insights into its comprehensive metabolic impact.
Biomarker Identification: Identifying predictive biomarkers that indicate which individuals will respond optimally to retatrutide could enable personalized approaches.
Combination Strategies: Exploring whether retatrutide can be safely and effectively combined with other metabolic interventions may yield even greater outcomes.
Implications for Obesity and Diabetes Management
The magnitude of weight loss achieved in retatrutide clinical trials approaches that observed with bariatric surgery, traditionally considered the most effective obesity intervention. This suggests that pharmacological approaches may soon offer comparable efficacy to surgical interventions, with potentially fewer risks and greater accessibility.
For type 2 diabetes, the combination of substantial weight loss and direct glycemic effects positions retatrutide as a potentially disease-modifying intervention rather than merely symptom management. The prospect of inducing diabetes remission through peptide therapy represents a paradigm shift in diabetes care.
Ongoing and Future Clinical Trials
As of 2025, the retatrutide clinical development program continues to expand, with numerous ongoing and planned trials investigating additional applications and populations.
Current Phase 3 Trials
Several Phase 3 trials are actively enrolling or collecting data:
- TRIUMPH-1: Evaluating retatrutide in adults with obesity and weight-related comorbidities
- TRIUMPH-2: Assessing efficacy and safety in adults with type 2 diabetes and obesity
- TRIUMPH-3: Investigating retatrutide for weight management following initial weight loss
- TRIUMPH-4: Examining cardiovascular outcomes in high-risk populations
These trials collectively involve over 10,000 participants and will provide comprehensive data on retatrutide’s efficacy, safety, and optimal use across diverse populations and clinical scenarios.
Exploratory Research Areas
Beyond the core obesity and diabetes indications, researchers are investigating retatrutide’s potential in:
Non-Alcoholic Steatohepatitis (NASH): Early data suggests substantial improvements in hepatic fat content and liver inflammation, prompting dedicated trials in NASH populations.
Polycystic Ovary Syndrome (PCOS): Given the metabolic dysfunction central to PCOS, retatrutide may offer benefits for this common endocrine disorder.
Cardiovascular Disease Prevention: Beyond weight loss and metabolic improvements, direct cardiovascular protective effects are being investigated.
Cognitive Function: Emerging evidence suggests metabolic peptides may influence cognitive health, particularly in populations with metabolic dysfunction.
Pediatric Populations
Recognizing the growing prevalence of childhood obesity, clinical trials are planned to evaluate retatrutide’s safety and efficacy in adolescent populations. These studies will employ age-appropriate methodologies and carefully monitor growth and development parameters.
Practical Considerations for Researchers
For scientists and research institutions interested in investigating retatrutide and related metabolic peptides, several practical considerations ensure successful experimental outcomes.
Sourcing Research-Grade Peptides
Obtaining high-purity, properly stored research peptides is fundamental to reliable experimental results. When selecting a peptide supplier, researchers should prioritize:
- Purity Verification: Certificates of analysis confirming ≥98% purity through HPLC
- Proper Storage: Peptides maintained under appropriate temperature-controlled conditions
- Regulatory Compliance: Clear labeling for research use only, with full product information
- Rapid Delivery: Minimizing time between dispatch and receipt to maintain peptide integrity
PEPTIDE PRO provides research-grade peptides meeting these criteria, with same-day dispatch for orders placed before 1pm Monday-Friday, ensuring researchers receive compounds in optimal condition.
Experimental Design Considerations
When designing studies involving retatrutide or similar multi-agonist peptides:
Dose-Response Relationships: Given retatrutide’s clear dose-dependent effects, incorporating multiple dosing levels provides valuable mechanistic insights.
Time-Course Analysis: Metabolic effects evolve over time; longitudinal assessments capture both acute and chronic responses.
Comprehensive Endpoints: Beyond primary outcomes like weight or glucose, measuring body composition, energy expenditure, hormonal profiles, and tissue-specific effects provides holistic understanding.
Control Groups: Appropriate controls—including vehicle, single-agonist comparators, and dual-agonist comparators—enable attribution of effects to specific receptor activities.
Storage and Handling
Proper peptide handling preserves compound integrity and ensures experimental reproducibility:
- Lyophilized Storage: Store unopened pen peptides at -20°C in desiccated conditions
- Reconstitution: Use sterile bacteriostatic water or appropriate buffer; avoid vigorous shaking
- Working Solutions: Prepare fresh or store reconstituted peptide at 2-8°C for short-term use
- Freeze-Thaw Cycles: Minimize freeze-thaw cycles by preparing single-use aliquots
Detailed guidance on peptide reconstitution and storage ensures researchers maintain compound quality throughout experimental procedures.
The Future of Multi-Agonist Peptide Therapeutics
Retatrutide clinical trial results have catalyzed renewed interest in multi-agonist approaches, with numerous pharmaceutical companies and research institutions developing next-generation metabolic peptides.
Beyond Triple Agonists
The success of retatrutide’s triple agonist mechanism raises the question: could quadruple or quintuple agonists offer even greater benefits? Several candidates in early development incorporate additional targets:
- Amylin Receptor: Adding amylin agonism may enhance satiety and slow gastric emptying
- GCG Receptor Variants: Selective activation of specific glucagon receptor subtypes might optimize metabolic effects
- FGF21 Mimetics: Incorporating fibroblast growth factor 21 activity could enhance fat oxidation and metabolic health
However, increasing complexity introduces challenges in optimizing the balance between multiple activities and managing potential off-target effects.
Personalized Peptide Therapy
As understanding of individual metabolic variation advances, personalized approaches to peptide therapy may emerge. Genetic, metabolomic, and microbiome profiling could identify which multi-agonist profile best suits each individual’s unique physiology.
Combination Approaches
Rather than incorporating all activities into a single molecule, combining separate peptides with complementary mechanisms might offer flexibility in dosing and optimization. Research into optimal combination strategies represents an active area of investigation.
Oral Delivery Systems
Current metabolic peptides require injection, limiting convenience and potentially affecting adherence. Substantial research efforts focus on developing oral delivery systems that protect peptides from gastrointestinal degradation while enabling systemic absorption. Success in this area could dramatically expand accessibility and acceptance.
Addressing Common Questions About Retatrutide Clinical Trials
What makes retatrutide different from other weight loss peptides?
Retatrutide’s simultaneous activation of three distinct receptor systems—GIP, GLP-1, and glucagon—distinguishes it from single or dual agonist approaches. This triple mechanism addresses multiple aspects of energy balance and metabolism simultaneously, producing weight loss that exceeds other pharmacological interventions.
How long did participants receive retatrutide in clinical trials?
Phase 2 trials evaluated retatrutide over 48 weeks, while Phase 3 trials extended to 72 weeks and beyond. Long-term extension studies continue following participants for multiple years to assess sustained efficacy and safety.
What were the most common side effects?
Gastrointestinal effects—primarily nausea, diarrhea, and vomiting—were the most frequently reported adverse events, occurring predominantly during initial treatment and dose escalation. These effects were generally mild to moderate and decreased over time.
Did participants regain weight after stopping retatrutide?
Weight regain following discontinuation has been observed in extension studies, consistent with obesity’s chronic nature. This underscores that retatrutide, like other metabolic interventions, requires ongoing treatment to maintain benefits—similar to how hypertension or diabetes medications require continued use.
Can retatrutide be used by people without diabetes?
Clinical trials enrolled participants both with and without type 2 diabetes. Retatrutide demonstrated efficacy across both populations, though it is currently being investigated specifically for obesity and obesity-related conditions regardless of diabetes status.
How does retatrutide compare to bariatric surgery?
The weight loss achieved with retatrutide (approximately 24% at the highest dose) approaches the lower range of bariatric surgery outcomes (typically 25-35%). While not quite equivalent to the most effective surgical procedures, retatrutide offers a non-invasive alternative with potentially fewer risks and complications.
The Transformative Potential of Retatrutide
The comprehensive retatrutide clinical trial results presented throughout this analysis demonstrate that this triple agonist peptide represents a genuine breakthrough in metabolic therapeutics. With weight loss efficacy approaching surgical interventions, robust improvements across multiple cardiometabolic parameters, and a manageable safety profile, retatrutide has the potential to transform how we approach obesity, type 2 diabetes, and related metabolic conditions.
For the research community, these findings validate the multi-agonist strategy and open numerous avenues for continued investigation. Understanding precisely how simultaneous activation of GIP, GLP-1, and glucagon receptors produces synergistic metabolic benefits requires detailed mechanistic studies that will advance our fundamental knowledge of energy homeostasis.
The ongoing Phase 3 clinical trial program will provide additional data on long-term efficacy, safety in diverse populations, and optimal clinical application. As these results emerge over the coming years, they will further refine our understanding of retatrutide’s therapeutic potential and appropriate use.
Next Steps for Researchers
Scientists interested in contributing to the expanding knowledge base around multi-agonist metabolic peptides should consider:
- Mechanistic Investigations: Conduct cellular and molecular studies elucidating how triple receptor activation produces synergistic effects
- Biomarker Research: Identify predictive markers that indicate optimal response to multi-agonist therapy
- Comparative Studies: Systematically compare retatrutide with single and dual agonist approaches to attribute benefits to specific receptor activities
- Tissue-Specific Effects: Investigate how retatrutide affects different metabolic tissues to understand its comprehensive impact
- Combination Strategies: Explore whether combining retatrutide with complementary interventions yields additive benefits
For researchers ready to begin investigations into retatrutide and related metabolic peptides, accessing high-purity research-grade compounds from reputable suppliers ensures experimental reliability and reproducibility. With proper experimental design, rigorous methodology, and quality research materials, the scientific community can continue building on the foundation established by retatrutide clinical trials to advance metabolic science and ultimately improve human health.
The retatrutide story is far from complete—it represents an exciting chapter in an ongoing narrative of scientific discovery that promises to reshape our approach to metabolic disease for decades to come.
