
When researchers first observed retatrutide’s profound effects on metabolic regulation, one mechanism stood out as particularly significant: its impact on gastric emptying. This triple-receptor agonist doesn’t just influence appetite through neural pathways—it fundamentally alters the rate at which the stomach processes and releases nutrients, creating a cascade of metabolic effects that distinguish it from earlier incretin-based therapies. Understanding retatrutide gastric emptying mechanisms has become essential for researchers investigating next-generation metabolic interventions and their broader physiological implications.
The relationship between retatrutide and gastric motility represents a fascinating intersection of peptide pharmacology, digestive physiology, and metabolic regulation. As laboratories worldwide examine this compound’s multi-receptor activity, the gastric emptying component emerges as a critical factor in its overall metabolic profile.
Key Takeaways
- Retatrutide delays gastric emptying through simultaneous activation of GLP-1, GIP, and glucagon receptors, creating a more pronounced effect than single or dual agonists
- Gastric emptying modulation contributes significantly to retatrutide’s appetite suppression and glucose regulation mechanisms in research models
- Triple-agonist activity produces distinct gastric motility patterns compared to semaglutide or tirzepatide, with implications for metabolic research
- Dose-dependent effects on gastric emptying rates have been observed in preclinical studies, informing optimal research protocols
- Understanding these mechanisms is essential for researchers working with retatrutide in metabolic and obesity research contexts
What Is Retatrutide and How Does It Affect Gastric Function?
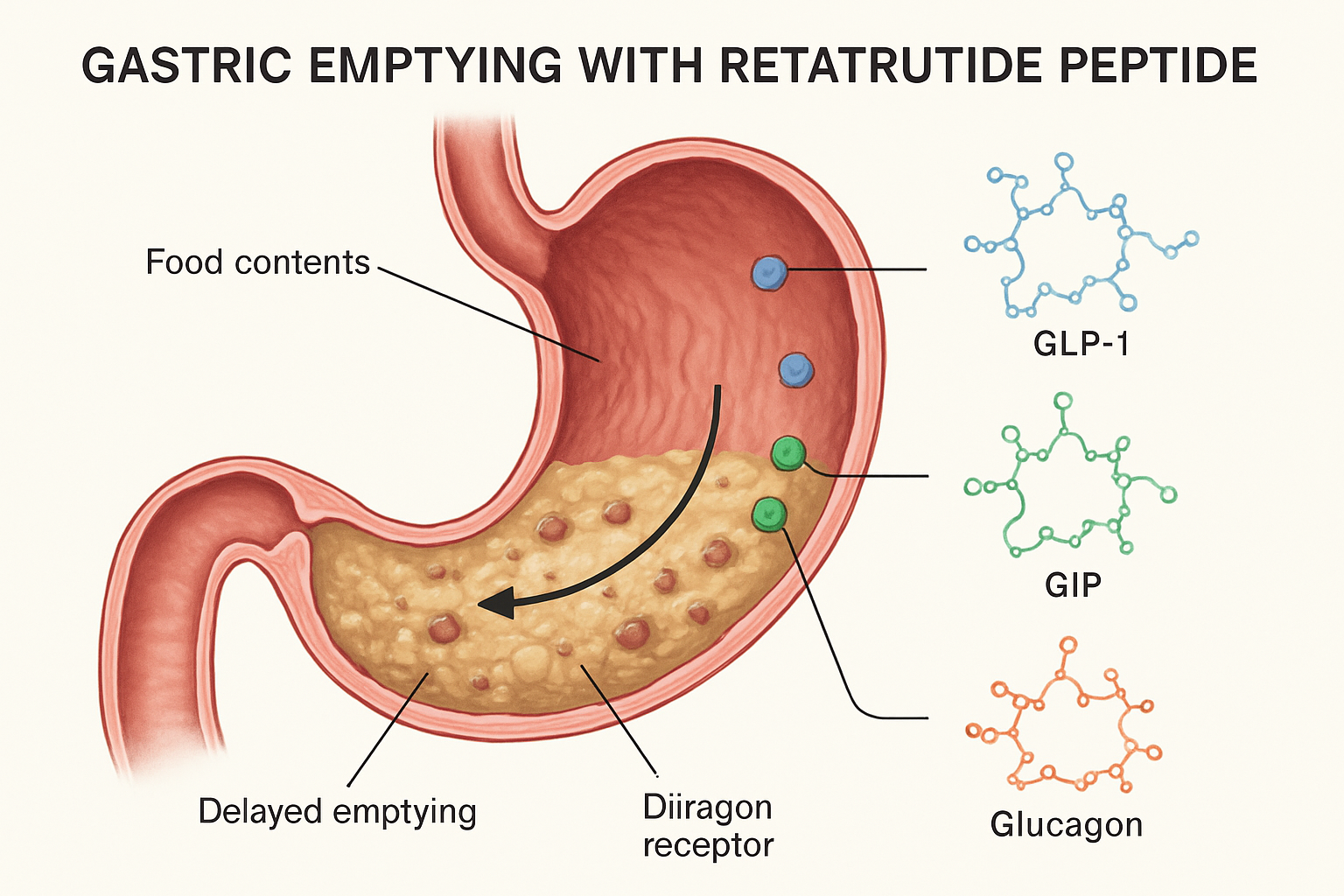
Retatrutide represents a significant advancement in peptide research, functioning as a triple-receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile creates a complex interplay of metabolic effects, with gastric emptying modulation serving as one of the primary mechanisms.
The Triple-Agonist Mechanism
Unlike earlier incretin-based compounds that target one or two receptors, retatrutide’s triple action creates a distinctive physiological signature:
| Receptor Target | Primary Gastric Effect | Metabolic Contribution |
|---|---|---|
| GLP-1 | Significant gastric emptying delay | Appetite suppression, glucose regulation |
| GIP | Modulated nutrient sensing | Enhanced incretin effect, lipid metabolism |
| Glucagon | Energy expenditure influence | Metabolic rate, hepatic glucose output |
The GLP-1 component is particularly relevant to gastric emptying, as GLP-1 receptor activation is well-established as a potent inhibitor of gastric motility. When retatrutide binds to GLP-1 receptors in the stomach and brainstem, it triggers a cascade of signals that reduce the contractile activity of gastric smooth muscle, effectively slowing the rate at which stomach contents move into the duodenum.
Gastric Emptying: A Critical Metabolic Checkpoint
Gastric emptying rate serves as a crucial regulator of postprandial glucose excursions and satiety signaling. When nutrients remain in the stomach for extended periods, several metabolic consequences occur:
✅ Extended satiety signals from gastric distension mechanoreceptors
✅ Reduced glucose absorption rate leading to improved glycemic control
✅ Enhanced incretin hormone release from intestinal L-cells
✅ Modulated ghrelin secretion affecting hunger signaling
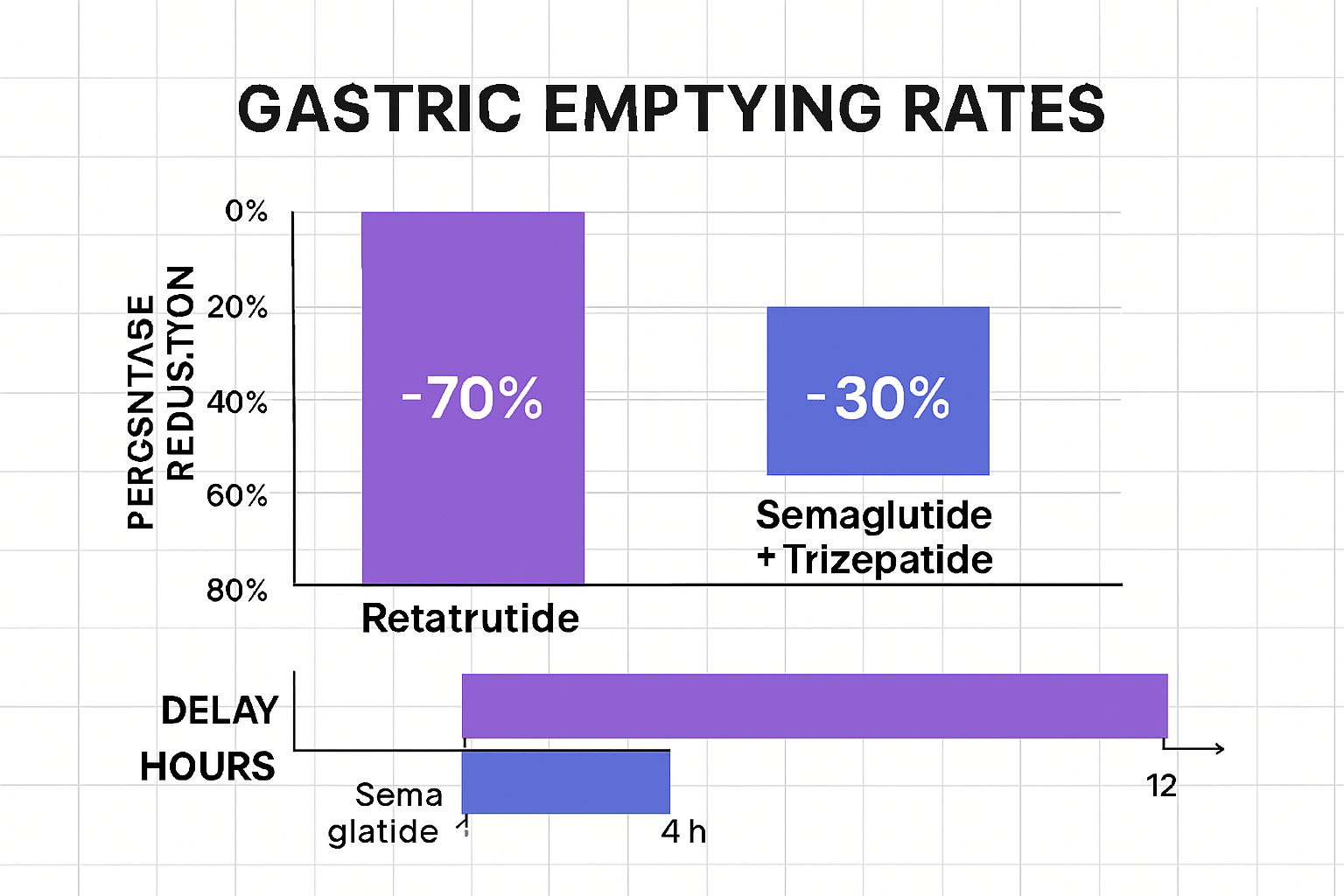
Research institutions investigating retatrutide have documented significant delays in gastric emptying across various experimental models. These delays appear more pronounced than those observed with single GLP-1 agonists, suggesting that the compound’s triple-agonist activity creates synergistic or additive effects on gastric motility.
For researchers sourcing high-purity research peptides for gastric emptying studies, understanding these fundamental mechanisms is essential for designing appropriate experimental protocols and interpreting results accurately.
The Science Behind Retatrutide Gastric Emptying Mechanisms
Understanding retatrutide gastric emptying requires examining the molecular and physiological pathways through which this peptide influences digestive motility. The mechanisms are multifaceted, involving both central and peripheral nervous system components.
Receptor Distribution and Gastric Motility Control
The stomach contains a complex network of receptor sites that respond to incretin hormones. GLP-1 receptors are found in:
- Gastric smooth muscle cells – directly influencing contractile activity
- Vagal afferent neurons – transmitting satiety signals to the brainstem
- Enteric nervous system – modulating local digestive reflexes
- Area postrema – central regulation of gastric function
When retatrutide activates these GLP-1 receptors, it initiates a coordinated reduction in gastric motility through multiple pathways simultaneously. This multi-level regulation explains why the gastric emptying delay is both robust and sustained throughout the compound’s active period.
Comparative Effects: Retatrutide vs. Other Incretin-Based Peptides
Research comparing retatrutide gastric emptying effects to other incretin-based compounds reveals important distinctions:
Semaglutide (GLP-1 agonist):
- Produces significant gastric emptying delay
- Effect primarily mediated through GLP-1 pathway
- Well-characterized dose-response relationship
- Researchers can explore semaglutide research formulations for comparative studies
Tirzepatide (GLP-1/GIP dual agonist):
- Demonstrates gastric emptying delay with dual-receptor activity
- GIP component may modulate GLP-1 effects
- Intermediate profile between single and triple agonists
- Available through specialized peptide research suppliers
Retatrutide (GLP-1/GIP/Glucagon triple agonist):
- Most pronounced gastric emptying delay observed in comparative studies
- Glucagon component adds energy expenditure dimension
- Complex receptor interaction profile
- Represents next-generation metabolic research compound
“The triple-agonist activity of retatrutide creates a gastric emptying profile that cannot be fully explained by simple additive effects of its individual receptor components. The receptor interactions appear to produce synergistic modulation of gastric motility that warrants further investigation.” — Research observation from metabolic peptide studies
Dose-Dependent Gastric Emptying Effects
Preclinical research has established clear dose-response relationships for retatrutide’s effects on gastric emptying. Higher doses produce:
- Greater magnitude of delay in gastric emptying rate
- Extended duration of gastric motility suppression
- More pronounced effects on postprandial glucose excursions
- Enhanced satiety signaling through prolonged gastric distension
These dose-dependent effects are critical for researchers designing experimental protocols. Selecting appropriate dosing regimens requires balancing the desired gastric emptying modulation against other metabolic parameters being investigated.
Temporal Dynamics of Gastric Emptying Modulation
The time course of retatrutide’s gastric emptying effects follows a predictable pattern:
Acute Phase (0-4 hours post-administration):
- Rapid onset of gastric motility suppression
- Peak effect typically observed 2-3 hours after administration
- Maximal delay in solid and liquid phase emptying
Sustained Phase (4-24 hours):
- Continued but gradually diminishing effect
- Return toward baseline gastric emptying rates
- Individual variation based on dosing and metabolic state
Chronic Administration:
- Some evidence of partial tachyphylaxis in gastric effects
- Maintained metabolic benefits despite modest adaptation
- Continued relevance to overall metabolic profile
Understanding these temporal dynamics helps researchers interpret experimental results and design appropriate measurement timepoints for gastric emptying assessments.
Research Applications and Experimental Considerations for Retatrutide Gastric Emptying Studies
For research institutions and laboratories investigating retatrutide gastric emptying mechanisms, several methodological considerations warrant careful attention. The complexity of gastric motility assessment requires sophisticated experimental design and appropriate measurement techniques.
Gastric Emptying Measurement Methodologies
Researchers employ various techniques to quantify gastric emptying rates in experimental models:
Scintigraphic Methods:
- Gold standard for quantitative gastric emptying assessment
- Radiolabeled test meals tracked through digestive system
- Provides detailed temporal resolution of emptying kinetics
- Requires specialized imaging equipment and protocols
Stable Isotope Breath Tests:
- Non-invasive alternative to scintigraphy
- Carbon-13 labeled substrates metabolized and measured in breath
- Useful for repeated measurements in longitudinal studies
- Less precise than scintigraphy but more accessible
Paracetamol Absorption Test:
- Indirect measure based on acetaminophen absorption rate
- Simple and cost-effective for screening studies
- Limited precision for detailed kinetic analysis
- Useful for comparative studies between conditions
Magnetic Resonance Imaging (MRI):
- Emerging technique with excellent spatial resolution
- Non-invasive with no radiation exposure
- Expensive and technically demanding
- Provides additional anatomical information
Experimental Design Considerations
When planning retatrutide gastric emptying research, several design elements require careful consideration:
📊 Test Meal Composition:
- Solid vs. liquid phase emptying shows different sensitivities
- Caloric density affects baseline emptying rates
- Macronutrient composition influences incretin response
- Standardization essential for reproducible results
📊 Dosing Protocols:
- Single-dose vs. chronic administration studies
- Timing of gastric emptying assessment relative to dosing
- Dose-escalation studies to establish response curves
- Comparison to appropriate control conditions
📊 Subject/Model Selection:
- Metabolic state influences gastric emptying baseline
- Age, sex, and other demographic factors affect results
- Disease models may show altered responses
- Appropriate sample sizes for statistical power
Integration with Broader Metabolic Research
Gastric emptying studies with retatrutide gain maximum value when integrated into comprehensive metabolic research protocols. Complementary measurements include:
- Glucose tolerance testing to correlate emptying delay with glycemic control
- Appetite and satiety assessments linking gastric effects to feeding behavior
- Incretin hormone profiling (GLP-1, GIP, GIP) to understand endocrine responses
- Energy expenditure measurements to capture glucagon receptor effects
Researchers working with research-grade retatrutide formulations should ensure their experimental design captures these multiple dimensions of metabolic regulation.
Quality Considerations for Research Peptides
The reliability of gastric emptying research depends fundamentally on peptide quality. Key quality parameters include:
| Quality Parameter | Research Importance | Verification Method |
|---|---|---|
| Purity (>98%) | Ensures consistent pharmacological activity | HPLC analysis, COA |
| Proper storage | Maintains peptide stability and potency | Temperature monitoring, lyophilization |
| Accurate reconstitution | Achieves intended dosing concentrations | Gravimetric verification, protocols |
| Batch consistency | Enables reproducible results across experiments | Batch documentation, testing |
Institutions conducting retatrutide research should source peptides from established research suppliers that provide comprehensive quality documentation and maintain rigorous storage protocols.
Regulatory and Ethical Framework
All retatrutide gastric emptying research must operate within appropriate regulatory frameworks:
⚠️ Research Use Only: Retatrutide is strictly for laboratory research applications
⚠️ Institutional Oversight: Appropriate committee approvals required
⚠️ Documentation: Comprehensive records of experimental protocols and results
⚠️ Safety Protocols: Proper handling, storage, and disposal procedures
These considerations ensure research integrity and compliance with applicable regulations governing peptide research.
Retatrutide Gastric Emptying: Clinical Implications and Research Frontiers
While retatrutide remains a research compound, understanding its gastric emptying effects provides valuable insights into potential metabolic applications and informs ongoing investigational work.
Metabolic Benefits Linked to Gastric Emptying Modulation
The gastric emptying delay produced by retatrutide contributes to several metabolic outcomes observed in research settings:
Glycemic Control:
- Slower nutrient absorption reduces postprandial glucose spikes
- Extended nutrient delivery improves insulin secretion patterns
- Enhanced incretin effect from prolonged intestinal nutrient exposure
- Reduced glycemic variability across feeding cycles
Appetite and Energy Intake:
- Prolonged gastric distension activates mechanoreceptor satiety signals
- Delayed emptying extends postprandial satiety period
- Reduced hunger perception between feeding episodes
- Decreased overall energy intake in feeding studies
Weight Regulation:
- Gastric emptying delay contributes to negative energy balance
- Synergistic with glucagon-mediated energy expenditure increase
- More pronounced effects than single-agonist comparators
- Sustained effects with chronic administration
Research Frontiers in Gastric Emptying Modulation
Several emerging research directions are expanding our understanding of retatrutide gastric emptying mechanisms:
Receptor Interaction Studies: Investigating how simultaneous GLP-1, GIP, and glucagon receptor activation creates unique gastric motility patterns that differ from simple additive effects of individual receptor components.
Microbiome Interactions: Exploring how altered gastric emptying rates influence gut microbiome composition and whether microbiome changes contribute to metabolic effects.
Neural Pathway Mapping: Detailed characterization of vagal and central nervous system pathways mediating retatrutide’s gastric effects using advanced neuroimaging and electrophysiology techniques.
Personalized Response Prediction: Identifying genetic, metabolic, or demographic factors that predict individual variation in gastric emptying response to retatrutide.
Combination Approaches: Investigating how retatrutide’s gastric effects interact with other metabolic interventions, including dietary modifications and complementary peptide therapies.
Comparative Pharmacology: Understanding the Competitive Landscape
Retatrutide exists within a rapidly evolving landscape of multi-agonist peptides. Understanding its gastric emptying profile relative to other compounds helps position it within metabolic research:
Survodutide (GLP-1/Glucagon dual agonist):
- Different receptor combination than retatrutide
- Distinct gastric emptying profile
- Researchers can access survodutide formulations for comparative studies
Mazdutide (GLP-1/Glucagon dual agonist):
- Alternative dual-agonist approach
- Unique pharmacokinetic properties
- Available through specialized peptide suppliers
Cagrilintide combinations:
- Amylin analog with complementary gastric effects
- Potential synergy with GLP-1 agonists
- Represents different mechanistic approach
This comparative context helps researchers select appropriate compounds for specific research questions and design informative head-to-head studies.
Practical Considerations for Laboratory Research
Researchers incorporating retatrutide gastric emptying studies into their laboratory work should consider several practical factors:
� Storage and Handling:
- Lyophilized peptides require -20°C or colder storage
- Reconstituted solutions have limited stability
- Proper reconstitution technique essential for accuracy
- Detailed protocols available from quality suppliers
� Experimental Timeline:
- Plan measurement timepoints based on pharmacokinetic profile
- Allow adequate washout periods between dosing in crossover designs
- Consider circadian influences on baseline gastric emptying
- Schedule experiments consistently to minimize variability
� Data Analysis:
- Use appropriate statistical models for gastric emptying curves
- Account for baseline individual variation
- Consider non-linear dose-response relationships
- Apply corrections for multiple comparisons when appropriate
� Documentation and Reproducibility:
- Maintain detailed records of peptide batch numbers
- Document reconstitution and dosing procedures
- Record all experimental conditions and deviations
- Archive data according to institutional policies
Researchers seeking to establish robust retatrutide gastric emptying research programs can benefit from consultation with experienced peptide suppliers who understand the technical requirements of these studies.
Future Directions and Emerging Research Questions
The study of retatrutide gastric emptying continues to evolve, with several important research questions remaining to be fully addressed:
Mechanistic Questions
Receptor Synergy vs. Additivity: Do the gastric emptying effects of retatrutide’s three receptor targets combine additively, or do receptor interactions create synergistic effects that exceed predictions based on individual components?
Tolerance Development: What is the time course of potential tolerance to gastric emptying effects with chronic administration, and do different receptor components show different tolerance patterns?
Individual Variability: What genetic, metabolic, or demographic factors explain the substantial individual variation observed in gastric emptying responses to retatrutide?
Translational Research Priorities
Biomarker Development: Can specific biomarkers predict individual gastric emptying responses to retatrutide, enabling more targeted research applications?
Optimal Dosing Strategies: What dosing regimens maximize beneficial metabolic effects while minimizing excessive gastric emptying delay that might compromise nutrient absorption?
Combination Approaches: How do retatrutide’s gastric effects interact with other metabolic interventions, and can combination approaches optimize overall metabolic outcomes?
Methodological Advances
Non-Invasive Assessment: Can emerging technologies provide accurate, non-invasive gastric emptying assessment suitable for repeated measurements in longitudinal studies?
Real-Time Monitoring: Will wireless motility capsules or other technologies enable continuous gastric emptying monitoring during retatrutide research?
Computational Modeling: Can mathematical models integrate gastric emptying data with other metabolic parameters to predict overall research outcomes?
Research Infrastructure and Resources
Advancing retatrutide gastric emptying research requires appropriate infrastructure and high-quality research materials. Key resources include:
🔬 High-Purity Peptides: Research-grade retatrutide with comprehensive quality documentation from established peptide suppliers
🔬 Gastric Emptying Assessment Capabilities: Access to scintigraphy, breath testing, or other validated measurement methodologies
🔬 Metabolic Phenotyping: Comprehensive assessment capabilities including glucose tolerance, hormone profiling, and energy expenditure
🔬 Data Analysis Expertise: Statistical and computational resources for complex gastric emptying kinetic analysis
🔬 Collaborative Networks: Connections to other research groups investigating incretin-based peptides and gastric physiology
Institutions building research programs in this area should ensure they have access to these essential resources before initiating comprehensive studies.
The Critical Role of Gastric Emptying in Retatrutide’s Metabolic Profile
Understanding retatrutide gastric emptying mechanisms represents far more than an academic exercise—it provides essential insights into how this novel triple-agonist peptide achieves its remarkable metabolic effects. The delayed gastric emptying produced by retatrutide’s simultaneous activation of GLP-1, GIP, and glucagon receptors creates a unique physiological signature that distinguishes it from earlier incretin-based compounds.
For researchers investigating metabolic regulation, obesity mechanisms, or next-generation peptide therapies, the gastric emptying component of retatrutide’s action offers valuable research opportunities. The mechanisms are complex, involving multiple receptor systems, neural pathways, and metabolic feedback loops that continue to yield new insights as research progresses.
Key Research Implications
The current understanding of retatrutide gastric emptying suggests several important implications for ongoing research:
✅ Multi-level regulation through peripheral and central pathways creates robust, sustained effects
✅ Dose-dependent responses enable tailored experimental designs for specific research questions
✅ Synergistic receptor interactions may produce effects exceeding simple additive predictions
✅ Integration with broader metabolic effects creates comprehensive metabolic regulation
✅ Methodological considerations require careful attention to ensure reproducible, meaningful results
Next Steps for Researchers
Institutions and laboratories interested in incorporating retatrutide gastric emptying research into their programs should consider:
1. Infrastructure Assessment: Evaluate available gastric emptying measurement capabilities and identify any gaps requiring development or collaboration.
2. Quality Peptide Sourcing: Establish relationships with reputable research peptide suppliers providing comprehensive quality documentation and technical support.
3. Protocol Development: Design detailed experimental protocols incorporating appropriate controls, measurement timepoints, and complementary metabolic assessments.
4. Collaborative Opportunities: Connect with other research groups investigating incretin-based peptides to share methodologies and insights.
5. Regulatory Compliance: Ensure all research protocols comply with institutional requirements and applicable regulations governing peptide research.
The field of metabolic peptide research continues to advance rapidly, with retatrutide representing one of the most promising compounds under investigation. Its unique gastric emptying profile contributes significantly to its overall metabolic effects and warrants continued detailed study.
Researchers seeking high-purity retatrutide and other research peptides for gastric emptying studies can explore the comprehensive catalogue available through PEPTIDE PRO, a trusted supplier of research-grade peptides with fast UK delivery and international shipping options. With rigorous quality control, detailed product documentation, and expert technical support, PEPTIDE PRO supports the research community in advancing our understanding of peptide pharmacology and metabolic regulation.
