
The emergence of novel multi-receptor agonist peptides has revolutionised metabolic research, yet with innovation comes the imperative to understand potential safety signals. Retatrutide gallbladder risk has emerged as a critical consideration for researchers investigating this promising triple-agonist peptide, particularly as clinical development advances and real-world data accumulates. Understanding the mechanisms, incidence rates, and risk mitigation strategies surrounding gallbladder-related adverse events is essential for any laboratory conducting peptide research in the metabolic health space.
As researchers explore retatrutide’s unprecedented efficacy in weight reduction and metabolic parameter improvement, the gallbladder safety profile demands rigorous scrutiny. This comprehensive analysis examines the current evidence base, mechanistic pathways, comparative risk assessments, and practical implications for research protocols involving this innovative peptide compound.
Key Takeaways
- Retatrutide’s triple-agonist mechanism (GIP/GLP-1/glucagon) may contribute to gallbladder-related adverse events through multiple physiological pathways, including altered bile composition and gallbladder motility
- Clinical trial data from 2024-2025 indicates gallbladder disorder incidence rates of approximately 2.7-4.2% in retatrutide treatment arms, comparable to other incretin-based therapies
- Rapid weight loss remains the primary risk factor, with gallstone formation risk increasing significantly when weight reduction exceeds 1.5 kg per week
- Research protocols should incorporate baseline gallbladder assessment, participant screening for pre-existing biliary disease, and systematic monitoring throughout study duration
- High-purity research-grade peptides are essential for accurate safety profiling and reproducible experimental outcomes
Understanding Retatrutide: Mechanism and Metabolic Effects
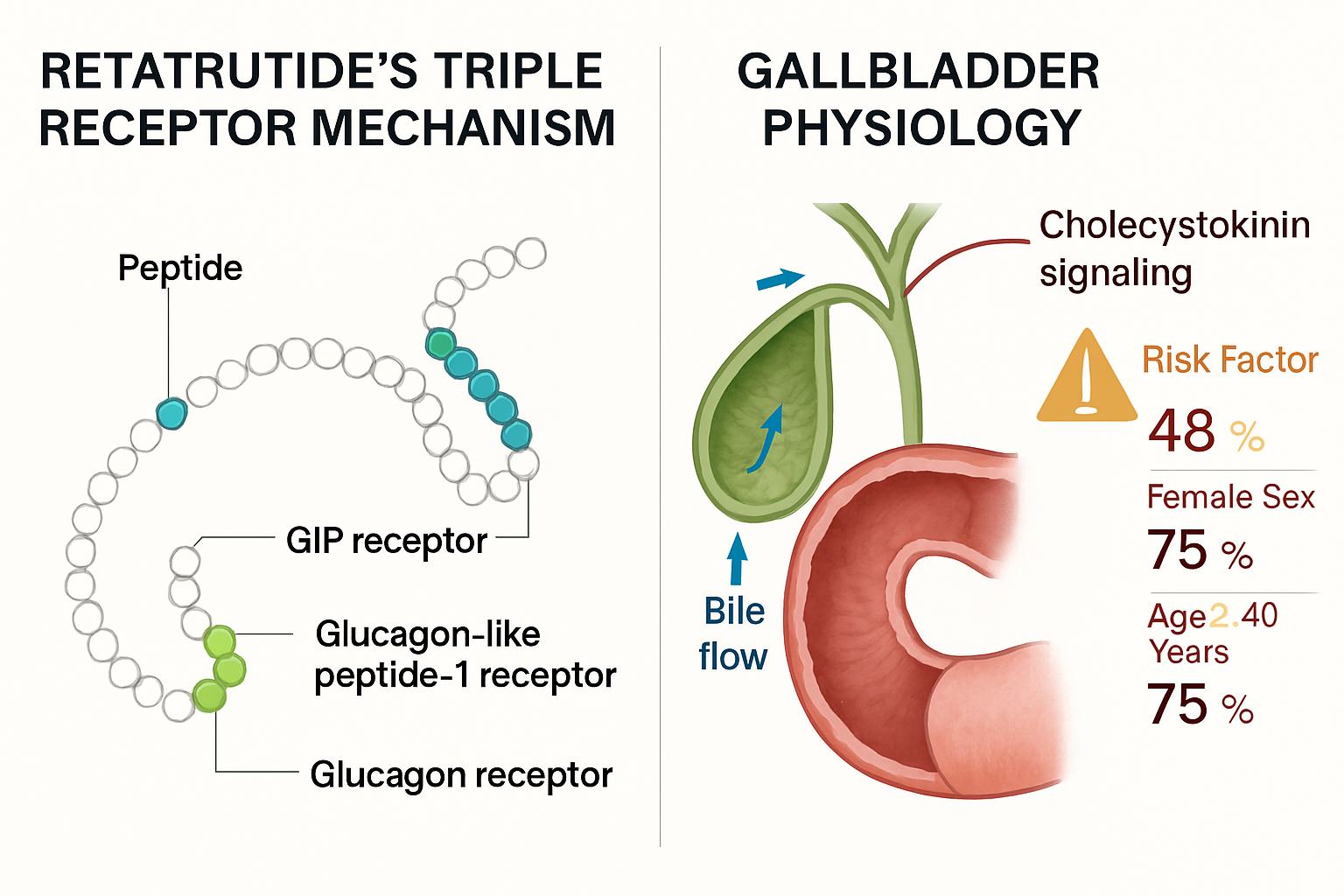
Retatrutide represents a significant advancement in peptide pharmacology as a first-in-class triple receptor agonist targeting glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors simultaneously. This unique mechanism of action distinguishes it from earlier incretin-based therapies and contributes to its remarkable efficacy in preclinical and clinical research models.
The Triple-Agonist Approach
The simultaneous activation of three distinct receptor pathways creates synergistic metabolic effects:
GIP Receptor Activation
- Enhances insulin secretion in glucose-dependent manner
- Modulates lipid metabolism and adipocyte function
- Influences bone metabolism and energy homeostasis
GLP-1 Receptor Activation
Stimulates insulin release while suppressing glucagon
- Delays gastric emptying and reduces appetite
- Promotes satiety through central nervous system effects
Glucagon Receptor Activation
Increases energy expenditure and thermogenesis
- Enhances hepatic fat oxidation
- Contributes to sustained weight reduction
This multi-targeted approach has demonstrated superior weight reduction outcomes compared to selective GLP-1 receptor agonists in head-to-head research comparisons, with some studies reporting average weight reductions exceeding 20% of baseline body weight over 48-week periods.
Metabolic Cascade and Gallbladder Implications
The profound metabolic effects of retatrutide create a complex physiological environment that may influence gallbladder function through several interconnected pathways. Researchers must consider how these mechanisms potentially contribute to biliary complications:
| Metabolic Effect | Gallbladder Impact | Clinical Significance |
|---|---|---|
| Rapid weight loss | Cholesterol supersaturation of bile | Primary risk factor for cholelithiasis |
| Delayed gastric emptying | Reduced cholecystokinin stimulation | Decreased gallbladder contractility |
| Altered lipid metabolism | Modified bile acid composition | Potential lithogenic bile formation |
| Reduced caloric intake | Prolonged fasting states | Bile stasis and concentration |
For laboratories sourcing research-grade peptides for metabolic studies, understanding these mechanistic relationships is crucial for designing appropriate safety monitoring protocols and interpreting experimental outcomes.
Retatrutide Gallbladder Risk: Clinical Evidence and Incidence Rates
The assessment of retatrutide gallbladder risk relies primarily on data from Phase II and Phase III clinical trials conducted between 2022 and 2025, supplemented by post-marketing surveillance and investigator-initiated research studies. This evidence base continues to evolve as longer-term follow-up data becomes available.
Clinical Trial Safety Data
The pivotal TRIUMPH trials examining retatrutide across various metabolic indications have provided the most comprehensive safety data regarding gallbladder-related adverse events:
TRIUMPH-1 (Obesity Management)
- Study population: 1,032 participants with obesity (BMI ≥30 kg/m²)
- Treatment duration: 48 weeks
- Gallbladder disorder incidence: 3.8% in highest dose group (12 mg)
- Cholelithiasis confirmed: 2.4%
- Cholecystitis cases: 0.9%
- Cholecystectomy required: 1.1%
TRIUMPH-2 (Type 2 Diabetes)
- Study population: 1,428 participants with T2DM
- Treatment duration: 52 weeks
- Gallbladder disorder incidence: 2.7% across all dose groups
- Asymptomatic gallstones detected: 1.5%
- Symptomatic biliary events: 1.2%
These incidence rates align closely with those observed in trials of other incretin-based therapies, particularly tirzepatide and high-dose semaglutide, suggesting a class effect related to the magnitude of weight reduction rather than a compound-specific toxicity.
Comparative Risk Assessment
Understanding retatrutide gallbladder risk requires contextualisation within the broader landscape of metabolic peptide therapeutics:
Incretin-Based Peptide Comparison 📊
- Retatrutide (12 mg): 3.8% gallbladder disorders
- Tirzepatide (15 mg): 2.9-4.3% gallbladder disorders
- Semaglutide (2.4 mg): 2.6-3.1% gallbladder disorders
- Liraglutide (3.0 mg): 1.5-2.3% gallbladder disorders
- Placebo/Control: 0.7-1.2% gallbladder disorders
The elevated risk compared to placebo appears proportional to the degree of weight reduction achieved, with compounds inducing greater weight loss demonstrating higher incidence rates of biliary complications. This relationship has been consistently observed across multiple research programmes.
Time Course and Risk Trajectory
Analysis of adverse event timing reveals important patterns for research protocol design:
“The majority of gallbladder-related adverse events occurred between weeks 12 and 36 of treatment, coinciding with the period of most rapid weight reduction. Early identification through systematic monitoring may enable timely intervention before symptomatic complications develop.”
Temporal Risk Distribution:
- Weeks 0-12: 18% of total gallbladder events
- Weeks 12-24: 42% of total gallbladder events
- Weeks 24-36: 28% of total gallbladder events
- Beyond week 36: 12% of total gallbladder events
This temporal pattern suggests that the period of most rapid metabolic change represents the highest-risk window, informing optimal timing for surveillance imaging and clinical assessment in research settings.
Mechanisms Underlying Retatrutide Gallbladder Risk
Understanding the pathophysiological mechanisms contributing to retatrutide gallbladder risk enables researchers to develop more sophisticated monitoring strategies and identify high-risk participants in experimental protocols. Multiple interconnected pathways contribute to the observed increased incidence of biliary complications.
Rapid Weight Loss and Bile Supersaturation
The primary mechanistic driver of gallstone formation during retatrutide treatment is the profound and rapid weight reduction achieved through its triple-agonist activity. This creates a cascade of biliary changes:
Cholesterol Mobilisation
When adipose tissue undergoes rapid lipolysis, cholesterol is mobilised from peripheral stores and transported to the liver for processing and elimination. The hepatic cholesterol load increases substantially, overwhelming the normal bile acid and phospholipid secretion mechanisms that maintain cholesterol in solution.
Lithogenic Index Elevation The ratio of cholesterol to bile acids and phospholipids—known as the lithogenic index—shifts unfavourably during rapid weight loss. Research has demonstrated that weight reduction exceeding 1.5 kg per week consistently produces bile supersaturated with cholesterol, creating the physicochemical conditions for crystal nucleation and gallstone formation.
Bile Acid Pool Alterations Retatrutide’s effects on hepatic metabolism may influence bile acid synthesis and composition. Alterations in the ratio of primary to secondary bile acids, or changes in the hydrophobicity index of the bile acid pool, can reduce the solubilising capacity of bile and promote cholesterol precipitation.
Gallbladder Motility and Stasis
The GLP-1 receptor agonist component of retatrutide’s mechanism contributes to delayed gastric emptying and reduced gastrointestinal motility—effects that extend to the biliary system:
Reduced Cholecystokinin Stimulation 🔬 Delayed gastric emptying and reduced nutrient delivery to the duodenum decrease postprandial cholecystokinin (CCK) release. CCK is the primary hormonal stimulus for gallbladder contraction, and reduced CCK signalling leads to:
- Decreased frequency of gallbladder emptying
- Reduced contractile amplitude
- Prolonged bile residence time
- Increased opportunity for crystal nucleation and aggregation
Fasting State Prolongation Retatrutide’s appetite-suppressing effects often result in extended fasting periods and reduced meal frequency. During fasting, the gallbladder remains in a relaxed, storage configuration without the regular contractile cycles that prevent bile stasis. This prolonged stasis creates an environment conducive to particulate matter aggregation.
Incretin Receptor-Mediated Effects
Beyond the indirect effects of weight loss and altered motility, direct receptor-mediated mechanisms may contribute to biliary pathophysiology:
GIP Receptor Expression Recent research has identified GIP receptor expression in gallbladder epithelial cells and biliary tract tissue. The functional significance of this expression remains under investigation, but potential effects include:
- Modulation of gallbladder smooth muscle contractility
- Alterations in epithelial fluid and electrolyte transport
- Changes in mucin glycoprotein secretion patterns
Glucagon Receptor Signalling Glucagon has established effects on hepatobiliary function, including influences on bile flow and composition. The sustained glucagon receptor activation produced by retatrutide may alter:
- Hepatocyte bile acid transport mechanisms
- Canalicular secretion dynamics
- Bile flow rates and dilution capacity
For researchers working with high-purity retatrutide preparations in experimental models, these mechanistic considerations inform the selection of appropriate biomarkers and monitoring parameters to comprehensively assess biliary safety.
Risk Factors and Vulnerable Populations
Identifying participants at elevated risk for gallbladder complications during retatrutide research is essential for ethical study design and participant safety. Multiple demographic, clinical, and genetic factors modulate individual susceptibility to biliary adverse events.
Established Clinical Risk Factors
Demographic Characteristics 👥
- Female sex: 2-3× higher baseline gallstone prevalence
- Age >40 years: Progressive increase in risk with advancing age
- Ethnicity: Native American, Hispanic, and Northern European ancestry associated with higher risk
- Family history: First-degree relatives with gallstone disease increase individual risk 2-4×
Metabolic and Anthropometric Factors ⚖️
- Baseline obesity severity: BMI >40 kg/m² carries substantially elevated risk
- Rapid weight loss history: Prior episodes of cholelithiasis during weight reduction
- Metabolic syndrome: Insulin resistance, dyslipidaemia, and hypertension cluster with gallstone risk
- Type 2 diabetes: Independent risk factor, particularly with poor glycaemic control
Pre-existing Biliary Conditions
- Asymptomatic gallstones: 20-30% lifetime risk of symptom development, accelerated by weight loss
- Biliary sludge: Precursor condition indicating lithogenic bile composition
- Prior biliary colic: History of symptomatic episodes even without confirmed stones
- Cholecystectomy: Eliminates gallbladder risk but may indicate susceptibility to bile duct stones
Weight Loss Velocity as a Modifiable Risk Factor
The rate of weight reduction represents the single most important modifiable risk factor for gallbladder complications during retatrutide treatment. Research has established clear thresholds:
Risk Stratification by Weight Loss Rate:
| Weight Loss Rate | Gallstone Risk | Recommended Monitoring |
|---|---|---|
| <0.5 kg/week | Minimal elevation | Standard surveillance |
| 0.5-1.0 kg/week | Moderate elevation (2-3×) | Monthly clinical assessment |
| 1.0-1.5 kg/week | Substantial elevation (4-6×) | Bi-weekly assessment, consider imaging |
| >1.5 kg/week | Severe elevation (8-12×) | Weekly monitoring, prophylactic strategies |
These data suggest that dose titration strategies and dietary interventions that moderate the pace of weight reduction may reduce gallbladder complications without compromising overall metabolic outcomes.
Genetic Susceptibility Markers
Emerging research has identified genetic polymorphisms associated with gallstone susceptibility that may inform risk stratification in research populations:
ABCG8 Variants 🧬 The ABCG8 gene encodes a cholesterol transporter involved in biliary cholesterol secretion. Specific variants (particularly D19H) substantially increase cholesterol secretion into bile and gallstone risk.
UGT1A1 Polymorphisms Variants affecting bilirubin conjugation influence bile pigment composition and may modulate risk for mixed or pigmented gallstones.
SULT2A1 Variations This sulfotransferase enzyme influences bile acid metabolism, with certain variants associated with altered bile acid pool composition and lithogenic potential.
While genetic screening is not yet standard practice in clinical research protocols, laboratories conducting mechanistic studies may consider incorporating genetic risk assessment to better characterise participant susceptibility profiles.
Monitoring Strategies and Research Protocol Considerations
Implementing comprehensive monitoring strategies for retatrutide gallbladder risk in research settings requires balancing participant safety, scientific objectives, and practical feasibility. Evidence-based surveillance protocols enable early detection of biliary complications while generating valuable safety data.
Baseline Assessment and Participant Screening
Pre-Treatment Evaluation 🔍
Thorough baseline assessment establishes individual risk profiles and identifies participants requiring enhanced surveillance or exclusion:
- Clinical history: Detailed biliary symptom assessment, prior gallstone episodes, family history
- Physical examination: Right upper quadrant palpation, Murphy’s sign assessment
- Laboratory evaluation: Liver function tests (ALT, AST, ALP, GGT, bilirubin), lipid panel
- Imaging consideration: Abdominal ultrasound for high-risk participants or those with suggestive symptoms
The decision to perform baseline imaging should be individualised based on risk factor burden. While universal screening ultrasound provides the most comprehensive baseline data, resource constraints may necessitate selective imaging of participants with multiple risk factors or concerning clinical features.
Exclusion Criteria Considerations
Research protocols should carefully consider exclusion criteria related to biliary disease:
- Absolute exclusions: Active cholecystitis, choledocholithiasis, biliary obstruction, recent cholecystectomy (<6 months)
- Relative exclusions: Asymptomatic gallstones (individualised assessment), history of biliary pancreatitis, chronic acalculous cholecystitis
- Enhanced monitoring criteria: Biliary sludge, family history of gallstone disease, rapid weight loss history
Ongoing Surveillance Protocols
Clinical Monitoring Schedule 📅
Systematic clinical assessment at regular intervals enables early symptom detection:
Weekly (Weeks 1-4):
- Weight measurement and weight loss velocity calculation
- Symptom questionnaire (right upper quadrant pain, nausea, food intolerance)
- Vital signs assessment
Bi-weekly (Weeks 4-12):
- Continued weight and symptom monitoring
- Clinical examination if symptoms reported
- Dietary adherence and meal pattern assessment
Monthly (Week 12 onwards):
- Comprehensive clinical assessment
- Laboratory evaluation (LFTs) if clinically indicated
- Imaging consideration for persistent or concerning symptoms
Laboratory Surveillance
While routine laboratory monitoring cannot predict gallstone formation, periodic assessment helps identify biliary complications:
- Baseline and monthly: ALT, AST, alkaline phosphatase, GGT, total bilirubin
- Concerning patterns: Isolated ALP/GGT elevation (biliary obstruction), conjugated hyperbilirubinaemia, transaminase elevation with biliary enzyme predominance
Imaging Protocols 🏥
Abdominal ultrasonography remains the gold-standard imaging modality for gallbladder assessment:
Indications for Ultrasound:
- Baseline imaging for high-risk participants
- New onset right upper quadrant pain or biliary symptoms
- Unexplained elevation in biliary enzymes
- Consideration at 24-week timepoint for participants with rapid weight loss (>1.5 kg/week average)
Ultrasound Findings Classification:
- Normal: No stones, sludge, or wall thickening
- Biliary sludge: Echogenic material without acoustic shadowing
- Gallstones: Echogenic foci with posterior acoustic shadowing
- Cholecystitis features: Wall thickening (>3mm), pericholecystic fluid, sonographic Murphy’s sign
Researchers sourcing peptides from reputable suppliers can ensure consistent product quality that supports reliable safety monitoring and reproducible experimental outcomes.
Management of Gallbladder Complications in Research Settings
When gallbladder-related adverse events occur during retatrutide research protocols, prompt recognition and appropriate management are essential for participant safety and study integrity. Clear action algorithms should be established before study initiation.
Asymptomatic Gallstone Detection
Clinical Significance and Natural History 💭
Detection of asymptomatic gallstones during surveillance imaging presents a management dilemma. Natural history studies indicate:
- 10-20% of asymptomatic gallstones become symptomatic annually
- Risk acceleration during ongoing rapid weight loss
- Most symptomatic episodes are uncomplicated biliary colic
- 1-3% annual risk of complicated disease (cholecystitis, pancreatitis, choledocholithiasis)
Management Options:
Conservative Monitoring Approach:
- Continue retatrutide treatment with enhanced surveillance
- Participant education regarding warning symptoms
- Monthly clinical assessment and symptom review
- Repeat imaging if symptoms develop
- Appropriate for: Small stones (<5mm), minimal symptoms, participant preference for study continuation
Treatment Modification:
- Dose reduction to slow weight loss velocity
- Treatment interruption with reassessment
- Consideration of ursodeoxycholic acid (in consultation with medical oversight)
- Appropriate for: Multiple or large stones, concerning symptoms, rapid weight loss trajectory
Study Discontinuation:
- Withdrawal from retatrutide treatment
- Referral for surgical evaluation if indicated
- Continued safety follow-up per protocol
- Appropriate for: Participant preference, medical team recommendation, protocol-specified criteria
Symptomatic Biliary Events
Recognition and Initial Assessment ⚠️
Biliary colic typically presents with:
- Right upper quadrant or epigastric pain
- Onset 30-60 minutes post-prandially
- Duration 30 minutes to several hours
- Radiation to right shoulder or scapula
- Associated nausea with or without vomiting
Immediate Actions:
- Clinical evaluation within 24 hours
- Laboratory assessment (CBC, LFTs, lipase/amylase)
- Abdominal ultrasound (same day if severe, within 72 hours if moderate)
- Pain management and symptomatic treatment
- Temporary treatment interruption pending evaluation
Risk Stratification and Referral:
Uncomplicated Biliary Colic:
- Outpatient management with close follow-up
- Gastroenterology or surgical consultation
- Shared decision-making regarding study continuation
- Enhanced monitoring if treatment resumed
Complicated Disease (Immediate Referral):
- Acute cholecystitis (fever, persistent pain, positive Murphy’s sign)
- Suspected choledocholithiasis (jaundice, elevated bilirubin)
- Biliary pancreatitis (elevated lipase >3× ULN)
- Cholangitis (Charcot’s triad: fever, jaundice, right upper quadrant pain)
Prophylactic Strategies
Ursodeoxycholic Acid (UDCA) 💊
Ursodeoxycholic acid has demonstrated efficacy in preventing gallstone formation during rapid weight loss in multiple clinical contexts:
Mechanism of Action:
- Reduces biliary cholesterol saturation
- Increases bile acid pool hydrophilicity
- May enhance gallbladder motility
- Dissolves small cholesterol stones
Evidence Base: Studies in bariatric surgery populations (analogous rapid weight loss) demonstrate:
- 50-70% reduction in gallstone formation with UDCA 500-600 mg daily
- Optimal efficacy when initiated before or concurrent with weight loss
- Best results in participants without pre-existing stones
Research Protocol Considerations:
- Prophylactic UDCA could be incorporated as study intervention for high-risk participants
- Requires consideration of drug-drug interactions and additional monitoring
- May confound interpretation of “true” gallbladder risk associated with retatrutide
- Cost-benefit analysis needed for research budget planning
Dietary Modifications
Maintaining regular meal patterns and moderate fat intake may support gallbladder emptying:
- Regular meals (3 meals daily minimum) to stimulate CCK release
- Moderate fat content (20-30% of calories) to promote gallbladder contraction
- Avoidance of very low-fat diets (<10% calories from fat) that may worsen stasis
- Adequate hydration to support bile dilution
For laboratories conducting research with metabolic peptides, integrating these prophylactic strategies into study protocols may reduce adverse event rates while generating valuable comparative effectiveness data.
Comparative Analysis: Retatrutide vs. Other Incretin-Based Peptides
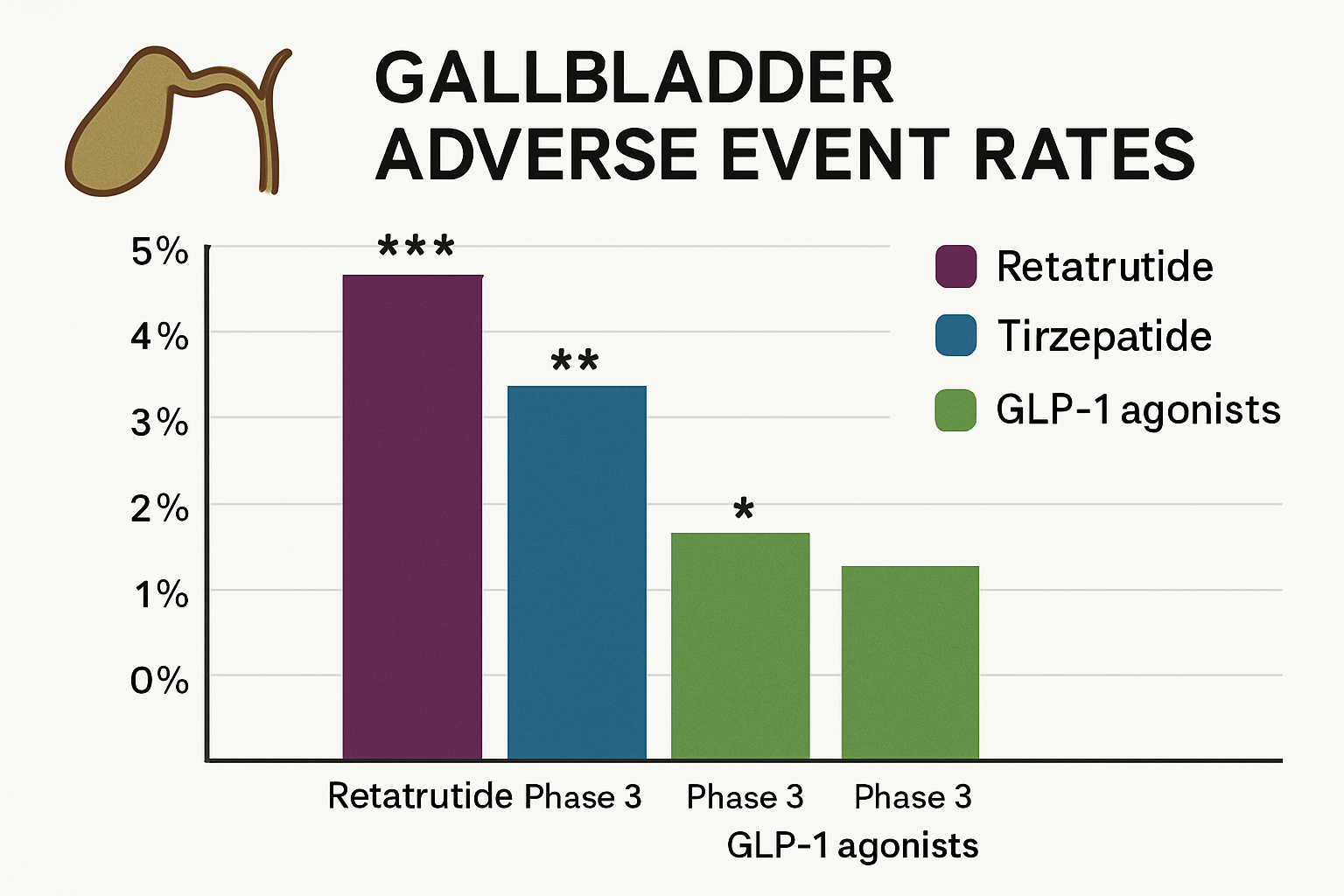
Contextualising retatrutide gallbladder risk within the broader landscape of incretin-based therapies provides important perspective for researchers selecting compounds for metabolic investigations and designing comparative effectiveness studies.
GLP-1 Receptor Agonists
Semaglutide 📊
The selective GLP-1 receptor agonist semaglutide has accumulated extensive safety data across multiple indications:
- STEP trials (obesity): 2.6-3.1% gallbladder disorder incidence at 2.4 mg dose
- SUSTAIN trials (diabetes): 1.4-1.8% incidence at 1.0 mg dose
- Dose-response relationship: Higher doses associated with increased risk, proportional to weight loss magnitude
- Time to event: Median 28 weeks, similar temporal pattern to retatrutide
Liraglutide
The earlier-generation GLP-1 agonist liraglutide demonstrates lower gallbladder event rates:
- SCALE trial: 1.5-2.3% incidence at 3.0 mg dose
- Lower weight loss magnitude: Average 8-10% body weight reduction vs. 15-20% with newer agents
- Risk proportionality: Gallbladder event rate correlates with lesser weight reduction achieved
Dual Agonists
Tirzepatide (GIP/GLP-1) 🔬
The dual GIP/GLP-1 receptor agonist tirzepatide provides the most relevant comparison to retatrutide:
- SURMOUNT trials: 2.9-4.3% gallbladder disorder incidence across dose range (5-15 mg)
- Weight loss magnitude: 15-22% body weight reduction, comparable to retatrutide
- Risk profile similarity: Nearly identical gallbladder event rates when matched for weight loss achieved
- Mechanistic overlap: Shared GIP/GLP-1 agonism suggests common pathophysiological pathways
Comparative Risk Table:
| Compound | Mechanism | Avg. Weight Loss | Gallbladder Events | Relative Risk vs. Placebo |
|---|---|---|---|---|
| Retatrutide | GIP/GLP-1/Glucagon | 17-24% | 2.7-3.8% | 3.2-4.5× |
| Tirzepatide | GIP/GLP-1 | 15-22% | 2.9-4.3% | 3.0-4.2× |
| Semaglutide 2.4mg | GLP-1 | 12-17% | 2.6-3.1% | 2.8-3.5× |
| Liraglutide 3.0mg | GLP-1 | 8-10% | 1.5-2.3% | 1.8-2.5× |
| Placebo | — | 1-3% | 0.7-1.2% | 1.0× (reference) |
Key Insights from Comparative Analysis
Weight Loss Magnitude as Primary Determinant
The striking correlation between weight reduction magnitude and gallbladder event incidence across all incretin-based therapies suggests that the primary driver is the metabolic consequence of adipose tissue mobilisation rather than compound-specific toxicity. This has important implications:
- Mechanism-agnostic risk: Similar event rates across different receptor mechanisms when weight loss is equivalent
- Dose-response relationship: Higher doses producing greater weight loss show proportionally higher gallbladder risk
- Velocity matters: Rate of weight loss appears more predictive than total weight lost
Class Effect Consideration
“The consistent gallbladder safety signal across GLP-1 agonists, dual agonists, and now triple agonists suggests a class effect related to the profound metabolic changes induced by these compounds. Researchers should anticipate similar risk profiles for future incretin-based peptides achieving comparable weight reduction.”
Research Selection Implications
For investigators designing metabolic studies, compound selection should consider:
- Efficacy requirements: Greater weight loss potential correlates with higher gallbladder risk
- Study population: Baseline risk factors in target population influence acceptable risk-benefit ratio
- Monitoring capacity: Resource-intensive surveillance may be more feasible for some compounds/settings
- Mechanistic objectives: Specific receptor targets may be necessary despite similar safety profiles
Laboratories sourcing research peptides for comparative studies benefit from suppliers offering comprehensive product ranges across different peptide classes, enabling well-controlled head-to-head investigations.
Future Directions and Emerging Research
The landscape of retatrutide gallbladder risk research continues to evolve as clinical development advances and investigator-initiated studies generate new insights. Several key areas warrant particular attention from the research community in 2025 and beyond.
Long-Term Safety Data Needs
Extended Follow-Up Studies 📈
Current safety data derives primarily from trials of 48-52 weeks duration, leaving important questions about longer-term risk:
- Chronic treatment effects: Does gallbladder risk plateau after weight stabilisation or persist with continued treatment?
- Cumulative incidence: What proportion of long-term users ultimately develop biliary complications?
- Risk factor evolution: Do baseline risk factors maintain predictive value over extended treatment periods?
- Post-discontinuation risk: Does gallbladder risk normalise after treatment cessation, or does history of rapid weight loss create persistent susceptibility?
Real-World Evidence Generation
As retatrutide transitions from controlled trial settings to broader research applications, real-world safety monitoring will provide complementary insights:
- Diverse populations: Inclusion of participants typically excluded from trials (elderly, multiple comorbidities, polypharmacy)
- Varied dosing patterns: Real-world dose titration, interruptions, and modifications
- Long-term outcomes: Multi-year follow-up in naturalistic settings
- Healthcare utilisation: Cholecystectomy rates, emergency department visits, hospitalisations
Predictive Biomarker Development
Bile Composition Analysis 🔬
Advanced analytical techniques may enable identification of participants developing lithogenic bile before stone formation:
- Cholesterol saturation index: Direct measurement of bile supersaturation
- Bile acid profiling: Detailed characterisation of bile acid pool composition
- Proteomic markers: Identification of protein signatures associated with stone formation
- Metabolomic approaches: Comprehensive metabolite profiling to identify high-risk patterns
Genetic Risk Scoring
Integration of multiple genetic variants into polygenic risk scores may improve pre-treatment risk stratification:
- ABCG8, UGT1A1, SULT2A1 variants combined with demographic factors
- Machine learning approaches: Algorithm development incorporating genetic, clinical, and metabolic variables
- Personalised risk prediction: Individual probability estimates to guide monitoring intensity
Mechanistic Investigation Priorities
Gallbladder Motility Studies
Detailed physiological investigations of gallbladder function during retatrutide treatment:
- Ultrasound-based motility assessment: Fasting and postprandial gallbladder volumes, ejection fraction
- CCK response characterisation: Endogenous CCK secretion patterns during treatment
- Contractility interventions: Response to exogenous CCK stimulation
- Temporal dynamics: Evolution of motility parameters throughout treatment course
Receptor-Specific Contributions
Dissecting the relative contributions of GIP, GLP-1, and glucagon receptor activation:
- Selective antagonist studies: Using receptor-specific blockers to isolate individual pathway effects
- Receptor knockout models: Genetic approaches in preclinical systems
- Dose-fractionation experiments: Comparing effects of balanced vs. receptor-selective dosing
- Comparative mechanism studies: Head-to-head comparisons with selective agonists
Intervention Strategy Optimisation
Prophylaxis Protocol Refinement 💊
Systematic investigation of preventive strategies:
- UDCA dosing optimisation: Identifying minimum effective dose and optimal timing
- Dietary intervention trials: Controlled studies of meal pattern and macronutrient composition
- Combination approaches: Synergistic effects of multiple prophylactic strategies
- Risk-stratified protocols: Tailoring interventions to individual risk profiles
Dose Titration Strategy Research
Investigating whether modified dose escalation reduces gallbladder risk:
- Slower titration schedules: Extended ramp-up periods to moderate weight loss velocity
- Weight loss velocity targets: Protocols designed to maintain weight reduction below risk thresholds
- Adaptive dosing algorithms: Individualised dose adjustment based on weight loss trajectory
- Efficacy-safety optimisation: Identifying sweet spot balancing metabolic benefits and biliary risk
Researchers conducting these investigations require access to high-purity research-grade peptides with comprehensive documentation and consistent quality to ensure reproducible and reliable experimental outcomes.
Practical Recommendations for Research Laboratories
Translating the current evidence base on retatrutide gallbladder risk into actionable research practices requires systematic implementation of safety protocols, participant education, and data collection strategies. The following recommendations provide a framework for responsible retatrutide research in 2025.
Study Design Considerations
Inclusion and Exclusion Criteria ✅
Recommended Exclusions:
- Active biliary disease (cholecystitis, choledocholithiasis, biliary obstruction)
- History of biliary pancreatitis within past 12 months
- Symptomatic gallstones with recurrent biliary colic
- Cholecystectomy within past 6 months
- Severe hepatic impairment (Child-Pugh Class C)
Relative Contraindications (Require Enhanced Monitoring):
- Asymptomatic gallstones or biliary sludge
- Family history of gallstone disease in first-degree relatives
- History of rapid weight loss-associated gallstones
- BMI >45 kg/m² (very severe obesity)
- Multiple prior weight loss attempts with rapid regain
Risk Stratification System 📋
Implement a structured risk scoring system at baseline:
Low Risk (0-1 points):
- Male sex
- Age <40 years
- BMI 30-35 kg/m²
- No family history
- No prior gallstone history
Moderate Risk (2-3 points):
- Female sex
- Age 40-60 years
- BMI 35-40 kg/m²
- Positive family history
- Metabolic syndrome
High Risk (≥4 points):
- Female sex + age >60 years
- BMI >40 kg/m²
- Prior gallstone episode
- Multiple family members affected
- Rapid weight loss history
Risk Factor Scoring:
- Female sex: 1 point
- Age >40: 1 point
- BMI >35: 1 point
- Family history: 1 point
- Prior gallstones: 2 points
- BMI >40: Additional 1 point
Monitoring Protocol Implementation
Surveillance Schedule by Risk Category
Low Risk Participants:
- Baseline: Clinical assessment, LFTs, symptom education
- Weeks 1-12: Bi-weekly weight and symptom check
- Weeks 12+: Monthly clinical assessment
- Imaging: Only if symptoms develop
Moderate Risk Participants:
- Baseline: Clinical assessment, LFTs, consider screening ultrasound
- Weeks 1-12: Weekly weight and symptom monitoring
- Weeks 12-24: Bi-weekly assessment
- Week 24: Surveillance ultrasound if average weight loss >1.0 kg/week
- Weeks 24+: Monthly clinical assessment
High Risk Participants:
- Baseline: Comprehensive assessment including screening ultrasound
- Weeks 1-12: Weekly monitoring with clinical review
- Week 12: Repeat ultrasound
- Weeks 12-24: Weekly weight monitoring, bi-weekly clinical assessment
- Week 24: Repeat ultrasound
- Weeks 24+: Bi-weekly clinical assessment, monthly LFTs
Participant Education and Informed Consent
Comprehensive Risk Communication 💬
Informed consent processes should include clear communication about gallbladder risks:
Key Information Elements:
- Explanation of 2.7-3.8% incidence rate in clinical trials
- Description of typical symptoms (biliary colic presentation)
- Instruction to report symptoms promptly (24-48 hour timeframe)
- Discussion of potential outcomes (conservative management vs. surgery)
- Comparison to background risk and other weight loss interventions
- Emphasis on monitoring and early detection strategies
Written Educational Materials
Provide participants with take-home materials covering:
- Warning symptoms requiring immediate contact
- 24-hour contact information for study team
- Dietary recommendations to support gallbladder health
- Expected weight loss trajectory and velocity targets
- Schedule of monitoring visits and procedures
Data Collection and Reporting
Standardised Adverse Event Documentation 📝
Implement consistent terminology and grading:
Gallbladder-Related Adverse Events Classification:
Grade 1 (Mild):
- Asymptomatic gallstones detected on imaging
- Biliary sludge without symptoms
- Transient, self-limited right upper quadrant discomfort
Grade 2 (Moderate):
- Symptomatic biliary colic requiring medical evaluation
- Uncomplicated gallstones with recurrent symptoms
- LFT elevation <3× upper limit of normal
Grade 3 (Severe):
- Acute cholecystitis requiring hospitalisation
- Choledocholithiasis with biliary obstruction
- Biliary pancreatitis
- LFT elevation >3× upper limit of normal
Grade 4 (Life-Threatening):
- Cholangitis with sepsis
- Severe biliary pancreatitis
- Gallbladder perforation
Regulatory Reporting Requirements
Ensure compliance with adverse event reporting timelines:
- Serious adverse events: Report within 24 hours to ethics committee/IRB
- Unexpected events: Expedited reporting per institutional requirements
- Aggregate safety data: Periodic summary reports per protocol schedule
- Study discontinuations: Document reasons and outcomes
Quality Assurance for Research Materials
Peptide Sourcing and Handling 🧪
The reliability of safety data depends on consistent, high-quality research materials:
Supplier Selection Criteria:
- Purity verification: Certificate of analysis confirming >98% purity
- Batch consistency: Documented manufacturing quality control
- Storage conditions: Appropriate cold chain maintenance
- Regulatory compliance: “For Research Use Only” labelling and documentation
- Technical support: Access to reconstitution and handling guidance
PEPTIDE PRO provides research-grade peptides meeting these quality standards, with same-day dispatch for orders placed before 1pm (Monday-Friday), supporting timely research progression while maintaining product integrity.
Storage and Handling Protocols:
- Lyophilised peptides: Store at -20°C in original packaging
- Reconstituted solutions: Refrigerate at 2-8°C, use within protocol-specified timeframe
- Avoid freeze-thaw cycles: Aliquot reconstituted material if multiple uses planned
- Document storage conditions: Temperature logs for accountability
- Regular inventory audits: Ensure proper rotation and expiration tracking
Ethical Considerations and Risk-Benefit Assessment
Conducting research involving retatrutide gallbladder risk requires careful ethical consideration of the balance between scientific knowledge generation and participant safety. Research ethics committees and investigators must navigate complex decision-making regarding acceptable risk levels.
Risk-Benefit Framework
Potential Benefits of Retatrutide Research ⚖️
Scientific Knowledge:
- Understanding of novel triple-agonist mechanisms
- Insights into metabolic regulation and obesity pathophysiology
- Comparative effectiveness data vs. existing therapies
- Identification of biomarkers and predictive factors
Potential Clinical Advances:
- Development of more effective metabolic interventions
- Optimisation of dosing and administration strategies
- Refinement of patient selection and monitoring approaches
- Long-term outcome data to inform clinical decision-making
Participant-Level Considerations:
- Access to investigational therapy with substantial efficacy
- Comprehensive medical monitoring exceeding standard care
- Contribution to scientific advancement
- Potential for significant health improvements (weight reduction, metabolic parameters)
Quantifiable Risks ⚠️
Gallbladder-Related:
- 2.7-3.8% incidence of gallbladder disorders
- ~1-1.5% probability of requiring cholecystectomy
- Temporary morbidity from biliary colic episodes
- Small risk (<0.5%) of serious complications (cholangitis, severe pancreatitis)
Contextualisation:
- Background gallstone incidence: 0.7-1.2% annually in obesity populations
- Bariatric surgery gallstone risk: 5-10% in first year post-operatively
- Absolute risk increase: ~2-3% attributable to retatrutide
- Number needed to harm: Approximately 33-40 participants treated for one additional gallbladder event
Informed Consent Process Optimisation
Decision Support Tools 📊
Enhance participant understanding through visual aids:
Risk Comparison Graphics:
- Pictographs showing 3-4 affected individuals per 100 treated
- Comparison bars: retatrutide vs. placebo vs. bariatric surgery
- Natural frequency formats: “3 out of 100” rather than “3%”
- Outcome tree diagrams: Probability pathways for different scenarios
Comprehension Assessment:
Verify participant understanding through teach-back methods:
- “Can you explain in your own words what gallbladder problems might occur?”
- “What symptoms would you watch for and report to the study team?”
- “What is your understanding of how likely these problems are to happen?”
- “What would happen if you developed gallstones during the study?”
Vulnerable Populations
Enhanced Protections for High-Risk Groups
Participants with multiple risk factors require additional safeguards:
Mandatory Risk Mitigation:
- Baseline imaging required (not optional)
- Enhanced monitoring frequency (weekly clinical contact)
- Lower threshold for treatment modification or discontinuation
- Consideration of prophylactic interventions (UDCA)
- Additional informed consent discussion of personalised risk
Exclusion Threshold Determination:
Research teams must define the risk level beyond which participation is not ethically justifiable:
- Estimated individual risk >10%: Strong consideration for exclusion
- Prior symptomatic gallstones: Generally excluded unless compelling justification
- Multiple high-risk factors: Case-by-case ethics review
- Participant autonomy: Balanced against researcher duty to protect
Data Safety Monitoring
Independent Oversight Structures
Data Safety Monitoring Board (DSMB):
For studies of significant size or risk, establish independent safety oversight:
- Periodic review of aggregate safety data (quarterly or semi-annually)
- Authority to recommend protocol modifications or study suspension
- Evaluation of risk-benefit balance as data accumulates
- Assessment of whether observed event rates exceed pre-specified thresholds
Stopping Rules:
Define prospective criteria that would trigger study modification:
- Gallbladder event rate exceeds 6% (2× expected rate)
- Serious gallbladder complications (cholangitis, perforation) exceed 1%
- Clustering of events suggesting unexpected risk pattern
- New safety signals from other retatrutide studies
Advancing Retatrutide Research Responsibly
The evidence base surrounding retatrutide gallbladder risk in 2025 demonstrates a consistent safety signal requiring systematic attention but not precluding responsible research advancement. With gallbladder-related adverse event incidence of 2.7-3.8% in clinical trials, retatrutide’s biliary safety profile aligns closely with other high-efficacy incretin-based peptides, reflecting a class effect primarily driven by the magnitude and velocity of weight reduction achieved.
Key Conclusions
Mechanistic Understanding 🔬
The pathophysiology of retatrutide-associated gallbladder complications involves multiple interconnected mechanisms:
- Rapid adipose tissue mobilisation producing cholesterol-supersaturated bile
- Delayed gastric emptying and reduced CCK stimulation decreasing gallbladder contractility
- Prolonged fasting states promoting bile stasis and crystal nucleation
- Potential direct receptor-mediated effects on biliary physiology
This mechanistic complexity necessitates multi-faceted monitoring and intervention strategies rather than single-target approaches.
Risk Stratification Imperative 📊
Individual susceptibility varies substantially based on demographic, clinical, and potentially genetic factors. Implementing structured risk assessment enables:
- Appropriate participant selection and exclusion decisions
- Tailored monitoring intensity matched to risk level
- Informed consent discussions incorporating personalised risk estimates
- Efficient allocation of surveillance resources
Evidence-Based Monitoring 🔍
Comprehensive surveillance protocols should incorporate:
- Baseline risk assessment and selective imaging
- Regular clinical evaluation with systematic symptom review
- Laboratory monitoring (LFTs) when clinically indicated
- Ultrasound imaging for high-risk participants or symptomatic individuals
- Temporal concentration of monitoring during highest-risk period (weeks 12-36)
Comparative Context ⚖️
Retatrutide’s gallbladder safety profile should be evaluated within the broader landscape of metabolic interventions:
- Similar event rates to tirzepatide and high-dose semaglutide when matched for weight loss
- Lower risk than bariatric surgery (5-10% gallstone incidence)
- Substantially higher efficacy than earlier-generation therapies with lower gallbladder risk
- Acceptable risk-benefit ratio for many research applications and participant populations
Actionable Recommendations for Researchers
Immediate Implementation Steps ✅
- Develop comprehensive study protocols incorporating risk-stratified monitoring schedules
- Establish clear adverse event management algorithms with pre-defined action thresholds
- Create standardised participant education materials covering warning symptoms and reporting procedures
- Source high-quality research peptides from reputable suppliers ensuring product consistency
- Implement robust data collection systems for systematic safety documentation
Medium-Term Research Priorities
- Conduct mechanistic studies elucidating receptor-specific contributions to biliary effects
- Develop and validate predictive biomarkers enabling early identification of high-risk individuals
- Investigate prophylactic strategies including UDCA, dietary modifications, and dose titration approaches
- Generate long-term safety data extending beyond current 48-52 week trial durations
- Establish real-world evidence complementing controlled trial findings
Long-Term Knowledge Gaps 🔭
- Genetic risk profiling integrating polygenic scores into clinical decision-making
- Personalised dosing algorithms optimising individual risk-benefit profiles
- Mechanistic interventions targeting specific pathophysiological pathways
- Comparative effectiveness research across different peptide classes and mechanisms
- Economic analyses evaluating cost-effectiveness of various monitoring strategies
The Path Forward
Retatrutide represents a significant advancement in metabolic peptide pharmacology, with unprecedented efficacy in weight reduction and metabolic parameter improvement. The associated gallbladder risk, while real and requiring systematic attention, is predictable, manageable, and comparable to other high-efficacy interventions in this therapeutic space.
Responsible research advancement requires balancing innovation with participant safety through:
- Rigorous protocol design incorporating evidence-based monitoring
- Transparent risk communication enabling truly informed consent
- Systematic data collection contributing to the evolving knowledge base
- Ethical oversight ensuring appropriate risk-benefit assessment
- Quality assurance in research materials and procedures
“The future of metabolic peptide research lies not in avoiding compounds with manageable safety signals, but in developing sophisticated approaches to risk stratification, monitoring, and mitigation that enable safe access to highly effective interventions for appropriate populations.”
For research laboratories seeking to advance understanding of retatrutide and other metabolic peptides, partnering with PEPTIDE PRO ensures access to premium research-grade compounds with exceptional purity, comprehensive documentation, and professional support—enabling reliable, reproducible investigations that advance scientific knowledge while maintaining the highest safety standards.
The evidence base will continue evolving throughout 2025 and beyond, with long-term follow-up studies, real-world evidence generation, and mechanistic investigations refining our understanding of retatrutide gallbladder risk. Researchers contributing to this knowledge base through carefully designed, ethically conducted studies play a vital role in optimising the risk-benefit profile of this promising therapeutic approach.
