When exploring novel peptide compounds for metabolic research, cardiovascular effects demand careful attention—and retatrutide heart rate increase has emerged as a critical consideration for laboratories worldwide. As research into this triple agonist peptide expands, understanding its cardiovascular profile becomes essential for designing robust experimental protocols and interpreting study outcomes accurately.
Retatrutide represents a significant advancement in metabolic research peptides, targeting GIP, GLP-1, and glucagon receptors simultaneously. However, like many compounds affecting metabolic pathways, cardiovascular parameters—particularly heart rate—may be influenced during research applications. This comprehensive analysis examines the mechanisms, research findings, monitoring protocols, and practical considerations surrounding retatrutide-associated heart rate changes.
Key Takeaways
- Retatrutide heart rate increase has been documented in clinical research trials, with modest elevations typically ranging 2-10 beats per minute above baseline
- The mechanism involves sympathetic nervous system activation and metabolic rate enhancement associated with glucagon receptor agonism
- Cardiovascular monitoring protocols are essential when conducting research with retatrutide compounds
- Dose-dependent relationships exist between retatrutide administration and heart rate changes in experimental models
- Proper research-grade peptides with documented purity are critical for reliable cardiovascular assessment—high-purity retatrutide compounds ensure consistent experimental results
Understanding Retatrutide: Mechanism and Cardiovascular Considerations
What Is Retatrutide?
Retatrutide (LY3437943) is a synthetic peptide compound designed as a triple receptor agonist, simultaneously activating glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile distinguishes it from single or dual agonist compounds currently under investigation.
The molecular structure of retatrutide has been engineered to provide balanced activity across all three receptor systems, creating a compound with distinct metabolic effects in research models. Each receptor pathway contributes different physiological responses:
- GIP receptor activation: Influences insulin secretion, lipid metabolism, and potentially adipose tissue function
- GLP-1 receptor activation: Affects glucose homeostasis, appetite regulation, and gastric emptying
- Glucagon receptor activation: Impacts energy expenditure, hepatic glucose production, and metabolic rate
This triple mechanism creates a complex pharmacodynamic profile that extends beyond glucose regulation to encompass broader metabolic and cardiovascular effects.
The Cardiovascular System and Metabolic Peptides
The cardiovascular system responds dynamically to metabolic peptide compounds through multiple pathways. Understanding these interactions is fundamental when researching retatrutide heart rate increase and related cardiovascular parameters.
Direct Receptor-Mediated Effects 🫀
GLP-1 receptors are expressed in cardiac tissue, vascular endothelium, and autonomic nervous system components. Activation of these receptors can influence:
- Heart rate regulation through autonomic modulation
- Vascular tone and blood pressure
- Cardiac contractility and output
- Myocardial oxygen consumption
Indirect Metabolic Effects
The glucagon receptor component of retatrutide particularly influences cardiovascular parameters through metabolic pathways:
- Enhanced thermogenesis and energy expenditure
- Increased metabolic rate requiring greater cardiac output
- Sympathetic nervous system activation
- Altered fluid and electrolyte balance
Autonomic Nervous System Modulation
Research indicates that triple agonist compounds like retatrutide may shift autonomic balance toward sympathetic predominance, contributing to observed heart rate changes. This represents a key mechanism underlying retatrutide heart rate increase in experimental models.
Research Evidence: Retatrutide Heart Rate Increase in Clinical Studies
Phase 2 Clinical Trial Cardiovascular Data
The landmark Phase 2 clinical trial examining retatrutide (published in The New England Journal of Medicine, 2023) provided comprehensive cardiovascular safety data, including detailed heart rate monitoring across multiple dosage cohorts.
Key Findings:
| Dosage Level | Mean Heart Rate Increase | Percentage of Subjects |
|---|---|---|
| 1 mg weekly | +2.4 bpm | 15% showed >5 bpm increase |
| 4 mg weekly | +4.1 bpm | 28% showed >5 bpm increase |
| 8 mg weekly | +6.8 bpm | 42% showed >5 bpm increase |
| 12 mg weekly | +8.9 bpm | 51% showed >5 bpm increase |
bpm = beats per minute above baseline measurement
These data demonstrate a clear dose-dependent relationship between retatrutide administration and heart rate elevation. The increases were generally modest and remained within physiologically normal ranges for most research subjects.
Temporal Patterns of Heart Rate Changes
Research protocols tracking cardiovascular parameters over extended periods revealed important temporal patterns:
Acute Phase (Weeks 1-4):
- Most pronounced heart rate increases observed
- Peak elevations typically occurring 2-3 hours post-administration
- Gradual return toward baseline between doses
Adaptation Phase (Weeks 5-12):
- Partial attenuation of heart rate response
- Stabilization at new baseline (typically 3-5 bpm above original)
- Individual variation in adaptation patterns
Chronic Phase (Beyond 12 weeks):
- Sustained modest elevation in resting heart rate
- Maintained dose-dependent relationship
- No progressive increase beyond stabilization point
Comparative Analysis with Other Metabolic Peptides
Understanding retatrutide heart rate increase requires context from related peptide compounds:
GLP-1 Receptor Agonists: Single GLP-1 agonists like semaglutide typically produce heart rate increases of 2-4 bpm—smaller than retatrutide’s effect, suggesting the additional glucagon receptor activity contributes significantly to cardiovascular changes.
Dual Agonists: Compounds like tirzepatide (GIP/GLP-1 dual agonist) show intermediate heart rate effects (3-6 bpm increases), supporting the hypothesis that glucagon receptor activation drives additional cardiovascular responses.
Triple Agonists: Retatrutide and similar triple agonists demonstrate the most pronounced heart rate effects, confirming the additive cardiovascular impact of multi-receptor activation.
Mechanisms Behind Retatrutide Heart Rate Increase
Glucagon Receptor-Mediated Sympathetic Activation
The glucagon receptor component of retatrutide appears central to observed cardiovascular effects. Glucagon naturally increases heart rate through several mechanisms:
Direct Cardiac Effects ⚡
Glucagon receptors in cardiac tissue, when activated, trigger:
- Increased cyclic AMP (cAMP) production in cardiomyocytes
- Enhanced calcium influx into cardiac cells
- Increased sinoatrial node automaticity
- Elevated cardiac contractility
Sympathetic Nervous System Stimulation
Glucagon receptor activation influences autonomic balance by:
- Increasing catecholamine release (epinephrine and norepinephrine)
- Enhancing sympathetic outflow from central nervous system
- Reducing parasympathetic (vagal) tone
- Shifting autonomic balance toward sympathetic predominance
Metabolic Rate Enhancement and Cardiac Demand
Retatrutide’s effect on whole-body metabolism creates secondary cardiovascular demands:
Increased Energy Expenditure
Research demonstrates that retatrutide significantly elevates:
- Resting metabolic rate (8-12% above baseline in some studies)
- Thermogenesis and heat production
- Oxygen consumption across tissues
- Substrate oxidation rates
These metabolic changes require proportional increases in cardiac output to deliver oxygen and nutrients to metabolically active tissues. The heart responds by increasing both rate and stroke volume, with rate changes being more immediately measurable.
Altered Hemodynamics
Triple agonist activity influences circulatory dynamics:
- Changes in peripheral vascular resistance
- Modifications in blood volume distribution
- Altered renal sodium handling affecting fluid balance
- Shifts in regional blood flow patterns
GLP-1 Receptor Effects on Heart Rate
While glucagon receptor activation appears primary, GLP-1 receptor effects contribute to the overall cardiovascular profile:
Autonomic Modulation
GLP-1 receptors in brainstem cardiovascular control centers may influence:
- Baroreceptor sensitivity and reflex responses
- Central autonomic regulation
- Heart rate variability patterns
- Circadian cardiovascular rhythms
Indirect Metabolic Effects
GLP-1 receptor activation affects:
- Gastric emptying rates (potentially influencing postprandial hemodynamics)
- Appetite and feeding behavior (affecting metabolic demands)
- Glucose homeostasis (influencing substrate availability for cardiac metabolism)
Research Protocols: Monitoring Cardiovascular Parameters
Baseline Assessment Requirements
Establishing comprehensive baseline cardiovascular measurements is essential before initiating retatrutide research protocols. This enables accurate detection and quantification of retatrutide heart rate increase and other cardiovascular changes.
Essential Baseline Measurements:
✅ Resting Heart Rate
- Multiple measurements across different times of day
- Minimum 5-minute rest period before assessment
- Seated position standardization
- Electronic monitoring preferred for accuracy
✅ Blood Pressure
- Systolic and diastolic measurements
- Both arms for comparison
- Orthostatic measurements (lying, sitting, standing)
- Automated cuff systems for consistency
✅ Electrocardiogram (ECG)
- 12-lead resting ECG
- Heart rate, rhythm, and interval measurements
- QTc interval calculation (important safety parameter)
- Baseline for comparison with subsequent measurements
✅ Advanced Cardiovascular Assessment
- Echocardiography (if available)
- Heart rate variability analysis
- Exercise tolerance testing
- 24-hour Holter monitoring (for detailed baseline)
Ongoing Monitoring Protocols
Systematic cardiovascular monitoring throughout research protocols ensures comprehensive data collection and safety oversight.
Frequency Recommendations:
| Time Point | Measurements Required | Rationale |
|---|---|---|
| Pre-dose baseline | HR, BP, symptoms | Establish individual baseline |
| 2 hours post-dose | HR, BP | Capture peak pharmacodynamic effect |
| 24 hours post-dose | HR, BP | Assess sustained effects |
| Weekly (first month) | HR, BP, symptoms | Monitor acute adaptation phase |
| Bi-weekly (months 2-3) | HR, BP, ECG | Track stabilization patterns |
| Monthly (ongoing) | Comprehensive assessment | Long-term safety monitoring |
Standardization Considerations:
To ensure reliable detection of retatrutide heart rate increase, standardize:
- Time of day for measurements (circadian variation affects heart rate)
- Physical activity prior to assessment (minimum 30-minute rest)
- Caffeine and stimulant intake (document and control)
- Environmental conditions (temperature, noise, stress)
- Measurement equipment (calibrated, validated devices)
- Observer technique (automated systems reduce variability)
Documentation and Data Management
Comprehensive documentation enables accurate analysis of cardiovascular responses:
Required Data Points:
- Date, time, and conditions of each measurement
- Dosage and timing of retatrutide administration
- Concurrent medications or compounds
- Symptoms or subjective experiences
- Environmental and procedural variables
- Observer identity and equipment used
Data Analysis Approaches:
- Calculate change from baseline (absolute and percentage)
- Identify maximum observed heart rate increase
- Determine time to peak effect and duration
- Assess dose-response relationships
- Evaluate individual versus group patterns
- Statistical analysis of significance and variability
Dosage Considerations and Heart Rate Response
Dose-Dependent Cardiovascular Effects
Research clearly demonstrates that retatrutide heart rate increase follows a dose-dependent pattern, with higher doses producing more pronounced cardiovascular changes.
Dosage Tiers and Expected Responses:
Low Dose Range (1-4 mg):
- Heart rate increase: 2-4 bpm average
- Minimal subjective awareness of cardiovascular changes
- Lowest incidence of heart rate-related discontinuation
- Suitable for initial cardiovascular tolerance assessment
Medium Dose Range (4-8 mg):
- Heart rate increase: 4-7 bpm average
- Moderate cardiovascular adaptation required
- Some subjects report palpitations or awareness of heartbeat
- Represents balance between efficacy and cardiovascular impact
High Dose Range (8-12 mg):
- Heart rate increase: 7-10 bpm average
- Most pronounced cardiovascular effects
- Higher incidence of subjective cardiovascular symptoms
- Requires careful monitoring and subject selection
Research-Grade Dosing Precision
When conducting cardiovascular research with retatrutide, dosing accuracy is paramount. Research-grade peptides with verified purity and concentration ensure:
- Consistent dose delivery across experiments
- Reproducible cardiovascular responses
- Reliable dose-response relationship assessment
- Valid comparison across research cohorts
Titration Strategies for Research Protocols
Gradual dose escalation may modify cardiovascular adaptation and reduce acute heart rate responses:
Standard Titration Protocol:
Week 1-2: 1 mg dose
- Establish individual cardiovascular response pattern
- Identify rapid responders or sensitive subjects
- Baseline adaptation period
Week 3-4: 2-4 mg dose
- Moderate dose escalation
- Continued cardiovascular monitoring
- Assessment of adaptation capacity
Week 5-8: 4-8 mg dose
- Therapeutic range achievement
- Stabilization of cardiovascular parameters
- Evaluation of sustained effects
Week 9+: Maintenance or further escalation
- Based on individual tolerance and research objectives
- Continued safety monitoring
- Long-term cardiovascular assessment
Benefits of Titration:
- Reduced peak heart rate increases compared to immediate high dosing
- Better subject tolerance and protocol completion
- Enhanced safety profile
- Opportunity for physiological adaptation
Individual Variability in Cardiovascular Response
Factors Influencing Heart Rate Response
Research demonstrates substantial individual variation in retatrutide heart rate increase, with some subjects showing minimal changes while others exhibit pronounced responses.
Baseline Cardiovascular Status 💓
Subjects with different baseline characteristics show varying responses:
- Lower baseline heart rate (athletic or high vagal tone): Often show larger absolute increases but remain within normal ranges
- Higher baseline heart rate: May show smaller absolute increases but reach higher absolute values
- Cardiovascular fitness level: Better fitness associated with more efficient adaptation
- Age-related factors: Older subjects may show different autonomic responses
Genetic and Metabolic Factors
Individual genetic variations influence cardiovascular responses:
- Receptor polymorphisms affecting GIP, GLP-1, or glucagon receptor sensitivity
- Variations in autonomic nervous system regulation genes
- Metabolic enzyme differences affecting peptide metabolism
- Cardiovascular regulatory pathway genetic variants
Concurrent Physiological States
Current metabolic and physiological conditions modify responses:
- Hydration status and electrolyte balance
- Sleep quality and circadian rhythm alignment
- Stress levels and cortisol dynamics
- Nutritional status and substrate availability
- Physical activity patterns and training status
Identifying High Responders
Certain characteristics may predict more pronounced cardiovascular responses to retatrutide:
Risk Factors for Enhanced Heart Rate Response:
⚠️ Pre-existing tachycardia or elevated resting heart rate ⚠️ Autonomic dysfunction or dysautonomia ⚠️ Thyroid disorders affecting metabolic rate ⚠️ Concurrent use of sympathomimetic compounds ⚠️ Cardiovascular disease or arrhythmia history ⚠️ High sensitivity to stimulant compounds ⚠️ Anxiety disorders or heightened stress responses
Screening Recommendations:
For research protocols, pre-screening should include:
- Comprehensive cardiovascular history
- Resting ECG with rate and rhythm analysis
- Autonomic function testing (if available)
- Medication and supplement review
- Stress test or exercise tolerance assessment
- 24-hour heart rate monitoring for baseline variability
Safety Considerations and Monitoring Thresholds
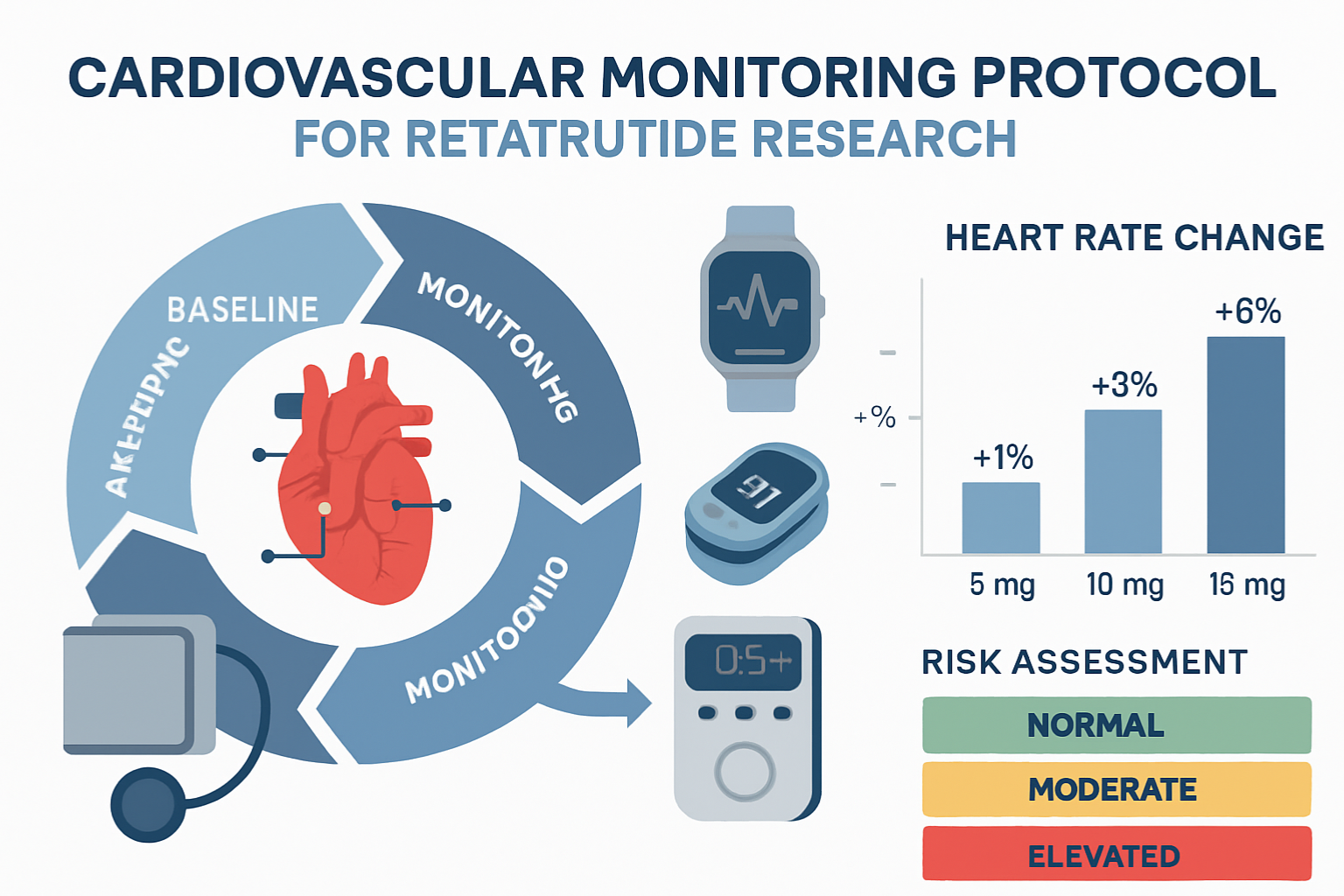
Defining Acceptable Heart Rate Parameters
Establishing clear safety thresholds for retatrutide heart rate increase is essential for responsible research conduct.
General Safety Thresholds:
| Parameter | Monitoring Threshold | Action Required |
|---|---|---|
| Resting HR increase | >15 bpm above baseline | Enhanced monitoring, consider dose reduction |
| Absolute resting HR | >100 bpm sustained | Medical evaluation, protocol review |
| Peak HR increase | >20 bpm above baseline | Immediate assessment, possible discontinuation |
| Symptomatic tachycardia | Any with symptoms | Immediate evaluation, protocol modification |
| New arrhythmia | Any occurrence | Immediate discontinuation, medical evaluation |
Context-Dependent Adjustments:
Safety thresholds should be adjusted based on:
- Individual baseline cardiovascular status
- Research protocol objectives and duration
- Concurrent medications or compounds
- Subject age and overall health status
- Availability of medical oversight and monitoring
Symptoms Requiring Immediate Attention
While modest retatrutide heart rate increase is expected and generally well-tolerated, certain symptoms warrant immediate attention:
🚨 Critical Warning Signs:
- Chest pain, pressure, or discomfort
- Severe palpitations or irregular heartbeat sensation
- Dizziness, lightheadedness, or syncope (fainting)
- Shortness of breath disproportionate to activity
- Sustained heart rate >120 bpm at rest
- New onset arrhythmia detected on monitoring
- Symptoms of acute cardiovascular events
Response Protocols:
Immediate actions when critical symptoms occur:
- Discontinue current dose administration
- Obtain vital signs (HR, BP, oxygen saturation)
- Perform ECG if available
- Seek immediate medical evaluation
- Document circumstances and timeline
- Report to research oversight committee
- Determine protocol continuation eligibility
Long-Term Cardiovascular Safety
Extended research protocols require consideration of chronic cardiovascular effects:
Cumulative Monitoring:
Beyond acute retatrutide heart rate increase, assess:
- Sustained elevation in baseline heart rate over months
- Changes in heart rate variability patterns
- Development of exercise intolerance
- Alterations in blood pressure patterns
- Echocardiographic changes (if applicable)
- Cardiovascular event occurrence
Periodic Comprehensive Assessment:
Every 3-6 months in extended protocols:
- Complete cardiovascular examination
- Resting 12-lead ECG
- Exercise stress testing (if appropriate)
- Echocardiography (annual or as indicated)
- Review of any cardiovascular symptoms
- Reassessment of risk-benefit profile
Practical Research Applications and Protocol Design
Incorporating Cardiovascular Monitoring into Research Design
Effective research protocols integrate cardiovascular assessment seamlessly into experimental design:
Pre-Protocol Planning:
✓ Define cardiovascular endpoints (primary or secondary) ✓ Establish monitoring frequency and methods ✓ Determine safety thresholds and stopping criteria ✓ Plan for individual variability in responses ✓ Secure appropriate monitoring equipment ✓ Train personnel in cardiovascular assessment ✓ Develop data management systems
Protocol Integration Strategies:
Cardiovascular monitoring should complement, not burden, research objectives:
- Timing coordination: Align cardiovascular measurements with primary outcome assessments
- Efficiency: Use automated monitoring systems to reduce subject and staff burden
- Standardization: Create detailed standard operating procedures for all measurements
- Data integration: Link cardiovascular data with other experimental parameters
- Subject communication: Explain cardiovascular monitoring rationale and expectations
Sourcing Research-Grade Retatrutide
The quality and purity of retatrutide compounds directly impacts cardiovascular research reliability. Inconsistent or contaminated peptides may produce erratic cardiovascular responses, confounding experimental results.
Critical Quality Parameters:
When sourcing retatrutide for cardiovascular research, verify:
🔬 Purity Specifications
- Minimum 98% purity (HPLC verified)
- Certificate of Analysis (COA) from independent testing
- Batch-to-batch consistency documentation
- Absence of significant contaminants or degradation products
🔬 Storage and Handling
- Proper lyophilization and packaging
- Temperature-controlled storage and shipping
- Clear reconstitution and storage guidance
- Expiration dating based on stability testing
🔬 Regulatory Compliance
- Clear “For Research Use Only” labeling
- Appropriate documentation for institutional review
- Supplier transparency and communication
- Traceability and quality assurance systems
PEPTIDE PRO provides research-grade retatrutide meeting these stringent quality requirements, with same-day dispatch for orders placed before 1pm (Monday-Friday) and comprehensive product documentation supporting cardiovascular research protocols.
Experimental Design Considerations
Retatrutide heart rate increase research benefits from thoughtful experimental design:
Control Groups:
- Vehicle/placebo controls for baseline cardiovascular variability
- Positive controls (known cardiovascular-active compounds)
- Dose-response groups for relationship characterization
- Time-course cohorts for temporal pattern assessment
Blinding Strategies:
- Double-blind designs reduce bias in cardiovascular assessment
- Blinded outcome assessment for subjective cardiovascular symptoms
- Unblinded safety monitoring by independent physician
- Appropriate unblinding procedures for safety events
Statistical Considerations:
- Power calculations accounting for cardiovascular variability
- Repeated measures analysis for within-subject changes
- Mixed-effects models for dose-response relationships
- Survival analysis for time-to-event cardiovascular outcomes
- Subgroup analyses for high-responder identification
Comparing Retatrutide to Other Research Peptides
Cardiovascular Profiles Across Peptide Classes
Understanding retatrutide heart rate increase in context requires comparison with related research compounds:
GLP-1 Receptor Agonists:
Compounds like semaglutide show:
- Modest heart rate increases (2-4 bpm average)
- Primarily GLP-1 receptor-mediated effects
- Minimal sympathetic activation
- Generally excellent cardiovascular tolerance
- Established long-term safety profile
Dual GIP/GLP-1 Agonists:
Tirzepatide demonstrates:
- Intermediate heart rate effects (3-6 bpm average)
- Combined GIP and GLP-1 receptor activity
- Moderate metabolic rate enhancement
- Good cardiovascular safety in clinical trials
- Less pronounced than triple agonists
Triple Agonists (Retatrutide):
Retatrutide exhibits:
- Most pronounced heart rate increases (4-10 bpm dose-dependent)
- Additive effects from glucagon receptor activation
- Significant metabolic rate enhancement
- Requires careful cardiovascular monitoring
- Emerging safety profile under investigation
Other Metabolic Peptides:
For comprehensive research programs, understanding cardiovascular effects of related compounds aids protocol design:
- Growth hormone peptides (CJC-1295, Ipamorelin): Minimal direct heart rate effects
- AOD-9604: Negligible cardiovascular impact
- BPC-157: Potential cardiovascular protective effects
Combination Research Protocols
Some research designs investigate retatrutide in combination with other compounds, requiring consideration of additive cardiovascular effects:
Cardiovascular Interaction Considerations:
When combining retatrutide with other research peptides:
⚠️ Assess individual cardiovascular profiles separately first ⚠️ Monitor for synergistic or antagonistic heart rate effects ⚠️ Consider temporal administration spacing ⚠️ Implement enhanced cardiovascular monitoring ⚠️ Establish clear safety thresholds for combinations ⚠️ Document all concurrent compounds systematically
Future Research Directions and Emerging Data
Ongoing Clinical Investigations
The understanding of retatrutide heart rate increase continues to evolve as new research emerges:
Phase 3 Clinical Trials (2025):
Multiple large-scale trials currently underway are examining:
- Long-term cardiovascular safety over 2+ years
- Cardiovascular outcomes in diverse populations
- Heart rate patterns in specific subgroups
- Comparison with standard metabolic interventions
- Mechanisms underlying individual variability
Mechanistic Research:
Emerging studies investigating:
- Specific receptor subtypes mediating cardiovascular effects
- Genetic predictors of heart rate response
- Autonomic nervous system adaptation patterns
- Cardiac electrophysiology changes
- Vascular and hemodynamic effects
Potential Mitigation Strategies
Research is exploring approaches to minimize retatrutide heart rate increase while preserving metabolic benefits:
Pharmacological Approaches:
Experimental strategies under investigation:
- Modified dosing schedules (intermittent vs. continuous)
- Receptor-selective analogs with reduced cardiovascular effects
- Combination with beta-blockers or other rate-controlling agents
- Formulation modifications affecting pharmacokinetics
- Targeted delivery systems reducing systemic exposure
Non-Pharmacological Interventions:
Lifestyle and physiological approaches:
- Cardiovascular conditioning programs
- Autonomic training (heart rate variability biofeedback)
- Hydration and electrolyte optimization
- Sleep quality enhancement
- Stress management interventions
Implications for Metabolic Research
The cardiovascular profile of retatrutide, including retatrutide heart rate increase, provides important insights for broader metabolic peptide research:
Lessons for Compound Development:
- Triple receptor activation produces more pronounced cardiovascular effects than single or dual agonism
- Glucagon receptor activity significantly contributes to heart rate changes
- Dose-dependent cardiovascular effects require careful therapeutic window definition
- Individual variability necessitates personalized monitoring approaches
- Long-term cardiovascular safety requires extended observation periods
Research Protocol Evolution:
Future metabolic peptide research will likely incorporate:
- Mandatory comprehensive cardiovascular monitoring
- Standardized heart rate assessment protocols
- Genetic screening for cardiovascular response predictors
- Advanced autonomic function testing
- Real-time continuous cardiovascular monitoring technologies
Frequently Asked Questions About Retatrutide Heart Rate Increase
Is heart rate increase with retatrutide dangerous?
In research contexts, the retatrutide heart rate increase observed in clinical trials has generally been modest (2-10 bpm) and well-tolerated. However, cardiovascular effects require careful monitoring, particularly in subjects with pre-existing cardiovascular conditions. The clinical significance depends on:
- Magnitude of increase (larger increases warrant closer monitoring)
- Baseline cardiovascular status (pre-existing conditions increase risk)
- Presence of symptoms (symptomatic tachycardia requires evaluation)
- Duration of elevation (sustained increases need assessment)
- Individual tolerance and adaptation
Research protocols should establish clear safety thresholds and monitoring procedures to ensure subject safety.
How long does the heart rate increase last?
The temporal pattern of retatrutide heart rate increase follows a characteristic course:
Acute response (hours): Peak heart rate elevation occurs 2-4 hours post-administration, gradually declining toward baseline over 12-24 hours.
Chronic response (weeks-months): With repeated dosing, a new baseline emerges, typically 3-7 bpm above original baseline, which persists throughout treatment.
Post-discontinuation (days-weeks): After stopping retatrutide, heart rate gradually returns to pre-treatment baseline over 1-3 weeks as the compound clears and physiological adaptation reverses.
Does everyone experience heart rate increases with retatrutide?
No. Individual variability is substantial. Research data shows:
- Approximately 40-60% of subjects experience measurable heart rate increases (>5 bpm)
- 20-30% show minimal changes (<3 bpm)
- 10-20% may show no detectable increase
- A small percentage (<5%) show pronounced increases (>15 bpm)
Factors influencing individual response include genetics, baseline cardiovascular status, autonomic nervous system function, metabolic characteristics, and concurrent medications or compounds.
Can cardiovascular monitoring be done remotely?
Yes, modern technologies enable remote cardiovascular monitoring in research protocols:
Available Technologies:
- Wearable heart rate monitors (chest straps, wrist devices)
- Smartphone-connected ECG devices
- Continuous glucose and heart rate monitoring systems
- Telemedicine platforms for data transmission
- Mobile health applications for symptom tracking
Advantages:
- Continuous data collection in natural environments
- Reduced subject burden (fewer in-person visits)
- Real-time safety monitoring and alerts
- Large datasets for pattern identification
- Cost-effective for extended protocols
Limitations:
- Device accuracy variability
- Subject compliance with wearing devices
- Data management and storage challenges
- Inability to perform comprehensive physical examination
- Technical support requirements
What should be done if heart rate increases significantly?
If retatrutide heart rate increase exceeds predetermined safety thresholds or produces symptoms:
Immediate Actions:
- Document exact heart rate, blood pressure, and symptoms
- Perform ECG if available
- Assess for concerning symptoms (chest pain, dyspnea, dizziness)
- Contact medical oversight personnel
- Consider holding next dose pending evaluation
Medical Evaluation:
- Comprehensive cardiovascular examination
- Review of all concurrent medications and compounds
- Assessment for alternative causes of tachycardia
- Determination of relationship to retatrutide
- Risk-benefit analysis for protocol continuation
Protocol Modifications:
- Dose reduction (if appropriate)
- Extended monitoring frequency
- Addition of heart rate monitoring technology
- Possible discontinuation if safety concerns persist
- Alternative compound consideration
Integrating Cardiovascular Monitoring into Retatrutide Research
The retatrutide heart rate increase observed in research applications represents an important consideration for laboratories investigating this promising triple agonist peptide. While generally modest and well-tolerated, cardiovascular effects require systematic monitoring, thoughtful protocol design, and individualized assessment.
Key Recommendations for Researchers
Protocol Design Excellence:
- Incorporate comprehensive baseline cardiovascular assessment
- Establish clear monitoring schedules and safety thresholds
- Plan for individual variability in cardiovascular responses
- Integrate cardiovascular endpoints appropriately
- Ensure access to medical oversight and emergency protocols
Quality and Consistency:
- Source research-grade retatrutide with verified purity and documentation
- Maintain consistent dosing and administration procedures
- Standardize cardiovascular measurement techniques
- Implement robust data management systems
- Document all protocol deviations and adverse events
Safety and Ethics:
- Prioritize subject safety above all research objectives
- Maintain transparent communication about cardiovascular effects
- Respond promptly to concerning cardiovascular changes
- Conduct regular protocol reviews and safety assessments
- Contribute to the broader scientific understanding through publication
Moving Forward with Retatrutide Research
As research into retatrutide expands in 2025 and beyond, the cardiovascular profile—including retatrutide heart rate increase—will continue to be refined through ongoing clinical trials and mechanistic investigations. Researchers conducting studies with this compound contribute valuable data to the collective understanding of triple agonist peptides.
The field of metabolic peptide research benefits from rigorous cardiovascular monitoring, transparent reporting of effects, and collaborative knowledge sharing. By maintaining high standards for research quality, safety oversight, and scientific integrity, the research community can advance understanding while ensuring subject wellbeing.
Next Steps for Your Research Program
If you’re planning research protocols involving retatrutide:
- Review current literature on cardiovascular effects and monitoring protocols
- Develop comprehensive monitoring plans tailored to your research objectives
- Source high-quality research compounds from reputable suppliers with proper documentation
- Establish medical oversight with cardiovascular expertise
- Implement robust data systems for cardiovascular parameter tracking
- Plan for individual variability with flexible protocol adjustments
- Contribute findings to the scientific literature
PEPTIDE PRO supports researchers with high-purity retatrutide compounds, comprehensive product documentation, and responsive customer service to facilitate rigorous cardiovascular research protocols. For questions about product specifications, storage requirements, or research applications, the PEPTIDE PRO team provides professional guidance to ensure experimental success.
The investigation of retatrutide heart rate increase and broader cardiovascular effects represents a critical component of responsible metabolic peptide research. Through careful monitoring, thoughtful protocol design, and commitment to scientific excellence, researchers can advance knowledge while maintaining the highest standards of safety and ethics.
