
When researchers first began investigating retatrutide’s triple-agonist mechanism targeting GLP-1, GIP, and glucagon receptors, the compound’s metabolic effects drew immediate attention. However, as clinical trials progressed, a pattern emerged that demanded equal scrutiny: retatrutide headache reports appeared consistently across study populations, prompting deeper investigation into this common adverse event. Understanding the mechanisms, prevalence, and management strategies for retatrutide-associated headaches has become essential for researchers working with this novel peptide compound.
The occurrence of headaches during retatrutide research protocols represents more than a mere inconvenience—it offers valuable insights into the compound’s neurological interactions and provides critical data for optimising research methodologies. As laboratories across the UK and internationally continue investigating this triple-agonist peptide, comprehensive documentation of adverse events like headaches contributes to the broader scientific understanding of metabolic peptide mechanisms.
Key Takeaways
- Retatrutide headache occurs in approximately 20-30% of research subjects during clinical trials, making it one of the most commonly reported adverse events
- The mechanism likely involves vascular changes, neurotransmitter modulation, and metabolic shifts associated with GLP-1, GIP, and glucagon receptor activation
- Headache severity typically decreases over time as physiological adaptation occurs, with most cases classified as mild to moderate intensity
- Research protocols can incorporate hydration optimisation, gradual dose escalation, and electrolyte monitoring to potentially reduce headache incidence
- Proper documentation of headache patterns contributes valuable data to the scientific understanding of retatrutide’s neurological effects
What Is Retatrutide? Understanding the Triple-Agonist Mechanism
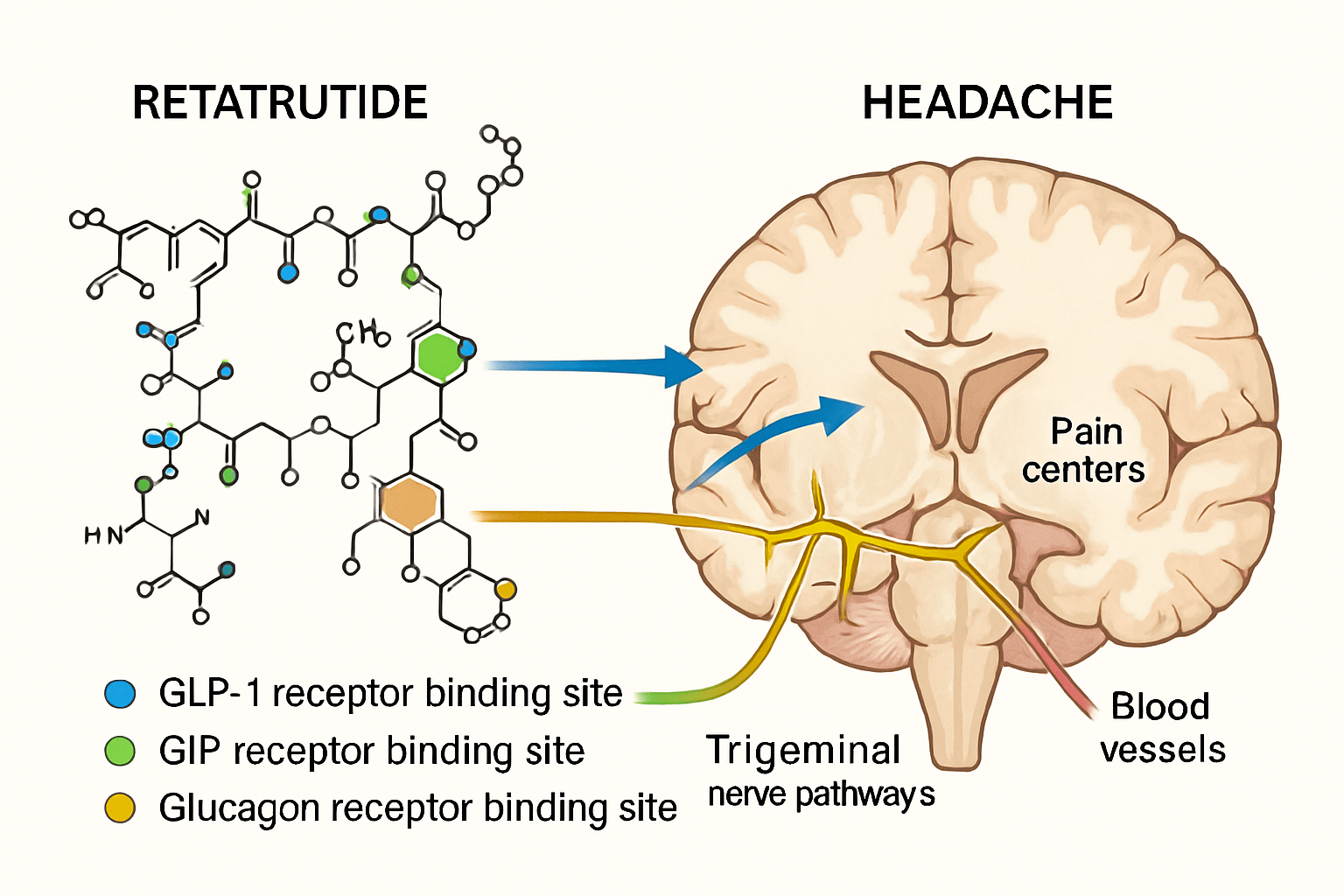
Retatrutide represents a significant advancement in peptide research, functioning as a triple receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile distinguishes it from earlier single or dual-agonist compounds and creates a complex cascade of metabolic effects that extend throughout multiple physiological systems.
The compound’s molecular structure allows it to bind with high affinity to all three target receptors, creating synergistic effects that researchers continue to investigate. At PEPTIDE PRO, research-grade retatrutide undergoes rigorous quality control to ensure consistency and purity for laboratory applications, enabling researchers to study these mechanisms with confidence in their materials.
The Receptor Activation Profile
Each receptor activation contributes distinct physiological responses:
- GLP-1 receptor activation: Influences insulin secretion, gastric emptying, appetite regulation, and potentially cardiovascular function
- GIP receptor activation: Modulates insulin response, lipid metabolism, and bone metabolism
- Glucagon receptor activation: Affects energy expenditure, hepatic glucose production, and thermogenesis
This multi-receptor engagement creates a comprehensive metabolic response that researchers hypothesise may contribute to the headache phenomenon through various pathways including altered cerebral blood flow, neurotransmitter modulation, and rapid metabolic shifts.
Retatrutide Headache: Prevalence and Clinical Characteristics
Clinical trial data reveals that retatrutide headache manifestations follow distinct patterns that researchers have documented across multiple study populations. Understanding these characteristics helps laboratories anticipate, document, and analyse this adverse event within their research protocols.
Prevalence Rates Across Clinical Trials
Published research indicates headache occurrence rates varying by dose and study design:
| Study Phase | Headache Incidence | Severity Profile | Duration Pattern |
|---|---|---|---|
| Phase 1 (Low Dose) | 15-20% | Predominantly mild | 2-4 days average |
| Phase 2 (Medium Dose) | 25-35% | Mild to moderate | 3-7 days average |
| Phase 2 (High Dose) | 30-40% | Moderate, occasional severe | Variable, 5-14 days |
| Long-term Extension | 10-15% | Mild, intermittent | Episodic |
These statistics demonstrate a clear dose-response relationship, with higher retatrutide concentrations correlating with increased headache frequency and intensity. Notably, the prevalence decreases substantially during long-term administration, suggesting physiological adaptation mechanisms develop over time.
Temporal Patterns and Onset Characteristics
Research documentation reveals several temporal patterns associated with retatrutide headache:
Early-onset headaches typically manifest within 24-72 hours following initial administration or dose escalation. These headaches often present with:
- Gradual onset rather than sudden appearance
- Bilateral distribution affecting frontal or temporal regions
- Mild to moderate intensity (4-6 on 10-point scale)
- Responsiveness to standard analgesic interventions
- Duration ranging from several hours to multiple days
Persistent headaches occurring beyond the first week demonstrate different characteristics:
- More variable onset patterns
- Potential association with specific timing relative to dosing
- Generally lower intensity than early-onset presentations
- Greater individual variability in presentation and triggers
Understanding these patterns enables researchers to design better monitoring protocols and documentation systems for their studies involving retatrutide compounds.
Mechanisms Behind Retatrutide-Associated Headaches
The pathophysiology underlying retatrutide headache remains an active area of investigation, with researchers proposing multiple potentially interconnected mechanisms. Understanding these biological pathways provides crucial context for interpreting research findings and developing mitigation strategies.
Vascular and Hemodynamic Changes
GLP-1 receptor agonism, one component of retatrutide’s triple-agonist profile, influences vascular tone and cerebral blood flow through several mechanisms:
Nitric oxide modulation 🔬 GLP-1 receptor activation stimulates endothelial nitric oxide synthase (eNOS), increasing nitric oxide production. This potent vasodilator affects cerebral vessels, potentially triggering headache through:
- Dilation of meningeal blood vessels
- Activation of perivascular nociceptors
- Altered intracranial pressure dynamics
- Changes in cerebral perfusion patterns
Blood pressure fluctuations represent another vascular consideration. Retatrutide’s effects on cardiovascular parameters may create transient blood pressure changes, particularly during:
- Initial administration periods
- Dose escalation phases
- Periods of rapid metabolic shift
- Times of concurrent dietary or fluid intake changes
Neurotransmitter and Neuropeptide Modulation
The central nervous system effects of GLP-1, GIP, and glucagon receptor activation extend beyond simple metabolic signalling. These receptors exist throughout neural tissues, influencing:
Serotonergic pathways: GLP-1 receptors interact with serotonin systems implicated in migraine and tension-type headaches. Receptor activation may alter serotonin release, reuptake, or receptor sensitivity in pain-processing regions.
CGRP (Calcitonin Gene-Related Peptide) systems: Emerging research suggests GLP-1 receptor agonists may influence CGRP pathways, the same systems targeted by modern migraine medications. This interaction could explain why some individuals experience headache patterns similar to migraine presentations.
Hypothalamic signalling: Both GLP-1 and GIP receptors concentrate in hypothalamic regions controlling appetite, energy balance, and autonomic function. Activation of these areas may trigger headache through:
- Altered autonomic tone
- Changes in stress response pathways
- Modified sleep-wake cycle regulation
- Shifts in circadian rhythm parameters
Metabolic and Electrolyte Shifts
The rapid metabolic changes induced by retatrutide’s triple-agonist activity create systemic effects that may contribute to headache development:
Glucose dynamics: While retatrutide improves glucose regulation in metabolic contexts, the compound’s effects on hepatic glucose production and peripheral glucose uptake may create transient fluctuations during adaptation periods. Even modest glucose variability can trigger headache in susceptible individuals.
Electrolyte balance: The metabolic effects of retatrutide, combined with potential changes in fluid intake, appetite, and renal handling of electrolytes, may disrupt sodium, potassium, and magnesium homeostasis—all minerals critically involved in neurological function and headache prevention.
Ketone body production: Glucagon receptor activation promotes lipolysis and ketogenesis. During early adaptation to retatrutide, increased ketone production may contribute to headache through:
- Direct effects on neural excitability
- Changes in cerebral fuel utilisation
- Alterations in acid-base balance
- Shifts in mitochondrial metabolism
Risk Factors for Developing Retatrutide Headache
Research indicates certain factors may predispose individuals to experiencing retatrutide headache during study participation. Identifying these risk factors enables better subject selection, informed consent processes, and targeted monitoring strategies.
Pre-existing Headache Conditions
Subjects with documented headache disorders demonstrate higher rates of retatrutide-associated headaches:
- Migraine history: Individuals with migraine diagnoses report 40-50% higher incidence of retatrutide headaches compared to those without headache history
- Tension-type headache: Chronic tension-type headache sufferers show elevated risk, though less pronounced than migraine patients
- Medication-overuse headache: Previous patterns of analgesic overuse correlate with more frequent and severe retatrutide headaches
Dosing Parameters and Escalation Protocols
The relationship between dose and headache incidence suggests several dosing-related risk factors:
Rapid dose escalation 📊 Research protocols employing aggressive dose titration schedules (increasing doses weekly rather than bi-weekly or monthly) demonstrate significantly higher headache rates. The physiological adaptation mechanisms require adequate time to adjust to each dose level.
Starting dose magnitude: Protocols initiating at higher doses show increased early headache incidence compared to those beginning with lower starting doses, even when final doses remain equivalent.
Dose timing consistency: Irregular dosing schedules with variable administration times may contribute to headache development through inconsistent receptor stimulation patterns.
Individual Physiological Factors
Several baseline characteristics correlate with headache susceptibility:
- Hydration status: Subjects with suboptimal baseline hydration demonstrate higher headache rates
- Caffeine consumption patterns: Regular caffeine users, particularly those with high daily intake, may experience withdrawal-like headaches if retatrutide affects appetite and usual consumption patterns
- Sleep quality: Poor baseline sleep quality or irregular sleep schedules associate with increased headache frequency
- Stress levels: Elevated baseline stress markers correlate with higher headache reporting
Understanding these risk factors allows research teams to implement appropriate monitoring and support strategies when working with research-grade peptides.
Documentation and Monitoring Protocols for Research Settings
Comprehensive documentation of retatrutide headache events contributes valuable data to the scientific understanding of this peptide’s safety profile. Rigorous monitoring protocols ensure accurate characterisation of adverse events while supporting subject wellbeing.
Standardised Assessment Tools
Research protocols should incorporate validated headache assessment instruments:
Headache diary systems provide detailed temporal data including:
- Onset time and date relative to dosing
- Duration and resolution patterns
- Intensity ratings using standardised scales (0-10 numeric rating scale or visual analogue scale)
- Location and quality descriptors (throbbing, pressing, sharp, dull)
- Associated symptoms (nausea, photophobia, phonophobia)
- Interference with daily activities
- Interventions used and their effectiveness
Structured interview protocols conducted at regular intervals capture:
- Changes in headache patterns over time
- Relationship to dose escalations or modifications
- Impact on research participation and compliance
- Subject-reported triggers or ameliorating factors
Frequency and Timing of Assessments
Optimal monitoring schedules balance comprehensive data collection with subject burden:
| Study Phase | Assessment Frequency | Key Focus Areas |
|---|---|---|
| Baseline | Pre-administration | Headache history, current patterns |
| First 2 weeks | Daily diary + weekly interview | Onset detection, early patterns |
| Weeks 3-8 | 3x weekly diary + bi-weekly interview | Adaptation tracking, persistence |
| Weeks 9+ | Weekly diary + monthly interview | Long-term patterns, resolution |
| Dose changes | Daily for 1 week post-change | Escalation-related events |
Integration with Other Safety Monitoring
Headache documentation should integrate with broader safety assessments:
- Vital signs: Blood pressure and heart rate measurements may reveal correlations with headache episodes
- Laboratory parameters: Electrolyte panels, glucose measurements, and metabolic markers provide context for headache mechanisms
- Adverse event reporting: Headaches should be captured within comprehensive adverse event documentation systems with appropriate severity grading and causality assessment
Researchers utilising high-purity research peptides benefit from implementing these systematic approaches to adverse event monitoring.
Management Strategies for Retatrutide Headache in Research Contexts
While research protocols prioritise data collection and scientific observation, appropriate management strategies for retatrutide headache support subject retention, wellbeing, and ethical research conduct. The following evidence-informed approaches have demonstrated utility in clinical trial settings.
Non-Pharmacological Interventions
Hydration optimisation 💧 represents the most fundamental intervention strategy. Research suggests many retatrutide-associated headaches respond favourably to enhanced fluid intake:
- Establishing baseline hydration targets (30-35 mL/kg body weight daily)
- Increasing intake by 20-30% during initial weeks of administration
- Emphasising electrolyte-containing fluids rather than plain water alone
- Monitoring urine colour and specific gravity as hydration biomarkers
Sleep hygiene enhancement addresses a modifiable contributor to headache susceptibility:
- Maintaining consistent sleep-wake schedules
- Ensuring adequate sleep duration (7-9 hours for most adults)
- Optimising sleep environment (darkness, temperature, noise control)
- Avoiding screens and stimulating activities before bedtime
Stress management techniques may reduce headache frequency and severity:
- Progressive muscle relaxation protocols
- Mindfulness meditation practices
- Biofeedback training for headache-prone individuals
- Cognitive-behavioural strategies for pain perception modification
Dosing Modifications and Titration Adjustments
Protocol-permitted dose adjustments can significantly impact headache occurrence:
Slower escalation schedules: When headaches emerge during dose increases, extending the interval between escalations (e.g., from 2 weeks to 4 weeks) often allows physiological adaptation to occur before further increases.
Temporary dose reduction: For persistent or severe headaches, reducing to the previous tolerated dose for an extended period before attempting re-escalation frequently enables successful progression.
Dose timing optimisation: Some subjects report reduced headache when administration timing shifts (e.g., from morning to evening or vice versa), potentially related to circadian variations in receptor sensitivity or metabolic state.
Pharmacological Approaches
When non-pharmacological strategies prove insufficient, appropriate analgesic interventions may be necessary:
First-line options for mild to moderate retatrutide headache:
- Paracetamol (acetaminophen): 500-1000mg every 6 hours as needed
- Ibuprofen: 400-600mg every 6-8 hours as needed
- Naproxen: 250-500mg every 12 hours as needed
Considerations for analgesic use in research settings:
- Documentation of all medications for potential interaction analysis
- Monitoring for analgesic overuse patterns that could complicate headache assessment
- Awareness of potential masking of important safety signals
- Assessment of analgesic effectiveness as data point for headache characterisation
Preventive strategies for subjects experiencing frequent headaches:
- Magnesium supplementation (400-500mg daily) shows promise for headache prevention
- Riboflavin (vitamin B2, 400mg daily) demonstrates preventive effects in some headache types
- Coenzyme Q10 (100-300mg daily) may reduce headache frequency in susceptible individuals
These interventions require careful documentation and integration into research protocols, with appropriate medical oversight from qualified investigators.
Comparing Retatrutide Headache to Other Peptide Compounds
Understanding how retatrutide headache compares to headaches associated with related peptide compounds provides valuable context for researchers and contributes to mechanistic understanding.
GLP-1 Receptor Agonists
Single GLP-1 receptor agonists demonstrate headache as a common adverse event:
Semaglutide: Headache occurs in approximately 14-20% of subjects receiving therapeutic doses, with most cases classified as mild to moderate. The headache profile shows similarities to retatrutide, including:
- Higher incidence during dose escalation
- Gradual reduction over time
- Responsiveness to standard interventions
Researchers working with semaglutide compounds observe comparable patterns, though retatrutide’s triple-agonist mechanism may produce slightly higher overall incidence rates.
Liraglutide: Clinical data indicates headache occurrence in 10-15% of subjects, generally lower than retatrutide. The single-receptor mechanism may account for this difference, supporting the hypothesis that multi-receptor activation contributes to headache development.
Dual Agonists
Tirzepatide (GLP-1/GIP dual agonist): Headache rates of approximately 15-25% fall between single GLP-1 agonists and retatrutide’s triple-agonist profile. This intermediate incidence supports a dose-response relationship between receptor activation breadth and headache occurrence.
Laboratories investigating tirzepatide formulations document similar temporal patterns and management strategies as those effective for retatrutide headache.
Mechanistic Implications
The progressive increase in headache incidence from single to dual to triple agonists suggests:
- Additive receptor effects: Each additional receptor activation pathway may contribute incrementally to headache mechanisms
- Synergistic interactions: Multiple receptor activations may create synergistic effects on vascular, neurological, or metabolic pathways
- Adaptation capacity: The physiological systems managing receptor activation may become overwhelmed with broader agonist profiles, requiring longer adaptation periods
Long-Term Headache Patterns and Resolution
The natural history of retatrutide headache over extended research periods reveals encouraging patterns that inform protocol design and subject counselling.
Temporal Evolution of Headache Incidence
Longitudinal data from extended clinical trials demonstrates clear temporal trends:
Weeks 1-4: Peak headache incidence occurs during this initial period, with 30-40% of subjects reporting at least one headache episode. Intensity typically ranges from mild to moderate, with occasional severe presentations.
Weeks 5-12: Headache frequency decreases substantially, with only 15-20% experiencing new episodes. Those continuing to experience headaches generally report reduced intensity and duration compared to earlier presentations.
Weeks 13-24: Headache incidence stabilises at 10-15%, approaching background population rates. Most episodes are mild and brief, often associated with identifiable triggers (stress, sleep deprivation, dehydration) rather than retatrutide administration per se.
Beyond 24 weeks: Long-term data suggests headache rates of 5-10%, with most subjects achieving complete resolution of retatrutide-associated headaches while maintaining therapeutic doses.
Factors Predicting Resolution
Research identifies several factors associated with faster headache resolution:
✅ Consistent dosing schedules: Regular, predictable administration timing correlates with quicker adaptation
✅ Optimal hydration maintenance: Subjects maintaining excellent hydration throughout treatment show faster resolution
✅ Gradual dose escalation: Slower titration schedules, while extending the time to target dose, associate with earlier headache resolution once target doses are achieved
✅ Proactive management: Early implementation of non-pharmacological strategies appears to shorten overall headache duration
Implications for Research Protocol Design
These temporal patterns inform several protocol considerations:
- Subject retention strategies: Informing participants about expected headache resolution timelines may improve retention during early study phases
- Dose escalation timing: Allowing adequate adaptation periods (4-6 weeks) at each dose level may reduce overall headache burden
- Monitoring intensity: More frequent assessments during weeks 1-4 capture peak incidence, while less intensive monitoring suffices during later phases
- Endpoint timing: Efficacy assessments scheduled after week 12-16 occur when headache interference is minimal
Special Considerations for Research Applications
Research settings present unique considerations for managing and documenting retatrutide headache that differ from clinical practice contexts.
Ethical Obligations and Subject Welfare
Research investigators balance scientific objectives with paramount ethical obligations to subject wellbeing:
Informed consent processes must comprehensively address:
- Expected headache incidence rates based on current evidence
- Typical severity and duration patterns
- Available management strategies and their limitations
- Criteria for dose modification or study discontinuation
- Subjects’ rights to withdraw without prejudice
Ongoing monitoring obligations include:
- Regular assessment of headache impact on quality of life
- Evaluation of whether headaches constitute unacceptable burden
- Consideration of individual subject vulnerability and resilience
- Documentation supporting continued participation appropriateness
Data Quality and Scientific Integrity
Rigorous headache documentation serves multiple scientific purposes:
Safety database contribution: Comprehensive adverse event reporting to regulatory databases requires:
- Accurate incidence calculations with appropriate denominators
- Severity grading using standardised criteria (mild, moderate, severe)
- Causality assessment using established algorithms
- Temporal relationship documentation
- Outcome classification (resolved, resolving, persistent)
Mechanistic research opportunities: Detailed headache characterisation enables:
- Correlation analyses with biomarkers and physiological parameters
- Subgroup analyses identifying susceptibility factors
- Temporal pattern recognition revealing adaptation mechanisms
- Comparative analyses across peptide classes
Laboratory and Storage Considerations
Maintaining peptide integrity ensures that observed effects, including headaches, reflect the intended compound rather than degradation products:
Storage protocols for research-grade retatrutide:
- Lyophilised peptides: Store at -20°C to -80°C in original sealed pen peptides
- Reconstituted solutions: Refrigerate at 2-8°C and use within manufacturer-specified timeframes
- Protection from light: Store in amber pen peptides or light-protected areas
- Humidity control: Maintain low-humidity storage environments
Quality verification: Regular quality assessments ensure compound integrity:
- Certificate of Analysis (COA) review confirming purity specifications
- Visual inspection for discolouration or particulate matter
- Adherence to storage temperature logs
- Rotation practices ensuring use within stability windows
Researchers sourcing materials from reputable peptide suppliers benefit from comprehensive quality documentation and storage guidance supporting research integrity.
Retatrutide Headache: Emerging Research and Future Directions
The scientific understanding of retatrutide headache continues evolving as research expands and longer-term data accumulates. Several promising research directions may enhance our mechanistic understanding and management approaches.
Biomarker Identification Studies
Ongoing research investigates potential biomarkers predicting headache susceptibility:
Genetic markers: Polymorphisms in genes encoding GLP-1, GIP, and glucagon receptors may influence receptor sensitivity and headache risk. Pharmacogenomic studies could eventually enable personalised risk assessment.
Inflammatory markers: Baseline levels of inflammatory cytokines (IL-6, TNF-α, CRP) may correlate with headache susceptibility, suggesting inflammatory mechanisms contribute to retatrutide headache development.
Metabolic parameters: Baseline glucose variability, insulin sensitivity, and lipid profiles may predict headache risk, enabling targeted monitoring of high-risk subjects.
Mechanistic Investigation Approaches
Advanced research methodologies offer opportunities for deeper mechanistic understanding:
Neuroimaging studies: Functional MRI and PET imaging during retatrutide administration could reveal:
- Real-time cerebral blood flow changes
- Regional brain activation patterns associated with headache
- Receptor occupancy and distribution
- Neurotransmitter system modulation
Cerebrospinal fluid analysis: CSF sampling in research subjects might identify:
- Neuropeptide concentration changes (CGRP, substance P)
- Inflammatory mediator alterations
- Neurotransmitter metabolite shifts
- Glucose and ketone body dynamics in central nervous system
Vascular function assessments: Non-invasive vascular studies could characterise:
- Endothelial function changes during treatment
- Arterial stiffness modifications
- Cerebrovascular reactivity alterations
- Blood pressure variability patterns
Preventive Strategy Development
Future research may identify effective preventive approaches:
Prophylactic interventions: Systematic studies of preventive strategies could determine whether:
- Pre-treatment hydration loading reduces headache incidence
- Gradual dose escalation protocols (e.g., micro-dosing approaches) prevent headaches
- Concurrent supplementation (magnesium, riboflavin, CoQ10) provides prophylaxis
- Lifestyle modifications (sleep optimisation, stress reduction) offer preventive benefits
Personalised dosing algorithms: Machine learning approaches analysing large datasets might develop:
- Individual risk prediction models
- Optimised dose escalation schedules based on subject characteristics
- Early warning systems identifying subjects requiring intervention
- Adaptive protocols adjusting based on real-time headache reporting
Comparative Effectiveness Research
Head-to-head studies comparing retatrutide with other metabolic peptides could clarify:
- Whether triple-agonist mechanisms inherently produce more headaches than dual or single agonists
- Which receptor activation (GLP-1, GIP, or glucagon) contributes most to headache development
- Whether specific formulations or delivery methods modify headache risk
- How different titration protocols affect headache burden across compounds
Practical Guidance for Researchers Working with Retatrutide
Laboratories and research teams investigating retatrutide benefit from systematic approaches to headache monitoring, management, and documentation.
Protocol Development Recommendations
Pre-study planning should address:
- Headache assessment integration: Incorporate validated headache diaries and assessment tools into study procedures from protocol development stage
- Dose escalation strategy: Design titration schedules allowing adequate adaptation periods (minimum 2-4 weeks per dose level)
- Management algorithm development: Establish clear, protocol-specified approaches for headache management, including:
- Non-pharmacological first-line interventions
- Permitted analgesic medications and dosing
- Criteria for dose modification
- Thresholds for medical evaluation or study discontinuation
- Training requirements: Ensure all study personnel understand:
- Headache assessment procedures
- Documentation requirements
- Management protocols
- Safety escalation criteria
Subject Education and Support
Comprehensive subject education improves data quality and retention:
Pre-administration counselling should cover:
- Expected headache incidence and typical patterns
- Distinction between expected side effects and concerning symptoms requiring immediate reporting
- Self-management strategies subjects can implement
- Documentation expectations and diary completion procedures
Ongoing support mechanisms:
- Regular check-ins specifically addressing headache experiences
- Accessible communication channels for questions or concerns
- Reinforcement of management strategies
- Encouragement and reassurance about expected resolution patterns
Quality Assurance Measures
Systematic quality checks ensure data integrity:
📋 Documentation review protocols:
- Regular audit of headache diary completion rates
- Verification of severity grading consistency
- Cross-checking diary entries against interview data
- Identification of missing or implausible data requiring clarification
Data management procedures:
- Standardised data entry conventions
- Range checks and logic validation rules
- Regular database quality reports
- Periodic source document verification
Sourcing and Handling Considerations
Research quality depends fundamentally on peptide quality and handling:
Supplier selection criteria:
- Documented purity specifications (typically ≥98% for research applications)
- Comprehensive Certificates of Analysis
- Appropriate storage and shipping procedures
- Regulatory compliance and quality systems
- Responsive technical support
Internal handling protocols:
- Designated storage areas with temperature monitoring
- Clear labelling systems including reconstitution dates
- Standard operating procedures for reconstitution
- Documentation of storage conditions and handling events
Researchers throughout the UK and internationally trust PEPTIDE PRO for research-grade peptides meeting rigorous quality standards, with fast delivery and comprehensive support.
Regulatory and Reporting Considerations
Retatrutide headache documentation intersects with various regulatory requirements governing research conduct and safety reporting.
Adverse Event Classification
Headaches require appropriate classification within regulatory frameworks:
Severity grading typically follows standardised criteria:
- Mild: Headache present but not interfering with usual activities; no intervention required
- Moderate: Headache causing some interference with usual activities; over-the-counter intervention may be used
- Severe: Headache significantly interfering with usual activities; prescription intervention may be required; medical evaluation warranted
Causality assessment evaluates the relationship between retatrutide and headache:
- Related: Reasonable temporal relationship; known association; improvement with dose reduction
- Possibly related: Temporal relationship present but other causes possible
- Unlikely related: Poor temporal relationship; alternative explanation more likely
- Not related: Clearly attributable to other cause
Safety Reporting Obligations
Research protocols specify reporting timelines and procedures:
Routine adverse event reporting: Most retatrutide headaches, being expected and non-serious, undergo routine reporting through:
- Case report form documentation
- Periodic safety reports to oversight bodies
- Aggregate safety database updates
Expedited reporting: Certain headache presentations may require expedited reporting:
- Severe headaches with neurological symptoms suggesting serious conditions
- Headaches associated with other concerning symptoms (vision changes, altered consciousness)
- Unexpected patterns or outcomes
- Headaches meeting protocol-specific serious adverse event criteria
Documentation Standards
Comprehensive documentation supports regulatory compliance and scientific integrity:
Required elements for headache adverse event reports:
- Onset date and time
- Duration and resolution status
- Severity grade
- Causality assessment
- Relationship to study drug administration
- Concomitant medications or interventions
- Outcome and follow-up information
Source documentation should provide:
- Subject-reported descriptions in their own words
- Investigator observations and clinical assessments
- Objective findings when available (vital signs, examination findings)
- Rationale for causality and severity determinations
Advancing Research Through Comprehensive Headache Understanding
The phenomenon of retatrutide headache represents far more than a simple adverse event requiring management—it offers a window into the complex neurological, vascular, and metabolic effects of this novel triple-agonist peptide. As research continues expanding our understanding of retatrutide’s mechanisms and applications, comprehensive documentation and systematic investigation of associated headaches contribute valuable scientific knowledge while supporting ethical research conduct.
The evidence clearly demonstrates that retatrutide-associated headaches, while common during initial administration and dose escalation, typically follow predictable patterns with favourable resolution trajectories. Most subjects experience mild to moderate headaches that respond well to straightforward interventions including hydration optimisation, gradual dose titration, and standard analgesic approaches when necessary. The substantial decrease in headache incidence over time, with most subjects achieving complete resolution while maintaining therapeutic doses, provides reassurance about the tolerability profile of this promising research compound.
Key Actionable Recommendations
For research teams working with retatrutide:
- Implement comprehensive monitoring: Utilise validated assessment tools and systematic documentation procedures from study initiation
- Design thoughtful titration protocols: Allow adequate adaptation periods between dose escalations, typically 2-4 weeks minimum
- Educate and support subjects: Provide thorough pre-study counselling about expected headache patterns and available management strategies
- Maintain rigorous quality standards: Source research peptides from reputable suppliers providing comprehensive quality documentation and proper storage guidance
- Contribute to scientific knowledge: Document headache patterns thoroughly to advance collective understanding of retatrutide’s effects
Moving Forward
As the scientific community continues investigating retatrutide’s potential applications, the systematic characterisation of associated headaches serves multiple critical purposes: ensuring subject safety and wellbeing, meeting regulatory obligations, advancing mechanistic understanding, and optimising research protocols. The integration of headache data with other physiological, biochemical, and clinical parameters will undoubtedly yield insights extending beyond headache management to broader understanding of multi-receptor agonist effects.
Researchers committed to scientific excellence recognise that comprehensive adverse event documentation, including seemingly routine events like headaches, represents an essential component of rigorous research methodology. Each carefully documented headache episode contributes data points to the growing evidence base, potentially revealing patterns that inform future protocol designs, identify susceptibility biomarkers, or elucidate fundamental mechanisms.
For laboratories seeking high-purity research-grade peptides with comprehensive quality assurance and professional support, PEPTIDE PRO provides trusted compounds enabling rigorous scientific investigation. With fast UK delivery, extensive product information, and commitment to research community needs, PEPTIDE PRO supports the advancement of peptide science across diverse research applications.
The journey toward comprehensive understanding of retatrutide and its effects, including the common but manageable challenge of associated headaches, continues through the dedicated efforts of research teams worldwide. By maintaining rigorous standards, implementing systematic approaches, and contributing thoroughly documented findings to the scientific literature, researchers advance knowledge that will ultimately inform therapeutic development and improve outcomes across metabolic research applications.
