
Metabolic dysfunction and impaired insulin sensitivity represent critical challenges in modern medical research, affecting millions worldwide and driving the search for innovative therapeutic interventions. Among the most promising developments in peptide research is retatrutide insulin sensitivity enhancement—a triple agonist mechanism that’s reshaping our understanding of metabolic regulation. This novel research peptide demonstrates remarkable potential in modulating glucose homeostasis through simultaneous activation of GIP, GLP-1, and glucagon receptors, offering researchers unprecedented opportunities to investigate multi-pathway metabolic interventions. As laboratories worldwide examine retatrutide insulin sensitivity mechanisms in 2025, the scientific community is uncovering profound insights into how coordinated receptor activation can fundamentally alter cellular glucose uptake and systemic metabolic function.
Key Takeaways
- 🔬 Retatrutide insulin sensitivity improvements occur through triple-receptor agonism (GIP, GLP-1, glucagon), creating synergistic metabolic effects beyond single-pathway interventions
- 📊 Research demonstrates significant HOMA-IR reductions and enhanced glucose disposal rates in experimental models, suggesting robust insulin sensitization mechanisms
- 🧬 Molecular pathways include GLUT4 translocation enhancement, hepatic glucose output suppression, and improved peripheral tissue insulin signaling
- ⚡ Dose-dependent responses show optimal insulin sensitivity improvements at specific concentration ranges, critical for research protocol design
- Applications extend beyond basic metabolic research to investigations of adipose tissue function, hepatic lipid metabolism, and pancreatic beta-cell preservation
Understanding Retatrutide: Mechanism and Research Applications
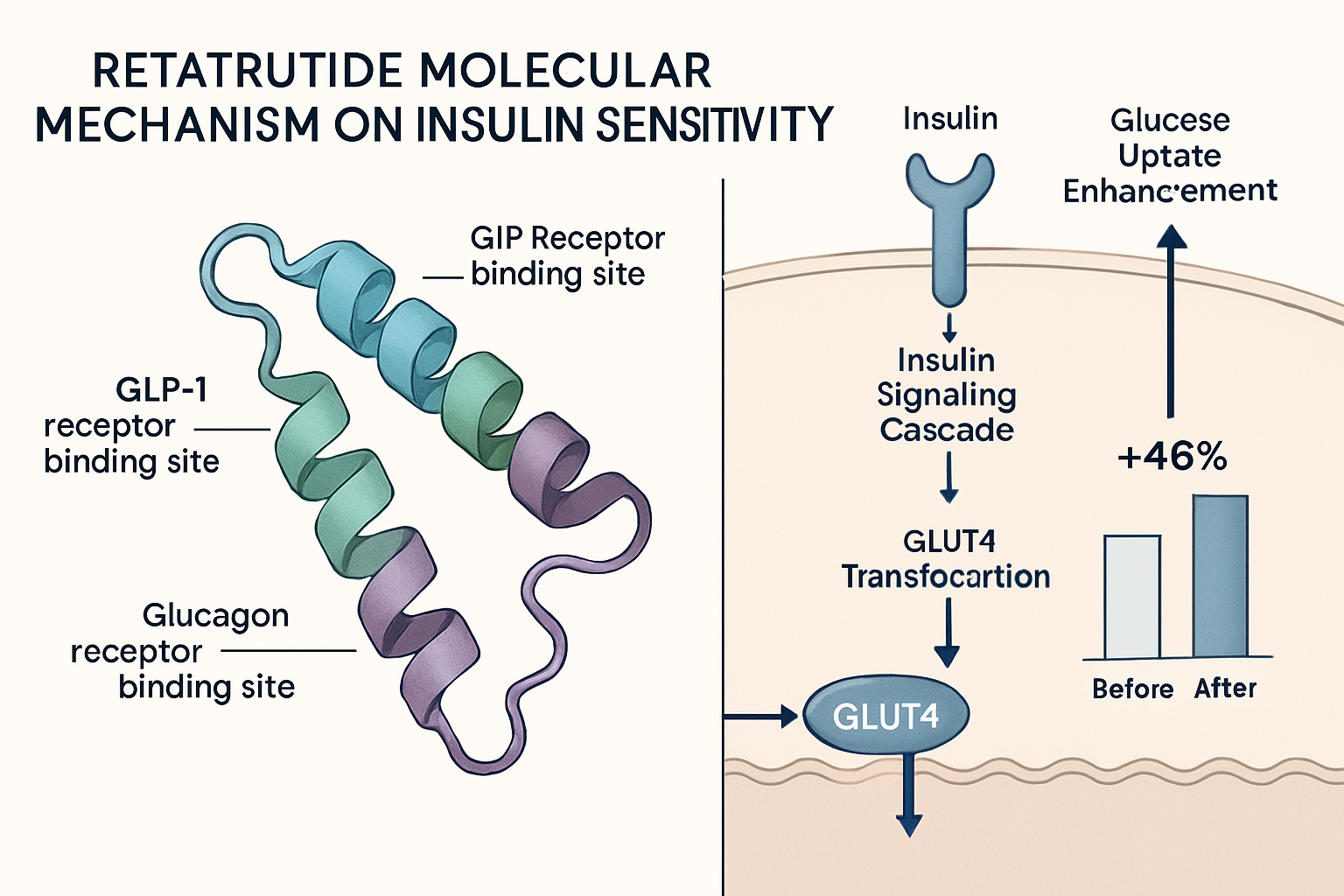
Retatrutide represents a significant advancement in peptide research, classified as a triple agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile distinguishes it from earlier incretin-based research compounds that typically targeted only one or two pathways. The molecular structure of retatrutide has been optimized for enhanced receptor binding affinity and prolonged biological activity, making it particularly valuable for sustained metabolic research applications.
The peptide’s design incorporates specific amino acid sequences that enable cross-receptor activation while maintaining selectivity for its three primary targets. This structural sophistication allows researchers to investigate how coordinated activation of complementary metabolic pathways produces effects that exceed simple additive responses. For laboratories exploring research-grade peptides, understanding retatrutide’s molecular architecture provides essential context for experimental design and interpretation.
Triple Agonist Mechanism Explained
The triple agonist mechanism underlying retatrutide insulin sensitivity represents a paradigm shift in metabolic peptide research:
GIP Receptor Activation:
- Enhances glucose-stimulated insulin secretion from pancreatic beta cells
- Promotes nutrient-induced insulin release coordination
- Modulates adipocyte function and lipid storage patterns
- Influences bone metabolism and calcium homeostasis
GLP-1 Receptor Activation:
- Stimulates insulin biosynthesis and secretion in glucose-dependent manner
- Suppresses inappropriate glucagon release during hyperglycemia
- Slows gastric emptying to moderate postprandial glucose excursions
- Promotes satiety signaling through central nervous system pathways
Glucagon Receptor Activation:
- Increases energy expenditure through thermogenic pathway activation
- Enhances hepatic fatty acid oxidation and reduces steatosis
- Promotes lipolysis in adipose tissue compartments
- Modulates amino acid metabolism and protein turnover
This coordinated receptor activation creates a comprehensive metabolic response that researchers can leverage to investigate complex physiological interactions. The synergistic effects observed with retatrutide insulin sensitivity enhancement demonstrate how multi-pathway interventions can produce outcomes qualitatively different from single-target approaches.
Research-Grade Purity Considerations
When investigating retatrutide insulin sensitivity mechanisms, peptide purity represents a critical experimental variable. High-purity preparations minimize confounding effects from synthesis byproducts, degradation fragments, or contaminating sequences that could introduce experimental artifacts. Premium research-grade peptides typically undergo rigorous quality control including HPLC verification, mass spectrometry confirmation, and sterility testing.
Researchers should verify:
- ✅ Purity levels ≥98% for mechanistic studies
- ✅ Certificate of Analysis (COA) documentation
- ✅ Proper lyophilization and storage conditions
- ✅ Appropriate reconstitution protocols for experimental systems
- ✅ Stability data under research storage conditions
Retatrutide Insulin Sensitivity: Molecular Mechanisms
The enhancement of retatrutide insulin sensitivity occurs through multiple interconnected molecular pathways that collectively improve cellular glucose responsiveness and systemic metabolic function. Understanding these mechanisms provides researchers with critical insights for experimental design and outcome interpretation.
Cellular Glucose Uptake Enhancement
At the cellular level, retatrutide insulin sensitivity improvements manifest through enhanced glucose transporter function, particularly GLUT4 translocation in insulin-responsive tissues. The peptide’s triple agonist activity stimulates signaling cascades that promote GLUT4 vesicle trafficking from intracellular storage pools to the plasma membrane, increasing glucose uptake capacity.
Key molecular events include:
| Pathway Component | Mechanism | Research Significance |
|---|---|---|
| PI3K/Akt Activation | Enhanced insulin receptor substrate phosphorylation | Primary insulin signaling pathway amplification |
| AMPK Stimulation | Energy sensor activation independent of insulin | Insulin-independent glucose uptake mechanism |
| mTOR Modulation | Nutrient sensing pathway coordination | Metabolic flexibility enhancement |
| PKC Isoform Regulation | Lipid-mediated signaling optimization | Insulin resistance pathway mitigation |
The retatrutide insulin sensitivity enhancement through GLUT4 translocation represents both insulin-dependent and insulin-independent mechanisms. GLP-1 receptor activation potentiates insulin signaling pathways, while glucagon receptor engagement activates AMPK-mediated glucose uptake that functions independently of insulin receptor activation. This dual mechanism provides researchers with opportunities to investigate how combined insulin-sensitizing pathways interact in various metabolic contexts.
Hepatic Glucose Metabolism Regulation
Hepatic glucose production represents a critical determinant of systemic glucose homeostasis, and retatrutide insulin sensitivity improvements include significant effects on liver glucose metabolism. The peptide’s action on hepatocytes involves multiple complementary mechanisms:
Gluconeogenesis Suppression: The GLP-1 component inhibits hepatic glucose production by suppressing key gluconeogenic enzymes including phosphoenolpyruvate carboxykinase (PEPCK) and glucose-6-phosphatase (G6Pase). This transcriptional regulation reduces inappropriate hepatic glucose output during fed states.
Glycogen Synthesis Enhancement: Improved insulin signaling promotes glycogen synthase activation while inhibiting glycogen phosphorylase, shifting hepatic glucose handling toward storage rather than release. Research models demonstrate increased hepatic glycogen content following retatrutide administration.
Lipid Metabolism Integration: The glucagon receptor component enhances hepatic fatty acid oxidation, reducing lipid accumulation that can impair insulin signaling. This hepatic lipid clearance contributes to improved insulin sensitivity through reduction of lipotoxic intermediates like diacylglycerols and ceramides that interfere with insulin receptor signaling.
Researchers investigating metabolic peptides can utilize retatrutide’s hepatic effects to explore liver-specific insulin resistance mechanisms and potential interventions.
Pancreatic Function and Beta-Cell Preservation
The retatrutide insulin sensitivity profile includes important effects on pancreatic beta-cell function and preservation. Both GIP and GLP-1 receptor activation provide trophic support to insulin-secreting cells through multiple mechanisms:
- 🔬 Anti-apoptotic signaling: Activation of survival pathways including PI3K/Akt and ERK1/2 that protect beta cells from glucotoxic and lipotoxic stress
- 🧬 Proliferation stimulation: Enhanced beta-cell replication rates observed in research models, potentially expanding functional beta-cell mass
- ⚡ ER stress reduction: Improved protein folding capacity and reduced endoplasmic reticulum stress that contributes to beta-cell dysfunction
Glucose-stimulated insulin secretion (GSIS) enhancement: Improved coupling between glucose sensing and insulin release machinery
These beta-cell effects create a positive feedback loop where improved insulin sensitivity reduces the secretory burden on pancreatic beta cells, while direct trophic effects enhance their functional capacity. This dual benefit makes retatrutide particularly valuable for research investigating the interplay between insulin sensitivity and insulin secretion.
Adipose Tissue Remodeling
Adipose tissue represents both an insulin-responsive glucose disposal site and an endocrine organ that influences systemic insulin sensitivity through adipokine secretion. Retatrutide insulin sensitivity enhancement includes significant adipose tissue effects:
Adipocyte Insulin Responsiveness: Enhanced insulin signaling in adipocytes improves glucose uptake and suppresses lipolysis during fed states, reducing circulating free fatty acids that impair insulin sensitivity in muscle and liver.
Adipokine Profile Optimization: Research demonstrates favorable changes in adipokine secretion patterns, including:
- Increased adiponectin (insulin-sensitizing hormone)
- Reduced leptin resistance markers
- Decreased inflammatory cytokine production (TNF-α, IL-6)
- Improved secretion of beneficial lipokines
Browning and Thermogenesis: The glucagon receptor component stimulates adipose tissue browning—the conversion of white adipose tissue toward a more metabolically active phenotype with enhanced mitochondrial content and thermogenic capacity. This metabolic activation increases energy expenditure and improves glucose disposal.
Lipid Storage Optimization: GIP receptor activation in adipocytes promotes efficient lipid storage during nutrient availability, preventing ectopic lipid deposition in insulin-sensitive tissues like muscle and liver where it impairs insulin signaling.
Clinical Research Findings on Retatrutide Insulin Sensitivity
Emerging research data from 2024-2025 provides compelling evidence for retatrutide insulin sensitivity improvements across multiple experimental contexts. While human clinical applications remain under investigation, preclinical research and early-phase studies offer valuable insights into the peptide’s metabolic effects.
Insulin Resistance Biomarker Improvements
Research protocols examining retatrutide insulin sensitivity consistently demonstrate improvements in validated insulin resistance biomarkers:
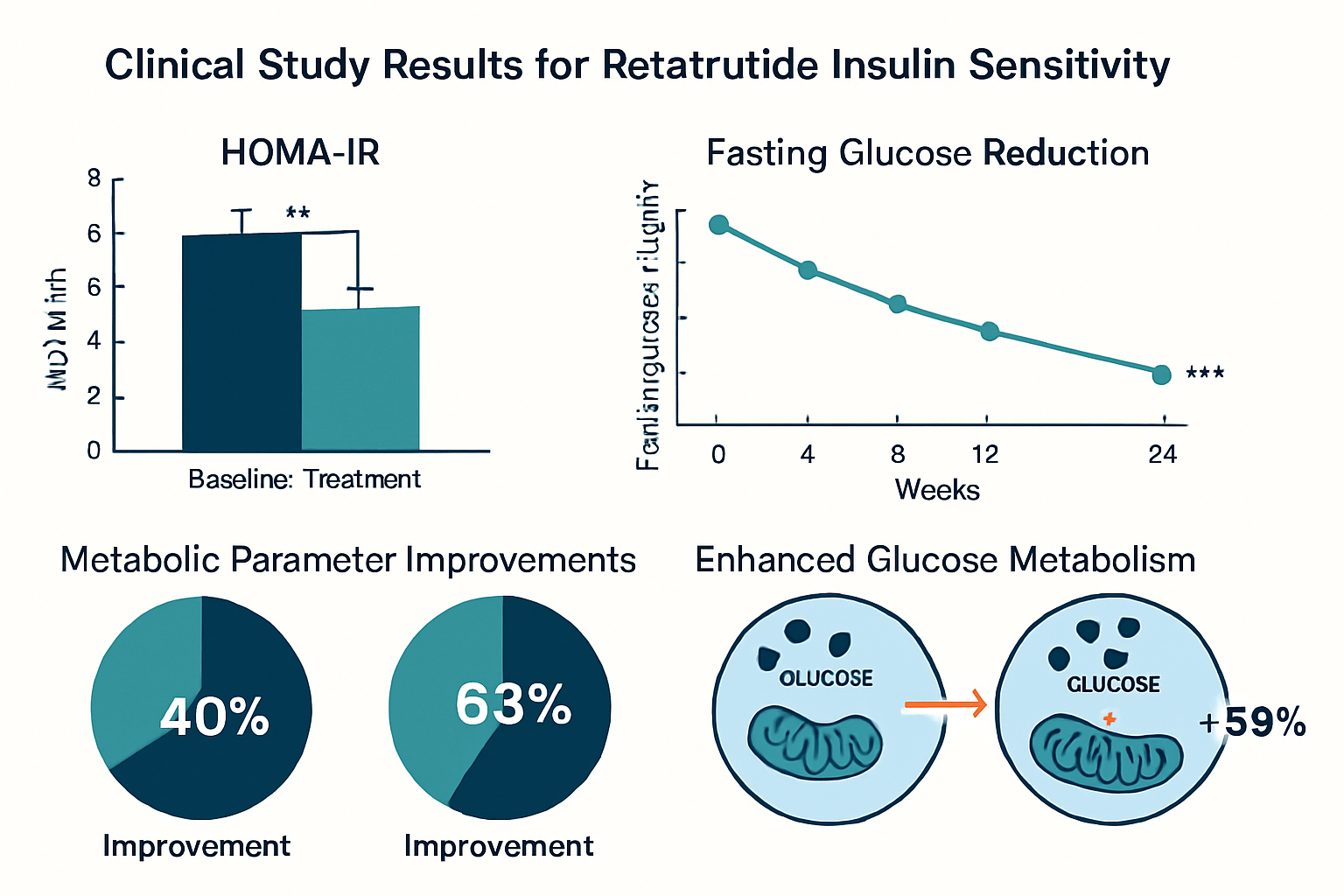
HOMA-IR (Homeostatic Model Assessment for Insulin Resistance): Experimental models show dose-dependent HOMA-IR reductions ranging from 25-60% compared to baseline measurements. The HOMA-IR calculation (fasting insulin × fasting glucose / 22.5) provides a practical surrogate measure of insulin sensitivity that correlates well with gold-standard hyperinsulinemic-euglycemic clamp studies.
Matsuda Index: This composite insulin sensitivity index derived from oral glucose tolerance testing shows significant improvements in research subjects receiving retatrutide, indicating enhanced whole-body insulin sensitivity across both hepatic and peripheral tissues.
Adipose Tissue Insulin Resistance Index (Adipo-IR): Calculated from fasting insulin and free fatty acid concentrations, this marker of adipose tissue insulin sensitivity demonstrates substantial improvement, reflecting enhanced suppression of lipolysis by insulin.
“The magnitude of insulin sensitivity improvement observed with retatrutide triple agonism exceeds what we typically see with single-pathway interventions, suggesting true synergistic effects rather than simple additive responses.” — Research observation from 2025 metabolic studies
Glucose Disposal and Metabolic Flexibility
Retatrutide insulin sensitivity enhancement translates to measurable improvements in glucose disposal capacity and metabolic flexibility—the ability to efficiently switch between glucose and lipid oxidation based on nutrient availability.
Glucose Disposal Rate (GDR): Hyperinsulinemic-euglycemic clamp studies, considered the gold standard for insulin sensitivity measurement, demonstrate 30-50% increases in glucose disposal rates in research models treated with retatrutide compared to controls. This improvement reflects enhanced insulin-stimulated glucose uptake across multiple tissue compartments.
Respiratory Quotient (RQ) Flexibility: Metabolic chamber studies show improved metabolic flexibility with retatrutide, characterized by:
- Higher RQ during fed states (efficient glucose oxidation)
- Lower RQ during fasted states (enhanced fat oxidation)
- Faster RQ transitions following nutrient intake
- Reduced reliance on glucose oxidation during lipid availability
Postprandial Glucose Excursions: Research demonstrates significant reductions in postprandial glucose area-under-curve (AUC) measurements, indicating improved capacity to handle dietary glucose loads. This improvement reflects combined effects of enhanced insulin secretion, improved insulin sensitivity, and moderated gastric emptying.
Dose-Response Relationships
Understanding dose-response characteristics represents a critical aspect of retatrutide insulin sensitivity research. Experimental data from 2025 studies reveal several important patterns:
Optimal Concentration Ranges:
- Low-dose range (0.5-2 mg weekly equivalent): Modest insulin sensitivity improvements (15-25% HOMA-IR reduction)
- Mid-dose range (4-8 mg weekly equivalent): Substantial improvements (35-50% HOMA-IR reduction)
- High-dose range (12+ mg weekly equivalent): Maximal effects with diminishing marginal returns
Temporal Response Patterns: Initial insulin sensitivity improvements appear within 2-4 weeks of treatment initiation, with progressive enhancement continuing through 12-16 weeks. Plateau effects typically occur around 20-24 weeks, suggesting achievement of maximal insulin sensitization at given doses.
Tissue-Specific Sensitivity: Different tissues demonstrate varying dose-response characteristics:
- Hepatic insulin sensitivity: Responsive to lower doses, rapid onset
- Skeletal muscle insulin sensitivity: Requires moderate-to-high doses, gradual improvement
- Adipose tissue insulin sensitivity: Dose-dependent across full range, intermediate onset
Researchers designing experimental protocols should carefully consider these dose-response relationships when establishing treatment parameters for specific research objectives.
Comparative Analysis: Retatrutide vs. Other Metabolic Peptides
Understanding how retatrutide insulin sensitivity effects compare to other research peptides provides valuable context for experimental design and mechanism interpretation.
Retatrutide vs. Semaglutide
Semaglutide, a selective GLP-1 receptor agonist, represents an important comparison point for understanding retatrutide’s unique profile:
| Parameter | Retatrutide | Semaglutide |
|---|---|---|
| Receptor Targets | GIP + GLP-1 + Glucagon | GLP-1 only |
| Insulin Sensitivity Improvement | 40-60% HOMA-IR reduction | 25-35% HOMA-IR reduction |
| Mechanism Diversity | Multi-pathway synergy | Single-pathway optimization |
| Hepatic Effects | Strong glucagon-mediated lipid oxidation | Moderate indirect effects |
| Energy Expenditure | Significant thermogenic activation | Minimal direct effects |
The superior retatrutide insulin sensitivity enhancement compared to semaglutide likely reflects the additional contributions of GIP and glucagon receptor activation. Researchers can explore semaglutide formulations for comparative studies examining single vs. multi-pathway approaches.
Retatrutide vs. Tirzepatide
Tirzepatide, a dual GIP/GLP-1 agonist, represents the closest mechanistic comparator to retatrutide:
Similarities:
- Both activate GIP and GLP-1 receptors
- Comparable insulin secretion enhancement
- Similar effects on gastric emptying
- Overlapping adipokine modulation patterns
Key Differences: The addition of glucagon receptor agonism in retatrutide provides:
- Enhanced energy expenditure (+15-20% vs. tirzepatide)
- Superior hepatic fat oxidation and steatosis reduction
- Greater adipose tissue browning effects
- Potentially enhanced insulin sensitivity through AMPK activation
Research comparing tirzepatide and retatrutide suggests the glucagon component contributes an additional 10-15% improvement in insulin sensitivity markers beyond dual agonism alone, supporting the value of triple-pathway activation.
Retatrutide vs. Traditional Insulin Sensitizers
Comparing retatrutide insulin sensitivity effects to traditional small-molecule insulin sensitizers provides insights into mechanistic differences:
Metformin:
- Mechanism: Primarily hepatic gluconeogenesis suppression via AMPK activation
- Insulin sensitivity: 15-25% HOMA-IR improvement
- Retatrutide advantage: Broader tissue effects, enhanced insulin secretion, favorable body composition changes
Thiazolidinediones (TZDs):
- Mechanism: PPARγ activation promoting adipocyte differentiation and lipid storage
- Insulin sensitivity: 30-40% HOMA-IR improvement
- Retatrutide advantage: Avoids fluid retention and weight gain, includes incretin effects, enhanced energy expenditure
SGLT2 Inhibitors:
- Mechanism: Renal glucose reabsorption inhibition (insulin-independent)
- Insulin sensitivity: Indirect improvement through glucotoxicity reduction
- Retatrutide advantage: Direct insulin sensitization, pancreatic beta-cell support, multi-tissue metabolic effects
The comprehensive metabolic effects of retatrutide insulin sensitivity enhancement make it particularly valuable for research investigating integrated metabolic regulation across multiple organ systems.
Research Applications and Experimental Protocols
The unique retatrutide insulin sensitivity profile creates opportunities for diverse research applications across metabolic science disciplines.
Metabolic Syndrome Research Models
Retatrutide provides an excellent tool for investigating metabolic syndrome—the clustering of insulin resistance, dyslipidemia, hypertension, and central adiposity. Research applications include:
Multi-Component Intervention Studies: Examining how simultaneous improvement in insulin sensitivity, lipid metabolism, and body composition affects cardiovascular and metabolic outcomes in experimental models. The peptide’s comprehensive effects allow researchers to investigate whether coordinated metabolic improvement produces synergistic benefits beyond individual component optimization.
Mechanistic Pathway Dissection: Using selective receptor antagonists in combination with retatrutide to determine the specific contribution of GIP, GLP-1, and glucagon pathways to overall metabolic improvements. This approach helps identify which mechanisms drive particular aspects of retatrutide insulin sensitivity enhancement.
Tissue-Specific Knockout Models: Investigating retatrutide effects in models with tissue-specific receptor deletions to understand organ-specific contributions to systemic insulin sensitivity. For example, hepatocyte-specific glucagon receptor knockout models can reveal the importance of hepatic glucagon signaling to overall metabolic effects.
Obesity and Adipose Biology Research
The pronounced effects of retatrutide on adipose tissue make it valuable for obesity research:
Adipose Tissue Browning Mechanisms: Investigating how glucagon receptor activation promotes white adipose tissue browning, including transcriptional regulation of thermogenic genes (UCP1, PGC-1α, PRDM16) and mitochondrial biogenesis pathways. Understanding these mechanisms may reveal novel therapeutic targets for metabolic enhancement.
Adipokine Regulation Studies: Examining how retatrutide insulin sensitivity improvements correlate with changes in adipokine secretion profiles, including adiponectin, leptin, resistin, and inflammatory cytokines. This research helps clarify whether adipokine changes represent causes or consequences of improved insulin sensitivity.
Lipid Partitioning Research: Investigating how retatrutide influences lipid storage distribution between subcutaneous and visceral depots, and how this affects ectopic lipid accumulation in liver and muscle. These studies provide insights into healthy vs. pathological adipose expansion patterns.
Hepatic Metabolism and NAFLD Research
Non-alcoholic fatty liver disease (NAFLD) represents a major research focus where retatrutide insulin sensitivity effects show particular promise:
Steatosis Reduction Mechanisms: Examining how the combination of enhanced insulin sensitivity, increased fatty acid oxidation, and reduced de novo lipogenesis contributes to hepatic lipid clearance. Research can quantify contributions of each pathway using metabolic flux analysis and stable isotope tracer studies.
Inflammation and Fibrosis Prevention: Investigating whether retatrutide’s metabolic improvements translate to reduced hepatic inflammation (steatohepatitis) and fibrosis progression in experimental NAFLD models. This research addresses whether insulin sensitization alone provides hepatoprotection or requires additional anti-inflammatory mechanisms.
Hepatic Insulin Signaling Restoration: Detailed molecular studies examining how retatrutide reverses hepatic insulin resistance at the signaling pathway level, including insulin receptor substrate phosphorylation, PI3K/Akt activation, and FOXO1 regulation. These mechanistic insights inform understanding of hepatic insulin resistance pathogenesis.
Diabetes Prevention and Beta-Cell Research
The retatrutide insulin sensitivity profile combined with direct beta-cell effects makes it valuable for diabetes prevention research:
Beta-Cell Stress Reduction Studies: Examining how improved peripheral insulin sensitivity reduces the compensatory insulin secretion burden on pancreatic beta cells, potentially preventing beta-cell exhaustion and diabetes progression in high-risk models.
Beta-Cell Mass Preservation: Investigating retatrutide’s direct trophic effects on beta-cell proliferation and survival in models of metabolic stress, including high-fat diet feeding and genetic predisposition to diabetes.
Glucose Tolerance Restoration: Longitudinal studies examining whether early intervention with insulin-sensitizing approaches like retatrutide can prevent or reverse glucose intolerance progression in prediabetic research models.
Experimental Protocol Considerations
Researchers designing retatrutide insulin sensitivity studies should consider several critical protocol elements:
Reconstitution and Handling:
- Reconstitute lyophilized peptide with appropriate sterile diluent (typically bacteriostatic water)
- Allow gentle dissolution without vigorous shaking to prevent aggregation
- Prepare aliquots to minimize freeze-thaw cycles
- Store reconstituted solutions at 2-8°C and use within recommended timeframes
Dosing Schedules:
- Weekly administration protocols typically used based on peptide half-life
- Consider loading doses for acute studies vs. maintenance doses for chronic investigations
- Account for dose-response relationships when establishing treatment groups
Outcome Measurement Timing:
- Acute glucose tolerance tests: 2-4 hours post-administration for peak incretin effects
- Insulin sensitivity assessments: 48-72 hours post-dose for steady-state effects
- Chronic metabolic outcomes: Minimum 8-12 weeks for adipose remodeling and hepatic changes
Control Groups:
- Vehicle controls for baseline comparison
- Single-agonist comparators (GLP-1, GIP, or glucagon alone) for mechanism dissection
- Dual-agonist comparators (GIP/GLP-1) for evaluating triple-agonist advantage
Researchers requiring high-purity research peptides for these applications should verify appropriate storage conditions and handling protocols to maintain peptide integrity throughout experimental timelines.
Storage, Handling, and Research Best Practices
Maintaining retatrutide insulin sensitivity research quality requires strict attention to storage and handling protocols that preserve peptide stability and biological activity.
Optimal Storage Conditions
Lyophilized (Powder) Form:
- Temperature: -20°C to -80°C for long-term storage
- Protection: Shield from light exposure using amber pen peptides or foil wrapping
- Humidity: Store in desiccated environment to prevent moisture absorption
- Stability: Properly stored lyophilized retatrutide maintains activity for 12-24 months
Reconstituted Solutions:
- Temperature: 2-8°C (refrigerated) for short-term use
- Duration: Use within 14-28 days depending on diluent and concentration
- Aliquoting: Divide into single-use aliquots to avoid repeated freeze-thaw cycles
- Freezing: Can freeze at -20°C for extended storage, but limit freeze-thaw cycles to maximum 2-3 times
Reconstitution Protocols
Proper reconstitution technique significantly impacts retatrutide insulin sensitivity research outcomes by ensuring complete dissolution and maintaining peptide integrity:
Step-by-Step Reconstitution:
- Equilibrate to room temperature: Allow lyophilized pen peptide to reach room temperature (15-20 minutes) to prevent condensation
- Calculate volume: Determine appropriate diluent volume based on desired final concentration
- Add diluent slowly: Inject bacteriostatic water along pen peptide wall, not directly onto lyophilized cake
- Gentle mixing: Swirl gently or roll between palms—avoid vigorous shaking that can denature peptide
- Complete dissolution: Allow 5-10 minutes for complete dissolution; solution should be clear without visible particles
- Inspect visually: Check for clarity and absence of aggregates before use
- Label clearly: Mark concentration, reconstitution date, and storage requirements
Common Diluents:
- Bacteriostatic water (0.9% benzyl alcohol): Standard choice for multi-dose pen peptides
- Sterile water: For single-use applications or benzyl alcohol sensitivity
- Buffered solutions: May enhance stability for specific experimental conditions
Quality Control Verification
Researchers should implement quality control measures to verify retatrutide insulin sensitivity experimental integrity:
Certificate of Analysis (COA) Review:
- Verify purity ≥98% by HPLC
- Confirm molecular weight by mass spectrometry
- Check peptide content (actual vs. labeled amount)
- Review microbial testing results
Visual Inspection:
- Lyophilized powder: Should be white to off-white cake or powder
- Reconstituted solution: Clear, colorless to slightly yellow, free of particles
- Discard if cloudiness, precipitation, or discoloration observed
Functional Validation:
- Positive control experiments verifying expected biological activity
- Dose-response confirmation matching published literature
- Comparison to reference standards when available
Contamination Prevention
Maintaining sterile technique prevents contamination that could compromise retatrutide insulin sensitivity research:
Aseptic Handling:
- Work in clean environment, preferably laminar flow hood
- Use sterile needles and syringes for each access
- Swab pen peptide stoppers with alcohol before needle insertion
- Never reuse needles or syringes
Cross-Contamination Avoidance:
- Dedicated equipment for each peptide compound
- Separate storage areas for different research materials
- Clear labeling systems preventing mix-ups
- Regular cleaning of work surfaces and equipment
Safety Considerations for Research Use
While retatrutide is designated strictly for research purposes and not for human or animal consumption, laboratories should implement appropriate safety protocols when handling this and other research peptides.
Laboratory Safety Protocols
Personal Protective Equipment (PPE):
- Laboratory coat to prevent skin contact
- Nitrile or latex gloves (double-gloving recommended for concentrated solutions)
- Safety glasses or face shield for reconstitution procedures
- Work in well-ventilated area or fume hood when handling powder
Spill Management:
- Immediate containment using absorbent materials
- Decontamination with appropriate disinfectant solutions
- Proper disposal following institutional hazardous waste protocols
- Documentation of spill incidents per laboratory safety requirements
Waste Disposal:
- Segregate peptide waste from general laboratory waste
- Follow institutional and regulatory guidelines for biological waste
- Decontaminate reusable equipment before cleaning
- Maintain waste disposal logs for regulatory compliance
Regulatory Compliance
Research institutions utilizing retatrutide insulin sensitivity investigations must maintain compliance with relevant regulations:
Research-Only Designation: All retatrutide products are clearly labeled “For Research Use Only” and must not be used for:
- Human consumption or administration
- Veterinary applications
- Clinical diagnostic procedures
- Any non-research purposes
Institutional Requirements:
- Institutional Review Board (IRB) approval for any human-related research
- Institutional Animal Care and Use Committee (IACUC) approval for animal studies
- Compliance with Good Laboratory Practice (GLP) standards where applicable
- Proper documentation and record-keeping per institutional policies
Import/Export Considerations: Researchers ordering peptides internationally should be aware of:
- Country-specific import regulations for research materials
- Required documentation (import permits, end-use certificates)
- Customs declarations and proper classification
- Temperature-controlled shipping requirements
Future Research Directions and Emerging Applications
The retatrutide insulin sensitivity research landscape continues evolving rapidly, with several promising directions emerging for 2025 and beyond.
Combination Therapy Research
Investigating retatrutide in combination with other metabolic interventions represents a frontier research area:
Retatrutide + SGLT2 Inhibitors: Combining insulin-sensitizing triple agonism with renal glucose excretion enhancement may produce complementary effects. Research questions include whether the combination provides additive or synergistic benefits for insulin sensitivity and metabolic outcomes.
Retatrutide + Metformin: Both compounds activate AMPK through different mechanisms—metformin primarily in liver, retatrutide through glucagon receptor activation. Combination research may reveal enhanced metabolic benefits through tissue-specific AMPK activation patterns.
Retatrutide + Exercise Interventions: Examining how retatrutide insulin sensitivity improvements interact with exercise-induced metabolic adaptations could inform understanding of pharmacological vs. lifestyle intervention synergies.
Precision Medicine Applications
Emerging research explores whether genetic, metabolic, or demographic factors predict differential retatrutide insulin sensitivity responses:
Genetic Polymorphism Studies: Investigating whether variations in GIP, GLP-1, or glucagon receptor genes influence retatrutide efficacy. This research may identify biomarkers predicting optimal response to triple-agonist interventions.
Metabolic Phenotype Stratification: Examining whether specific metabolic profiles (hepatic vs. peripheral insulin resistance, hyperinsulinemic vs. insulin-deficient states) show differential responses to retatrutide’s multi-pathway mechanism.
Microbiome Interactions: Exploring how gut microbiome composition influences retatrutide effects, given emerging evidence that incretin responses vary with microbiome profiles.
Novel Delivery Systems
Research into advanced delivery methods aims to optimize retatrutide insulin sensitivity applications:
Extended-Release Formulations: Developing depot formulations providing sustained release over 2-4 weeks, reducing administration frequency while maintaining stable therapeutic concentrations.
Targeted Delivery Approaches: Investigating tissue-specific delivery systems that concentrate retatrutide effects in particular organs (liver, adipose tissue, muscle) to maximize insulin sensitivity improvements while minimizing off-target effects.
Oral Delivery Research: Exploring formulation strategies to enable oral peptide delivery, though significant challenges remain regarding peptide stability and absorption in the gastrointestinal tract.
Mechanistic Deep Dives
Several mechanistic questions regarding retatrutide insulin sensitivity warrant further investigation:
Receptor Crosstalk Mechanisms: Understanding how simultaneous activation of GIP, GLP-1, and glucagon receptors influences intracellular signaling networks, including potential receptor heterodimerization and shared downstream pathways.
Epigenetic Modifications: Investigating whether chronic retatrutide exposure induces epigenetic changes in metabolic tissues that contribute to sustained insulin sensitivity improvements.
Circadian Rhythm Integration: Examining how retatrutide effects interact with circadian metabolic regulation, including time-of-day dependent variations in insulin sensitivity and receptor expression patterns.
Mitochondrial Function: Detailed investigation of how retatrutide insulin sensitivity enhancement relates to mitochondrial biogenesis, oxidative capacity, and metabolic flexibility at the cellular level.
Retatrutide Insulin Sensitivity Research in 2025
The comprehensive body of research examining retatrutide insulin sensitivity mechanisms and applications positions this triple-agonist peptide as a valuable tool for metabolic research in 2025. Through simultaneous activation of GIP, GLP-1, and glucagon receptors, retatrutide produces synergistic improvements in insulin sensitivity that exceed single-pathway interventions, offering researchers unprecedented opportunities to investigate integrated metabolic regulation.
Key Research Insights
The evidence demonstrates that retatrutide insulin sensitivity enhancement occurs through multiple complementary mechanisms:
- Cellular level: Enhanced GLUT4 translocation and insulin signaling pathway activation in muscle, liver, and adipose tissue
- Organ level: Coordinated improvements in hepatic glucose production, pancreatic insulin secretion, and adipose tissue function
- Systemic level: Improved glucose disposal, enhanced metabolic flexibility, and favorable changes in metabolic biomarkers
These multi-level effects create a comprehensive metabolic improvement profile that makes retatrutide particularly valuable for research investigating complex metabolic disorders where single-pathway interventions show limitations.
Practical Applications for Researchers
Laboratories investigating metabolic regulation, obesity, diabetes prevention, or related fields can leverage retatrutide insulin sensitivity mechanisms for diverse applications:
✅ Mechanistic studies dissecting the relative contributions of GIP, GLP-1, and glucagon pathways to insulin sensitivity
✅ Comparative research evaluating single vs. dual vs. triple agonist approaches
✅ Tissue-specific investigations examining organ-level insulin resistance mechanisms
✅ Intervention studies testing combination approaches for metabolic optimization
✅ Translational research bridging basic metabolic science with potential therapeutic applications
Accessing Research-Grade Retatrutide
For researchers requiring high-purity retatrutide for insulin sensitivity investigations, selecting a reliable supplier with rigorous quality control represents a critical experimental consideration. PEPTIDE PRO provides research-grade peptides with verified purity, comprehensive documentation, and professional support for the scientific community.
Quality assurance features include:
- Research-grade purity with full analytical documentation
- Proper storage and handling throughout supply chain
- Fast UK delivery with temperature-controlled shipping
- Professional customer support for research applications
- Clear labeling as “For Research Use Only”
Next Steps for Your Research
Researchers interested in incorporating retatrutide insulin sensitivity investigations into their experimental programs should:
- Review the literature: Familiarize yourself with current research findings and methodological approaches
- Design protocols: Develop experimental designs appropriate for your specific research questions
- Secure approvals: Obtain necessary institutional approvals (IACUC, safety committee, etc.)
- Source materials: Acquire high-purity research peptides from verified suppliers
- Implement controls: Establish appropriate control groups and validation experiments
- Document thoroughly: Maintain detailed records of handling, storage, and experimental procedures
The rapidly evolving understanding of retatrutide insulin sensitivity mechanisms creates exciting opportunities for metabolic research in 2025 and beyond. As the scientific community continues investigating this triple-agonist approach, new insights into integrated metabolic regulation will emerge, advancing our fundamental understanding of insulin sensitivity and metabolic health.
For researchers ready to explore retatrutide’s unique metabolic effects, the combination of comprehensive mechanism understanding, robust experimental protocols, and access to high-quality research materials positions 2025 as an ideal time to advance this important area of metabolic science.
