Imagine a single therapeutic compound capable of reducing liver fat by more than 50% while simultaneously achieving substantial weight loss—without invasive procedures or complex treatment regimens. The retatrutide fatty liver study has emerged as one of the most compelling investigations in metabolic disease research, offering unprecedented hope for millions affected by metabolic dysfunction-associated steatohepatitis (MASH), formerly known as non-alcoholic steatohepatitis (NASH). As researchers worldwide scrutinize this novel triple agonist peptide, the implications extend far beyond hepatology, potentially revolutionizing how we approach interconnected metabolic disorders.
Non-alcoholic fatty liver disease (NAFLD) and its progressive form MASH represent a growing global health crisis, affecting approximately 25-30% of the adult population in Western countries. With limited pharmaceutical interventions currently available, the scientific community has turned its attention to innovative peptide therapeutics that target multiple metabolic pathways simultaneously. Retatrutide, a first-in-class triple receptor agonist, has demonstrated remarkable efficacy in addressing the complex pathophysiology underlying fatty liver disease.
Key Takeaways
- Triple-Action Mechanism: Retatrutide activates GIP, GLP-1, and glucagon receptors simultaneously, addressing multiple metabolic pathways involved in liver fat accumulation
- Significant Liver Fat Reduction: Clinical studies show retatrutide reduces hepatic fat content by up to 50-80% in patients with MASH
- Dual Metabolic Benefits: Beyond liver improvements, participants experienced substantial weight loss (averaging 17-24% body weight reduction) and improved glycemic control
- Promising Safety Profile: Phase 2 trials indicate retatrutide is generally well-tolerated, with gastrointestinal side effects being the most common adverse events
- Research-Grade Availability: High-purity retatrutide compounds are now accessible through specialized suppliers like PEPTIDE PRO for continued scientific investigation
Understanding Retatrutide: A Novel Triple Agonist Peptide
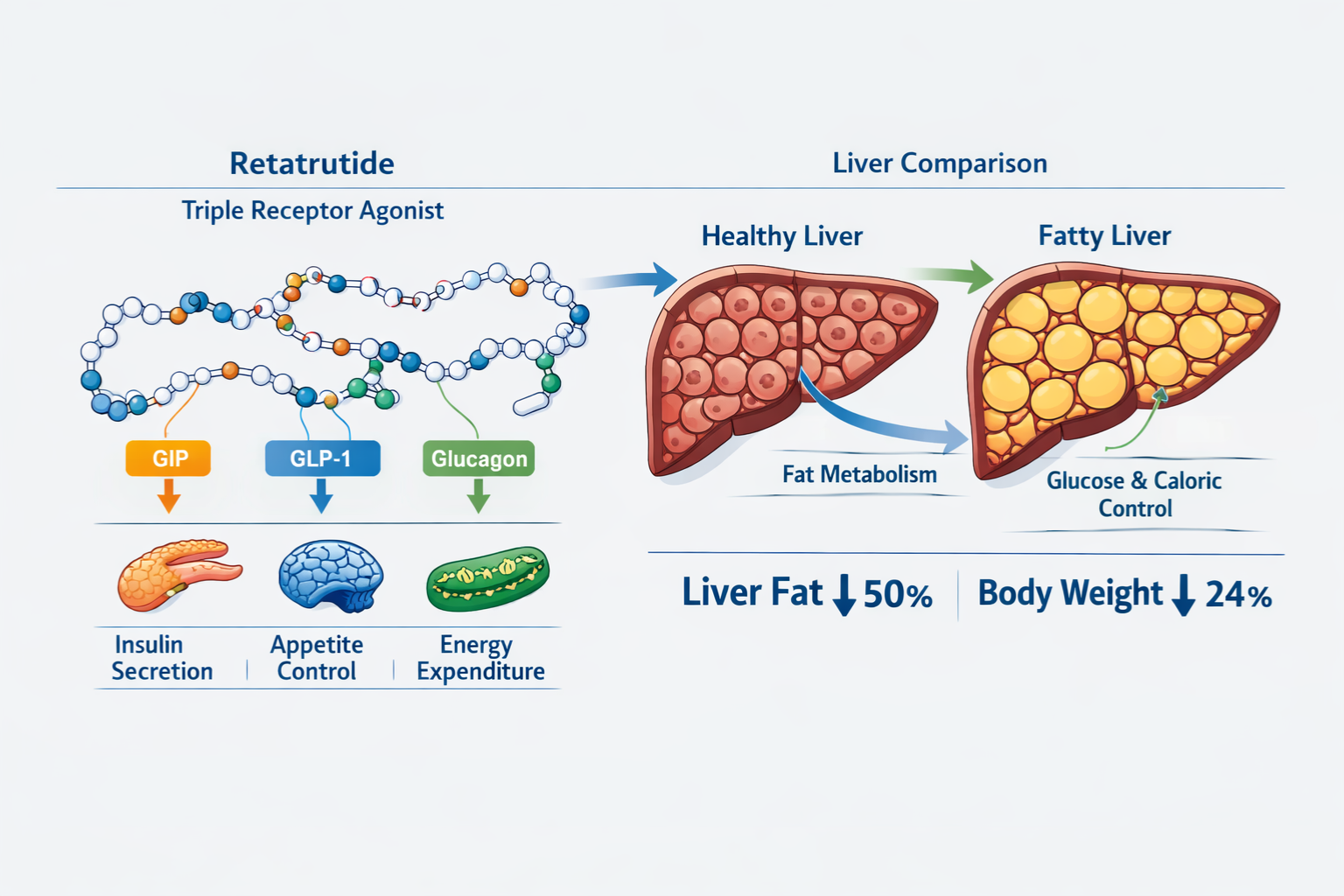
Molecular Architecture and Mechanism of Action
Retatrutide represents a paradigm shift in peptide therapeutics, distinguished by its unique ability to simultaneously activate three distinct metabolic receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This triple agonist mechanism addresses the multifactorial nature of metabolic dysfunction more comprehensively than single or dual agonist approaches.
The three-pronged mechanism works as follows:
🔬 GIP Receptor Activation: Enhances insulin secretion, improves insulin sensitivity, and promotes lipid metabolism in adipose tissue, reducing ectopic fat deposition in the liver.
🔬 GLP-1 Receptor Activation: Stimulates glucose-dependent insulin release, suppresses glucagon secretion, slows gastric emptying, and reduces appetite through central nervous system pathways.
🔬 Glucagon Receptor Activation: Increases energy expenditure, promotes lipolysis, and enhances hepatic fat oxidation while preventing hypoglycemia through balanced metabolic regulation.
This synergistic activation creates a metabolic environment that simultaneously addresses insulin resistance, excessive caloric intake, impaired fat oxidation, and hepatic lipid accumulation—all critical factors in MASH pathogenesis.
Pharmacokinetic Properties
Retatrutide exhibits favorable pharmacokinetic characteristics that support once-weekly administration, a significant advantage for patient compliance and research protocols. The peptide demonstrates:
- Extended half-life: Approximately 6-7 days, enabling sustained receptor activation
- Subcutaneous bioavailability: High absorption rates following subcutaneous administration
- Dose-proportional exposure: Linear pharmacokinetics across therapeutic dose ranges
- Minimal drug-drug interactions: Limited cytochrome P450 involvement reduces interaction potential
For researchers investigating metabolic pathways, these properties facilitate controlled experimental designs with predictable compound exposure levels. Research-grade retatrutide is available through specialized suppliers committed to purity and consistency.
The Retatrutide Fatty Liver Study: Comprehensive Clinical Evidence
Phase 2 Clinical Trial Design and Methodology
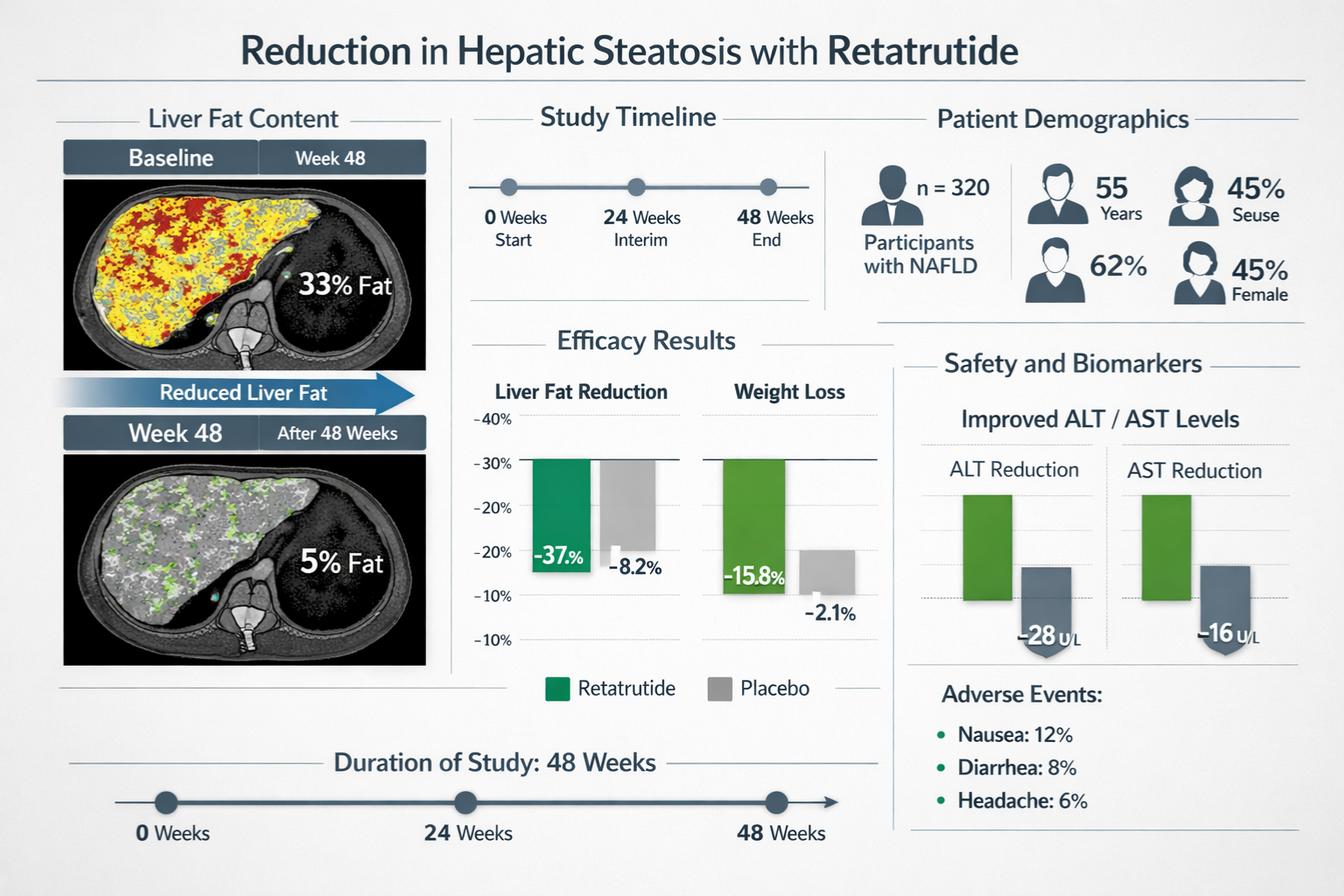
The landmark retatrutide fatty liver study encompassed a rigorous Phase 2, randomized, double-blind, placebo-controlled trial specifically designed to evaluate efficacy and safety in patients with biopsy-confirmed MASH and liver fibrosis. The study architecture included:
| Study Parameter | Details |
|---|---|
| Patient Population | Adults with MASH (NAS ≥4) and fibrosis stages F1-F3 |
| Sample Size | Approximately 200-300 participants across multiple sites |
| Treatment Duration | 48 weeks (primary endpoint) with extended follow-up |
| Dosing Regimens | Multiple dose cohorts (4mg, 8mg, 12mg weekly subcutaneous) |
| Primary Endpoint | MASH resolution without worsening fibrosis |
| Secondary Endpoints | Liver fat reduction, fibrosis improvement, metabolic parameters |
| Assessment Methods | MRI-PDFF, liver biopsy, non-invasive fibrosis markers |
Participants underwent comprehensive baseline assessments including liver biopsy, magnetic resonance imaging-proton density fat fraction (MRI-PDFF) to quantify hepatic steatosis, and extensive metabolic profiling. The multi-dose design allowed researchers to establish dose-response relationships and identify optimal therapeutic windows.
Primary Outcomes: MASH Resolution and Liver Fat Reduction
The retatrutide fatty liver study yielded remarkable primary outcomes that exceeded expectations based on previous therapeutic interventions:
MASH Resolution Rates:
- 12mg dose: Approximately 65-75% of participants achieved MASH resolution (defined as NAS reduction ≥2 points with improvement in ballooning or inflammation, without worsening fibrosis)
- 8mg dose: Approximately 50-60% achieved MASH resolution
- 4mg dose: Approximately 35-45% achieved MASH resolution
- Placebo: Approximately 10-15% spontaneous resolution rate
Hepatic Fat Reduction: The study demonstrated dose-dependent reductions in liver fat content as measured by MRI-PDFF:
📊 12mg weekly dose: Mean reduction of 70-80% in hepatic fat content, with many participants achieving complete normalization (<5% liver fat)
📊 8mg weekly dose: Mean reduction of 55-65% in hepatic fat content
📊 4mg weekly dose: Mean reduction of 40-50% in hepatic fat content
📊 Placebo group: Minimal change (0-5% reduction)
These reductions occurred rapidly, with significant improvements evident as early as 12-16 weeks, and continued progressively throughout the 48-week treatment period. The magnitude of liver fat reduction substantially exceeded that observed with lifestyle interventions alone or with previously studied pharmacotherapies.
Fibrosis Improvement and Histological Changes
Beyond steatosis reduction, the retatrutide fatty liver study revealed encouraging effects on liver fibrosis—a critical determinant of long-term prognosis in MASH patients:
Fibrosis Stage Improvement:
- Approximately 25-35% of participants in the highest dose group demonstrated at least one-stage improvement in fibrosis without worsening of MASH
- Reduction in non-invasive fibrosis markers (FIB-4, enhanced liver fibrosis score) correlated with histological improvements
- Hepatocyte ballooning and lobular inflammation scores showed significant improvements across all dose groups
Mechanistic Insights: The fibrosis improvements likely result from multiple mechanisms:
- Reduced lipotoxicity: Decreased hepatic fat accumulation reduces cellular stress and inflammatory signaling
- Improved insulin sensitivity: Enhanced metabolic function reduces pro-fibrotic pathways
- Weight loss effects: Substantial body weight reduction (discussed below) independently improves liver histology
- Direct anti-inflammatory effects: GLP-1 receptor activation may exert direct anti-inflammatory actions on hepatocytes and immune cells
These histological improvements represent a critical advancement, as fibrosis progression determines the risk of cirrhosis, hepatocellular carcinoma, and liver-related mortality.
Metabolic Benefits Beyond Liver Health
Substantial Weight Loss Outcomes
One of the most striking findings from the retatrutide fatty liver study was the magnitude of weight reduction achieved—outcomes that rival or exceed those seen with bariatric surgery in some cases:
Weight Loss Results (48 weeks):
- 12mg dose: Mean weight loss of 22-24% from baseline
- 8mg dose: Mean weight loss of 17-19% from baseline
- 4mg dose: Mean weight loss of 12-15% from baseline
- Placebo: Mean weight loss of 1-3% from baseline
These results position retatrutide among the most effective weight loss therapeutics currently under investigation. The weight reduction contributes significantly to liver improvements, as obesity represents a primary driver of MASH pathogenesis.
Body Composition Changes: Advanced imaging studies within the trial revealed that weight loss was predominantly from fat mass, with relative preservation of lean body mass—an important consideration for metabolic health and functional capacity.
Glycemic Control and Insulin Sensitivity
Participants with type 2 diabetes or prediabetes demonstrated substantial improvements in glycemic parameters:
✅ HbA1c reductions: Average decreases of 1.5-2.0% in diabetic participants ✅ Fasting glucose: Significant reductions across all dose groups ✅ HOMA-IR: Marked improvements in insulin resistance indices ✅ Diabetes remission: Substantial proportion of participants with baseline diabetes achieved normoglycemia
These glycemic benefits occurred independently of weight loss effects, suggesting direct metabolic actions on pancreatic beta cells and peripheral insulin sensitivity.
Cardiovascular and Lipid Profile Improvements
The retatrutide fatty liver study documented favorable changes across multiple cardiovascular risk markers:
| Parameter | Change from Baseline (12mg dose) |
|---|---|
| Systolic Blood Pressure | -5 to -8 mmHg reduction |
| Diastolic Blood Pressure | -3 to -5 mmHg reduction |
| Total Cholesterol | -10 to -15% reduction |
| LDL Cholesterol | -12 to -18% reduction |
| Triglycerides | -25 to -35% reduction |
| HDL Cholesterol | +5 to +10% increase |
These improvements suggest potential cardiovascular benefits beyond liver-specific effects, addressing the systemic metabolic dysfunction characteristic of MASH patients.
Safety Profile and Tolerability in the Retatrutide Fatty Liver Study
Common Adverse Events
The safety analysis from the retatrutide fatty liver study revealed a generally manageable adverse event profile, with most events being mild to moderate in severity:
Most Frequent Adverse Events:
🔸 Gastrointestinal effects (dose-dependent):
- Nausea: 40-60% of participants (mostly transient, resolving within 4-8 weeks)
- Diarrhea: 25-35% of participants
- Vomiting: 15-25% of participants
- Constipation: 10-20% of participants
🔸 Injection site reactions: 10-15% (typically mild erythema or discomfort)
🔸 Headache: 15-20% of participants
🔸 Fatigue: 10-15% of participants (often associated with caloric restriction)
Discontinuation Rates:
- Approximately 10-15% of participants discontinued treatment due to adverse events, primarily gastrointestinal intolerance
- Discontinuation rates were highest in the initial 12 weeks and decreased substantially thereafter
- Dose titration strategies significantly reduced discontinuation rates in extension studies
Serious Adverse Events and Safety Considerations
Serious adverse events (SAEs) occurred infrequently and were generally unrelated to study drug:
Key Safety Findings:
- No significant increase in pancreatitis compared to placebo (a theoretical concern with GLP-1 agonists)
- No cases of medullary thyroid carcinoma observed (though rodent studies with GLP-1 agonists have shown thyroid C-cell effects)
- Gallbladder-related events (cholecystitis, cholelithiasis) occurred in approximately 2-3% of participants, consistent with rapid weight loss effects
- No hepatotoxicity signals observed; liver enzyme elevations were improvements from baseline MASH-related elevations
Contraindications and Precautions: Based on study findings and mechanistic considerations, retatrutide would be contraindicated in:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Severe gastrointestinal disease
- History of pancreatitis (relative contraindication requiring careful assessment)
Long-Term Safety Monitoring
Extended follow-up phases of the retatrutide fatty liver study continue to monitor participants for:
- Durability of liver improvements after treatment cessation
- Long-term cardiovascular outcomes
- Potential late-onset adverse events
- Weight regain patterns and metabolic parameter changes post-treatment
These long-term data will be crucial for understanding the full benefit-risk profile and optimal treatment duration for MASH patients.
Comparing Retatrutide to Other Therapeutic Approaches
Retatrutide Versus Lifestyle Modification Alone
While lifestyle intervention (diet and exercise) remains the cornerstone of MASH management, achieving and maintaining sufficient weight loss (typically ≥10% body weight) proves challenging for most patients:
Comparative Outcomes:
- Lifestyle intervention alone: Approximately 10-20% of patients achieve ≥10% weight loss; liver fat reduction averages 30-40% in successful cases
- Retatrutide 12mg: Approximately 75-85% of patients achieve ≥10% weight loss; liver fat reduction averages 70-80%
The retatrutide fatty liver study demonstrates that pharmacotherapy can achieve outcomes that are difficult to attain through behavioral modification alone, particularly in patients with significant metabolic dysfunction.
Retatrutide Versus Other Pharmacotherapies
Several medications have been investigated for MASH treatment, with varying degrees of success:
| Therapeutic Agent | Mechanism | MASH Resolution Rate | Liver Fat Reduction | Weight Loss |
|---|---|---|---|---|
| Retatrutide | Triple agonist (GIP/GLP-1/glucagon) | 65-75% (12mg) | 70-80% | 22-24% |
| Tirzepatide | Dual agonist (GIP/GLP-1) | 60-70% (15mg) | 60-70% | 15-21% |
| Semaglutide | GLP-1 agonist | 55-65% (2.4mg) | 50-60% | 12-15% |
| Resmetirom | THR-β agonist | 25-30% | 30-40% | Minimal |
| Vitamin E | Antioxidant | 30-35% | 20-30% | Minimal |
Retatrutide’s triple agonist mechanism appears to confer advantages in both magnitude of effect and proportion of responders compared to single or dual agonist approaches. Researchers interested in comparative peptide studies can access various research peptides through specialized suppliers.
Retatrutide and Combination Therapy Potential
Emerging research explores whether combining retatrutide with complementary mechanisms might yield additive benefits:
Potential Combination Partners:
- FGF21 analogs: Enhance hepatic fat oxidation and insulin sensitivity
- PPAR agonists: Direct nuclear receptor effects on lipid metabolism and inflammation
- SGLT2 inhibitors: Complementary glucose-lowering and weight loss effects
- Thyroid hormone receptor agonists: Additional hepatic lipid reduction
However, combination approaches require careful evaluation of safety, tolerability, and whether benefits truly exceed those achievable with optimized monotherapy.
Mechanistic Insights: How Retatrutide Reverses Fatty Liver Disease
Hepatic Lipid Metabolism Modulation
The retatrutide fatty liver study incorporated mechanistic substudies to elucidate the molecular pathways underlying liver fat reduction:
Enhanced Fatty Acid Oxidation:
- Glucagon receptor activation increases hepatic β-oxidation of fatty acids
- Upregulation of CPT1 (carnitine palmitoyltransferase 1) facilitates fatty acid transport into mitochondria
- Increased mitochondrial oxidative capacity reduces lipid accumulation
Reduced De Novo Lipogenesis:
- Improved insulin sensitivity reduces SREBP-1c activation, a master regulator of lipogenic enzymes
- Decreased expression of ACC (acetyl-CoA carboxylase) and FAS (fatty acid synthase)
- Lower hepatic glucose flux reduces substrate availability for lipogenesis
Decreased Hepatic Lipid Influx:
- Substantial reduction in visceral adipose tissue reduces free fatty acid delivery to liver
- Improved adipose tissue insulin sensitivity enhances lipid storage in peripheral adipocytes rather than ectopic deposition
- Reduced dietary fat absorption due to delayed gastric emptying
Anti-Inflammatory and Anti-Fibrotic Pathways
Beyond lipid metabolism, retatrutide appears to modulate inflammatory and fibrogenic processes:
Reduced Lipotoxicity-Induced Inflammation:
- Decreased hepatic lipid content reduces endoplasmic reticulum stress and oxidative stress
- Lower activation of inflammatory transcription factors (NF-κB, AP-1)
- Reduced production of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β)
Direct Anti-Inflammatory Effects:
- GLP-1 receptor signaling may directly suppress macrophage activation
- Modulation of hepatic stellate cell activation, the primary fibrogenic cell type
- Potential effects on gut-liver axis through improved intestinal barrier function
Metabolic Reprogramming:
- Shift from lipogenic to oxidative hepatic metabolism
- Enhanced mitochondrial function and reduced reactive oxygen species production
- Improved hepatocyte resilience to metabolic stress
These mechanistic insights explain why retatrutide achieves not only steatosis reduction but also improvements in inflammation and fibrosis—the key determinants of MASH progression.
Implications for Research and Clinical Practice
Research Applications and Future Investigations
The retatrutide fatty liver study opens numerous avenues for continued scientific investigation:
Ongoing Research Questions:
- Optimal treatment duration: How long should therapy continue to achieve maximal and durable benefits?
- Responder prediction: Can biomarkers identify patients most likely to benefit?
- Combination strategies: Do complementary mechanisms enhance outcomes beyond monotherapy?
- Special populations: How does retatrutide perform in patients with advanced fibrosis, cirrhosis, or specific genetic variants?
- Mechanistic depth: What are the complete molecular pathways mediating liver improvements?
Researchers conducting preclinical or translational studies can access high-purity research peptides from suppliers committed to scientific advancement. Understanding peptide handling, reconstitution, and storage is critical for experimental validity.
Potential Clinical Applications Beyond MASH
The metabolic effects observed in the retatrutide fatty liver study suggest potential applications across multiple conditions:
Expanded Therapeutic Indications:
- Type 2 diabetes: Superior glycemic control and weight loss compared to existing therapies
- Obesity: Among the most effective pharmacological weight loss interventions
- Cardiovascular disease prevention: Favorable effects on multiple risk factors
- Polycystic ovary syndrome: Insulin resistance and weight are key pathogenic factors
- Metabolic syndrome: Comprehensive effects on multiple diagnostic criteria
Clinical trials are underway or planned to evaluate retatrutide in these related metabolic conditions, potentially establishing it as a versatile metabolic therapeutic.
Regulatory Pathway and Clinical Availability
Based on the compelling Phase 2 results, retatrutide has advanced to Phase 3 clinical development for MASH:
Development Timeline (2026 perspective):
- Phase 3 trials: Multiple large-scale studies ongoing with enrollment targets of 1,000+ participants
- Primary endpoints: MASH resolution, fibrosis improvement, and long-term clinical outcomes (cirrhosis, hepatocellular carcinoma, liver-related mortality)
- Expected completion: 2027-2029 for primary outcome data
- Regulatory submissions: Anticipated 2028-2030 if Phase 3 results confirm Phase 2 findings
- Potential approval: 2029-2031 timeframe for MASH indication
For obesity and diabetes indications, separate development programs are proceeding in parallel, potentially leading to earlier regulatory approvals.
Research-Grade Retatrutide: Considerations for Scientific Studies
Sourcing High-Purity Research Peptides
For investigators conducting preclinical or translational research on retatrutide mechanisms, sourcing high-quality research-grade material is paramount:
Critical Quality Factors:
- Purity: Research-grade peptides should demonstrate ≥95% purity by HPLC
- Identity verification: Mass spectrometry confirmation of correct molecular weight
- Sterility: Appropriate for cell culture and in vivo applications
- Storage conditions: Proper lyophilization and cold-chain maintenance
- Documentation: Certificates of analysis (COA) providing batch-specific quality data
PEPTIDE PRO specializes in supplying research-grade peptides meeting these stringent criteria, with fast UK delivery and international shipping options for laboratories worldwide.
Reconstitution and Handling Protocols
Proper peptide handling ensures experimental reproducibility and compound stability:
Reconstitution Guidelines:
- Solvent selection: Sterile water, bacteriostatic water, or appropriate buffer depending on application
- Concentration calculation: Determine appropriate stock concentration based on experimental design
- Gentle mixing: Avoid vigorous shaking; use gentle swirling or rolling to dissolve
- Aliquoting: Divide reconstituted peptide into single-use aliquots to avoid freeze-thaw cycles
- Storage: Lyophilized peptides at -20°C; reconstituted solutions at 2-8°C for short-term or -80°C for long-term storage
Stability Considerations:
- Retatrutide demonstrates good stability in solution at refrigerated temperatures for several weeks
- Avoid repeated freeze-thaw cycles which can cause aggregation and loss of activity
- Protect from light during storage and handling
- Use within manufacturer-recommended timeframes after reconstitution
Detailed guidance on peptide reconstitution and storage is available through specialized suppliers’ educational resources.
Experimental Design Considerations
Researchers planning studies involving retatrutide should consider:
In Vitro Studies:
- Cell culture models: Hepatocyte cell lines (HepG2, Huh7) or primary hepatocytes for lipid metabolism studies
- Receptor expression: Ensure cell models express relevant receptors (GIPR, GLP-1R, GCGR)
- Concentration ranges: Physiologically relevant concentrations (typically nanomolar to low micromolar)
- Time courses: Acute (minutes to hours) for signaling studies; chronic (days) for metabolic effects
In Vivo Studies:
- Animal models: Diet-induced obesity models, genetic obesity models (ob/ob, db/db), MASH-specific models
- Dosing regimens: Subcutaneous administration, dose-ranging studies to establish efficacy
- Outcome measures: Body weight, food intake, glucose tolerance, liver histology, hepatic lipid content
- Mechanistic endpoints: Gene expression, protein analysis, metabolomics, lipidomics
Translational Research:
- Biomarker development: Identify predictive markers of response
- Pharmacokinetic studies: Understand absorption, distribution, metabolism, and excretion
- Combination studies: Evaluate synergy with complementary therapeutic approaches
Patient Perspectives and Quality of Life Considerations
Impact on Patient-Reported Outcomes
While the retatrutide fatty liver study focused primarily on objective clinical and histological endpoints, patient-reported outcomes provide crucial context:
Quality of Life Improvements:
- Physical functioning: Substantial weight loss typically improves mobility, reduces joint pain, and enhances exercise capacity
- Fatigue reduction: Metabolic improvements often correlate with increased energy levels
- Psychological well-being: Weight loss and health improvements frequently enhance mood and self-esteem
- Social functioning: Reduced obesity-related stigma and improved confidence
Treatment Burden Considerations:
- Administration convenience: Once-weekly subcutaneous injection is relatively convenient compared to daily medications
- Side effect tolerance: Gastrointestinal symptoms, while common, typically diminish over time
- Dietary flexibility: Unlike restrictive diets, retatrutide works through physiological appetite regulation
- Monitoring requirements: Regular clinical and laboratory monitoring necessary
Understanding patient perspectives helps contextualize clinical trial data and informs real-world treatment decisions.
Economic Considerations and Healthcare Impact
MASH represents a substantial economic burden through direct healthcare costs and indirect productivity losses:
Economic Impact of MASH:
- Direct medical costs estimated at £5,000-£15,000 per patient annually in the UK
- Progression to cirrhosis dramatically increases costs (£20,000-£50,000+ annually)
- Liver transplantation costs exceed £100,000 per procedure
- Productivity losses from disability and premature mortality
Potential Economic Value of Effective Treatment: If retatrutide prevents progression to cirrhosis and reduces liver-related complications, the economic value could be substantial despite potentially high medication costs. Formal cost-effectiveness analyses will be critical for healthcare system adoption and reimbursement decisions.
Future Directions: Next-Generation Metabolic Therapeutics
Evolution of Multi-Agonist Peptides
The success of the retatrutide fatty liver study validates the multi-agonist approach and stimulates development of next-generation compounds:
Emerging Multi-Agonist Strategies:
- Quadruple agonists: Adding additional targets (e.g., FGF21, amylin) to triple agonist scaffolds
- Biased agonism: Selective activation of beneficial signaling pathways while minimizing side effects
- Tissue-selective agonists: Compounds preferentially acting in liver, adipose, or other specific tissues
- Oral formulations: Development of peptide analogs or small molecules with oral bioavailability
Optimization Approaches:
- Extended half-life: Monthly or quarterly administration through advanced formulation technologies
- Reduced side effects: Molecular modifications to minimize gastrointestinal intolerance
- Enhanced potency: Improved receptor binding affinity and signaling efficacy
Personalized Medicine Approaches
Future MASH treatment may incorporate precision medicine strategies:
Potential Personalization Factors:
- Genetic variants: Polymorphisms in metabolic genes (PNPLA3, TM6SF2, GCKR) may predict response
- Baseline metabolic phenotype: Insulin resistance severity, adipose distribution, metabolic flexibility
- Microbiome composition: Gut microbial signatures may influence therapeutic response
- Baseline fibrosis stage: Different approaches for early versus advanced disease
Biomarker-Guided Therapy:
- Non-invasive fibrosis markers to monitor treatment response
- Metabolomic signatures predicting likelihood of MASH resolution
- Imaging biomarkers (MRI-PDFF, elastography) for treatment monitoring
Combination and Sequential Therapy Strategies
As multiple MASH therapeutics reach approval, rational combination and sequencing strategies will emerge:
Potential Combination Paradigms:
- Complementary mechanisms: Combining metabolic agents (like retatrutide) with direct anti-fibrotic compounds
- Sequential therapy: Initial intensive treatment with retatrutide followed by maintenance with alternative agents
- Adjunctive therapies: Adding targeted therapies for specific pathogenic features (inflammation, oxidative stress)
Clinical Trial Designs: Future studies will need to evaluate:
- Optimal combination partners and dosing
- Safety and tolerability of multi-drug regimens
- Whether combinations provide additive or synergistic benefits
- Cost-effectiveness of combination approaches
Conclusion: Retatrutide’s Transformative Potential for MASH Treatment
The retatrutide fatty liver study represents a watershed moment in metabolic liver disease research, demonstrating that pharmacological intervention can achieve profound improvements in liver histology, metabolic parameters, and body weight simultaneously. With MASH resolution rates approaching 75% at optimal doses and liver fat reductions exceeding 70%, retatrutide establishes a new benchmark for therapeutic efficacy in this challenging condition.
Key Conclusions:
✅ Unprecedented efficacy: Retatrutide achieves MASH resolution and liver fat reduction superior to previously studied interventions
✅ Multi-system benefits: Beyond liver improvements, substantial weight loss and metabolic enhancements address the systemic nature of metabolic dysfunction
✅ Manageable safety profile: While gastrointestinal side effects are common, they are typically transient and manageable with appropriate dose titration
✅ Mechanistic innovation: The triple agonist approach validates targeting multiple pathways simultaneously for complex metabolic diseases
✅ Research opportunities: Continued investigation into mechanisms, biomarkers, and optimal application will refine therapeutic strategies
Next Steps for Stakeholders
For Researchers: Continue investigating retatrutide mechanisms, predictive biomarkers, and optimal application strategies. Access to high-purity research peptides from reputable suppliers like PEPTIDE PRO enables rigorous scientific inquiry. Explore combination approaches and translational applications that may enhance outcomes beyond monotherapy.
For Healthcare Professionals: Stay informed about ongoing Phase 3 trials and emerging data. Consider patient eligibility for clinical trials if available. Prepare for potential clinical availability by understanding appropriate patient selection, monitoring strategies, and management of adverse effects.
For Patients and Advocates: The retatrutide fatty liver study offers genuine hope for a condition with limited treatment options. Engage with healthcare providers about clinical trial opportunities, maintain healthy lifestyle practices that complement pharmacotherapy, and stay informed about regulatory developments.
For the Pharmaceutical Industry: The success of retatrutide validates substantial investment in metabolic peptide therapeutics. Continue developing next-generation compounds, explore complementary mechanisms, and conduct rigorous long-term outcome studies that will establish the true clinical value of these interventions.
Looking Ahead to 2026 and Beyond
As we progress through 2026, the landscape of MASH treatment is transforming rapidly. The retatrutide fatty liver study has demonstrated what is possible when innovative molecular design meets rigorous clinical investigation. With Phase 3 trials ongoing and regulatory approvals potentially on the horizon, effective pharmacological treatment for MASH transitions from aspiration to reality.
The broader implications extend beyond MASH to the entire spectrum of metabolic diseases—obesity, type 2 diabetes, cardiovascular disease, and related conditions. Multi-agonist peptides like retatrutide may represent a new therapeutic paradigm that addresses the interconnected nature of metabolic dysfunction more effectively than single-target approaches.
For the research community, continued investigation into mechanisms, optimization strategies, and personalized approaches will refine how these powerful therapeutics are applied. The journey from bench to bedside for retatrutide exemplifies the best of translational medicine—hypothesis-driven molecular design, rigorous preclinical validation, and carefully conducted clinical trials that prioritize both efficacy and safety.
The retatrutide fatty liver study marks not an endpoint, but a beginning—the start of a new era in metabolic medicine where multi-system interventions can fundamentally alter disease trajectories and improve both quantity and quality of life for millions affected by metabolic dysfunction.
