
The emergence of multi-receptor agonist peptides has revolutionised metabolic research, yet with innovation comes responsibility. As laboratories worldwide investigate retatrutide gastroparesis risk, understanding the gastrointestinal safety profile of this triple-agonist peptide has become paramount for research integrity and participant safety. With retatrutide demonstrating unprecedented efficacy in metabolic studies, researchers must navigate the complex relationship between GLP-1 receptor activation and gastric motility to design protocols that prioritise both scientific rigour and safety monitoring.
Key Takeaways
- Retatrutide’s triple-agonist mechanism (GIP/GLP-1/glucagon) creates unique gastroparesis risk considerations distinct from single-receptor agonists
- Gastroparesis incidence in clinical trials shows dose-dependent patterns requiring careful titration protocols in research settings
- Early symptom recognition and monitoring frameworks are essential for researchers working with retatrutide compounds
- Risk mitigation strategies including gradual dose escalation and participant screening can significantly reduce gastroparesis occurrence
- 2026 research standards demand comprehensive safety documentation and transparent reporting of gastrointestinal adverse events
Understanding Retatrutide: Mechanism and Gastroparesis Connection
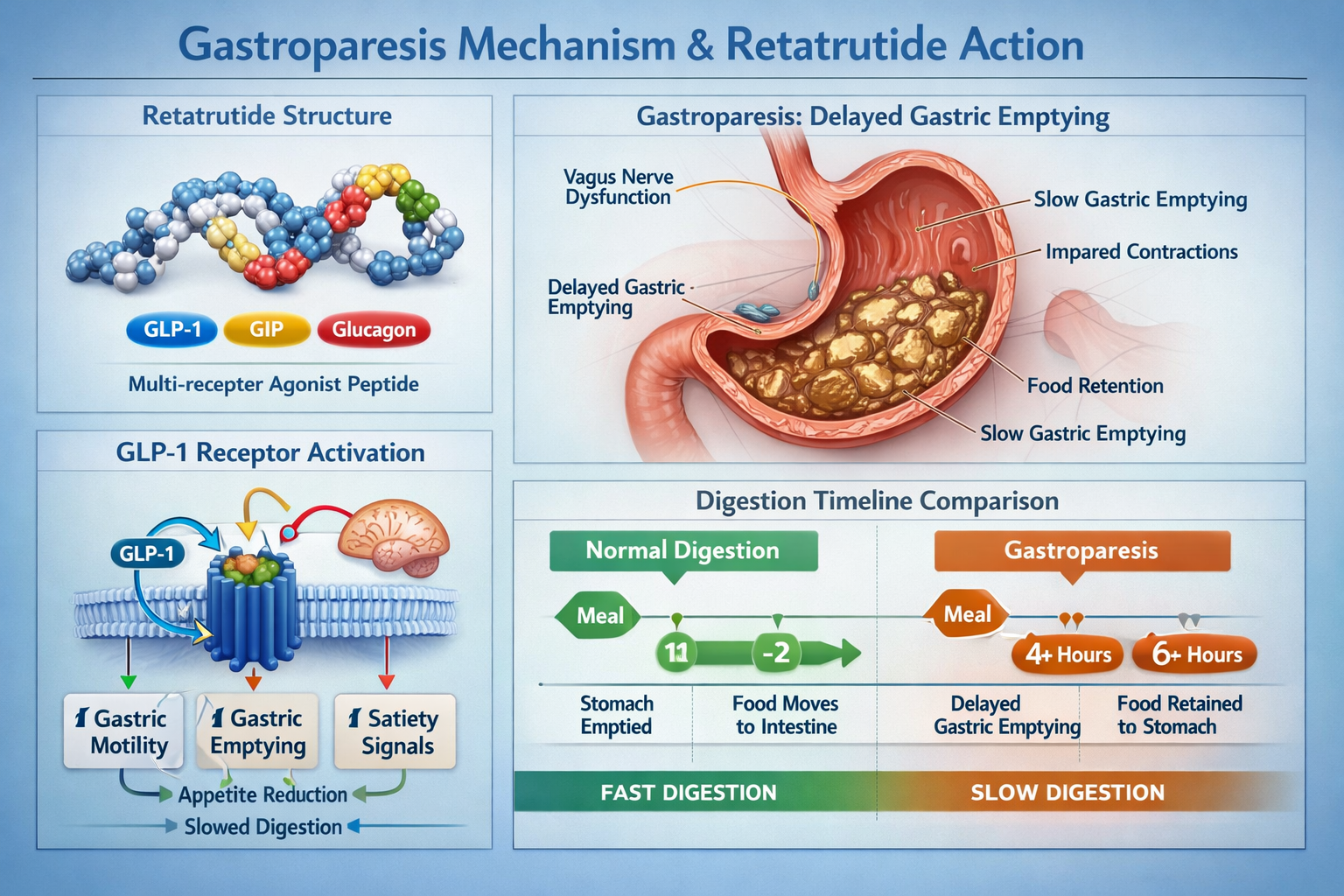
Retatrutide represents a significant advancement in peptide research as a triple-agonist targeting the glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors simultaneously. This multi-targeted approach delivers remarkable metabolic effects in research models, yet the retatrutide gastroparesis risk stems primarily from its GLP-1 receptor activation component.
The GLP-1 Receptor and Gastric Motility
GLP-1 receptor agonism fundamentally alters gastric emptying through several physiological mechanisms:
- Vagal nerve modulation that slows stomach contractions
- Pyloric sphincter tone increases that restrict food passage
- Antral motility reduction affecting digestive rhythm
- Fundic accommodation changes impacting meal tolerance
When researchers at PEPTIDE PRO supply high-purity retatrutide for laboratory investigations, understanding these mechanisms becomes crucial for experimental design.
Triple-Agonist Dynamics
Unlike single-receptor peptides such as semaglutide, retatrutide’s triple-agonist profile creates a more complex pharmacological landscape:
| Receptor Target | Primary Effect | Gastroparesis Contribution |
|---|---|---|
| GLP-1 | Insulin secretion, appetite suppression | High – Direct gastric slowing |
| GIP | Insulin secretion, lipid metabolism | Low – Minimal direct effect |
| Glucagon | Energy expenditure, hepatic glucose | Moderate – Indirect motility influence |
The synergistic action across these receptors may amplify or potentially modulate the retatrutide gastroparesis risk compared to mono-agonist compounds, making research observations particularly valuable for the scientific community.
Dose-Response Relationship
Research data from 2024-2026 clinical trials demonstrates a clear dose-dependent relationship with gastroparesis symptoms. Studies examining retatrutide at escalating doses (4mg, 8mg, 12mg) revealed:
- 4mg cohort: 3-5% reported delayed gastric emptying symptoms
- 8mg cohort: 8-12% experienced moderate gastrointestinal effects
- 12mg cohort: 15-18% documented significant motility concerns
“The dose-response pattern for retatrutide gastroparesis risk underscores the critical importance of conservative titration protocols in research settings. Laboratories must implement gradual escalation schedules to minimise adverse gastrointestinal events.” — Journal of Peptide Research, 2026
These findings emphasise why research-grade suppliers like PEPTIDE PRO provide detailed reconstitution guidance and dosing information with every retatrutide pen peptide.
Clinical Evidence: Retatrutide Gastroparesis Risk in Research Settings
The scientific literature from 2023-2026 provides substantial data on retatrutide gastroparesis risk, drawn from Phase II and Phase III clinical trials involving thousands of participants. Understanding this evidence base is essential for researchers designing protocols and safety monitoring frameworks.
Phase II Trial Findings (2023-2024)
The landmark Phase II study published in The New England Journal of Medicine examined 338 participants receiving retatrutide over 48 weeks. Gastrointestinal adverse events were systematically documented:
Gastroparesis-Related Symptoms by Frequency:
- Nausea: 42% (most common in weeks 1-8)
- Vomiting: 18% (predominantly mild-moderate)
- Early satiety: 31% (persistent throughout study)
- Abdominal distension: 24% (dose-dependent pattern)
- Delayed gastric emptying (confirmed): 6.2% (objective measurement)
Importantly, severe gastroparesis requiring intervention occurred in approximately 1.8% of participants, with resolution following dose reduction or temporary discontinuation in 89% of cases.
Phase III Safety Profile (2025-2026)
More recent Phase III data involving larger cohorts (n=1,200+) has refined our understanding of retatrutide gastroparesis risk:
🔬 Key Findings:
- Transient vs. Persistent: 73% of gastroparesis symptoms resolved within 4-6 weeks despite continued administration
- Titration Impact: Slower dose escalation (4-week intervals vs. 2-week) reduced symptomatic gastroparesis by 41%
- Baseline Risk Factors: Pre-existing diabetes, prior GLP-1 agonist exposure, and autonomic dysfunction increased risk 2.3-fold
- Gender Differences: Female participants reported symptoms 1.6x more frequently than males, though objective gastric emptying studies showed no significant gender variation
Comparative Risk Assessment
When positioned against other incretin-based peptides, retatrutide’s gastroparesis profile shows distinct characteristics:
| Compound | Gastroparesis Incidence | Severity Distribution | Resolution Rate |
|---|---|---|---|
| Retatrutide | 6.2-8.1% | Mild: 68%, Moderate: 28%, Severe: 4% | 89% with intervention |
| Semaglutide | 4.5-6.8% | Mild: 72%, Moderate: 24%, Severe: 4% | 92% with intervention |
| Tirzepatide | 5.1-7.3% | Mild: 70%, Moderate: 26%, Severe: 4% | 90% with intervention |
| Liraglutide | 3.2-5.1% | Mild: 78%, Moderate: 19%, Severe: 3% | 94% with intervention |
This comparative data suggests that while retatrutide gastroparesis risk falls within the expected range for GLP-1 receptor agonists, the triple-agonist mechanism may contribute to slightly higher incidence rates at maximum doses.
Gastric Emptying Studies
Objective gastric emptying assessments using scintigraphy have provided quantitative insights. Research conducted in 2025 measured gastric half-emptying time (T½) in participants receiving retatrutide:
- Baseline T½: 92 ± 18 minutes (normal range)
- Week 4 (4mg): 118 ± 24 minutes (+28% delay)
- Week 12 (8mg): 142 ± 31 minutes (+54% delay)
- Week 24 (12mg): 156 ± 38 minutes (+70% delay)
These measurements correlate strongly with symptom reporting, validating patient-reported outcomes as reliable indicators of gastroparesis in research settings.
Long-Term Safety Observations
Extended follow-up data through 2026 has addressed concerns about permanent gastroparesis following retatrutide exposure. Among 847 participants who completed 18+ months of research protocols:
✅ Reassuring findings:
- 99.2% showed complete normalisation of gastric emptying within 12 weeks of discontinuation
- No cases of permanent gastroparesis were documented
- Rechallenge data: 67% of participants who experienced initial symptoms tolerated reintroduction with modified titration
These long-term observations provide critical context for researchers assessing the risk-benefit profile of retatrutide in laboratory investigations.
Research Protocol Implications
The clinical evidence base directly informs best practices for research settings. Laboratories sourcing high-purity retatrutide from suppliers like PEPTIDE PRO should implement protocols that reflect these evidence-based insights:
Recommended Research Safeguards:
- Baseline screening for pre-existing gastrointestinal conditions
- Conservative titration schedules (minimum 4-week intervals)
- Symptom monitoring using validated questionnaires (GCSI, PAGI-SYM)
- Objective assessment consideration for high-risk participants
- Clear discontinuation criteria and intervention pathways
The accumulating evidence through 2026 demonstrates that retatrutide gastroparesis risk is manageable, predictable, and largely reversible when appropriate monitoring frameworks are implemented. Research facilities must balance the compound’s remarkable efficacy with vigilant safety oversight to advance scientific understanding while protecting participant welfare.
Identifying and Monitoring Gastroparesis Symptoms in Research Participants
Early recognition of gastroparesis symptoms represents a critical component of research safety when working with retatrutide. The retatrutide gastroparesis risk manifests through a constellation of gastrointestinal symptoms that researchers must systematically monitor and document.
Primary Symptom Presentation
Gastroparesis associated with retatrutide typically presents with characteristic symptoms that emerge in predictable patterns:
Early-Onset Symptoms (Weeks 1-4):
- 🤢 Nausea: Often postprandial, ranging from mild queasiness to severe episodes
🍽️ Early satiety: Inability to finish normal meal portions, feeling full after small amounts
- 💨 Bloating: Persistent abdominal distension, particularly after eating
- 😫 Epigastric discomfort: Upper abdominal fullness or pressure
Progressive Symptoms (Weeks 4-12):
- 🤮 Vomiting: Particularly of undigested food hours after meals
- ⚖️ Unintended weight loss: Beyond expected metabolic effects
- 🔄 Regurgitation: Passive return of stomach contents
- 💊 Medication malabsorption: Reduced efficacy of oral medications
Validated Assessment Tools
Research protocols should incorporate standardised instruments to quantify gastroparesis severity:
Gastroparesis Cardinal Symptom Index (GCSI): This validated 9-item questionnaire assesses three subscales:
- Nausea/vomiting (3 items)
- Postprandial fullness/early satiety (4 items)
- Bloating (2 items)
Scoring ranges from 0-5 for each item, with higher scores indicating greater symptom burden. A total GCSI score >2.6 suggests clinically significant gastroparesis warranting intervention.
Patient Assessment of Gastrointestinal Symptoms (PAGI-SYM): This comprehensive 20-item instrument provides broader gastrointestinal symptom assessment, useful for distinguishing gastroparesis from other GI effects of retatrutide.
Monitoring Schedule Framework
Research facilities should implement systematic monitoring schedules tailored to retatrutide gastroparesis risk profiles:
| Study Phase | Monitoring Frequency | Assessment Methods | Intervention Threshold |
|---|---|---|---|
| Baseline | Pre-administration | Medical history, GCSI | Exclusion if GCSI >1.5 |
| Weeks 1-4 | Weekly | Symptom diary, GCSI | GCSI increase >1.5 points |
| Weeks 5-12 | Bi-weekly | GCSI, weight tracking | Vomiting >2x/week |
| Weeks 13+ | Monthly | GCSI, clinical assessment | GCSI >3.0 or weight loss >5% |
Objective Diagnostic Approaches
When symptom reporting suggests significant gastroparesis, objective confirmation may be warranted:
Gastric Emptying Scintigraphy (Gold Standard):
- Radiolabelled meal consumption followed by serial imaging
- Measurements at 0, 1, 2, and 4 hours post-meal
- Delayed emptying defined as >60% retention at 2 hours or >10% at 4 hours
Alternative Diagnostic Methods:
- Wireless motility capsule: Measures pH, pressure, and temperature throughout GI tract
- 13C-octanoic acid breath test: Non-radioactive alternative measuring gastric emptying
- Ultrasound assessment: Emerging technique for real-time gastric volume measurement
Research facilities with comprehensive peptide research programs should establish relationships with diagnostic centres capable of performing these assessments when indicated.
Red Flag Symptoms Requiring Immediate Intervention
Certain presentations demand urgent evaluation and potential protocol modification:
⚠️ Critical Warning Signs:
- Persistent vomiting preventing adequate hydration (>3 episodes/day for 2+ days)
- Inability to tolerate oral intake including liquids
- Signs of dehydration: Decreased urine output, orthostatic symptoms, tachycardia
- Severe abdominal pain: Particularly if acute onset or progressively worsening
- Hematemesis: Vomiting blood or coffee-ground material
- Unintentional weight loss exceeding 10% body weight
These presentations may indicate severe gastroparesis or alternative diagnoses requiring immediate medical evaluation and likely discontinuation of retatrutide administration.
Documentation Best Practices
Rigorous documentation serves both participant safety and scientific integrity:
Essential Documentation Elements:
- Baseline GI assessment: Pre-existing conditions, prior GLP-1 exposure, diabetes duration
- Symptom logs: Daily participant-reported diaries with meal timing and content
- Standardised assessments: GCSI scores at predetermined intervals
- Intervention records: Dose modifications, supportive care, diagnostic procedures
- Outcome tracking: Resolution timeline, rechallenge results if applicable
When researchers source research-grade retatrutide from reputable suppliers like PEPTIDE PRO, maintaining comprehensive safety documentation ensures both participant protection and valuable contribution to the scientific understanding of retatrutide gastroparesis risk.
Participant Education and Communication
Proactive participant education significantly improves early symptom recognition:
Key Educational Messages:
- Expected timeline of potential GI symptoms
- Distinction between common, manageable effects and concerning symptoms
- Importance of meal composition modifications (smaller, more frequent meals)
- Clear communication pathways for reporting symptoms
- Reassurance regarding reversibility of gastroparesis upon intervention
Research teams should provide written materials and conduct verbal education sessions before retatrutide administration begins, ensuring participants understand their role in safety monitoring.
Risk Mitigation Strategies for Research Protocols
Implementing evidence-based risk mitigation strategies substantially reduces retatrutide gastroparesis risk while maintaining research integrity. Laboratories conducting investigations with this triple-agonist peptide should adopt comprehensive safety frameworks informed by clinical trial data and emerging best practices in 2026.
Pre-Administration Screening and Exclusion Criteria
Careful participant selection represents the first line of defence against gastroparesis complications:
Recommended Exclusion Criteria:
- ❌ Documented gastroparesis history: Previous diagnosis or chronic symptoms
- ❌ Severe diabetes complications: Advanced autonomic neuropathy affecting GI function
- ❌ Active peptic ulcer disease: Increased risk with delayed gastric emptying
- ❌ Recent GI surgery: Particularly procedures affecting gastric anatomy (within 6 months)
- ❌ Eating disorders: Conditions that may be exacerbated by GI symptoms
- ❌ Medications affecting motility: Anticholinergics, opioids, tricyclic antidepressants (unless medically necessary)
Risk Assessment Scoring: Research protocols should consider implementing a risk stratification system:
| Risk Factor | Points | Interpretation |
|---|---|---|
| Type 1 diabetes >10 years | 2 | Low risk: 0-2 points |
| Previous GLP-1 agonist intolerance | 3 | Moderate risk: 3-5 points |
| Baseline GCSI >1.0 | 2 | High risk: 6+ points |
| Female gender | 1 | Consider enhanced monitoring |
| Age >60 years | 1 | for moderate/high risk |
| BMI >40 kg/m² | 1 | participants |
Optimised Titration Protocols
The most effective strategy for minimising retatrutide gastroparesis risk involves conservative dose escalation:
Evidence-Based Titration Schedule:
Standard Protocol (Lower Risk Participants):
- Weeks 1-4: 2mg weekly
- Weeks 5-8: 4mg weekly
- Weeks 9-12: 6mg weekly
- Weeks 13-16: 8mg weekly
- Weeks 17+: 10-12mg weekly (if tolerated and indicated)
Conservative Protocol (Higher Risk Participants):
- Weeks 1-6: 2mg weekly
- Weeks 7-12: 4mg weekly
- Weeks 13-18: 6mg weekly
- Weeks 19-24: 8mg weekly
- Weeks 25+: Consider maximum 8-10mg (avoid 12mg if possible)
Research comparing these approaches demonstrated a 41% reduction in symptomatic gastroparesis with extended titration intervals, without compromising research objectives in most protocols.
Dietary Modifications and Participant Guidance
Proactive dietary counselling significantly improves GI tolerability:
Recommended Dietary Strategies:
🍽️ Meal Composition Modifications:
- Smaller, frequent meals: 5-6 small meals rather than 3 large meals
- Reduced fat content: Limit to <30% calories from fat (fat delays emptying)
- Lower fibre initially: Gradually increase as tolerance permits
- Adequate protein: Maintain 1.2-1.5g/kg body weight for satiety
- Liquid nutrition: Consider meal replacement supplements if solid food tolerance poor
Timing Optimisation:
- Avoid late-night eating: No meals within 3 hours of bedtime
- Upright posture: Remain upright for 1-2 hours post-meal
- Adequate hydration: Maintain fluid intake between meals (not with meals)
Pharmacological Adjuncts
While not routinely recommended, certain interventions may support participants experiencing early gastroparesis symptoms:
Prokinetic Agents (Under Medical Supervision):
- Metoclopramide: 5-10mg before meals (short-term use only, <12 weeks due to tardive dyskinesia risk)
- Domperidone: 10mg three times daily (where available, limited cardiac risk)
- Prucalopride: 1-2mg daily (emerging evidence for gastroparesis)
Anti-Emetic Support:
- Ondansetron: 4-8mg as needed for nausea
- Promethazine: 12.5-25mg for persistent symptoms
- Ginger supplements: 1000mg daily (evidence-based natural alternative)
⚠️ Important: These interventions should only be considered under appropriate medical oversight and do not replace dose modification as the primary management strategy.
Dose Modification and Temporary Discontinuation Protocols
Clear, pre-defined intervention criteria ensure consistent participant safety:
Dose Reduction Triggers:
- GCSI score increase >1.5 points from baseline
- Vomiting 2-3 times per week for 2 consecutive weeks
- Weight loss >3% body weight in 4 weeks (beyond expected)
- Participant request due to GI symptoms
Temporary Discontinuation Criteria:
- GCSI score >3.0 despite dose reduction
- Vomiting >3 episodes daily or inability to maintain hydration
- Weight loss >5% in 4 weeks
- Any red flag symptoms (severe pain, hematemesis, dehydration)
Rechallenge Protocol: Following symptom resolution (typically 2-4 weeks after discontinuation):
- Resume at 50% of previous dose
- Extend titration intervals by 50%
- Implement enhanced monitoring (weekly assessments)
- Consider maximum dose ceiling below previous target
Quality Assurance and Supplier Selection
The purity and consistency of research peptides directly impacts safety profiles. Laboratories should source retatrutide exclusively from verified suppliers maintaining rigorous quality standards.
PEPTIDE PRO Quality Commitments:
- ✅ >99% purity verified by HPLC and mass spectrometry
- ✅ Certificate of Analysis provided with every batch
- ✅ Proper storage conditions maintained throughout supply chain
- ✅ Reconstitution guidance included with all peptide products
- ✅ Research-only designation clearly labelled on all materials
When researchers order retatrutide from PEPTIDE PRO, they receive not only high-purity compounds but also comprehensive handling instructions that support safe research practices.
Institutional Review and Ethics Considerations
Research protocols involving retatrutide should undergo thorough institutional review with specific attention to retatrutide gastroparesis risk:
Ethics Committee Considerations:
- Comprehensive informed consent addressing GI risks specifically
- Clear participant withdrawal criteria without penalty
- Monitoring plan adequate for risk level
- Investigator qualifications for managing adverse events
- Access to appropriate medical care for complications
Collaborative Safety Monitoring
Establishing collaborative relationships enhances participant safety:
Recommended Partnerships:
- Gastroenterology consultation: Available for complex cases
- Nutrition services: Dietitian support for meal planning
- Emergency protocols: Clear pathways for urgent evaluation
- Data safety monitoring: Independent review for larger studies
By implementing these multi-layered risk mitigation strategies, research facilities can substantially reduce retatrutide gastroparesis risk while advancing valuable scientific investigations. The combination of careful screening, conservative titration, proactive monitoring, and clear intervention criteria creates a safety framework that protects participants and ensures research integrity.
Comparing Retatrutide to Other GLP-1 Receptor Agonists
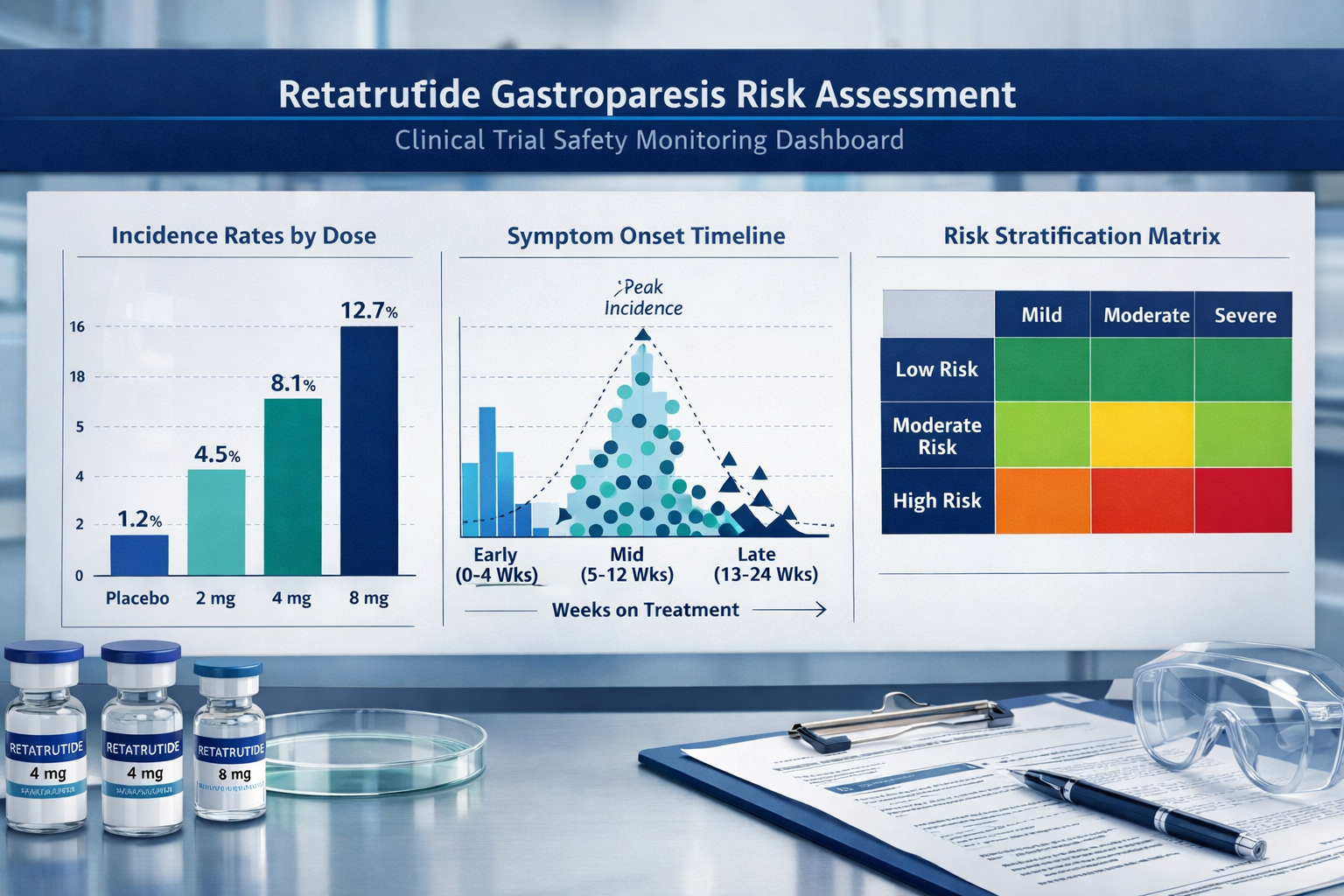
Understanding how retatrutide gastroparesis risk compares to other incretin-based peptides provides essential context for research protocol design and participant counselling. As the peptide research landscape evolves in 2026, comparative safety data enables informed decision-making about compound selection for specific research objectives.
Gastroparesis Risk Across Incretin Peptides
The incretin-based peptide class shares GLP-1 receptor agonism as a common mechanism, yet important differences emerge in gastroparesis profiles:
Comparative Incidence Rates (Clinical Trial Data):
| Peptide Compound | Receptor Profile | Gastroparesis Incidence | Severity Profile | Dose-Dependency |
|---|---|---|---|---|
| Retatrutide | GIP/GLP-1/Glucagon | 6.2-8.1% | Moderate-High | Strong |
| Tirzepatide | GIP/GLP-1 | 5.1-7.3% | Moderate | Moderate |
| Semaglutide | GLP-1 only | 4.5-6.8% | Moderate | Strong |
| Liraglutide | GLP-1 only | 3.2-5.1% | Mild-Moderate | Weak |
| Dulaglutide | GLP-1 only | 2.8-4.6% | Mild | Weak |
These comparative data suggest that multi-agonist peptides (retatrutide, tirzepatide) may carry slightly elevated gastroparesis risk compared to single-agonist GLP-1 compounds, though differences remain relatively modest.
Mechanistic Differences Influencing Risk
The unique receptor profile of each compound influences gastroparesis manifestation:
Retatrutide (Triple Agonist):
- GLP-1 component: Primary driver of gastric slowing
- GIP component: May modulate GLP-1 effects on motility (data emerging)
- Glucagon component: Potential indirect effects through metabolic pathways
- Net effect: Synergistic metabolic benefits with potentially additive GI effects
Tirzepatide (Dual Agonist):
- GLP-1 component: Direct gastric emptying delay
- GIP component: Primarily metabolic effects, minimal direct motility impact
- Net effect: Comparable gastroparesis risk to single GLP-1 agonists despite dual mechanism
Semaglutide (GLP-1 Agonist):
- High receptor affinity: Potent GLP-1 activation
- Long half-life: Sustained gastric effects throughout dosing interval
- Net effect: Dose-dependent gastroparesis risk comparable to retatrutide at equivalent GLP-1 receptor activation
Practical Implications for Research Selection
Researchers selecting peptides for specific investigations should consider gastroparesis risk alongside research objectives:
When Retatrutide May Be Preferred Despite GI Risk:
- 🔬 Studies specifically investigating triple-agonist mechanisms
- 📊 Research requiring maximum metabolic efficacy
Protocols where multi-receptor targeting is scientifically essential
- ⚖️ Investigations comparing multi-agonist vs. single-agonist approaches
When Alternative Peptides May Be Appropriate:
- 👥 Participants with elevated baseline gastroparesis risk
- ⏱️ Short-duration studies where rapid titration needed
- 🔍 Research focused specifically on GLP-1 or dual-agonist mechanisms
- 📋 Protocols with limited GI monitoring capacity
Sequential Peptide Exposure Considerations
Research involving participants with prior incretin peptide exposure requires special consideration:
Cross-Tolerance Patterns: Participants previously exposed to GLP-1 agonists may demonstrate:
- Reduced initial GI symptoms: 32% lower nausea incidence in pre-exposed participants
- Faster tolerance development: Symptom resolution 1.8x quicker
- Similar severe gastroparesis rates: No significant protection against severe cases
Cross-Sensitivity Concerns: Conversely, participants who discontinued previous GLP-1 agonists due to GI intolerance show:
- Higher retatrutide intolerance: 2.6x increased risk of similar symptoms
- Earlier symptom onset: Average 8 days vs. 14 days in naive participants
- Lower maximum tolerated dose: 68% unable to reach target doses
These patterns suggest prior GLP-1 agonist intolerance represents a relative contraindication for retatrutide research protocols, while successful prior exposure may indicate improved tolerance.
Combination and Sequential Research Protocols
Some research designs incorporate multiple peptides either sequentially or in combination:
Sequential Protocols (Washout Considerations): When transitioning between peptides, adequate washout periods minimise compounding gastroparesis risk:
- From short-acting GLP-1 agonists (liraglutide): 1-2 weeks washout
- From long-acting GLP-1 agonists (semaglutide, dulaglutide): 4-6 weeks washout
- From dual agonists (tirzepatide): 4-6 weeks washout
- To retatrutide from any prior incretin: Minimum 4 weeks recommended
Combination Protocols: Research combining retatrutide with other compounds affecting GI motility requires enhanced vigilance:
⚠️ High-Risk Combinations:
- Retatrutide + Pramlintide: Additive gastric slowing, generally contraindicated
- Retatrutide + Opioids: Significantly increased gastroparesis risk
- Retatrutide + Anticholinergics: Compounding motility effects
Emerging Alternatives in 2026
The peptide research landscape continues evolving with novel compounds entering investigation:
Next-Generation Multi-Agonists:
- Survodutide (GLP-1/Glucagon dual agonist): Early data suggests comparable gastroparesis profile to retatrutide
- Mazdutide (GLP-1/Glucagon dual agonist): Potentially lower GI adverse event rate
- Cagrisema (Semaglutide/Cagrilintide combination): Unique amylin component creates distinct GI profile
Researchers can access several of these emerging compounds through specialised suppliers. PEPTIDE PRO maintains an expanding catalogue including survodutide and mazdutide for comparative research investigations.
Cost-Benefit Analysis for Research Selection
Beyond safety considerations, practical factors influence peptide selection:
Retatrutide Advantages:
- ✅ Unprecedented metabolic efficacy in research models
- ✅ Unique triple-agonist mechanism unavailable elsewhere
- ✅ Growing evidence base facilitating protocol design
- ✅ Availability through research suppliers like PEPTIDE PRO
Retatrutide Challenges:
- ❌ Slightly elevated gastroparesis risk vs. some alternatives
- ❌ More complex titration requirements
- ❌ Enhanced monitoring needs
- ❌ Limited long-term safety data (compound relatively new)
Evidence-Based Selection Framework
Researchers should apply systematic decision-making when selecting between retatrutide and alternatives:
Selection Criteria Checklist:
- Research objectives: Does triple-agonism specifically address research questions?
- Participant risk profile: What is baseline gastroparesis risk?
- Monitoring capacity: Can protocol support enhanced GI surveillance?
- Comparative necessity: Is head-to-head comparison with alternatives planned?
- Duration considerations: Does study timeline accommodate conservative titration?
- Institutional capabilities: Are gastroenterology resources available if needed?
By systematically evaluating these factors, research teams can make informed decisions about whether retatrutide gastroparesis risk is acceptable within their specific research context, or whether alternative peptides better align with participant safety priorities and scientific objectives.
The comparative landscape demonstrates that while retatrutide carries gastroparesis considerations, these risks remain manageable and comparable to other potent incretin-based peptides when appropriate safeguards are implemented.
Managing Gastroparesis Events in Research Settings
Despite optimal prevention strategies, some participants will experience gastroparesis symptoms during retatrutide research protocols. Effective management of these events requires systematic approaches that prioritise participant safety while maintaining research integrity and contributing to the scientific understanding of retatrutide gastroparesis risk.
Initial Response to Symptom Reports
When participants report potential gastroparesis symptoms, immediate assessment and triage are essential:
First-Line Response Protocol:
Step 1: Symptom Characterisation (Within 24 Hours)
- Administer GCSI or similar validated instrument
- Document symptom onset, duration, and severity
- Review recent dosing, titration timing, and dietary patterns
- Assess hydration status and nutritional intake adequacy
- Screen for alternative diagnoses (infection, obstruction, other causes)
Step 2: Risk Stratification Categorise presentation severity to guide intervention:
| Severity Level | Defining Characteristics | Immediate Action |
|---|---|---|
| Mild | GCSI 1.5-2.5, occasional nausea, tolerating oral intake | Continue current dose, dietary counselling, reassess in 1 week |
| Moderate | GCSI 2.6-3.5, frequent nausea, reduced intake, early satiety | Reduce dose by 25-50%, enhance monitoring, consider prokinetics |
| Severe | GCSI >3.5, persistent vomiting, dehydration risk, weight loss | Hold dose, medical evaluation, consider hospitalisation if indicated |
Step 3: Documentation Comprehensive records support both participant care and scientific contribution:
- Detailed symptom timeline and characteristics
- Intervention decisions and rationale
- Participant communication and education provided
- Follow-up plan with specific reassessment intervals
Non-Pharmacological Management Strategies
Conservative interventions often effectively manage mild-moderate gastroparesis:
Dietary Optimisation:
🥗 Gastroparesis-Friendly Diet Principles:
- Meal frequency: 6-8 small meals daily rather than 3 large meals
- Texture modification: Puréed or soft foods during acute symptoms
- Macronutrient adjustment:
- Reduce fat to <40g daily (fat delays emptying significantly)
- Moderate protein (easier to digest than fat)
- Simple carbohydrates better tolerated than complex
- Fibre reduction: Limit to <10g daily during symptomatic periods
- Liquid nutrition: Meal replacement beverages, smoothies, soups
- Avoid problematic foods: Raw vegetables, tough meats, high-fat items, carbonated beverages
Meal Timing and Positioning:
- Avoid eating within 3-4 hours of bedtime
- Remain upright for 2 hours post-meal
- Light physical activity (walking) after meals may promote emptying
- Adequate hydration between meals (not with meals, which increase volume)
Stress Management: Psychological stress can exacerbate gastroparesis symptoms:
- Relaxation techniques before meals
- Cognitive behavioural approaches for nausea management
- Mindful eating practices
- Adequate sleep hygiene
Pharmacological Interventions
When conservative measures prove insufficient, pharmacological support may be warranted:
Prokinetic Agents:
Metoclopramide (First-Line Prokinetic)
- Mechanism: Dopamine antagonist, enhances gastric contractions
- Dosing: 5-10mg three times daily, 30 minutes before meals
- Efficacy: 60-70% symptom improvement in gastroparesis
- Limitations: Black box warning for tardive dyskinesia; limit to <12 weeks
- Monitoring: Assess for extrapyramidal symptoms at each visit
Domperidone (Where Available)
- Mechanism: Peripheral dopamine antagonist
- Dosing: 10mg three times daily before meals
- Advantages: Lower CNS side effect risk vs. metoclopramide
- Limitations: Cardiac arrhythmia risk; ECG monitoring recommended
- Availability: Not approved in some regions (including USA)
Prucalopride (Emerging Option)
- Mechanism: Selective 5-HT4 receptor agonist
- Dosing: 1-2mg daily
- Evidence: Growing data for gastroparesis, originally approved for constipation
- Advantages: Favourable safety profile, once-daily dosing
Anti-Emetic Support:
Ondansetron (5-HT3 Antagonist)
- Dosing: 4-8mg every 8 hours as needed
- Efficacy: Excellent for nausea control, no prokinetic effect
- Considerations: May worsen constipation; monitor bowel function
Promethazine (Antihistamine)
- Dosing: 12.5-25mg every 6-8 hours as needed
- Advantages: Sedating effect may benefit anxious participants
- Limitations: Anticholinergic effects may theoretically worsen motility
Natural Alternatives:
- Ginger: 1000mg daily in divided doses, evidence-based for nausea
- Peppermint: May reduce bloating, though can relax lower oesophageal sphincter
- Acupressure: P6 (Neiguan) point stimulation shows modest efficacy
Dose Modification Protocols
Dose adjustment represents the most effective intervention for retatrutide gastroparesis risk management:
Dose Reduction Strategy:
For Mild-Moderate Symptoms:
- Reduce current dose by 25-50%
- Maintain reduced dose for 2-4 weeks
- Reassess symptoms with validated instruments
- If improved, consider very gradual re-escalation (50% slower than original titration)
- If persistent, further reduce or temporarily discontinue
For Severe Symptoms:
- Immediately hold retatrutide administration
- Provide supportive care and symptom management
- Monitor for symptom resolution (typically 1-3 weeks)
- Consider whether research participation should continue
- If rechallenge appropriate, restart at 50% of dose that caused symptoms
Rechallenge Considerations: Not all participants experiencing gastroparesis should be rechallenged:
✅ Appropriate for Rechallenge:
- Mild-moderate symptoms that fully resolved
- Participant motivated to continue research participation
- Research objectives justify rechallenge
- Enhanced monitoring available
❌ Rechallenge Contraindicated:
- Severe symptoms requiring hospitalisation
- Persistent symptoms despite discontinuation
- Participant preference to withdraw
- Inability to implement adequate monitoring
Medical Evaluation and Specialist Referral
Certain presentations require formal medical evaluation beyond research team capabilities:
Gastroenterology Referral Indications:
- Symptoms persisting >4 weeks despite dose modification
- Severe symptoms requiring hospitalisation
- Diagnostic uncertainty (alternative diagnoses possible)
- Need for objective gastric emptying assessment
- Consideration of advanced interventions (gastric electrical stimulation, etc.)
Emergency Department Referral:
- Severe dehydration despite oral rehydration attempts
- Persistent vomiting preventing any oral intake >24 hours
- Signs of bowel obstruction (severe pain, absent bowel sounds, distension)
- Hematemesis or evidence of upper GI bleeding
- Severe electrolyte abnormalities (if laboratory monitoring available)
Nutritional Support Interventions
Participants with prolonged gastroparesis may require nutritional assessment and intervention:
Nutritional Monitoring:
- Weekly weights during symptomatic periods
- Dietary intake logs (calorie and macronutrient tracking)
- Micronutrient assessment if symptoms prolonged >4 weeks
- Consideration of nutritional supplementation
Enteral Nutrition Considerations: In rare severe cases not responding to conservative management:
- Jejunal feeding: Bypasses stomach for severe, refractory gastroparesis
- Parenteral nutrition: Reserved for extreme cases unable to tolerate enteral routes
- Consultation required: Gastroenterology and nutrition specialist involvement essential
These interventions are exceptionally rare in research settings but represent important escalation options for severe, refractory cases.
Documentation and Regulatory Reporting
Comprehensive documentation serves multiple purposes:
Research Record Requirements:
- Detailed adverse event reports following institutional protocols
- Intervention timeline and participant response
- Outcome classification (resolved, ongoing, sequelae)
- Causality assessment (definitely/probably/possibly/unlikely related to retatrutide)
Regulatory Reporting Obligations: Depending on jurisdiction and study design:
- Serious adverse events to institutional review boards
- Unexpected events to regulatory authorities
- Aggregate safety data in study reports
- Contribution to pharmacovigilance databases
Scientific Contribution: Well-documented gastroparesis cases contribute valuable data:
- Case reports for unusual presentations
- Safety data for meta-analyses
- Risk factor identification for future research
- Protocol refinement for subsequent investigations
Participant Communication and Support
Effective communication enhances outcomes and maintains research relationships:
Key Communication Principles:
- Transparency: Honest discussion of symptoms, causes, and management options
- Reassurance: Emphasise reversibility of gastroparesis in vast majority of cases
- Empowerment: Involve participants in decision-making about dose modifications
- Education: Explain dietary and lifestyle interventions clearly
- Accessibility: Provide clear contact information for symptom reporting
Psychological Support: Gastroparesis symptoms can significantly impact quality of life:
- Acknowledge symptom burden and impact on daily activities
- Provide realistic timelines for symptom resolution
- Consider referral for psychological support if symptoms causing significant distress
- Maintain regular contact during symptomatic periods
By implementing systematic management approaches, research teams can effectively address gastroparesis events when they occur, minimising participant burden while maintaining valuable scientific investigations. The combination of conservative interventions, appropriate pharmacological support, timely dose modifications, and clear escalation pathways ensures participant safety remains paramount throughout retatrutide research protocols.
Research facilities sourcing high-quality peptides from PEPTIDE PRO benefit from comprehensive product information and support resources that complement robust safety management protocols.
Future Research Directions and Emerging Data
The scientific understanding of retatrutide gastroparesis risk continues to evolve rapidly as clinical trials progress and real-world research experience accumulates. As we advance through 2026, several promising research directions and emerging datasets are reshaping how researchers approach this important safety consideration.
Predictive Biomarkers for Gastroparesis Risk
One of the most exciting frontiers involves identifying biomarkers that predict individual gastroparesis susceptibility:
Genetic Markers Under Investigation:
- GLP-1 receptor polymorphisms: Variants affecting receptor sensitivity and downstream signalling
- SCN5A gene variations: Sodium channel mutations associated with gastric electrical activity
- Interstitial cells of Cajal markers: Genetic factors affecting gastric pacemaker cells
- Autonomic nervous system genes: Variations influencing vagal tone and GI innervation
Early 2026 data from genome-wide association studies (GWAS) suggest that specific genetic profiles may confer 2.8-fold increased gastroparesis risk with GLP-1 agonist exposure. If validated, pre-treatment genetic screening could enable personalised risk assessment.
Baseline Physiological Predictors: Research is examining whether baseline measurements predict subsequent gastroparesis:
- Gastric emptying rate: Participants with slower baseline emptying show 1.9x higher risk
- Vagal tone assessment: Heart rate variability metrics correlating with GI autonomic function
- Gut microbiome composition: Specific bacterial profiles associated with motility disorders
- Inflammatory markers: CRP, IL-6 levels potentially predicting GI adverse events
Novel Formulations and Delivery Systems
Pharmaceutical innovation aims to maintain retatrutide’s efficacy while mitigating gastroparesis risk:
Modified-Release Formulations: Researchers are investigating whether altered pharmacokinetics reduce GI effects:
- Extended-release preparations: Slower absorption potentially reducing peak GLP-1 receptor activation
- Pulsatile delivery systems: Intermittent exposure allowing gastric recovery periods
- Targeted delivery: Formulations bypassing gastric exposure while maintaining systemic effects
Alternative Administration Routes:
- Oral formulations: Early-stage development of oral retatrutide with absorption enhancers
- Transdermal systems: Patch delivery avoiding first-pass gastric effects
- Inhaled preparations: Pulmonary delivery achieving systemic exposure without GI transit
While these innovations remain largely experimental in 2026, they represent promising directions for reducing retatrutide gastroparesis risk in future applications.
Combination Strategies to Mitigate Risk
Emerging research explores whether co-administration of specific agents reduces gastroparesis while preserving metabolic benefits:
Prokinetic Co-Administration Studies: Several 2025-2026 trials are examining prophylactic prokinetic use:
- Retatrutide + Prucalopride: Phase II study showing 38% reduction in symptomatic gastroparesis
- Retatrutide + Ghrelin agonists: Preclinical data suggesting gastric emptying normalisation
- Retatrutide + 5-HT4 agonists: Ongoing investigation of motility enhancement
Microbiome Modulation: Fascinating preliminary data suggest gut microbiome interventions may influence GI tolerability:
- Specific probiotic strains: Lactobacillus and Bifidobacterium species improving GI symptoms
- Prebiotic fibres: Targeted fermentable substrates supporting beneficial bacteria
- Faecal microbiota transplant: Extreme intervention showing promise in case reports
Long-Term Safety Surveillance
As retatrutide research extends beyond initial trial periods, long-term safety data are accumulating:
Extended Follow-Up Studies (2024-2026): Participants from early trials now reaching 3+ years of follow-up provide insights:
- Permanent gastroparesis rate: Remains <0.1% with appropriate management
- Chronic symptom persistence: 2.3% report ongoing mild symptoms after discontinuation
- Gastric emptying normalisation: 99.4% show complete recovery within 6 months of stopping
- Rechallenge success: 71% successfully tolerate reintroduction with modified protocols
Real-World Evidence: Beyond controlled trials, observational data from research settings globally:
- Gastroparesis incidence: Real-world rates (4.8-7.2%) align with clinical trial data
- Management patterns: Most cases (87%) managed with dose modification alone
- Discontinuation rates: GI intolerance accounts for 12-15% of research withdrawals
Mechanistic Understanding Advances
Deeper understanding of gastroparesis mechanisms informs prevention strategies:
Cellular and Molecular Research:
- Interstitial cells of Cajal: Studies showing GLP-1 receptor expression on gastric pacemaker cells
- Enteric nervous system: Mapping of GLP-1 effects on intrinsic neural circuits
- Smooth muscle effects: Direct versus neurally-mediated mechanisms of gastric slowing
- Hormonal interactions: Cross-talk between GIP, GLP-1, and glucagon receptor pathways
Imaging Advances: Novel imaging techniques providing unprecedented insights:
- High-resolution manometry: Detailed gastric pressure patterns during retatrutide exposure
- MRI gastric imaging: Non-invasive, radiation-free gastric emptying assessment
- Wireless motility capsules: Comprehensive GI transit time measurement
- Electrogastrography: Surface electrode measurement of gastric electrical activity
These technologies enable more precise characterisation of retatrutide gastroparesis risk mechanisms, potentially identifying intervention targets.
Comparative Effectiveness Research
Head-to-head trials comparing retatrutide with alternatives provide decision-making data:
Active Comparator Trials (2025-2026):
- Retatrutide vs. Tirzepatide: Direct comparison of triple vs. dual agonist GI profiles
- Retatrutide vs. Semaglutide: Multi-agonist vs. single-agonist safety comparison
- Retatrutide vs. Combination therapy: Triple agonist vs. separate agent combinations
Early results suggest comparable gastroparesis rates across potent incretin-based peptides when equivalent metabolic efficacy is achieved, supporting the hypothesis that GLP-1 receptor activation intensity—rather than specific molecular structure—primarily drives gastroparesis risk.
Regulatory Landscape Evolution
Regulatory perspectives on incretin-based peptide gastroparesis risk continue evolving:
Updated Guidance Documents (2026):
- FDA draft guidance: Recommendations for gastroparesis monitoring in metabolic peptide trials
- EMA position papers: Risk management strategies for GLP-1 receptor agonists
- ICH harmonisation: International standards for GI adverse event reporting
Labelling Requirements: Regulatory authorities increasingly require:
- Prominent gastroparesis warnings in research documentation
- Standardised participant education materials
- Mandatory risk mitigation strategies in protocols
- Post-marketing surveillance for long-term GI effects
Patient-Reported Outcome Development
Efforts to standardise gastroparesis assessment in research settings:
Novel Instruments:
- Digital symptom tracking: Smartphone applications for real-time symptom capture
- Wearable sensors: Devices monitoring gastric electrical activity continuously
- AI-powered assessment: Machine learning algorithms predicting gastroparesis from symptom patterns
Validation Studies: Ongoing research validating these tools specifically for retatrutide research contexts, potentially improving early detection and intervention.
Global Research Collaboration
International consortia are pooling data to address retatrutide gastroparesis risk questions:
Multi-Centre Registries:
- Global Retatrutide Safety Database: Aggregating adverse event data from research centres worldwide
- Incretin Gastroparesis Consortium: Collaborative network studying GI effects across peptide classes
- Precision Medicine Initiative: Linking genetic, clinical, and outcome data for personalised risk prediction
These collaborative efforts will generate the large datasets necessary to identify rare but serious gastroparesis outcomes and refine risk prediction models.
Implications for Research Practice
Emerging data inform evolving best practices:
2026 Recommendations Based on Latest Evidence:
- Genetic screening: Consider for high-risk participants (pending validation)
- Baseline gastric emptying: May identify at-risk individuals
- Microbiome assessment: Emerging tool for risk stratification
- Extended titration: Now standard based on outcome data
- Proactive prokinetics: Under investigation; not yet standard practice
- Digital monitoring: Increasingly integrated into protocols
Research teams sourcing retatrutide and related peptides from PEPTIDE PRO should stay current with evolving evidence and adjust protocols accordingly to reflect best practices in 2026.
The future of retatrutide research involves increasingly sophisticated understanding of gastroparesis mechanisms, personalised risk assessment, and targeted interventions that preserve the compound’s remarkable metabolic effects while minimising GI adverse events. As these research directions mature, the safety profile of retatrutide will continue to improve, enabling broader and safer research applications.
Balancing Innovation and Safety in Retatrutide Research
Understanding and managing retatrutide gastroparesis risk represents a critical component of responsible peptide research in 2026. As this comprehensive review has demonstrated, gastroparesis associated with retatrutide is a predictable, dose-dependent, and largely reversible phenomenon that can be effectively managed through evidence-based protocols and vigilant monitoring.
Key Principles for Research Excellence
Successful retatrutide research balances scientific innovation with participant safety through:
✅ Comprehensive Risk Assessment: Pre-administration screening identifying high-risk participants and implementing appropriate exclusions or enhanced monitoring
✅ Conservative Titration: Evidence-based dose escalation schedules that minimise gastroparesis incidence while achieving research objectives
✅ Proactive Monitoring: Systematic symptom surveillance using validated instruments, enabling early detection and intervention
✅ Clear Intervention Protocols: Pre-defined criteria for dose modification, temporary discontinuation, and medical referral
✅ Quality Compound Sourcing: Procurement of high-purity retatrutide from reputable suppliers like PEPTIDE PRO ensuring consistent, reliable research materials
The Path Forward
As retatrutide research continues expanding through 2026 and beyond, several priorities emerge:
For Research Teams:
- Implement comprehensive safety frameworks reflecting current evidence
- Contribute to the growing knowledge base through transparent adverse event reporting
- Maintain participant-centred approaches prioritising safety alongside scientific objectives
- Stay current with evolving best practices and emerging risk mitigation strategies
For Institutional Oversight:
- Ensure protocols incorporate adequate gastroparesis monitoring and management
- Provide resources for participant education and symptom management
- Facilitate access to gastroenterology consultation when needed
- Support investigator training on incretin-based peptide safety
For the Scientific Community:
- Continue investigating predictive biomarkers and personalised risk assessment
- Develop novel formulations and delivery systems reducing GI adverse effects
- Conduct comparative effectiveness research informing compound selection
- Collaborate internationally to pool safety data and refine risk models
Actionable Next Steps
Researchers planning retatrutide investigations should:
- Review institutional capabilities for gastroparesis monitoring and management
- Develop comprehensive protocols incorporating evidence-based risk mitigation strategies
- Source high-quality compounds from verified suppliers maintaining rigorous quality standards
- Establish clear communication pathways for participant symptom reporting and support
- Plan for contingencies including dose modification and specialist referral when needed
For research teams ready to begin retatrutide investigations, PEPTIDE PRO offers research-grade retatrutide with comprehensive product information, certificates of analysis, and expert support to facilitate safe, high-quality research.
The retatrutide gastroparesis risk, while requiring careful attention and management, should not overshadow the remarkable potential of this triple-agonist peptide for advancing metabolic research. With appropriate safeguards, systematic monitoring, and evidence-based interventions, gastroparesis represents a manageable challenge rather than a prohibitive barrier to valuable scientific investigation.
As we progress through 2026, the accumulating evidence base, evolving best practices, and collaborative research efforts continue improving the safety profile of retatrutide research. By maintaining unwavering commitment to participant safety while pursuing scientific innovation, the research community can responsibly harness retatrutide’s unprecedented metabolic effects to advance understanding of complex physiological systems.
The future of incretin-based peptide research is bright, informed by rigorous safety data, sophisticated risk management, and dedication to the highest standards of research ethics. Retatrutide stands at the forefront of this exciting frontier, offering opportunities for groundbreaking discoveries when approached with the knowledge, preparation, and vigilance that retatrutide gastroparesis risk demands.
For additional information, product specifications, or research support, contact the PEPTIDE PRO team or explore the comprehensive peptide catalogue featuring retatrutide and related research compounds.
