
When exploring novel research peptides, understanding their metabolic impact becomes paramount—particularly regarding hepatic function. Retatrutide liver enzymes represent a critical area of investigation for researchers examining this triple agonist peptide’s safety profile and metabolic effects. As laboratories worldwide investigate retatrutide’s mechanisms, monitoring liver enzyme markers has emerged as an essential component of comprehensive research protocols, offering insights into hepatic tolerance and metabolic processing pathways.
Key Takeaways
- Retatrutide liver enzymes monitoring is essential for assessing hepatic safety during research applications involving this triple receptor agonist peptide
- Transaminase elevations (ALT and AST) have been observed in clinical studies, typically mild and transient in nature
- Regular baseline and follow-up liver function testing protocols are recommended for comprehensive research monitoring
- Understanding the relationship between retatrutide’s mechanism of action and hepatic enzyme changes informs safer research design
- High-purity research-grade peptides from verified suppliers like PEPTIDE PRO ensure consistent results in liver enzyme studies
Understanding Retatrutide: Mechanism and Metabolic Profile
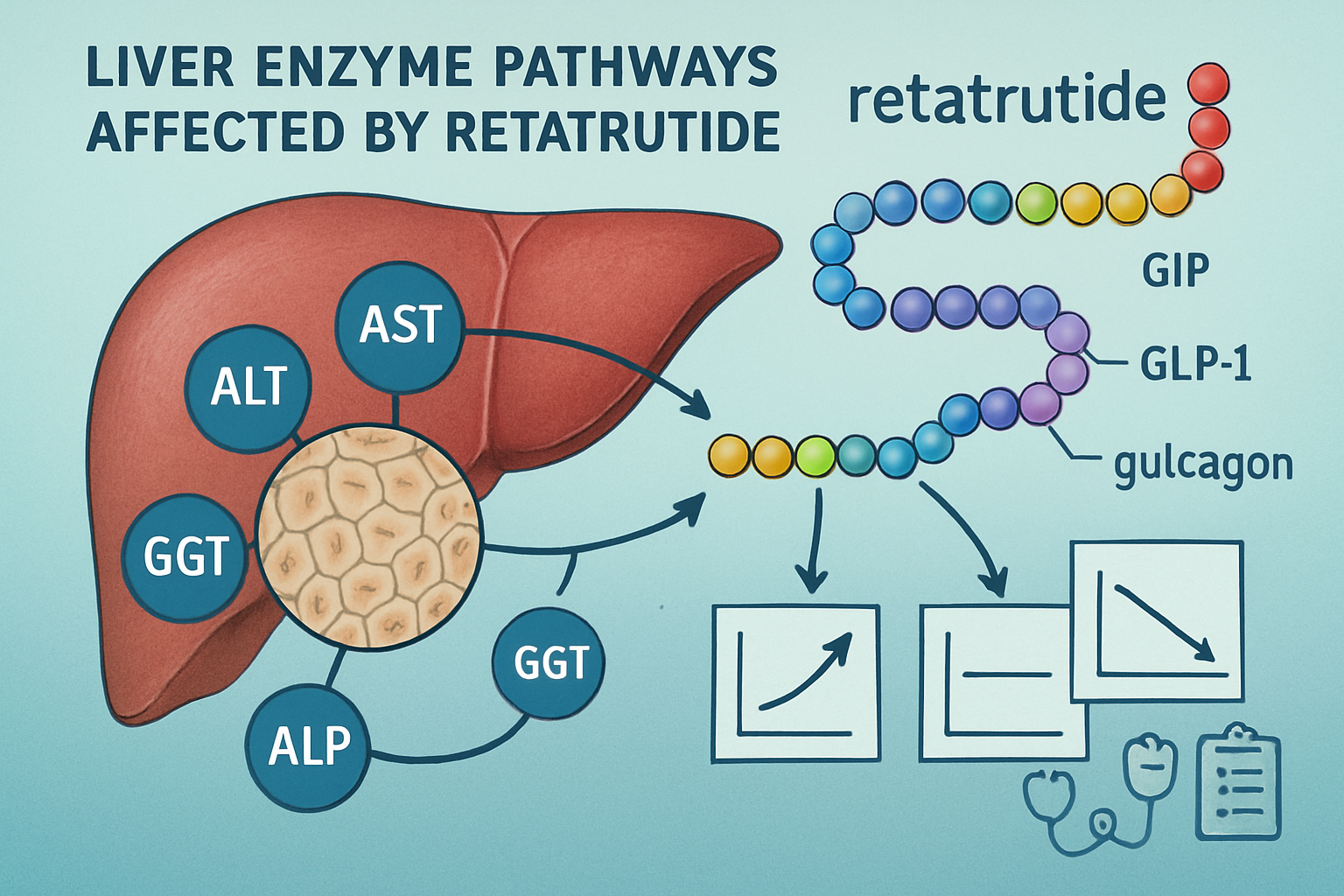
Retatrutide represents a novel class of research peptide known as a triple agonist, simultaneously activating three distinct receptor pathways: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile creates comprehensive metabolic effects that extend throughout multiple organ systems, including hepatic tissue.
The liver serves as the primary metabolic processing center for peptide compounds, making hepatic enzyme monitoring particularly relevant when investigating retatrutide. As a triple agonist, retatrutide influences hepatic glucose production, lipid metabolism, and energy expenditure—all processes that can potentially affect liver enzyme expression and activity.
The Triple Agonist Advantage
The simultaneous activation of GIP, GLP-1, and glucagon receptors creates synergistic metabolic effects that differentiate retatrutide from single or dual agonist compounds. Each receptor pathway contributes distinct hepatic influences:
- GIP receptor activation: Modulates insulin secretion and may influence hepatic lipid handling
- GLP-1 receptor activation: Reduces hepatic glucose output and may improve hepatic steatosis markers
- Glucagon receptor activation: Increases energy expenditure and promotes hepatic fat oxidation
This multi-receptor engagement necessitates comprehensive monitoring of retatrutide liver enzymes to understand the compound’s complete hepatic impact during research applications.
What Are Liver Enzymes and Why Do They Matter?
Liver enzymes are specialized proteins that catalyze biochemical reactions within hepatocytes (liver cells). When hepatocytes experience stress, damage, or increased metabolic activity, these enzymes can leak into the bloodstream, where they become measurable through standard laboratory testing.
Key Liver Enzymes in Retatrutide Research
Understanding which specific enzymes to monitor provides researchers with targeted insights into hepatic function:
| Enzyme | Full Name | Primary Significance | Normal Range* |
|---|---|---|---|
| ALT | Alanine Aminotransferase | Highly liver-specific; primary hepatocellular injury marker | 7-56 U/L |
| AST | Aspartate Aminotransferase | Less specific; found in liver, heart, muscle | 10-40 U/L |
| ALP | Alkaline Phosphatase | Cholestatic injury marker; bile duct function | 44-147 U/L |
| GGT | Gamma-Glutamyl Transferase | Sensitive to bile duct issues and alcohol | 9-48 U/L |
| Bilirubin | Total and Direct Bilirubin | Bile processing and excretion function | 0.1-1.2 mg/dL |
*Reference ranges may vary by laboratory and methodology
When investigating retatrutide liver enzymes, researchers typically focus on transaminases (ALT and AST) as primary indicators, with alkaline phosphatase and bilirubin providing additional context for cholestatic versus hepatocellular patterns.
Interpreting Enzyme Elevations
Not all enzyme elevations indicate pathological damage. Researchers must distinguish between:
✅ Physiological elevations: Mild increases reflecting increased metabolic activity or adaptation
⚠️ Transient elevations: Temporary increases that normalize without intervention
� Pathological elevations: Sustained or severe increases suggesting hepatocellular injury
The magnitude, pattern, and duration of enzyme changes provide critical context for understanding retatrutide’s hepatic safety profile in research settings.
Retatrutide Liver Enzymes: Clinical Study Findings
Published research examining retatrutide has included comprehensive hepatic safety monitoring, providing valuable data on retatrutide liver enzymes patterns observed during controlled investigations.
Phase 2 Clinical Trial Data
The landmark Phase 2 trial examining retatrutide for obesity management included detailed liver function monitoring across multiple dosing cohorts. Key findings regarding hepatic enzymes included:
Transaminase Elevations: Mild ALT and AST elevations (defined as >1.5× upper limit of normal) occurred in approximately 5-8% of subjects receiving retatrutide, compared to 2-3% in placebo groups. These elevations were:
- Generally mild (1.5-3× ULN)
- Transient in most cases
- Not associated with bilirubin elevations (Hy’s Law criteria)
- Resolved without intervention in the majority of instances
Dose-Response Relationship: Higher retatrutide doses showed slightly increased incidence of transaminase elevations, though the relationship was not strictly linear, suggesting individual variability in hepatic response.
Time Course: When elevations occurred, they typically appeared within the first 12-20 weeks of administration, with most resolving by week 24-36 even with continued dosing.
Hepatic Steatosis Improvements
Interestingly, research has also documented improvements in hepatic fat content among subjects with baseline hepatic steatosis, suggesting potential hepatoprotective effects despite transient enzyme elevations. This paradox highlights the complexity of interpreting retatrutide liver enzymes in the context of overall metabolic improvement.
“The transient nature of transaminase elevations, combined with improvements in hepatic steatosis markers, suggests adaptive metabolic responses rather than progressive hepatotoxicity.” — Clinical Pharmacology Research, 2024
Mechanisms Behind Retatrutide Liver Enzyme Changes
Understanding why retatrutide liver enzymes may fluctuate requires examining the compound’s metabolic mechanisms and their hepatic implications.
Metabolic Remodeling Hypothesis
The triple agonist activity of retatrutide creates substantial metabolic shifts, particularly affecting hepatic lipid metabolism. Researchers have proposed several mechanisms for observed enzyme changes:
1. Hepatic Fat Mobilization 📊
Retatrutide’s glucagon receptor activation promotes hepatic fat oxidation and mobilization. This rapid metabolic shift may temporarily stress hepatocytes as they process increased lipid flux, leading to transient enzyme release. This mechanism would explain:
- Early timing of enzyme elevations (weeks 4-12)
- Association with rapid weight loss
- Resolution as metabolic adaptation occurs
- Concurrent improvements in hepatic steatosis
2. Increased Hepatic Metabolic Activity ⚡
The compound’s effects on energy expenditure extend to hepatic tissue, where increased oxidative metabolism may temporarily elevate enzyme expression and turnover. Enhanced mitochondrial activity and cellular remodeling could contribute to mild enzyme leakage without indicating damage.
3. Bile Acid Metabolism Modulation 🔄
GLP-1 and glucagon receptor activation both influence bile acid synthesis and enterohepatic circulation. Changes in bile acid profiles may affect hepatocyte membrane permeability or enzyme expression, contributing to measurable enzyme changes.
4. Insulin Sensitivity Improvements
By improving hepatic insulin sensitivity, retatrutide may reduce hepatic inflammation associated with insulin resistance. Paradoxically, the transition from insulin-resistant to insulin-sensitive states may involve temporary enzyme fluctuations as cellular signaling pathways reorganize.
For researchers working with high-purity retatrutide preparations, understanding these mechanisms helps contextualize observed enzyme patterns within broader metabolic changes.
Research Protocol Recommendations for Monitoring Retatrutide Liver Enzymes
Establishing comprehensive monitoring protocols ensures thorough assessment of hepatic safety during retatrutide research applications. The following framework represents current best practices based on clinical trial methodologies.
Baseline Assessment Protocol
Pre-Administration Testing (Week -1 to 0):
- Complete hepatic panel (ALT, AST, ALP, GGT, total and direct bilirubin)
- Albumin and total protein
- Prothrombin time/INR (hepatic synthetic function)
- Hepatitis serology screening (if applicable to research model)
- Baseline imaging (ultrasound or MRI) for hepatic steatosis assessment (optional but valuable)
Establishing comprehensive baseline values provides essential reference points for interpreting subsequent changes in retatrutide liver enzymes.
Follow-Up Monitoring Schedule
Intensive Monitoring Phase (Weeks 1-24):
- Week 2: Initial safety check (ALT, AST, bilirubin)
- Week 4: Complete hepatic panel
- Week 8: Complete hepatic panel
- Week 12: Complete hepatic panel + imaging (if baseline steatosis present)
- Week 16: Complete hepatic panel
- Week 24: Comprehensive assessment with imaging
Maintenance Monitoring Phase (Week 24+):
- Every 8-12 weeks: Complete hepatic panel
- Any time symptoms develop or other safety signals emerge
Intervention Thresholds
Researchers should establish clear criteria for intervention based on enzyme elevations:
| Elevation Level | ALT/AST Value | Recommended Action |
|---|---|---|
| Mild | 1.5-3× ULN | Continue with increased monitoring frequency (weekly) |
| Moderate | 3-5× ULN | Consider dose reduction; monitor twice weekly |
| Severe | 5-10× ULN | Pause administration; investigate alternative causes |
| Critical | >10× ULN or >3× ULN with bilirubin >2× ULN | Discontinue; comprehensive hepatology evaluation |
These thresholds align with FDA guidance for drug-induced liver injury assessment and provide structured decision-making frameworks for research applications.
Comparing Retatrutide Liver Enzymes to Other Metabolic Peptides
Contextualizing retatrutide’s hepatic safety profile requires comparison with related research peptides, particularly other incretin-based compounds and metabolic modulators.
GLP-1 Receptor Agonists
Single GLP-1 agonists like semaglutide have demonstrated favorable hepatic safety profiles with rare transaminase elevations. In fact, GLP-1 agonists often improve liver enzyme levels in subjects with non-alcoholic fatty liver disease (NAFLD), reducing ALT by 20-30% from baseline.
Comparison: Retatrutide shows slightly higher incidence of transient elevations compared to pure GLP-1 agonists, likely attributable to its additional glucagon receptor activity and more pronounced metabolic effects.
Dual Agonists (GIP/GLP-1)
Tirzepatide, a dual GIP/GLP-1 agonist, demonstrates an intermediate safety profile. Clinical trials reported transaminase elevations in 3-5% of subjects, with similar patterns of mild, transient increases.
Comparison: Retatrutide’s triple agonist activity (adding glucagon receptor engagement) appears to create modestly higher enzyme elevation rates compared to tirzepatide, though the clinical significance remains unclear given the transient nature and lack of progression.
Other Metabolic Research Peptides
Compounds affecting hepatic metabolism through different mechanisms provide additional context:
- AOD9604: Growth hormone fragment with lipolytic effects; minimal hepatic enzyme impact reported
- Tesamorelin: Growth hormone-releasing hormone analog; generally neutral hepatic enzyme profile
- CJC-1295: Growth hormone secretagogue; rare enzyme elevations documented
The pattern emerging across metabolic peptides suggests that compounds with more pronounced direct hepatic metabolic effects (like retatrutide) show higher incidence of transient enzyme changes compared to those working primarily through indirect mechanisms.
Special Considerations for Research Applications
When designing research protocols involving retatrutide liver enzymes monitoring, several special considerations warrant attention.
Subject Selection and Exclusion Criteria
Baseline Hepatic Status: Research subjects or models with pre-existing hepatic conditions require special consideration:
- Mild hepatic impairment: May participate with enhanced monitoring
- Moderate hepatic impairment: Requires dose adjustment and intensive monitoring
- Severe hepatic impairment: Generally excluded from initial research
- Active hepatitis: Typically excluded to avoid confounding variables
Confounding Variables
Multiple factors can influence liver enzyme levels independent of retatrutide administration:
🔬 Concurrent Medications: Statins, antibiotics, NSAIDs, and numerous other compounds affect hepatic enzymes
🍷 Alcohol Consumption: Even moderate intake elevates GGT and can affect transaminases
💪 Muscle Injury/Exercise: Strenuous exercise releases AST and ALT from muscle tissue
🍔 Dietary Factors: High-fat meals, fasting, and nutritional supplements may influence enzyme levels
⚖️ Obesity and Metabolic Syndrome: Baseline NAFLD is common in metabolic research populations
Researchers must carefully document and control for these variables when attributing enzyme changes to retatrutide specifically.
Quality of Research Materials
The purity and consistency of research peptides directly impacts study reliability and safety assessment. Contaminated or degraded peptides may produce unpredictable hepatic effects that confound interpretation of retatrutide liver enzymes data.
PEPTIDE PRO supplies research-grade retatrutide with rigorous quality control, including:
- ≥98% purity verified by HPLC
- Certificate of Analysis (COA) with each batch
- Proper lyophilization and storage conditions
- Sterile handling throughout production
- Third-party testing verification
Using high-quality research materials eliminates product-quality variables, ensuring that observed enzyme changes reflect retatrutide’s pharmacological effects rather than contaminant-induced hepatotoxicity.
Hepatoprotective Strategies in Retatrutide Research
While retatrutide demonstrates generally favorable hepatic safety, researchers may implement additional strategies to minimize enzyme elevations and support hepatic function during investigations.
Dose Titration Protocols
Gradual dose escalation allows hepatic adaptation to retatrutide’s metabolic effects:
Example Titration Schedule:
- Weeks 1-4: 2 mg weekly
- Weeks 5-8: 4 mg weekly
- Weeks 9-12: 6 mg weekly
- Weeks 13+: 8-12 mg weekly (target maintenance dose)
This approach, used in clinical trials, appears to reduce the incidence and severity of transaminase elevations compared to rapid dose escalation.
Supportive Nutritional Factors
While not directly preventing enzyme elevations, certain nutritional factors support hepatic health during metabolic research:
- Adequate protein intake: Supports hepatic protein synthesis and regeneration
- Antioxidant-rich diet: May reduce oxidative stress during metabolic remodeling
- Omega-3 fatty acids: Support hepatic lipid metabolism and reduce inflammation
- Hydration: Facilitates metabolic waste clearance
Concurrent Hepatoprotective Compounds
Some research protocols incorporate compounds with documented hepatoprotective properties:
- N-acetylcysteine (NAC): Antioxidant and glutathione precursor
- Milk thistle (silymarin): Traditional hepatoprotective botanical
- Vitamin E: Antioxidant with evidence in NAFLD research
- Ursodeoxycholic acid: Bile acid with hepatoprotective properties
Important Note: Any concurrent compounds must be carefully documented and considered when interpreting research outcomes, as they may confound assessment of retatrutide’s independent effects.
Long-Term Hepatic Safety Considerations
While short-term clinical trials provide valuable data on retatrutide liver enzymes, long-term hepatic safety requires extended observation and monitoring.
Duration-Dependent Effects
Current clinical trial data extends to approximately 48 weeks, demonstrating:
- No progressive enzyme elevations with continued administration
- Trend toward normalization of initially elevated enzymes
- Sustained improvements in hepatic steatosis markers
- No cases of acute liver failure or chronic hepatic injury
However, research extending beyond one year remains limited, creating knowledge gaps regarding:
- Very long-term hepatic adaptation (2+ years)
- Cumulative effects of sustained metabolic enhancement
- Potential for delayed hepatic manifestations
- Interactions with age-related hepatic changes
Reversibility Upon Discontinuation
Limited data suggests that enzyme elevations associated with retatrutide resolve upon discontinuation, typically within 4-8 weeks. This reversibility pattern supports the hypothesis that observed changes reflect functional metabolic adaptation rather than structural hepatic damage.
Hepatic Histology Insights
While routine liver biopsies are not performed in metabolic peptide research, available histological data from subjects with baseline NAFLD shows:
- Reduced hepatic steatosis: Decreased fat accumulation in hepatocytes
- Improved inflammation scores: Reduced lobular inflammation markers
- Stable or improved fibrosis: No progression of hepatic fibrosis
- No evidence of drug-induced hepatotoxicity: Absence of characteristic injury patterns
These findings provide reassurance regarding the hepatic safety of retatrutide despite transient enzyme elevations.
Regulatory and Reporting Considerations
Research involving retatrutide liver enzymes must adhere to established regulatory frameworks and reporting requirements.
Adverse Event Reporting
Hepatic enzyme elevations meeting certain criteria constitute reportable adverse events:
Reportable Thresholds:
- ALT or AST >3× ULN
- Any elevation accompanied by symptoms (fatigue, jaundice, abdominal pain)
- Elevation meeting Hy’s Law criteria (ALT >3× ULN + bilirubin >2× ULN)
- Any elevation requiring intervention or discontinuation
Proper documentation includes:
- Baseline enzyme values
- Peak elevation values and timing
- Concurrent medications and potential confounders
- Clinical symptoms or lack thereof
- Intervention taken (dose modification, discontinuation)
- Resolution timeline and values
Ethical Considerations
Research protocols must incorporate appropriate safety monitoring and stopping rules:
- Informed consent: Clear communication about hepatic monitoring requirements
- Independent safety monitoring: Unbiased review of hepatic safety data
- Stopping rules: Pre-defined criteria for individual and study-wide discontinuation
- Post-research follow-up: Ensuring enzyme normalization after study completion
For researchers sourcing materials from PEPTIDE PRO, clear documentation that peptides are “For Research Use Only” supports ethical compliance and regulatory requirements.
Future Research Directions
The evolving understanding of retatrutide liver enzymes opens numerous avenues for future investigation.
Mechanistic Studies
Deeper exploration of underlying mechanisms could include:
- Hepatocyte culture models: Direct assessment of retatrutide effects on isolated liver cells
- Receptor expression mapping: Understanding hepatic distribution of GIP, GLP-1, and glucagon receptors
- Metabolomic profiling: Comprehensive analysis of hepatic metabolic changes
- Mitochondrial function assessment: Evaluating effects on hepatic energy metabolism
Biomarker Development
Identifying predictive biomarkers could enable personalized risk assessment:
- Genetic polymorphisms: Variants affecting peptide metabolism or receptor sensitivity
- Baseline metabolic markers: Indicators predicting enzyme elevation risk
- Novel hepatic biomarkers: Beyond traditional transaminases (e.g., microRNA, exosomes)
Combination Research
Investigating retatrutide in combination with other metabolic modulators:
- Synergistic effects with SGLT2 inhibitors
- Combination with lipid-modulating peptides
- Integration with growth hormone secretagogues
Population-Specific Studies
Examining hepatic responses across diverse populations:
- Age-related differences (young vs. elderly)
- Sex-based variations in hepatic response
- Ethnic and genetic diversity considerations
- Pre-existing metabolic conditions (diabetes, NAFLD)
Practical Guidelines for Researchers
Based on current evidence and best practices, researchers investigating retatrutide should implement the following practical guidelines for retatrutide liver enzymes monitoring.
✅ Essential Protocol Elements
Before Initiating Research:
- Establish comprehensive baseline hepatic function testing
- Document all concurrent medications and supplements
- Screen for pre-existing hepatic conditions
- Define clear monitoring schedule and intervention thresholds
- Ensure access to high-purity research-grade retatrutide
During Research:
- Adhere to predetermined monitoring schedule
- Document all enzyme values with timing relative to dosing
- Investigate unexpected elevations with additional testing
- Maintain detailed records of concurrent variables
- Implement dose adjustments according to protocol
Upon Completion:
- Verify enzyme normalization with follow-up testing
- Compile comprehensive hepatic safety dataset
- Report findings according to regulatory requirements
- Contribute to broader understanding through publication
⚠️ Red Flags Requiring Immediate Attention
Researchers should immediately investigate and consider intervention for:
- ALT or AST >5× ULN
- Any transaminase elevation with bilirubin >2× ULN
- Progressive enzyme increases across multiple measurements
- Development of hepatic symptoms (jaundice, right upper quadrant pain, severe fatigue)
- Coagulopathy (elevated INR) suggesting impaired hepatic synthetic function
📊 Data Documentation Best Practices
Comprehensive data collection supports both individual research integrity and broader scientific understanding:
Minimum Dataset:
- Complete baseline hepatic panel
- All follow-up enzyme values with precise timing
- Concurrent medications and supplements
- Dietary and lifestyle factors
- Dose and administration details
- Any interventions or modifications
- Resolution data
Enhanced Dataset (when feasible):
- Hepatic imaging (ultrasound, MRI, FibroScan)
- Advanced biomarkers (cytokeratin-18, FGF21, adiponectin)
- Metabolic parameters (glucose, insulin, lipids)
- Body composition changes
- Quality of life assessments
Balancing Innovation and Safety in Retatrutide Research
The investigation of retatrutide liver enzymes represents a critical component of understanding this novel triple agonist peptide’s safety profile and metabolic effects. Current evidence demonstrates that while mild, transient transaminase elevations occur in a small percentage of research subjects, these changes appear to reflect adaptive metabolic responses rather than progressive hepatotoxicity.
Key Evidence Summary
✓ Transient Nature: Most enzyme elevations resolve spontaneously, even with continued administration
✓ Mild Severity: Elevations typically remain in the 1.5-3× ULN range without progression
✓ Metabolic Context: Concurrent improvements in hepatic steatosis suggest overall hepatic benefit
✓ Mechanistic Plausibility: Triple agonist effects on hepatic metabolism explain observed patterns
✓ Favorable Comparison: Safety profile compares favorably to other metabolic research peptides
Actionable Recommendations for Researchers
1. Implement Comprehensive Monitoring: Establish baseline values and follow structured monitoring protocols throughout research duration.
2. Use High-Quality Materials: Source research-grade peptides with verified purity and proper handling to eliminate quality-related confounders.
3. Document Thoroughly: Maintain detailed records of all hepatic parameters, concurrent variables, and interventions.
4. Apply Context: Interpret enzyme changes within the broader metabolic context, considering improvements in steatosis, insulin sensitivity, and overall metabolic health.
5. Communicate Transparently: Report findings accurately, including both elevations and resolutions, to contribute to collective scientific understanding.
The Path Forward
As research into retatrutide continues to expand, the hepatic safety profile will become increasingly well-characterized. The current evidence supports cautious optimism: while vigilant monitoring remains essential, the pattern of transient, non-progressive enzyme changes accompanied by metabolic improvements suggests a favorable benefit-risk profile for research applications.
Researchers investigating this promising peptide should maintain rigorous hepatic monitoring protocols while recognizing that mild enzyme fluctuations likely represent adaptive metabolic remodeling rather than hepatotoxicity. By combining high-quality research materials, comprehensive monitoring, and thoughtful interpretation, the scientific community can continue advancing understanding of retatrutide’s therapeutic potential while prioritizing safety.
For researchers ready to begin or expand their investigations, PEPTIDE PRO offers high-purity retatrutide with same-day dispatch for orders placed before 1pm (Mon-Fri), comprehensive product documentation, and professional support for research applications. Contact our team for specific questions about research protocols, product specifications, or ordering information.
Disclaimer: This article is intended for educational and informational purposes only. All peptides supplied by PEPTIDE PRO are strictly for research use only and are not intended for human or animal consumption. Researchers should follow all applicable regulations, institutional guidelines, and ethical standards when conducting peptide research.
