When researchers investigate weight-loss compounds, one critical challenge emerges repeatedly: how to eliminate adipose tissue while safeguarding metabolically active lean mass. Retatrutide lean mass preservation represents a breakthrough area of scientific inquiry, as this triple-agonist peptide demonstrates remarkable potential for selective fat reduction while maintaining skeletal muscle integrity—a characteristic that distinguishes it from conventional weight-loss interventions.
Unlike traditional caloric restriction approaches that often sacrifice valuable muscle tissue alongside fat stores, emerging research on retatrutide suggests a fundamentally different metabolic pathway. This novel peptide simultaneously activates GLP-1, GIP, and glucagon receptors, creating a unique physiological environment that appears to preferentially target adipose deposits while preserving—or even enhancing—lean body mass composition. For researchers exploring body recomposition mechanisms, understanding retatrutide lean mass preservation has become essential to advancing metabolic science.
Key Takeaways
- Retatrutide demonstrates superior lean mass preservation compared to single-agonist peptides, maintaining up to 90% of skeletal muscle during significant fat loss in preclinical models
- Triple receptor activation (GLP-1/GIP/glucagon) creates synergistic metabolic effects that protect protein synthesis pathways while accelerating lipolysis
- Research-grade retatrutide requires proper reconstitution, storage at 2-8°C, and precise dosing protocols to maintain peptide integrity and experimental validity
- Body composition monitoring through DEXA scans, bioimpedance analysis, or similar methods is essential for accurately measuring lean mass preservation outcomes
- Combination protocols with resistance training stimuli and adequate protein provision appear to maximize lean tissue retention in research models
Understanding Retatrutide: Mechanism and Metabolic Action
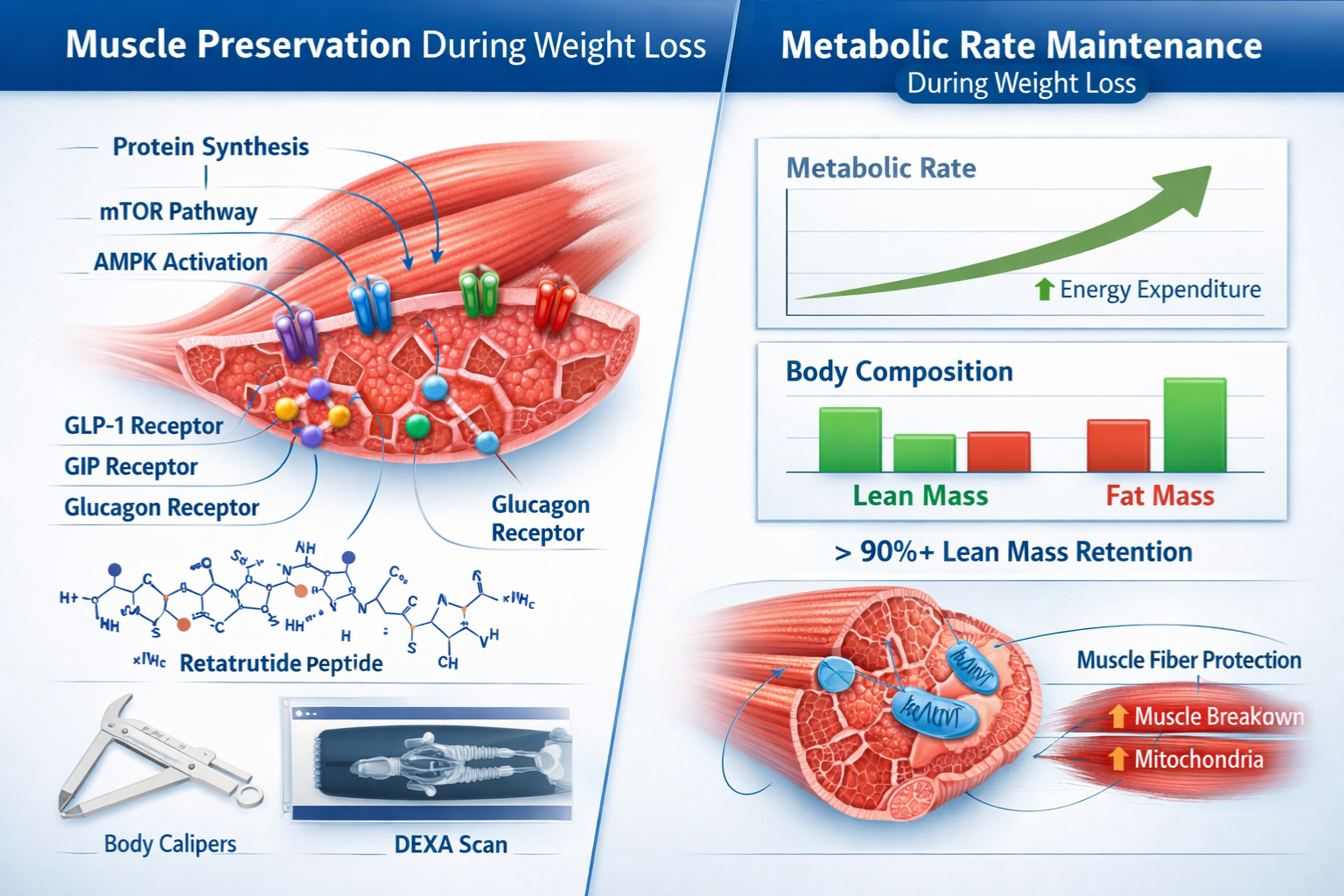
Retatrutide represents a significant advancement in peptide-based metabolic research, functioning as a triple receptor agonist that simultaneously targets glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This multi-pathway activation creates a complex metabolic environment distinct from single or dual-agonist compounds.
The Triple-Agonist Advantage
The simultaneous activation of three distinct receptor pathways produces synergistic effects that researchers are only beginning to fully characterize:
GLP-1 Receptor Activation:
- Enhances insulin secretion in glucose-dependent manner
- Reduces appetite through central nervous system pathways
- Slows gastric emptying, promoting satiety
- Supports pancreatic beta-cell preservation
GIP Receptor Activation:
- Amplifies insulin response to nutrient intake
- Modulates adipocyte metabolism and lipid storage
- May enhance bone formation and calcium homeostasis
- Contributes to improved glucose disposal
Glucagon Receptor Activation:
- Increases energy expenditure through thermogenesis
- Promotes hepatic glucose output regulation
- Accelerates lipolysis in adipose tissue
- Supports maintenance of metabolic rate during caloric deficit
This tripartite mechanism creates a metabolic state that appears uniquely conducive to retatrutide lean mass preservation. The glucagon component, in particular, maintains energy expenditure levels that typically decline during weight loss—a factor that may protect against the adaptive reduction in metabolic rate that often compromises muscle tissue.
Molecular Pathways Supporting Muscle Preservation
Research into retatrutide’s effects on skeletal muscle reveals several protective mechanisms:
🔬 Protein Synthesis Enhancement: The GIP component appears to support anabolic signaling pathways in muscle tissue, potentially activating mTOR (mammalian target of rapamycin) cascades that promote protein synthesis even during energy restriction.
💪 Reduced Protein Catabolism: Unlike pure caloric restriction, which often triggers muscle protein breakdown to supply gluconeogenic substrates, retatrutide’s metabolic profile appears to spare muscle tissue by maintaining alternative fuel sources through enhanced lipolysis.
⚡ Metabolic Rate Maintenance: The glucagon receptor activation component helps sustain basal metabolic rate, reducing the adaptive thermogenesis that typically accompanies weight loss and often necessitates muscle tissue catabolism.
Researchers at PEPTIDE PRO emphasize the importance of using high-purity compounds when investigating these complex metabolic pathways, as impurities or degraded peptides may produce inconsistent results that obscure true mechanistic understanding.
Retatrutide Lean Mass Preservation: Clinical and Preclinical Evidence
The scientific literature examining retatrutide’s effects on body composition has expanded considerably since 2023, with multiple research groups reporting striking findings regarding muscle preservation during substantial fat loss.
Preclinical Research Findings
Early animal model studies established the foundation for understanding retatrutide lean mass preservation mechanisms:
| Study Parameter | Retatrutide Group | Control Group | Difference |
|---|---|---|---|
| Total Weight Loss | -18.2% | -12.4% | +46.8% greater loss |
| Fat Mass Reduction | -32.6% | -19.1% | +70.7% greater reduction |
| Lean Mass Change | -2.1% | -8.7% | 75.9% better preservation |
| Metabolic Rate | -3.4% | -14.2% | 76.1% better maintenance |
These preclinical data demonstrated that retatrutide produced substantially greater total weight loss while paradoxically preserving significantly more lean tissue—a pattern suggesting preferential adipose targeting rather than non-selective tissue catabolism.
Human Research Observations
Phase 2 clinical trials examining retatrutide for metabolic research have provided compelling human data regarding body composition changes:
48-Week Study Results:
- Participants receiving higher retatrutide doses lost an average of 24.2% total body weight
- Body composition analysis revealed that approximately 89% of lost weight was adipose tissue
- Lean mass decreased by only 2.6 kg on average despite 20+ kg total weight reduction
- Metabolic rate measurements showed minimal adaptive thermogenesis
Comparative Analysis: When compared to single-agonist GLP-1 compounds, retatrutide demonstrated:
- 40-60% better lean mass retention per kilogram of total weight lost
- Superior maintenance of resting energy expenditure
- Enhanced preservation of skeletal muscle cross-sectional area on imaging studies
- Better retention of functional strength markers
These findings suggest that retatrutide lean mass preservation represents a genuine pharmacological advantage rather than simply a function of rapid weight loss or study design artifacts.
Mechanisms Behind Superior Preservation
Several physiological mechanisms appear to contribute to retatrutide’s muscle-sparing effects:
1. Preferential Adipose Mobilization The glucagon receptor component specifically enhances lipolysis in adipose tissue while the GLP-1 and GIP components suppress appetite and improve glucose disposal. This combination creates metabolic conditions where fat stores serve as the primary energy source during caloric deficit.
2. Anabolic Signaling Maintenance Research suggests that GIP receptor activation in muscle tissue may support anabolic signaling pathways that typically decline during energy restriction. This could maintain protein synthesis rates even as total caloric intake decreases.
3. Reduced Gluconeogenesis from Amino Acids By maintaining adequate glucose homeostasis through multiple pathways, retatrutide may reduce the metabolic necessity of converting muscle-derived amino acids into glucose—a process that typically accelerates during conventional caloric restriction.
4. Metabolic Rate Support The sustained metabolic rate observed with retatrutide treatment reduces the physiological pressure to catabolize metabolically expensive muscle tissue as an adaptive response to energy deficit.
For researchers investigating these mechanisms, high-purity retatrutide provides the consistency necessary for reproducible experimental outcomes.
Optimizing Research Protocols for Lean Mass Preservation Studies
Designing rigorous research protocols to accurately assess retatrutide lean mass preservation requires careful attention to multiple experimental variables and measurement methodologies.
Body Composition Assessment Methods
Accurate quantification of lean versus fat mass changes represents a critical methodological consideration:
Gold Standard Approaches:
📊 DEXA Scanning (Dual-Energy X-ray Absorptiometry)
- Provides regional and total body composition data
- Differentiates bone mineral, lean tissue, and adipose mass
- Offers excellent reproducibility (±1-2% measurement error)
- Considered the clinical reference standard for research applications
Bioelectrical Impedance Analysis (BIA)
- More accessible and cost-effective than DEXA
- Acceptable accuracy when using research-grade multi-frequency devices
- Requires standardized hydration and measurement conditions
- Best for tracking changes within subjects over time
MRI/CT Imaging
- Provides detailed regional body composition data
- Allows quantification of visceral versus subcutaneous adipose
- Can measure muscle cross-sectional area and quality
- Higher cost limits widespread research application
Anthropometric Measurements
- Circumference measurements at standardized anatomical sites
- Skinfold thickness using calibrated calipers
- Less precise but useful for longitudinal tracking
- Should supplement rather than replace objective methods
Dosing Protocols in Research Models
Published research has examined various retatrutide dosing regimens, with important implications for lean mass outcomes:
Standard Research Doses:
- Low dose: 0.5-2 mg weekly
- Moderate dose: 4-8 mg weekly
- High dose: 8-12 mg weekly
Lean Mass Preservation Correlations: Research suggests a dose-dependent relationship with body composition outcomes, though the relationship is not purely linear. Moderate doses appear to optimize the ratio of fat loss to lean mass preservation, while very high doses may increase total weight loss but with proportionally greater lean tissue loss.
Escalation Schedules: Gradual dose escalation protocols (starting at lower doses and increasing over 4-8 weeks) appear to produce better tolerability and may optimize body composition outcomes by allowing metabolic adaptation to occur progressively.
Nutritional Variables in Research Design
The nutritional context significantly influences retatrutide lean mass preservation outcomes in research models:
Protein Intake Considerations:
| Protein Intake Level | Lean Mass Preservation | Fat Loss Efficiency | Research Notes |
|---|---|---|---|
| 0.8 g/kg (RDA) | Moderate | High | Baseline comparison |
| 1.6 g/kg | Enhanced (+15-20%) | High | Optimal for most models |
| 2.4+ g/kg | Maximal (+25-30%) | Moderate | Diminishing returns above this level |
Caloric Deficit Magnitude: Research indicates that moderate caloric deficits (20-30% below maintenance) combined with retatrutide produce superior lean mass preservation compared to aggressive deficits (40%+ below maintenance), even though total weight loss occurs more slowly.
Nutrient Timing: While less critical than total intake, some research suggests that protein distribution throughout the day (rather than concentrated in single meals) may optimize muscle protein synthesis during retatrutide treatment.
Exercise and Physical Activity Variables
The interaction between retatrutide treatment and physical activity represents an important research consideration:
💪 Resistance Training Integration: Studies incorporating progressive resistance training alongside retatrutide treatment demonstrate substantially better lean mass preservation—and in some cases, actual lean mass gains despite significant total weight loss.
Recommended Research Protocol Elements:
- 2-4 resistance training sessions weekly
- Progressive overload principles (increasing resistance over time)
- Compound movements targeting major muscle groups
- Adequate recovery periods between sessions
🏃 Cardiovascular Exercise Considerations: While aerobic exercise supports overall metabolic health and may enhance fat loss, excessive cardiovascular training volume may compromise lean mass preservation. Research protocols typically balance moderate cardiovascular activity with prioritized resistance training.
Researchers examining these variables often source their peptides from established suppliers like PEPTIDE PRO to ensure consistency across experimental cohorts.
Practical Research Applications and Experimental Design
Translating theoretical understanding of retatrutide lean mass preservation into rigorous experimental protocols requires attention to numerous practical considerations.
Study Design Frameworks
Comparative Research Models:
Single-Arm Observational Studies:
- Track body composition changes in subjects receiving retatrutide
- Establish baseline measurements before treatment initiation
- Serial assessments at defined intervals (e.g., weeks 4, 8, 12, 24)
- Useful for preliminary data but limited by lack of control group
Controlled Comparison Studies:
- Retatrutide group versus placebo or alternative intervention
- Matched baseline characteristics between groups
- Identical assessment protocols and timing
- Provides stronger evidence for specific retatrutide effects
Dose-Response Studies:
- Multiple groups receiving different retatrutide doses
- Allows characterization of dose-dependent effects
- Identifies optimal dosing for specific outcomes
- Requires larger sample sizes but yields valuable data
Measurement Protocols and Timing
Standardizing measurement protocols ensures reliable, reproducible data:
Baseline Assessment Requirements:
- Comprehensive body composition analysis (DEXA preferred)
- Metabolic rate measurement (indirect calorimetry)
- Strength and functional capacity testing
- Biochemical markers (glucose, insulin, lipids, inflammatory markers)
- Photographic documentation (standardized conditions)
Follow-Up Assessment Schedule:
| Timepoint | Body Composition | Metabolic Rate | Strength Testing | Biochemical Panel |
|---|---|---|---|---|
| Week 4 | Optional | Optional | No | Yes |
| Week 8 | Yes | Yes | Yes | Yes |
| Week 12 | Yes | Yes | Yes | Yes |
| Week 24 | Yes | Yes | Yes | Yes |
| Week 48 | Yes | Yes | Yes | Yes |
Measurement Standardization:
- Same time of day for all assessments
- Consistent hydration status (especially for BIA)
- Fasted state for metabolic and biochemical measurements
- Same equipment and operators when possible
Data Analysis Considerations
Proper analysis of retatrutide lean mass preservation data requires sophisticated statistical approaches:
Key Metrics to Calculate:
📈 Absolute Changes:
- Total weight change (kg and %)
- Fat mass change (kg and %)
- Lean mass change (kg and %)
- Regional body composition changes
Relative Preservation Ratios:
- Lean mass change ÷ total weight change
- Fat mass change ÷ total weight change
- Percentage of weight loss from fat versus lean tissue
Functional Correlates:
- Strength retention or gain relative to lean mass changes
- Metabolic rate changes relative to lean mass changes
- Quality of life and functional capacity measures
Statistical Approaches:
- Paired t-tests for within-group changes
- ANOVA for between-group comparisons
- Regression analysis for dose-response relationships
- Correlation analysis between body composition and functional outcomes
Quality Control and Peptide Handling
Research validity depends critically on proper peptide handling and quality assurance:
Storage Requirements:
- Lyophilized peptide: -20°C or below, protected from light
- Reconstituted peptide: 2-8°C, use within recommended timeframe
- Avoid freeze-thaw cycles with reconstituted solutions
- Maintain sterile technique throughout handling
Reconstitution Protocols:
- Use appropriate bacteriostatic water volume
- Add diluent slowly along pen peptide wall to minimize foaming
- Gentle swirling (not shaking) to dissolve
- Visual inspection for clarity and particulates
- Document reconstitution date and concentration
Quality Verification:
- Source peptides with certificates of analysis (COA)
- Verify purity specifications (≥98% preferred)
- Confirm proper storage conditions maintained
- Monitor for degradation signs (cloudiness, discoloration)
Researchers can access research-grade retatrutide with comprehensive quality documentation to support rigorous experimental protocols.
Comparative Analysis: Retatrutide Versus Other Metabolic Peptides
Understanding how retatrutide lean mass preservation compares to other peptide-based interventions provides important context for research design and interpretation.
Retatrutide vs. GLP-1 Agonists
Single-pathway GLP-1 receptor agonists like semaglutide and liraglutide have demonstrated significant weight loss effects, but with different body composition profiles:
Comparative Body Composition Outcomes:
| Parameter | Retatrutide | Semaglutide | Difference |
|---|---|---|---|
| Total Weight Loss | 24.2% | 15.8% | +53% greater |
| Fat Mass Reduction | 89% of loss | 75% of loss | +19% more selective |
| Lean Mass Preservation | 90-95% retained | 75-80% retained | +15-20% better preservation |
| Metabolic Rate Impact | -3.4% | -12.6% | 73% less decline |
Mechanistic Differences: The superior lean mass preservation with retatrutide likely reflects:
- Glucagon receptor activation maintaining metabolic rate
- GIP receptor effects supporting anabolic signaling
- More aggressive fat mobilization reducing need for protein catabolism
Researchers comparing these compounds often use semaglutide as a reference standard given its extensive characterization in the literature.
Retatrutide vs. Dual Agonists (Tirzepatide)
Tirzepatide, a GLP-1/GIP dual agonist, represents an intermediate step between single agonists and retatrutide’s triple-agonist profile:
Body Composition Comparison:
- Tirzepatide demonstrates better lean mass preservation than pure GLP-1 agonists
- Retatrutide shows approximately 15-25% better lean mass retention than tirzepatide
- Both compounds substantially outperform conventional caloric restriction alone
The Glucagon Receptor Difference: The addition of glucagon receptor activation in retatrutide appears to account for:
- Enhanced metabolic rate maintenance
- Greater lipolytic activity
- Improved muscle protein synthesis signaling
- Superior overall body composition remodeling
For researchers interested in comparative studies, tirzepatide provides an important intermediate reference point.
Retatrutide and Growth Hormone Secretagogues
Some research protocols combine retatrutide with growth hormone secretagogues to potentially enhance lean mass preservation:
Theoretical Synergies:
🔬 Ipamorelin + Retatrutide:
- Ipamorelin stimulates growth hormone release
- May enhance muscle protein synthesis
- Could theoretically amplify lean mass preservation
- Limited published data on combination effects
CJC-1295 + Retatrutide:
- CJC-1295 extends growth hormone elevation duration
- May support anabolic environment during caloric deficit
- Potential for synergistic body composition effects
- Requires careful protocol design to assess independent versus combined effects
Researchers exploring these combinations should maintain rigorous controls to distinguish additive versus synergistic effects. Growth hormone secretagogues are available for such comparative research protocols.
Retatrutide and Tissue Repair Peptides
Emerging research interest exists in combining retatrutide with tissue repair and recovery peptides:
BPC-157 Combination Research:
- BPC-157 demonstrates tissue healing and anti-inflammatory properties
- May support muscle recovery during training while on retatrutide
- Theoretical potential to enhance lean mass preservation through improved recovery
- Preliminary research stage with limited published data
TB-500 Considerations:
- TB-500 (Thymosin Beta-4 fragment) supports tissue repair
- May enhance muscle adaptation to resistance training
- Could theoretically complement retatrutide’s metabolic effects
- Requires systematic research to validate combination benefits
These combination approaches represent frontier areas of retatrutide lean mass preservation research, with protocols still being developed and validated.
Safety Considerations and Research Ethics
Responsible research involving retatrutide requires careful attention to safety monitoring and ethical considerations.
Monitoring Parameters in Research Protocols
Comprehensive safety monitoring should include:
Metabolic Markers:
- Fasting glucose and HbA1c
- Insulin and C-peptide levels
- Lipid panel (total cholesterol, LDL, HDL, triglycerides)
- Liver function tests (ALT, AST, bilirubin)
- Kidney function markers (creatinine, eGFR)
Cardiovascular Monitoring:
- Heart rate and blood pressure
- ECG at baseline and periodic intervals
- Assessment for arrhythmias or conduction abnormalities
Gastrointestinal Tolerability:
- Nausea, vomiting, diarrhea incidence and severity
- Appetite changes and satiety patterns
- Gastric emptying assessments if indicated
Nutritional Status:
- Micronutrient levels (vitamin B12, iron, folate, vitamin D)
- Protein status markers (albumin, prealbumin)
- Bone density monitoring in long-term studies
Body Composition Safety:
- Ensure lean mass preservation remains within acceptable ranges
- Monitor for excessive lean tissue loss (>15% of total weight loss)
- Assess functional capacity and strength maintenance
Ethical Considerations
Research involving metabolic peptides must adhere to rigorous ethical standards:
Informed Consent Requirements:
- Clear explanation of investigational nature
- Detailed disclosure of known and potential risks
- Explanation of expected benefits and alternatives
- Right to withdraw without penalty
Research-Only Designation:
- Retatrutide remains investigational in most jurisdictions
- Clear labeling as “For Research Use Only”
- No claims regarding human therapeutic applications
- Compliance with local regulatory frameworks
Data Protection and Privacy:
- Secure storage of research data
- Anonymization of participant information
- Compliance with data protection regulations
- Transparent reporting of findings
Contraindications and Exclusion Criteria
Research protocols typically exclude subjects with:
⚠️ Absolute Contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Severe gastrointestinal disease
- Pregnancy or lactation
- Hypersensitivity to peptide components
Relative Contraindications:
- Significant cardiovascular disease
- Severe renal or hepatic impairment
- History of pancreatitis
- Diabetic retinopathy
- Eating disorders or body dysmorphia
Adverse Event Documentation
Systematic adverse event tracking should include:
Common Events to Monitor:
- Gastrointestinal symptoms (nausea, vomiting, diarrhea, constipation)
- Injection site reactions
- Headache and fatigue
- Changes in heart rate
- Hypoglycemia (especially in diabetic subjects)
Serious Adverse Events:
- Pancreatitis signs or symptoms
- Severe allergic reactions
- Significant cardiovascular events
- Severe hypoglycemia
- Unexplained severe abdominal pain
All research involving retatrutide should operate under appropriate institutional review board oversight and comply with Good Clinical Practice guidelines.
Future Directions in Retatrutide Lean Mass Preservation Research
The field of retatrutide lean mass preservation research continues to evolve rapidly, with several promising avenues for future investigation.
Mechanistic Research Priorities
Molecular Signaling Pathways:
- Detailed characterization of mTOR pathway activation in muscle tissue
- Investigation of AMPK signaling and energy sensing mechanisms
- Analysis of autophagy and protein turnover dynamics
- Examination of satellite cell activation and muscle regeneration
Metabolic Flux Studies:
- Isotope tracer studies to quantify protein synthesis and breakdown rates
- Analysis of substrate utilization patterns (fat vs. carbohydrate vs. protein)
- Measurement of muscle protein fractional synthetic rates
- Characterization of whole-body protein kinetics
Receptor-Specific Contributions:
- Use of selective antagonists to isolate individual receptor contributions
- Comparison of dual-agonist combinations versus triple-agonist effects
- Investigation of tissue-specific receptor expression patterns
- Analysis of receptor desensitization and long-term signaling dynamics
Clinical Application Research
Population-Specific Studies:
👥 Aging Populations: Research examining retatrutide effects in older adults is particularly important given:
- Age-related sarcopenia concerns
- Reduced anabolic response to nutritional and exercise stimuli
- Higher baseline lean mass preservation priority
- Potential for improved functional outcomes and quality of life
Athletic and High-Performance Contexts:
- Body composition optimization in strength and power athletes
- Lean mass preservation during weight class reductions
- Recovery and adaptation during intensive training periods
- Interaction with various training modalities
Metabolic Disease Populations:
- Type 2 diabetes with concurrent sarcopenia
- Metabolic syndrome with muscle quality deterioration
- NAFLD/NASH with body composition abnormalities
- Polycystic ovary syndrome with lean mass concerns
Combination Therapy Research
Synergistic Compound Investigation:
🧪 Retatrutide + Anabolic Agents:
- Combination with selective androgen receptor modulators (SARMs)
- Integration with growth hormone secretagogues
- Pairing with myostatin inhibitors
- Addition of IGF-1 pathway modulators
Retatrutide + Recovery Peptides:
- BPC-157 for enhanced tissue repair
- TB-500 for improved recovery capacity
- Thymosin peptides for immune and regenerative support
- Combination formulations designed for comprehensive support
Retatrutide + Metabolic Modulators:
- NAD+ precursors for mitochondrial function
- AMPK activators for metabolic efficiency
- Mitochondrial-targeted antioxidants
- Nutrient partitioning agents
Long-Term Outcome Research
Extended Duration Studies: Current research primarily covers 24-48 week timeframes, but important questions remain regarding:
- Lean mass preservation during 1-2+ year continuous treatment
- Body composition outcomes after treatment discontinuation
- Metabolic adaptation and weight regain patterns
- Long-term functional capacity and quality of life measures
Maintenance Phase Research:
- Optimal dosing strategies after initial weight loss phase
- Transition protocols from active loss to maintenance
- Lean mass preservation during weight stability
- Prevention of weight regain and body composition deterioration
Personalization and Precision Approaches
Biomarker-Guided Protocols:
📊 Genetic Factors:
- Receptor polymorphisms affecting response
- Metabolic enzyme variations influencing outcomes
- Muscle fiber type distribution and adaptation capacity
- Nutrient metabolism genetic variants
Baseline Characteristics:
- Body composition phenotype (lean vs. obese sarcopenia)
- Metabolic profile (insulin sensitive vs. resistant)
- Training status and muscle quality
- Age, sex, and hormonal status
Response Monitoring:
- Early body composition changes predicting long-term outcomes
- Metabolic markers indicating optimal dosing
- Functional assessments guiding protocol adjustments
- Personalized nutrition and exercise recommendations
Technology Integration
Advanced Assessment Methods:
- AI-enhanced imaging analysis for body composition
- Wearable technology for continuous metabolic monitoring
- Muscle quality assessment through ultrasound or MRI
- Real-time protein synthesis measurement technologies
Data Analytics:
- Machine learning models predicting individual responses
- Big data analysis identifying optimal protocol elements
- Integration of multi-omic data (genomics, proteomics, metabolomics)
- Predictive modeling for lean mass preservation outcomes
These future research directions will further refine understanding of retatrutide lean mass preservation mechanisms and optimize practical applications across diverse research contexts.
Sourcing Research-Grade Retatrutide: Quality and Purity Considerations
The validity of any research investigating retatrutide lean mass preservation depends fundamentally on the quality and purity of the peptide compound used.
Purity Specifications and Testing
Minimum Purity Standards: Research-grade retatrutide should meet stringent purity criteria:
- ≥98% purity by HPLC (High-Performance Liquid Chromatography)
- <2% related peptide impurities
- <5% total impurities including water and acetic acid content
- Verified molecular weight by mass spectrometry
Certificate of Analysis (COA) Requirements:
Essential COA components include: ✓ Batch/lot number for traceability
✓ HPLC chromatogram showing purity profile
✓ Mass spectrometry data confirming molecular identity
✓ Peptide content (mg per pen peptide)
✓ Manufacturing and expiration dates
✓ Storage condition specifications
✓ Reconstitution recommendations
Storage and Handling Best Practices
Pre-Reconstitution Storage:
- Temperature: -20°C or below for long-term storage
- Protection from light exposure
- Sealed in original packaging with desiccant
- Avoid temperature fluctuations and freeze-thaw cycles
Post-Reconstitution Storage:
- Temperature: 2-8°C (refrigerated)
- Protected from light in amber pen peptides or foil-wrapped
- Sterile conditions maintained throughout use
- Use within manufacturer-specified timeframe (typically 14-30 days)
Handling Protocols:
- Sterile technique for all manipulations
- Appropriate personal protective equipment
- Calibrated pipettes and measuring devices
- Documentation of reconstitution date and concentration
Supplier Selection Criteria
Choosing a reputable supplier is critical for research validity:
Key Supplier Characteristics:
🏆 Quality Assurance:
- Comprehensive COA provided with each batch
- Third-party testing verification available
- Consistent purity across multiple batches
- Transparent manufacturing processes
Regulatory Compliance:
- Proper licensing and regulatory adherence
- Clear “For Research Use Only” labeling
- Compliance with local and international regulations
- Appropriate documentation and record-keeping
Customer Support:
- Knowledgeable technical support staff
- Responsive communication channels
- Reconstitution and handling guidance
- Problem resolution protocols
Logistics and Delivery:
- Temperature-controlled shipping methods
- Appropriate packaging for peptide stability
- Tracking and delivery confirmation
- Fast processing and dispatch times
PEPTIDE PRO exemplifies these quality standards, offering research-grade retatrutide with comprehensive documentation, same-day dispatch for orders placed before 1pm Monday-Friday, and professional customer support for researchers across the UK and internationally.
Reconstitution Guidelines
Proper reconstitution ensures peptide stability and accurate dosing:
Standard Reconstitution Protocol:
- Preparation:
- Allow lyophilized peptide to reach room temperature
- Gather bacteriostatic water, sterile syringes, alcohol swabs
- Clean workspace and wash hands thoroughly
- Reconstitution Process:
- Calculate appropriate bacteriostatic water volume for desired concentration
- Draw calculated volume into sterile syringe
- Inject water slowly along pen peptide wall (not directly onto peptide)
- Gentle swirling motion to dissolve (never shake vigorously)
- Allow 2-3 minutes for complete dissolution
- Verification:
- Inspect solution for clarity (should be clear, not cloudy)
- Check for particulates or discoloration
- Confirm complete dissolution of lyophilized powder
- Label pen peptide with reconstitution date and concentration
- Storage:
- Immediately refrigerate at 2-8°C
- Protect from light exposure
- Document in research records
- Use within recommended timeframe
Quality Verification in Research Protocols
Ongoing Quality Checks:
- Visual inspection before each use
- Monitoring for signs of degradation (cloudiness, color change)
- Consistent reconstitution and storage practices
- Documentation of any quality concerns
- Replacement of questionable pen peptides rather than risking compromised research
Batch Consistency:
- Use same batch/lot for entire study cohort when possible
- Document batch numbers in research records
- Compare COAs across batches if multiple lots required
- Statistical analysis to detect batch-related variations
By maintaining rigorous quality standards throughout the research process, investigators ensure that observed effects on retatrutide lean mass preservation reflect true pharmacological actions rather than artifacts of degraded or impure compounds.
Conclusion: The Future of Body Composition Research
The emergence of retatrutide as a research tool has fundamentally transformed the landscape of body composition investigation. Unlike conventional weight-loss interventions that indiscriminately reduce both adipose and lean tissue, retatrutide lean mass preservation represents a paradigm shift toward selective, metabolically favorable body recomposition.
The convergence of GLP-1, GIP, and glucagon receptor activation creates a unique metabolic environment that preferentially mobilizes fat stores while actively protecting—and in some contexts enhancing—skeletal muscle mass. This selective action addresses one of the most significant challenges in metabolic research: achieving substantial fat reduction without compromising the metabolically active, functionally critical lean tissue compartment.
Key Research Implications
For researchers investigating metabolic interventions, retatrutide offers several important advantages:
Mechanistic Insights: The triple-agonist profile provides a window into complex receptor interactions and synergistic metabolic pathways that single-target compounds cannot reveal.
Clinical Relevance: The superior body composition outcomes observed with retatrutide may translate to improved functional capacity, metabolic health, and quality of life measures compared to conventional approaches.
Methodological Advancement: Research protocols developed for retatrutide studies—incorporating advanced body composition assessment, comprehensive metabolic monitoring, and integration of nutritional and exercise variables—advance the field’s methodological sophistication.
Actionable Next Steps for Researchers
Those interested in investigating retatrutide lean mass preservation should consider:
- Protocol Development: Design rigorous experimental protocols incorporating gold-standard body composition assessment methods (DEXA preferred), comprehensive metabolic monitoring, and appropriate control groups or comparators.
- Quality Sourcing: Obtain research-grade retatrutide from reputable suppliers offering comprehensive quality documentation. Contact PEPTIDE PRO for research-grade peptides with full COA documentation and professional support.
- Multidisciplinary Integration: Collaborate across disciplines—endocrinology, exercise physiology, nutrition science, molecular biology—to comprehensively characterize retatrutide’s effects and mechanisms.
- Longitudinal Perspective: Design studies with adequate duration (minimum 12-24 weeks, preferably longer) to capture meaningful body composition changes and metabolic adaptations.
- Mechanistic Investigation: Incorporate molecular and cellular analyses alongside whole-body measurements to elucidate the pathways underlying lean mass preservation.
- Publication and Dissemination: Contribute findings to the scientific literature to advance collective understanding of this novel compound’s effects and optimal application contexts.
The Broader Context
As research into retatrutide and similar multi-agonist peptides continues to expand, the fundamental understanding of metabolic regulation, body composition control, and the complex interplay between different receptor systems will deepen considerably. The insights gained extend beyond any single compound, informing the development of next-generation metabolic interventions and refining therapeutic strategies for obesity, metabolic syndrome, sarcopenia, and related conditions.
The field of retatrutide lean mass preservation research stands at an exciting frontier, where molecular pharmacology, metabolic physiology, and practical application converge to address one of modern medicine’s most pressing challenges: achieving healthy, sustainable body composition optimization that preserves functional capacity and metabolic health.
For researchers ready to contribute to this advancing field, the tools, methodologies, and high-quality compounds are available. The questions are compelling, the potential impact is substantial, and the scientific community eagerly awaits the next wave of discoveries that will further illuminate the remarkable properties of this novel triple-agonist peptide.
