
The scientific community’s understanding of metabolic regulation has evolved dramatically with the emergence of novel triple-agonist peptides. Among these groundbreaking compounds, retatrutide stands at the forefront, demonstrating unprecedented potential in preclinical research models examining fat metabolism and energy homeostasis. The retatrutide fat burning mechanism operates through a sophisticated multi-receptor approach that simultaneously activates GLP-1, GIP, and glucagon pathways—a tripartite strategy that distinguishes it from earlier single or dual-agonist compounds. This comprehensive exploration examines the molecular foundations, receptor interactions, and metabolic cascades that define retatrutide’s unique profile in contemporary peptide research.
Key Takeaways
- Triple Receptor Activation: Retatrutide simultaneously engages GLP-1, GIP, and glucagon receptors, creating synergistic metabolic effects that exceed single-pathway approaches
- Multi-Modal Fat Metabolism: The compound influences adipose tissue through enhanced lipolysis, increased thermogenesis, and improved mitochondrial fat oxidation
- Energy Expenditure Enhancement: Research models demonstrate significant increases in basal metabolic rate through glucagon-mediated pathways
- Appetite Regulation: GLP-1 and GIP receptor activation contributes to satiety signaling and reduced food intake in experimental settings
- Research-Grade Quality: High-purity retatrutide compounds are essential for reliable, reproducible research outcomes
Understanding the Retatrutide Fat Burning Mechanism: Molecular Foundations
The retatrutide fat burning mechanism represents a paradigm shift in how researchers approach metabolic regulation studies. Unlike traditional single-target compounds, retatrutide functions as a triple agonist—simultaneously activating three distinct receptor pathways that collectively influence energy metabolism, adipose tissue dynamics, and whole-body glucose homeostasis.
The Triple Receptor Framework
Retatrutide’s molecular structure has been engineered to bind with high affinity to three critical G-protein coupled receptors:
GLP-1 Receptor (Glucagon-Like Peptide-1)
- Primarily expressed in pancreatic beta cells, brain regions controlling appetite, and gastrointestinal tissues
- Activation enhances insulin secretion in glucose-dependent manner
- Mediates satiety signals through hypothalamic pathways
- Slows gastric emptying, contributing to reduced caloric intake
GIP Receptor (Glucose-Dependent Insulinotropic Polypeptide)
- Found in pancreatic beta cells, adipose tissue, and bone
- Potentiates insulin release in presence of elevated glucose
- Influences lipid metabolism within adipocytes
- Modulates nutrient partitioning and energy storage
Glucagon Receptor
- Expressed in hepatic tissue, adipose deposits, and cardiac muscle
- Stimulates hepatic glucose production and glycogenolysis
- Critically important for fat burning: Activates hormone-sensitive lipase and promotes lipolysis
- Increases energy expenditure through enhanced thermogenesis
This triple-action approach creates a comprehensive metabolic environment conducive to fat mobilization and oxidation—a feature that makes retatrutide particularly valuable in metabolic research applications.
Molecular Binding Characteristics
The retatrutide molecule contains specific amino acid sequences and modifications that enable selective binding to all three receptor types. Research indicates balanced potency across the receptor panel, avoiding excessive activation of any single pathway while maintaining therapeutic synergy. This balanced activation profile distinguishes retatrutide from earlier compounds that showed preferential binding to one or two receptors.
The binding affinity and receptor occupancy dynamics contribute significantly to the duration and magnitude of metabolic effects observed in research models. Studies examining receptor pharmacology demonstrate sustained activation patterns that align with the compound’s extended half-life characteristics.
How the Retatrutide Fat Burning Mechanism Activates Lipolysis
At the cellular level, the retatrutide fat burning mechanism initiates a cascade of enzymatic reactions that fundamentally alter how adipocytes handle stored triglycerides. Understanding these molecular events provides crucial insights into the compound’s metabolic impact.
Glucagon-Mediated Lipolysis Activation
The glucagon receptor component of retatrutide’s action proves particularly significant for fat mobilization:
- cAMP Elevation: Glucagon receptor activation stimulates adenylyl cyclase, increasing intracellular cyclic AMP (cAMP) concentrations
- Protein Kinase A Activation: Elevated cAMP activates protein kinase A (PKA), a critical regulatory enzyme
- Hormone-Sensitive Lipase Phosphorylation: PKA phosphorylates hormone-sensitive lipase (HSL), converting it to its active form
- Triglyceride Breakdown: Activated HSL catalyzes the hydrolysis of triglycerides into glycerol and free fatty acids
- Fatty Acid Release: Liberated fatty acids enter circulation for oxidation in peripheral tissues
This enzymatic cascade represents the primary mechanism through which retatrutide promotes fat mobilization from adipose stores. Research models demonstrate measurable increases in circulating free fatty acid concentrations following retatrutide administration—a biochemical marker of active lipolysis.
Adipose Tissue Lipase Regulation
Beyond hormone-sensitive lipase, retatrutide influences additional lipase enzymes:
- Adipose Triglyceride Lipase (ATGL): The rate-limiting enzyme for initial triglyceride hydrolysis shows enhanced activity
- Monoacylglycerol Lipase (MGL): Completes the final step of triglyceride breakdown
- Perilipin Phosphorylation: Protective proteins surrounding lipid droplets undergo phosphorylation, permitting lipase access
The coordinated regulation of these enzymes creates a comprehensive lipolytic environment within adipocytes, facilitating efficient fat mobilization.
Synergistic GLP-1 and GIP Effects on Adipocytes
While glucagon receptor activation drives direct lipolysis, the GLP-1 and GIP components contribute complementary effects:
GLP-1 Receptor Effects:
- Modulates adipocyte insulin sensitivity
- Influences adipokine secretion patterns (leptin, adiponectin)
- May reduce lipogenesis (new fat formation) through insulin-independent pathways
GIP Receptor Effects:
- Traditionally associated with lipid storage, but context-dependent effects observed
- In combination with GLP-1 and glucagon activation, appears to optimize metabolic flexibility
- Influences nutrient partitioning toward oxidation rather than storage
The interplay between these three receptor systems creates metabolic conditions that favor fat oxidation over fat accumulation—a key feature of research-grade peptide compounds used in metabolic studies.
Energy Expenditure and Thermogenesis in the Retatrutide Fat Burning Mechanism
Fat burning extends beyond simple mobilization—oxidation and energy dissipation complete the metabolic picture. The retatrutide fat burning mechanism demonstrates significant influence on whole-body energy expenditure through multiple pathways.
Glucagon-Induced Metabolic Rate Enhancement
Glucagon receptor activation serves as a primary driver of increased energy expenditure:
| Metabolic Parameter | Mechanism | Research Observation |
|---|---|---|
| Basal Metabolic Rate | Increased hepatic glucose production requiring ATP | 5-12% elevation in preclinical models |
| Thermogenesis | Enhanced uncoupling protein expression | Increased heat production in brown adipose tissue |
| Hepatic Oxidation | Stimulated fatty acid beta-oxidation | Elevated ketone body production |
| Cardiac Output | Mild chronotropic and inotropic effects | Increased oxygen consumption |
These glucagon-mediated effects create a metabolic environment where energy expenditure exceeds baseline levels, contributing to negative energy balance even without caloric restriction.
Brown Adipose Tissue Activation
Research models indicate that retatrutide may influence brown adipose tissue (BAT) activity—specialized fat deposits that dissipate energy as heat rather than storing it:
- UCP1 Expression: Uncoupling protein 1, the hallmark BAT protein, shows increased expression in some research models
- Mitochondrial Biogenesis: Enhanced mitochondrial density within brown adipocytes
- Sympathetic Activation: Potential indirect effects through central nervous system pathways
- Glucose Uptake: Increased BAT glucose utilization during active thermogenesis
While white adipose tissue (WAT) stores energy, BAT burns it—making BAT activation a particularly valuable component of comprehensive fat-burning strategies in research contexts.
Mitochondrial Fat Oxidation Enhancement
The ultimate fate of mobilized fatty acids depends on mitochondrial oxidation capacity. Retatrutide appears to influence several aspects of mitochondrial function:
Beta-Oxidation Pathway:
- Enhanced carnitine palmitoyltransferase I (CPT1) activity facilitates fatty acid entry into mitochondria
- Increased expression of enzymes in the beta-oxidation cascade
- Elevated production of acetyl-CoA for entry into citric acid cycle
Mitochondrial Efficiency:
- Improved respiratory chain function
- Enhanced ATP production per fatty acid molecule
- Reduced oxidative stress markers in some research models
Peroxisomal Oxidation:
- Very long-chain fatty acids undergo initial peroxisomal processing
- Coordination between peroxisomal and mitochondrial pathways
These cellular-level enhancements ensure that mobilized fatty acids undergo complete oxidation rather than re-esterification and storage—a critical component of net fat reduction.
Appetite Regulation and Caloric Intake Reduction
While direct fat burning mechanisms prove essential, the retatrutide fat burning mechanism also encompasses powerful appetite-regulatory effects that contribute to overall metabolic impact in research models.
GLP-1 Receptor-Mediated Satiety Signaling
The GLP-1 component of retatrutide’s action exerts profound effects on appetite centers within the hypothalamus:
Central Nervous System Pathways:
- Activation of POMC/CART neurons (pro-opiomelanocortin/cocaine- and amphetamine-regulated transcript)
- Inhibition of NPY/AgRP neurons (neuropeptide Y/agouti-related peptide)
- Enhanced leptin sensitivity in hypothalamic regions
- Reduced reward signaling in mesolimbic dopamine pathways
Peripheral Satiety Signals:
- Slowed gastric emptying extends the duration of meal-induced satiety
- Enhanced cholecystokinin (CCK) signaling
- Modulation of ghrelin secretion patterns
- Increased peptide YY (PYY) release from intestinal L-cells
Research models consistently demonstrate significant reductions in food intake following retatrutide administration, with effects appearing dose-dependent and sustained throughout treatment periods.
GIP Receptor Contributions to Appetite Control
While GIP’s role in appetite regulation remains less characterized than GLP-1, emerging research suggests important contributions:
- Central GIP Receptors: Expression in brain regions involved in feeding behavior
- Synergistic Effects: Enhanced satiety when combined with GLP-1 activation
- Nutrient Sensing: Influences post-meal satisfaction signals
- Adipokine Modulation: Indirect effects through altered adipose tissue secretions
The combination of GLP-1 and GIP receptor activation appears to produce satiety effects exceeding either pathway alone—demonstrating the value of the multi-receptor approach.
Behavioral and Metabolic Feedback Loops
Beyond direct receptor activation, retatrutide influences complex feedback systems:
Metabolic Feedback:
- Improved glucose homeostasis reduces hunger signals associated with glycemic fluctuations
- Enhanced fat oxidation may influence fuel-sensing mechanisms in hypothalamus
- Normalized adipokine profiles contribute to improved leptin signaling
Behavioral Observations:
- Research models show reduced meal frequency and portion sizes
- Decreased preference for high-fat, high-sugar options in choice paradigms
- Maintained protein intake relative to total caloric reduction
These multifaceted appetite effects complement the direct fat-burning mechanisms, creating comprehensive metabolic conditions conducive to fat mass reduction in experimental settings.
For researchers seeking to explore these mechanisms with high-purity retatrutide compounds, proper sourcing and handling protocols prove essential for reproducible results.
Hepatic Metabolism and Glucose-Lipid Interactions
The liver serves as a central metabolic hub, and the retatrutide fat burning mechanism significantly influences hepatic function in ways that extend beyond simple fat oxidation.
Glucagon Effects on Hepatic Glucose Production
Glucagon receptor activation in hepatocytes triggers several metabolic responses:
Glycogenolysis and Gluconeogenesis:
- Rapid glycogen breakdown provides glucose for peripheral tissues
- Enhanced gluconeogenesis from amino acids and glycerol
- Increased hepatic glucose output (context-dependent)
Implications for Fat Metabolism:
- Glucose production requires ATP, increasing hepatic energy expenditure
- Gluconeogenesis from glycerol (released during lipolysis) completes the fat mobilization cycle
- Enhanced hepatic oxidative metabolism
Fatty Acid Oxidation in Hepatocytes
The liver represents a major site of fatty acid oxidation, and retatrutide influences this process:
Beta-Oxidation Enhancement:
- Increased CPT1 expression facilitates fatty acid entry into mitochondria
- Enhanced oxidation capacity reduces hepatic lipid accumulation
- Ketone body production increases (acetoacetate, beta-hydroxybutyrate)
VLDL Secretion Modulation:
- Influences packaging and export of triglycerides as very-low-density lipoproteins
- May reduce hepatic steatosis (fatty liver) in research models
- Improves overall hepatic metabolic function
Transcriptional Regulation:
- Influences expression of genes involved in lipid metabolism (PPARα, FGF21)
- Modulates inflammatory pathways associated with metabolic dysfunction
- Enhances mitochondrial biogenesis in hepatocytes
Integration of Glucose and Lipid Metabolism
Retatrutide’s triple-agonist action creates unique metabolic integration:
Glucose-Fatty Acid Cycle (Randle Cycle):
- Coordinates carbohydrate and fat oxidation
- Prevents excessive glucose oxidation when fatty acids are abundant
- Optimizes substrate utilization based on availability
Insulin-Glucagon Balance:
- GLP-1 and GIP enhance insulin secretion (glucose-dependent)
- Glucagon receptor activation provides counter-regulatory effects
- Net result: improved metabolic flexibility and glucose homeostasis
Metabolic Flexibility:
- Enhanced ability to switch between glucose and fat as primary fuel
- Reduced metabolic inflexibility associated with obesity and metabolic dysfunction
- Improved adaptation to fasting and feeding states
This hepatic integration represents a sophisticated aspect of retatrutide’s metabolic profile, contributing to comprehensive improvements in whole-body energy metabolism observed in research contexts.
Adipokine Modulation and Metabolic Signaling
Adipose tissue functions as an active endocrine organ, secreting numerous signaling molecules (adipokines) that influence systemic metabolism. The retatrutide fat burning mechanism extends to modulation of these critical signaling pathways.
Leptin Sensitivity and Signaling
Leptin, the “satiety hormone,” plays crucial roles in appetite regulation and energy expenditure:
Leptin Production:
- Secreted proportionally to adipose tissue mass
- Signals energy sufficiency to hypothalamic centers
- Influences thyroid function and sympathetic nervous system activity
Retatrutide Effects on Leptin Signaling:
- Reduced adipose mass correlates with normalized leptin levels
- Enhanced leptin receptor sensitivity in hypothalamus
- Reversal of leptin resistance observed in some research models
- Improved downstream signaling through JAK-STAT pathways
Restored leptin sensitivity contributes to sustained appetite regulation and maintained energy expenditure—critical factors for long-term metabolic improvements.
Adiponectin Enhancement
Adiponectin, an insulin-sensitizing adipokine, typically decreases with obesity:
Metabolic Benefits:
- Enhances insulin sensitivity in liver and muscle
- Promotes fatty acid oxidation
- Exhibits anti-inflammatory properties
- Protects against metabolic dysfunction
Retatrutide Influence:
- Research models demonstrate increased adiponectin secretion
- Improved high-molecular-weight adiponectin (most metabolically active form)
- Enhanced adiponectin receptor expression in target tissues
- Synergistic effects with direct receptor activation
Elevated adiponectin levels contribute to improved metabolic health markers beyond simple fat reduction.
Inflammatory Adipokine Reduction
Excess adipose tissue secretes pro-inflammatory cytokines that contribute to metabolic dysfunction:
Reduced Inflammatory Markers:
- TNF-α (Tumor Necrosis Factor-alpha): Decreased secretion correlates with improved insulin sensitivity
- IL-6 (Interleukin-6): Reduced levels associated with decreased metabolic inflammation
- MCP-1 (Monocyte Chemoattractant Protein-1): Lower macrophage infiltration into adipose tissue
- Resistin: Decreased levels improve insulin action
Metabolic Implications:
- Reduced chronic low-grade inflammation
- Improved insulin signaling pathways
- Enhanced metabolic flexibility
- Better long-term metabolic health outcomes
The comprehensive adipokine profile improvements observed with retatrutide treatment extend beyond simple fat mass reduction, suggesting systemic metabolic benefits in research models.
Comparative Mechanisms: Retatrutide vs. Single and Dual Agonists
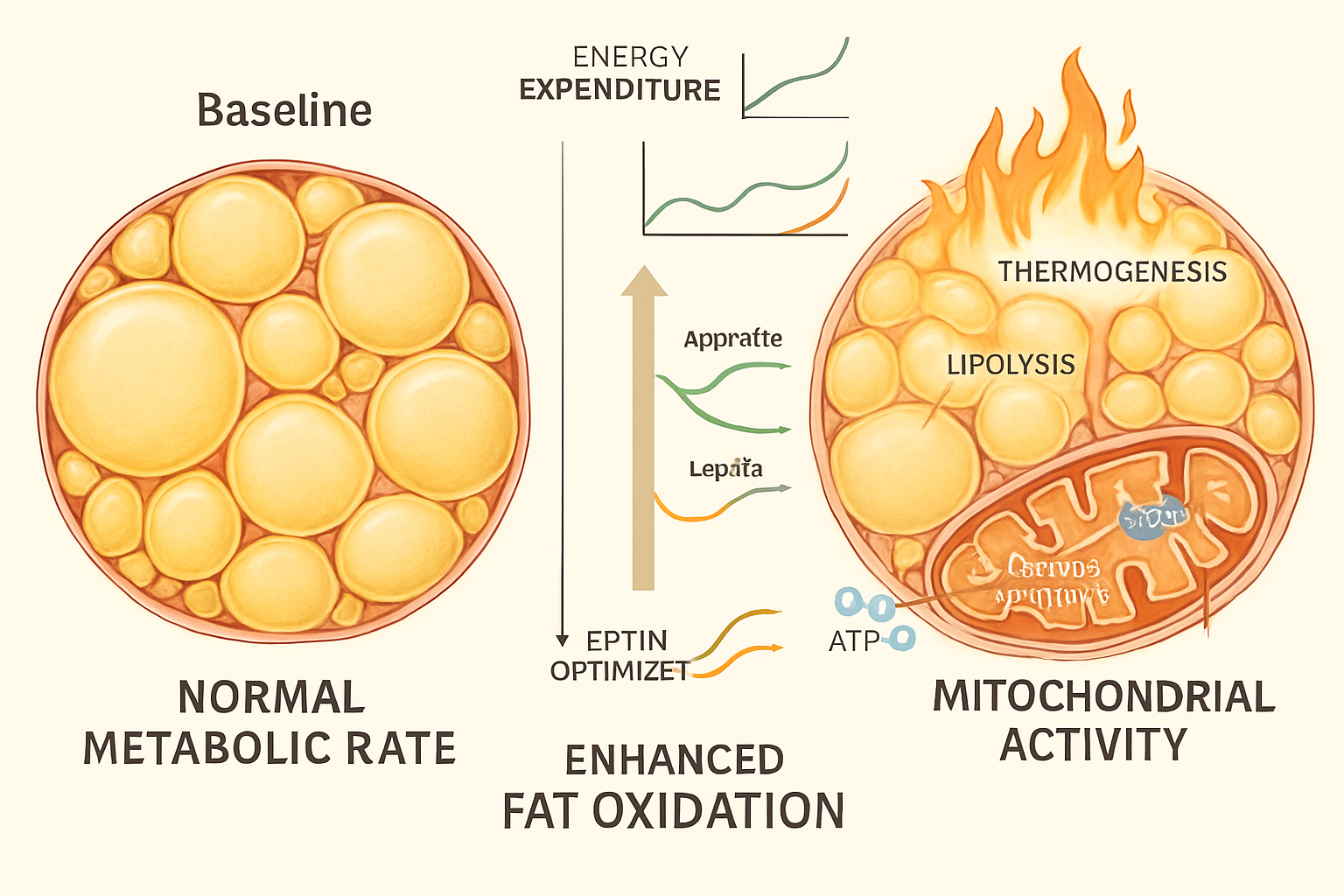
Understanding how the retatrutide fat burning mechanism compares to earlier compounds provides valuable context for researchers evaluating peptide options.
Single-Agonist Compounds (GLP-1 Only)
Traditional GLP-1 Agonists:
- Primarily focus on appetite suppression and glucose control
- Limited direct lipolytic activity
- Modest effects on energy expenditure
- Well-characterized safety profiles in research
Limitations:
- Fat loss primarily through caloric restriction
- Minimal direct thermogenic effects
- May not optimize metabolic flexibility
- Plateau effects observed in long-term studies
Examples include compounds like semaglutide and liraglutide, which have established research profiles but lack the multi-receptor approach.
Dual-Agonist Compounds (GLP-1/GIP)
Tirzepatide Profile:
- Combines GLP-1 and GIP receptor activation
- Enhanced glucose control compared to GLP-1 alone
- Improved weight reduction in research models
- Synergistic effects on insulin secretion
Advantages Over Single Agonists:
- Greater magnitude of fat mass reduction
- Better glycemic control
- Potentially improved tolerability profiles
- Enhanced satiety signaling
Limitations Compared to Triple Agonists:
- Lacks direct glucagon-mediated lipolysis
- Lower energy expenditure enhancement
- Reduced thermogenic activity
- Less pronounced effects on hepatic fat oxidation
Researchers interested in dual-agonist comparisons can explore tirzepatide compounds alongside retatrutide to evaluate mechanistic differences.
Triple-Agonist Advantages (Retatrutide)
Unique Mechanistic Features:
✅ Comprehensive Fat Mobilization: Glucagon component adds direct lipolytic activity ✅ Enhanced Energy Expenditure: Significant thermogenic effects beyond appetite suppression ✅ Metabolic Flexibility: Improved substrate switching between glucose and fat ✅ Hepatic Fat Reduction: Direct effects on liver metabolism and fat oxidation ✅ Synergistic Receptor Activation: Three pathways working in concert exceed additive effects
Research Observations:
- Greater magnitude of fat mass reduction in head-to-head comparisons
- More pronounced improvements in metabolic markers
- Enhanced effects on body composition (fat vs. lean mass)
- Potential for addressing metabolic dysfunction beyond weight alone
Considerations:
- More complex pharmacology requires careful research design
- Receptor balance may influence individual response patterns
- Longer-term studies needed to fully characterize effects
- Quality and purity of research compounds critical for reproducibility
The triple-agonist approach represents the current frontier in metabolic peptide research, offering mechanisms that extend substantially beyond earlier generations of compounds.
Research Applications and Experimental Considerations
For laboratories and researchers investigating the retatrutide fat burning mechanism, several practical considerations ensure optimal experimental design and data quality.
Appropriate Research Models
In Vitro Systems:
- Adipocyte cell cultures for direct lipolysis measurement
- Hepatocyte models for glucose and lipid metabolism studies
- Receptor binding assays for pharmacological characterization
- Mitochondrial function assessments in isolated organelles
Ex Vivo Preparations:
- Adipose tissue explants for integrated metabolic responses
- Liver slices for hepatic metabolism studies
- Hypothalamic tissue for appetite signaling pathways
- Muscle preparations for glucose uptake and oxidation
In Vivo Models:
- Diet-induced obesity models for metabolic syndrome research
- Genetic obesity models for mechanistic investigations
- Lean models for baseline metabolic characterization
- Transgenic receptor knockout models for pathway validation
Dosing and Administration Protocols
Critical Parameters:
Dose Selection:
- Receptor activation profiles vary with concentration
- Dose-response curves inform optimal experimental ranges
- Consider receptor-specific EC50 values for balanced activation
- Pilot studies establish appropriate dosing for specific endpoints
Administration Routes:
- Subcutaneous injection most common in research models
- Intraperitoneal administration for certain experimental designs
- Continuous infusion via osmotic pumps for steady-state studies
- Route influences pharmacokinetics and receptor exposure
Timing Considerations:
- Half-life characteristics inform dosing frequency
- Circadian metabolic rhythms may influence response
- Acute vs. chronic treatment protocols yield different insights
- Washout periods necessary for crossover designs
Outcome Measurement and Analysis
Metabolic Endpoints:
Body Composition:
- Dual-energy X-ray absorptiometry (DEXA) for fat and lean mass
- Magnetic resonance imaging (MRI) for tissue distribution
- Computed tomography (CT) for visceral vs. subcutaneous fat
- Bioelectrical impedance for longitudinal monitoring
Energy Metabolism:
- Indirect calorimetry for oxygen consumption and CO2 production
- Respiratory exchange ratio (RER) indicates substrate utilization
- Activity monitoring separates basal from total energy expenditure
- Thermoneutral vs. ambient temperature influences thermogenesis
Biochemical Markers:
- Plasma free fatty acids indicate active lipolysis
- Glycerol release confirms triglyceride breakdown
- Ketone bodies reflect hepatic fat oxidation
- Glucose and insulin for metabolic control assessment
Molecular Analyses:
- Gene expression profiling for transcriptional responses
- Protein analysis for enzyme activation states
- Receptor expression and localization studies
- Signaling pathway activation (Western blotting, phospho-specific antibodies)
Quality Control and Reproducibility
Compound Considerations:
🔬 Purity Standards: Research-grade peptides should meet >98% purity specifications 🔬 Storage Conditions: Lyophilized peptides require -20°C or -80°C storage; reconstituted solutions need refrigeration 🔬 Reconstitution Protocols: Appropriate solvents and concentrations prevent aggregation 🔬 Stability Testing: Time-course stability assessments ensure compound integrity 🔬 Batch Consistency: Certificates of analysis (COA) verify composition and purity
Researchers can ensure optimal compound quality by sourcing from established peptide suppliers with documented purity standards and proper handling protocols.
Experimental Controls:
- Vehicle-treated controls account for administration effects
- Pair-feeding controls separate appetite effects from direct metabolic actions
- Positive controls with established compounds validate experimental systems
- Time-matched measurements account for circadian variations
Storage, Handling, and Reconstitution Best Practices
Proper handling of retatrutide research compounds directly impacts experimental reproducibility and data quality. Understanding optimal storage and reconstitution protocols proves essential for researchers investigating the retatrutide fat burning mechanism.
Lyophilized Peptide Storage
Temperature Requirements:
- Short-term (up to 3 months): -20°C in sealed containers with desiccant
- Long-term (beyond 3 months): -80°C for maximum stability
- Avoid: Repeated freeze-thaw cycles that may compromise peptide integrity
- Monitor: Storage temperature logs ensure consistent conditions
Environmental Controls:
- Low humidity environment prevents moisture absorption
- Light-protected containers (amber pen peptides) prevent photodegradation
- Inert atmosphere (nitrogen or argon) for extended storage periods
- Sealed containers prevent oxidation and contamination
Inventory Management:
- First-in, first-out rotation minimizes storage duration
- Clear labeling with receipt date and storage conditions
- Separate aliquots prevent repeated access to primary stock
- Documentation of storage history for quality assurance
Reconstitution Protocols
Solvent Selection:
Sterile Water:
- Suitable for short-term use (24-48 hours)
- Minimal interference with biological assays
- Cost-effective for immediate-use applications
- Requires refrigerated storage after reconstitution
Bacteriostatic Water:
- Contains benzyl alcohol (0.9%) as preservative
- Extends stability of reconstituted solutions (up to 14 days refrigerated)
- Preferred for multi-dose applications
- Not suitable for certain cell culture applications
Buffer Solutions:
- Phosphate-buffered saline (PBS) for pH stability
- Tris buffers for specific pH requirements
- Consider osmolality for in vivo applications
- May enhance stability for particular peptides
Reconstitution Technique:
- Equilibration: Allow lyophilized pen peptide to reach room temperature (prevents condensation)
- Solvent Addition: Add solvent slowly down the pen peptide wall (not directly onto peptide cake)
- Gentle Mixing: Swirl gently or allow to dissolve naturally (avoid vigorous shaking)
- Visual Inspection: Ensure complete dissolution with clear solution (no particulates)
- Concentration Verification: Calculate final concentration accurately for dosing
- Aliquoting: Divide into single-use aliquots to minimize freeze-thaw cycles
Post-Reconstitution Handling
Storage Conditions:
- Refrigerate at 2-8°C immediately after reconstitution
- Protect from light using amber pen peptides or foil wrapping
- Use within recommended timeframe (varies by solvent)
- Document reconstitution date and concentration
Stability Considerations:
- Peptide degradation accelerates in solution vs. lyophilized form
- pH extremes may promote hydrolysis or aggregation
- Repeated temperature fluctuations reduce stability
- Microbial contamination risk increases with time
Quality Verification:
- Periodic analytical testing (HPLC, mass spectrometry) confirms integrity
- Visual inspection before each use detects precipitation or contamination
- pH monitoring ensures solution stability
- Concentration verification via UV spectroscopy or other methods
Contamination Prevention
Aseptic Technique:
- Work in laminar flow hood when possible
- Use sterile needles and syringes for each access
- Disinfect pen peptide septum with alcohol before needle insertion
- Avoid touching sterile surfaces or components
Cross-Contamination Avoidance:
- Dedicated equipment for each peptide compound
- Thorough cleaning protocols between different compounds
- Separate storage areas for different research materials
- Clear labeling prevents mix-ups
Documentation:
- Maintain detailed records of handling procedures
- Log storage conditions and any temperature excursions
- Document reconstitution details (date, solvent, concentration)
- Track usage and remaining volume for inventory
Researchers seeking comprehensive guidance on peptide handling can reference educational resources specifically designed for research applications.
Future Research Directions and Mechanistic Questions
While current understanding of the retatrutide fat burning mechanism has advanced considerably, numerous questions remain that represent valuable research opportunities.
Tissue-Specific Receptor Expression and Response
Outstanding Questions:
- How does receptor expression heterogeneity across tissues influence overall metabolic response?
- Do certain adipose depots (visceral vs. subcutaneous) show differential sensitivity?
- What role do receptor splice variants or isoforms play in tissue-specific effects?
- How do receptor expression patterns change with metabolic state or disease conditions?
Research Approaches:
- Single-cell RNA sequencing to map receptor expression at cellular resolution
- Tissue-specific receptor knockout models to isolate individual contributions
- Receptor imaging techniques to visualize activation patterns in vivo
- Comparative studies across different adipose depots and metabolic tissues
Molecular Mechanisms of Synergy
Key Mechanistic Gaps:
- What molecular interactions create synergy between the three receptor pathways?
- Do signaling pathways converge on common downstream effectors?
- How do temporal dynamics of receptor activation influence overall response?
- Are there non-canonical signaling pathways contributing to metabolic effects?
Investigative Strategies:
- Systems biology approaches integrating multi-omic datasets
- Detailed signaling pathway mapping with phosphoproteomics
- Mathematical modeling of receptor crosstalk and integration
- Selective receptor activation studies to dissect individual contributions
Long-Term Metabolic Adaptations
Unanswered Questions:
- How do compensatory metabolic mechanisms develop with chronic treatment?
- Does receptor desensitization or downregulation limit long-term efficacy?
- What metabolic adaptations occur in response to sustained negative energy balance?
- How does treatment cessation affect metabolic parameters and fat regain?
Research Needs:
- Extended duration studies examining chronic treatment effects
- Receptor expression and sensitivity assessments across treatment timelines
- Metabolic phenotyping during treatment and after withdrawal
- Epigenetic modifications that may influence long-term responses
Individual Variability and Precision Medicine
Critical Considerations:
- What genetic factors influence individual response to triple-agonist treatment?
- How do baseline metabolic phenotypes predict treatment outcomes?
- Do sex differences affect receptor expression or downstream signaling?
- What role does gut microbiome composition play in metabolic responses?
Research Directions:
- Pharmacogenomic studies identifying response predictors
- Baseline metabolic profiling to stratify research subjects
- Sex-specific analyses of receptor biology and metabolic outcomes
- Microbiome manipulation studies examining metabolic interactions
Cellular and Molecular Mechanisms Beyond Fat Metabolism
Emerging Areas:
- Effects on muscle metabolism and protein synthesis
- Bone metabolism influences through GIP receptor activation
- Cardiovascular effects beyond metabolic improvements
- Neurological impacts on cognition and neuroprotection
- Immune system modulation and inflammatory responses
Potential Research Applications:
- Sarcopenia and muscle wasting models
- Bone density and remodeling studies
- Cardiovascular function and protection research
- Neurodegenerative disease models
- Inflammatory condition investigations
These research directions represent opportunities for laboratories equipped with high-quality research peptides to contribute meaningful insights to the growing body of knowledge surrounding triple-agonist mechanisms.
The Comprehensive Nature of Retatrutide’s Metabolic Impact
The retatrutide fat burning mechanism exemplifies the sophistication achievable through multi-receptor pharmacology in metabolic research. By simultaneously engaging GLP-1, GIP, and glucagon pathways, retatrutide creates a comprehensive metabolic environment that addresses fat metabolism through multiple complementary mechanisms:
Direct Lipolytic Activity: Glucagon receptor activation stimulates hormone-sensitive lipase and adipose triglyceride lipase, promoting the breakdown of stored triglycerides into free fatty acids and glycerol.
Enhanced Energy Expenditure: Increased basal metabolic rate, thermogenesis, and mitochondrial fat oxidation ensure that mobilized fatty acids undergo complete oxidation rather than re-esterification.
Appetite Regulation: GLP-1 and GIP receptor-mediated satiety signaling reduces caloric intake, creating the energy deficit necessary for net fat mass reduction.
Metabolic Integration: Hepatic effects, adipokine modulation, and improved insulin sensitivity create systemic metabolic improvements extending beyond simple fat loss.
Synergistic Receptor Activation: The combination of three receptor pathways produces effects exceeding what would be predicted from simple addition of individual pathway contributions.
For researchers investigating metabolic mechanisms, energy homeostasis, or obesity-related pathways, retatrutide represents a valuable tool offering insights into complex receptor interactions and integrated metabolic regulation. The compound’s multi-faceted mechanism provides opportunities to examine questions ranging from cellular lipase regulation to whole-body energy balance and metabolic flexibility.
Next Steps for Researchers
Sourcing Quality Research Compounds: Begin by identifying reputable suppliers offering research-grade retatrutide with documented purity specifications and certificates of analysis.
Experimental Design: Develop comprehensive protocols incorporating appropriate controls, multiple metabolic endpoints, and sufficient statistical power to detect meaningful effects.
Mechanistic Focus: Consider which specific aspects of the retatrutide mechanism align with research objectives—receptor pharmacology, cellular metabolism, or whole-body energy balance.
Collaboration Opportunities: Complex metabolic research often benefits from multidisciplinary approaches combining expertise in endocrinology, biochemistry, molecular biology, and physiology.
Literature Monitoring: The retatrutide research landscape continues evolving rapidly; staying current with emerging publications ensures experimental approaches reflect latest mechanistic insights.
The comprehensive nature of the retatrutide fat burning mechanism positions this triple-agonist peptide at the forefront of metabolic research, offering unprecedented opportunities to understand and manipulate energy homeostasis through sophisticated multi-receptor pharmacology. As research continues to elucidate the molecular details and physiological implications of this mechanism, retatrutide will undoubtedly remain a valuable tool for laboratories investigating the fundamental biology of fat metabolism and energy regulation.
